Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (15): 2421-2426.doi: 10.3969/j.issn.2095-4344.1134
Previous Articles Next Articles
Mechanism and clinical research advance of muscle atrophy: thinking based on the 65th Annual Meeting of American College of Sports Medicine
Wang Minjia1, Qi Ziyi2, Zhu Weihua1, Sun Junzhi1
- (1Chengdu Sports Institute, Chengdu 610041, Sichuan Province, China; 2Chang’an University, Xi’an 710064, Shaanxi Province, China)
-
Received:2018-11-07Online:2019-05-28Published:2019-05-28 -
Contact:Sun Junzhi, PhD, Associate professor, Chengdu Sports Institute, Chengdu 610041, Sichuan Province, China -
About author:Wang Minjia, PhD, Associate professor, Chengdu Sports Institute, Chengdu 610041, Sichuan Province, China
CLC Number:
Cite this article
Wang Minjia1, Qi Ziyi2, Zhu Weihua1, Sun Junzhi1. Mechanism and clinical research advance of muscle atrophy: thinking based on the 65th Annual Meeting of American College of Sports Medicine[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(15): 2421-2426.
share this article
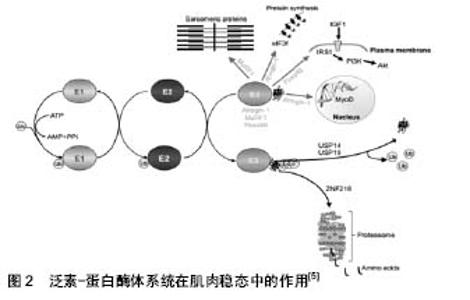
2.1 肌肉萎缩的分子机制 萎缩被定义为组织或器官由于细胞收缩而减小的大小,细胞大小的减少是由细胞器、细胞质和蛋白质的损失引起的。文章讨论关于肌肉萎缩最新的发现和新兴的概念与在生理和病理条件控制肌肉萎缩的途径,主要有泛素蛋白酶体系统和自噬溶酶体通路,这是2个最重要的细胞蛋白水解系统,控制肌肉中蛋白质周转。这些系统在肌肉病理学中的参与以及控制它们的活性的信号通路,仅在最近几年才被揭开,并且证据表明这两个过程在调节整体肌肉稳态中起着关键性的作用。 2.1.1 细胞蛋白水解系统 (1)泛素-蛋白酶体系统:在肌肉中,泛素蛋白酶体系统需要在肌肉活动变化时去除肌节蛋白。它是由泛素、泛素活化酶E1、泛素结合酶E2s、泛素蛋白连接酶E3s、26S蛋白酶体和泛素解离酶,对靶蛋白的整个降解过程不是单一分离而是一种级联反应,见图2。肌肉质量的减少与下列因素有关:①泛素与肌肉蛋白的结合增加;②蛋白酶体ATP-依赖性活性增加;③蛋白质分解能有效地被蛋白酶体抑制剂阻断;④编码泛素的转录物上调,如:一些泛素结合酶(E2),一些泛素蛋白连接酶(E3)和几个蛋白酶体亚基[1]。"
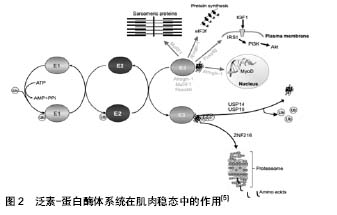

E1酶激活ATP裂解后的泛素蛋白,之后泛素从E1转移到E2酶类的成员,最终的泛素化反应是由E3酶类的成员催化的。E3与E2和蛋白底物结合,诱导泛素从E2转移到底物,一旦底物是聚泛素化的,它被停靠在蛋白酶体上降解,注意,聚泛素链可以通过去泛素化酶泛素特异性加工蛋白酶(USPS)除去。该系统的组成部分,有助于肌肉萎缩的描述。ZNF216参与了肌肉萎缩过程中泛素化蛋白蛋白酶体的识别和传递。AtROGNI-1调节MyoD转录因子和eIF3F的半衰期,这是蛋白质合成的关键。Fbxo40调节IRS1的半衰期,IRS1是IGF1/胰岛素信号传导的重要因子,而MuRF1调节几种肌节蛋白的半衰期。E3泛素连接酶被描绘成绿色,箭头指向它们的底物。注意泛素连接酶可以具有不同的细胞定位并能穿梭到细胞核中。 另外,Alfred L.Goldberg和David J.Glass等发现的开创性基因表达谱分析,比较不同肌肉萎缩模型中的基因表达,可以识别萎缩的肌肉中通常出现上调或下调的一组基因[2]。在不同的疾病模型(包括糖尿病、癌症恶病质、慢性肾功能衰竭、禁食和去神经)均发现肌肉萎缩共同点,通常表达下调的基因被认为调节肌肉成分的损失被称为“萎缩相关(atrophy-related)基因”或“萎缩基因”(atrogenes)[3]。此外,有研究发现编码最强烈诱导的基因是2种特异性泛素连接酶atrogin1(也被称为MAFbx)和MuRF1[4],值得注意的是,至少在去神经支配过程中,这2种基因的最大激活需要转录因子肌原蛋白(myogenin,肌肉发育的重要调控因子)[4]。同时推测,一些其他 E3s在萎缩期间激活,促进可溶性细胞蛋白的清除和限制同化过程[5-6]。 (2)自噬溶酶体通路:细胞自噬是广泛存在于真核细胞内的一种溶酶体介导的蛋白质降解途径。细胞在应答环境胁迫压力下(如细胞应激、营养缺乏、氨基酸饥饿和细胞因子),将胞内物质包裹,送入溶酶体中消化并释放小分子重新循环用于细胞生理活动,从而维持细胞的正常代谢和生存[7]。根据细胞物质运到溶酶体内的途径不同,通常分为3类:巨自噬(mac-roautophagy)、微自噬(microautophagy)和分子伴侣介导的自噬(chaperone-mediated autophagy)。巨自噬的特征是有独特的双层膜结构,包裹胞质成分与溶酶体融合[5],见图3。溶酶体能通过分解大量蛋白和细胞器来保证细胞在营养不足情况下存活,这一过程被称为自噬作用。研究发现,FoxO3转录因子引起了骨骼肌细胞的自噬过程。FoxO3控制着与自噬相关的基因的转录,其中包括LC3和Bnip3,而Bnip3能调节FoxO3在自噬过程中所起的作用,以上过程不受蛋白酶体抑制剂的影响。巨自噬在维持骨骼肌内环境稳态中起着相当重要的作用。自噬缺陷与多种骨骼肌疾病相关[8],过高或过低的巨自噬水平均对骨骼肌正常生理功能有负面影响,自噬过低会引起骨骼肌纤维虚弱无力,而自噬过度激活则会引起骨骼肌质量下降。巨自噬缺陷还会引起骨骼肌糖、脂代谢异常,胰岛素抵抗,造成耐力运动能力下降等[9]。 2.1.2 调整肌肉量的信号通路 有部分研究揭示了肌肉萎缩是一个由特定的信号通路和转录程序控制从而调节肌肉量的活跃过程。 (1)insulin/IGF-1-PI3K-Akt(PKB)/mTOR途径:胰岛素(insulin)或胰岛素样因子1与受体结合,其可以激活PI3K-Akt/FoxO及骨骼肌中特殊的泛素连接酶,通过胰岛素受体底物1(IRS-1)相关的PI3K-Akt-mTOR激活途径,进而促进蛋白的合成。但胰岛素样因子1对泛素蛋白酶体途径也起反向调节作用[10]。当处于胰岛素抵抗或者胰岛素样因子1缺乏的状态下,PI3K-Akt 通路受限,蛋白水解系统启动,导致肌肉蛋白的降解[11]。故Akt 下游的FoxO、Atrogin-1/MAFbx和MuRF1在该种情况下也能诱发肌肉分解致肌萎缩[12]。"
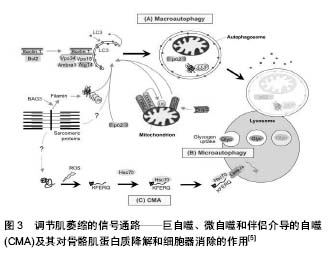
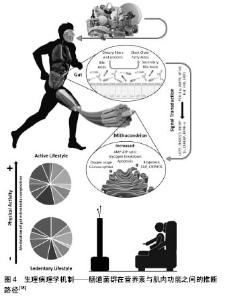
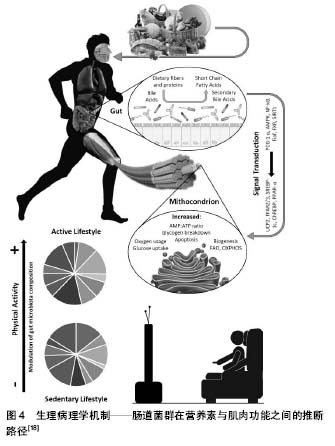
(2)炎症因子-核因子κB途径:有研究发现,核因子κB转录因子在免疫和炎症介质中起重要作用,在骨骼肌中也有表达,它们介导炎症因子。的确,肌萎缩时存在许多因子,但其中只有肿瘤坏死因子α能激活核因子κB并对肌肉萎缩和恶病质产生影响,见图3。即在非活性状态下,核因子κB通过一系列被称为IkB的抑制性蛋白在细胞质中被隔离,在肿瘤坏死因子α的作用下,IkB激酶复合物使IkB磷酸化,导致其泛素化和蛋白酶体降解。这造成了核因子κB的核转位和核因子κB介导的基因转录的激活[13],使其迅速进入细胞核并使转录因子FoxO 启动,导致MuRF1表达,进而引起肌肉蛋白的降解[14-15],发生肌萎缩。同样,肿瘤坏死因子α还可通过核因子κB途径,抑制MyoD等肌肉分化生长因子mRNA水平,导致骨骼肌分化中断,影响受损肌肉组织的修复[16]。 2.1.3 肠道-肌肉轴——骨骼肌质量调控的新途径 肠道菌群是近年来医学界研究的热点,其中“肠-脑轴”是学者研究的着重发力点。而和运动紧密相关的骨骼肌的关系,最近的研究发现:①体育锻炼影响肠道菌群组成,益生菌的干预影响肌肉的功能。机体中可能存在“肠道-肌肉轴(Gut-Muscle Axis)”,肠道菌群可作为营养对肌肉细胞作用的中介;②“肠道-肌肉轴”联通了肠道微生物群与骨骼肌健康之间的交叉对话,该轴对于调节衰老骨骼肌质量,组成和功能的变化起着重要的作用。 Gregory[17]报道机体衰老与肌肉质量和功能(肌肉减少症)的减少有关,肠道菌群正在成为与年龄相关的肌肉衰退的潜在因素。随年龄增长,肠道通透性增加,导致肠道微生物群的生态失调,促进内毒素和其他微生物产物进入循环;进入循环后,脂多糖和其他微生物因子促进炎症信号传导和骨骼肌变化,这是衰老肌肉表型的标志;研究指出营养不良和活动不足是老年人原发性肌少症的主要病理生理因素;肠道菌群通过代谢营养物质产生叶酸、甜菜碱、色氨酸、短链脂肪酸等,影响骨骼肌功能;粪便菌群的组成、物种丰度、多样性与老年人的虚弱及残疾相关,粪便菌群组成变化可能与肌肉功能及肌肉量的下降相关;锻炼影响肠道菌群组成,益生菌的干预影响肌肉功能,见图4。这些都提示在机体中可能存在“肠道-肌肉轴”,肠道菌群可作为营养对肌肉细胞作用的中介;Owen等[19]提出专业运动员的肠道菌群多样性比普通人丰富,因此更有利的促进了生理和代谢健康。但需要排除专业运动员或者专业健身人员饮食的特殊性,以确定是否是运动本身在菌群改变中发挥了核心作用。"
| [1] Lecker H, Goldberg L,Mitch E. Protein degradation by the ubiquitin-proteasome pathway in normal and disease states. J. Am. Soc. Nephrol.2006;17:1807-1819. [2] Gomes D, Lecker H, Jagoe T, et al. Atrogin-1, a muscle-specific F-box protein highly expressed during muscle atrophy. Proc Natl Acad Sci USA.2001;98:14440-14445. [3] Sacheck M, Hyatt P, Raffaello A, et al. Rapid disuse and denervation atrophy involve transcriptional changes similar to those of muscle wasting during systemic diseases. FASEB J. 2007;21:140-155. [4] Bodine C, Latres E, Baumhueter S, et al. Identification of ubiquitin ligases required for skeletal muscle atrophy. Science.2001; 294: 1704-1708. [5] Bonaldo P, Sandri M. Cellular and molecular mechanisms of muscle atrophy. Disease Models and Mechanisms. 2013;6:25-39. [6] Cohen S, Zhai B, Gygi P,et al. Ubiquitylation by Trim32 causes coupled loss of desmin, Z-bands, and thin filaments in muscle atrophy. J Cell Biol. 2012;198:575-589. [7] Mizushima N, Levine B, Cuervo AM, et al. Autophagy fights disease through cellular self-digestion. Nature.2008;451(7182): 1069-1075. [8] Holton JL, Beesley C, Jackson M,et al. Autophagic vacuolar myopathy in twin girls. Neuropathol Appl Neurobiol. 2006;32(3): 253-259. [9] He C, Bassik MC, Moresi V, et al. Exercise-induced BCL2- regulated autophagy is required for muscle glucose homeostasis. Nature.2012;481(7382):511-515.?[10] Nystrom G, Pruznak A, Huber D, et al. Local insulin- like growth factor I prevents sepsis- induced muscle atrophy. Metabolism. 2009;58(6):787-797. [11] Durham WJ, Dillon EL, Sheffield-Moore M. Inflammatory burden and amino acid metabolism in cachexia. Curr Opin Clin Nutr Metab Care. 2009;12:72-77. [12] Léger B, Cartoni R, Praz M, et al. Akt signalling through GSK- 3beta, m TOR and Foxo1 is involved in human skeletal muscle hypertrophy and atrophy. J Physiol.2006;576(Pt 3):923-933. [13] Peterson M, Bakkar N,Guttridge C. NF-κB signaling in skeletal muscle health and disease. Curr Top Dev Biol. , 2011;96:85-11. [14] Ladner KJ, Caligiuri MA, Guttridge DC. Tumor Necrosis Factor-regulated Biphasic Activation of NF-kB Is Required for Cytokine-induced Loss of Skeletal Muscle Gene Products. J Biol Chem.2003;278:2294-2303. [15] Moylan JS, Smith JD, Chambers MA, et al. TNF induction of atrogin-1/MAFbx mRNA depends on Foxo4 expression but not AKT-Foxo1/3 signaling. Am J Physiol Cell Physiol, 2008;295: C989-993. [16] Coletti D, Moresi V, Adamo S, et al. Tumor necrosis factor-alpha gene transfer induces cachexia and inhibits muscle regeneration. Genesis.2005;43:120-128. [17] Gregory J. Gut Microbiota Contribute to Age-Related Changes in Skeletal Muscle Size, Composition, and Function: Biological Basis for a Gut-Muscle Axis. Calcif Tissue Int. 2018;102(4):433-442.[18] Ticinesi A, Lauretani F, Milani C, et al. Aging Gut Microbiota at the Cross-Road between Nutrition, Physical Frailty, and Sarcopenia: Is Therea Gut–Muscle Axis? . Nutrients.2017;9(12):1303. [19] Cronin O, O'Sullivan O, Barton W, et al. Gut microbiota: implications for sports and exercise medicine. Br J Sports Med. 2017;51(9):700-701. [20] Cruz-Jentoft AJ, Baeyens JP, Bauer JM, et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing. 2010;39(4):412-423. [21] Evans WJ, Campbell WW. Sarcopenia and age-related changes in body com- position and functional capacity. J Nutr.1993;123: 465-468. [22] Fried LP, Tangen CM, Walston J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001; 56(3):M146-156.. [23] Marcus RL, Addison O, Dibble LE, et al. Intramuscular adipose tissue, sarcopenia, and mobility function in older individuals. J Aging Res. 2012;2012:629637. [24] Goodpaster BH, Thaete FL, Simoneau JA, Kelley DE. Subcutaneous abdominal fat and thigh muscle composition predict insulin sensitivity independently of visceral fat. Diabetes. 1997;46:1579-1585. [25] Akito Y, Takashi K, Hiroki S. Effects of 24 Months Resistance and Endurance Training On Muscle Quality. Quantity and Physical Functions In Elderly With Long-term Care. [26] Hiroshi A, Keitaro K, Hiroaki K, et al. Leg-press resistance training during 20 days of 6° head-down-tilt bed rest prevents muscle deconditioning. Eur J Appl Physiol.2000;82:30-38. [27] Akito Y, Takashi K, Hiroki S, et al. Effect of 12-month resistance and endurance training on quality, quantity, and function of skeletal muscle in older adults requiring long-term care. Experimental Gerontology.2017;98:230-237. [28] Norheim KL, Cullum CK, Andersen JL, et al. Inflammation Relates to Resistance Training–induced Hypertrophy in Elderly Patients. Med Sci Sports Exerc. 2017;49(6):1079-1085. [29] Koun Y, Akito Y, Shigetoshi S. Muscle atrophy and recovery of individual thigh muscles as measured by magnetic resonance imaging scan during treatment with cast for ankle or foot fracture. J Orthop Surg. 2017;25(3):1-10. [30] Lantto I, Heikkinen J, Flinkkila T, et al. A prospective randomized trial comparing surgical and nonsurgical treatments of acute Achilles ten- don ruptures. Am J Sports Med. 2016;44(9): 2406-2414. [31] Heikkinen J, Lantto I, Flinkkila T. Soleus Atrophy Is Common?After the Nonsurgical Treatment?of Acute Achilles Tendon Ruptures. Am J Sports Med. 2017;45(6):1395-1404. [32] Jessee MB, Buckner SL, Mattocks KT. Very Low Load Resistance Exercise Is Augmented By Blood Flow Restriction In The Lower Body. MEDICINE & SCIENCE IN SPORTS & EXERCISE® ACSM, 2018;49(5):S240. [33] Douris PC, Donoghue J. Blood Flow RestrictionTraining and Functional Improvements in a Single Subject with Parkinson Disease. MEDICINE & SCIENCE IN SPORTS & EXERCISE® ACSM, 2018;49(5):S44. [34] Pellegrinelli V, Rouault C, Rodriguez-Cuenca S, et al. Human Adipocytes Induce Inflammation and Atrophy in Muscle Cells During Obesity. Diabetes.2015;64(9):3121-3134. [35] Sullivan BP, JA. Weiss JA, Garner RT. Altered Skeletal Muscle IGF-1 and miR-206 at Rest and Following Resistance Exercise in Obese Humans. MEDICINE & SCIENCE IN SPORTS & EXERCISE® ACSM, 2018;49(5):S98. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||