Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (4): 606-612.doi: 10.3969/j.issn.2095-4344.1043
Previous Articles Next Articles
Bone morphologic protein signaling pathway in bone regeneration and repair: accurate regulation and treatment targets
Liu Junyin1, Feng Wei1, Xie Yingchun1, Li Yuwan2, Zeng Jitao1, Liu Ziming2, Tu Xiaolin1
- 1Laboratory for Bone Development and Regeneration, Institute of Life Sciences, Chongqing Medical University, Chongqing 400016, China; 2Department of Joint Surgery, the First Affiliated Hospital of Chongqing Medical University, Chongqing 630014, China
-
Online:2019-02-08Published:2019-02-08 -
Contact:Tu Xiaolin, MD, Professor, Laboratory for Bone Development and Regeneration, Institute of Life Sciences, Chongqing Medical University, Chongqing 400016, China -
About author:Liu Junyin, Master candidate, Laboratory for Bone Development and Regeneration, Institute of Life Sciences, Chongqing Medical University, Chongqing 400016, China Feng Wei, Master candidate, Laboratory for Bone Development and Regeneration, Institute of Life Sciences, Chongqing Medical University, Chongqing 400016, China Liu Junyin and Feng Wei contributed equally to this work. -
Supported by:the National Natural Science Foundation of China, No. 8167090813 (to TXL)
CLC Number:
Cite this article
Liu Junyin, Feng Wei, Xie Yingchun, Li Yuwan, Zeng Jitao, Liu Ziming, Tu Xiaolin. Bone morphologic protein signaling pathway in bone regeneration and repair: accurate regulation and treatment targets[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(4): 606-612.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
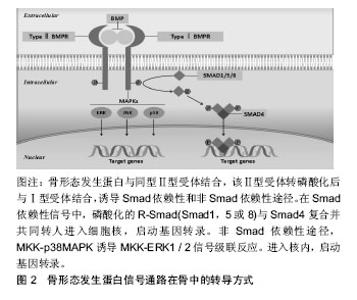
2.1 骨形态发生蛋白在骨再生修复中的作用 在14种骨形态发生蛋白亚型中,骨形态发生蛋白2,4,5,6,7,9都有促进成骨的作用[1-2]。其中骨形态发生蛋白2,7经FDA批准在临床上用来治疗颅面部的骨缺损、骨折不愈合以及脊柱融合[3-4]。据报道,人类骨形态发生蛋白2基因突变引起的疾病称为Brachydactyly type A2 (BDA2; OMIM 112600),主要表现为食指中段发育不良,可伴有小指发育不全。短期内观察骨形态发生蛋白2异位表达可诱导骨形成[5]。骨形态发生蛋白2靶向敲除后,小鼠骨骼正常发育,这说明骨形态发生蛋白2并不影响胚胎小鼠长骨的发育,但随着年龄增长,骨形态发生蛋白2敲除的小鼠与正常小鼠相比,易发生自发性骨折并伴有骨折后长期不愈合,并且由于血管生成和心脏发育中的显著缺陷而导致突变致死[6]。Tan等[7]发现杂合骨形态发生蛋白2敲除小鼠模型中与矮小身材和骨骼异常的人类表型的相似性,表明骨形态发生蛋白2的单倍体不足可能是具有包含骨形态发生蛋白2的预测截短变体和缺失的个体中的主要表型决定因素。骨形态发生蛋白7基因缺失的小鼠在出生后不久就出现死亡,小鼠尸体解剖发现,小鼠肋骨未对齐,剑突形状改变,13肋及颅骨尺寸显著缩小,多趾等一系列骨骼异常[8],但骨形态发生蛋白7调节骨稳态的机制并不明确。1998年Solloway等[9]为了评估体内骨形态发生蛋白6信号的作用,使用靶向基因构建了骨形态发生蛋白6突变的小鼠。产生的突变鼠骨架与野生小鼠相比并无明显差异,然而,在妊娠晚期胚胎中的骨骼发育情况显示胸骨骨架的矿化与野生性小鼠相比出现延迟,但在发育中的长骨和胸骨中的原位杂交结果表明骨形态发生蛋白6在重叠结构域中表达,这表明骨形态发生蛋白2可以功能性补偿在骨形态发生蛋白6突变所造成的骨骼异常。骨形态发生蛋白3由成骨细胞和骨细胞产生,是骨形态发生蛋白家族中唯一反向调节骨量的骨形态发生蛋白亚型[10]。2001年Daluiski等以非洲爪蟾胚胎为模型,发现骨形态发生蛋白3通过抑制骨形态发生蛋白2介导的Msx2并阻断骨形态发生蛋白2介导的骨祖细胞分化成成骨细胞从而来调控成骨活性。生长分化因子5,6和7(分别也称为骨形态发生蛋白14,13和12)不具有强大的成骨性。例如,有学者使用基于腺病毒在C2C12和C3H10T1/2细胞表达,表明这些生长分化因子配体在体内不会诱导强大的碱性磷酸酶活性[11-13]。 2.2 骨形态发生蛋白复合支架材料促骨再生、修复骨缺损 由创伤或骨病所导致的骨缺损是临床上常见的疾病,仅在美国每年估计发生10万例新的骨不连病例[14],并且每年使用自体移植物或同种异体移植物进行超过200万次骨移植手术[15]。利用3D打印技术构建人工骨已成为当前对于骨缺损修复的热点方向。骨形态发生蛋白分子可以促进骨形成,Bze等[16]在尤卡坦小型猪的胫骨中制备了一个临界尺寸的骨折,使用超声递送骨形态发生蛋白6质粒DNA到骨折部位,使骨形态发生蛋白6的瞬时表达和分泌定位于骨折区域。通过微型计算机断层扫描和生物力学分析显示,超声介导的骨形态发生蛋白6基因递送在治疗后6周,骨折动物骨折完全功能性愈合,而对照动物中不愈合明显。此研究结果表明,超声介导的骨形态发生蛋白6向内源性间充质祖细胞的传递可以有效地治疗大型动物的不愈合骨折。在兔股骨缺损模型中利用人源性骨形态发生蛋白2负载介孔二氧化硅/磷酸钙骨水泥介质3D的多孔支架,具有增强血管化和成骨特性[17]。并且通过贻贝激发的聚多巴胺对三维印刷多孔支架进行表面改性后,使得骨形态发生蛋白2负载3D打印介质的成骨分化效应增强[18]。Huang等[19]将介孔硅酸钙支架与骨形态发生蛋白2混合作为3D打印机的原料,发现复合骨形态发生蛋白2后的3DMesocs/PCL支架在骨中显示出更为优良的生物相容性和物理性质。在兔颅骨的临界尺寸缺陷中,在三磷酸钙支架中掺杂骨形态发生蛋白2时,可实现骨再生及骨缺损桥接有关参数的显著优化[20]。骨组织工程支架的添加制造是实现个性化骨组织工程的关键要素,通过以上可以得知,骨形态发生蛋白作为骨生物活性的添加剂,在修复骨缺损以及构建人工骨组织工程中起到了至关重要的作用。 2.3 骨形态发生蛋白在软骨修复中的作用 骨形态发生蛋白虽然在长骨骨细胞中分泌合成,但它的功能并不局限于长骨。除了促进间充质干细胞向成骨细胞谱系分化外,骨形态发生蛋白在软骨内成骨以及促进软骨形成具有重要的作用。骨形态发生蛋白不仅可刺激软骨细胞成熟而且能增强软骨细胞功能,增加软骨细胞中Ⅱ型和Ⅹ型胶原蛋白的表达,以及促进生长板培养物中硫酸盐在糖胺聚糖中的渗入[21-22]。骨形态发生蛋白促进软骨细胞的增殖和分化的机制是通过高表达SOX9,SOX9进而促进软骨中心形成和Ⅱ型、Ⅹ型胶原蛋白的产生[23-25]。文献报道显示,骨形态发生蛋白7可抑制关节软骨的退化,在小鼠膝关节骨性关节炎的模型中,向小鼠关节腔内注射外源性骨形态发生蛋白7,结果显示骨形态发生蛋白7通过降低促炎因子白细胞介素1β,升高抗炎因子白细胞介素10来延缓骨性关节炎的发生及发展[26-28]。Ozeki等[29]在大鼠关节软骨退变模型实验中发现,通过使用骨形态发生蛋白7处理的跟腱移植物可促进半月板再生并对大块半月板缺损有一定的治疗效果。而在软骨细胞内特异性提高骨形态发生蛋白2,对健康的软骨本身并无影响,但是在骨性关节炎的小鼠软骨细胞内提高骨形态发生蛋白2含量,会导致软骨周围骨赘形成[30],但Simson等[31]认为骨形态发生蛋白2可增强软骨形成,且使用硫酸软骨素水凝胶定位骨形态发生蛋白2可以增强关节软骨组织的形成。骨形态发生蛋白4在发育中的四肢中表达,会导致软骨细胞数量及基质软骨的增加,从而进一步可能诱导关节融合[32]。通过以上研究可得知,骨形态发生蛋白各亚型在骨骼发育和骨折范围之外的软骨内骨化所涉及到的生物过程尤为重要,仔细观察在骨形态发生蛋白植入后骨与软骨再生修复的诱导过程,能够发现骨形态发生蛋白介导的骨形成与正常骨骼发生过程具有等效性,很明显骨形态发生蛋白是生物级联反应的发起者,涉及到多种细胞和信号事件,最终导致了功能性骨组织的产生。 2.4 Smad依赖性途径 骨形态发生蛋白信号在骨中与之相关的传导信号主要是Smad依赖性途径(骨形态发生蛋白配体、受体和Smad分子)以及非经典Smad非依赖性途径(丝裂素活化蛋白激酶P38MAPK途径)来调控在骨形成和骨发育中干细胞分化,见图2。"
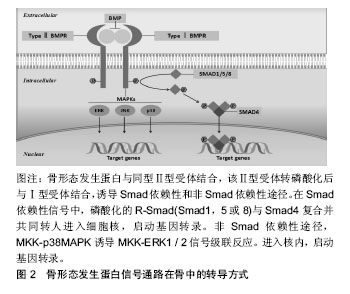
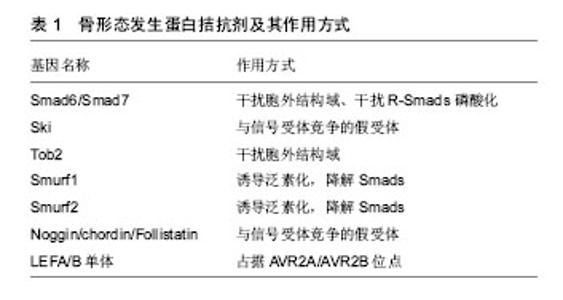
骨形态发生蛋白信号主要通过与Ⅰ型和Ⅱ型骨形态发生蛋白受体结合形成丝苏氨酸四聚体复合物,激活受体磷酸化,转录入核,后者与其他控制基因表达的转录因子结合。Smad蛋白根据其在每条信号转导通路中的不同作用分为3种[33]。受体激活的Smad (receptor-regulated Smad,R-Ssmad)、通用Smad (common-mediator Smad,Co-Smad)和抑制性Smad (inhibitory Smad,I-Smad)。骨形态发生蛋白经典转导途径是通过Smad分子介导,主要依赖于Smad1/5/8 (R-Ssmad) 与Smad4(Co-Smad)聚合后转录Runx2的表达,从而来调节成骨细胞的相关基因表达[34-36]。Smad1是重要的骨形态发生蛋白促进成骨的调节蛋白,在成骨细胞中特异性敲除Smad1,骨形态发生蛋白信号被阻断,成骨分化停滞,骨量减少[37]。破坏骨形态发生蛋白信号转导Smad5的小鼠发育出多种胚胎缺陷,导致颅面及神经管的发育异常[36-37]。Smad1和Smad5协同作用的发挥可调节软骨内骨形成,软骨细胞内特异性敲除Smad1会导致颅骨生长发育的延迟以及软骨生长板的缩短[36-37],而Smad1和Smad5的同时敲除会导致更为严重的软骨内骨发育异常,而Smad1/5/8的同时敲除不会加重软骨的发育异常,这也反向证明了两种分子在调节软骨内骨形成的协同调节作用[38]。Smad4是骨形态发生蛋白与转化生长因子β的共同转录分子,与Smad家族同源性较低,在它的C端功能域没有磷酸化位点。与Smad家族中的其他成员相互作用并形成稳定的异源三聚体。这种异源三聚体的形成是Ⅰ型受体家族信号转导所必须的,骨形态发生蛋白导致的成骨细胞的分化依赖于Smad4[39],缺失Smad4不在对骨形态发生蛋白产生应答。在人类基因中激活突变Smad4引起的疾病为Myhre综合征,患者主要表现为以短小为特征的发育障碍,此类患者通常身材矮小、手脚短、面部畸形、肌肉发达肥大、耳聋并且存在认知延迟。Smad4对于骨稳态的调控是通过调节成骨细胞和骨细胞活性,在成骨细胞中特异性敲除Smad4,发现Smad4通过调节成骨细胞凋亡和矿化以及降低破骨细胞活性从而来增加骨量。2013年时,有学者发现在成骨细胞中敲除Smad4,影响骨基质形成,骨量降低,且小鼠出现严重的生长停滞以及牙齿咬合不良且小鼠难以成活到8周。综上所述,可以看出Smad4对于骨稳态的调控功能是巨大的,但是Smad4如何对于骨稳态进行调控,暂无统一明确的标准,仍需进一步的研究。 2.5 骨形态发生蛋白受体 骨形态发生蛋白受体有3种,包括1A型骨形态发生蛋白受体(BMPR1A/ALK3)、1B型骨形态发生蛋白受体(BMPR1B/ALK6)和I型激活受体(AcvR1/ALK2)。骨形态发生蛋白对于成骨作用的发挥,主要依靠这3种骨形态发生蛋白受体。进展性纤维化发育不良是一种罕见的遗传性疾病,其特征为异位骨化(HO)[40-41],即除正常生理钙化外,在骨骼组织以外发生的钙盐沉积。这种改变通常发生于骨骼的肌腱附着处、韧带、血管壁、骨筋膜等处。在小鼠和人的体内激活突变AcvR1可以导致小鼠的异位骨化[42],2015年Sarah 等[43]发现将AcvR1第206位精氨酸突变为组氨酸可以诱导异位骨形成,表型复制人类进行性骨化性纤维发育不良并且此诱导作用可以被拮抗骨形态发生蛋白配体的激活素家族所阻断,这意味着激活素A的人抗体可能是一种潜在的进行性骨化性纤维发育不良的治疗方法。且AcvR1对于促进上皮-间质的转化以及异位成骨是通过经典的骨形态发生蛋白-Smads途径介导的[44]。研究发现在小鼠体内注射突变型ALK5,也可诱导异位骨化[45],复制人体进行性骨化性纤维发育不良表型。而LDN-193189作为一种骨形态发生蛋白1型受体的拮抗剂,可以抑制骨形态发生蛋白通过ALK2信号的转导,降低异位骨化。2008年Yu等[46]证明,单独的ALK2突变并不能诱导异位骨化,ALK2诱导的异位骨化还依赖于炎性环境。另一方面,通过过表达DN-BMPRⅡ或者在成骨细胞中敲除骨形态发生蛋白1A型受体,可导致骨量减少[47-48]。3种骨形态发生蛋白受体在长骨发育中的作用通常是可重复的。1A型骨形态发生蛋白受体和1B型骨形态发生蛋白受体在软骨的生长发育和长骨的生长发育中的作用基本相同,并且二者有协同调节的作 用[49]。Zou等[50]单独敲除1A型骨形态发生蛋白受体或1B型骨形态发生蛋白受体均可导致小鼠胚胎期软骨发育不良,小鼠四肢处于软骨前状态,并且成软骨标志物SOX9及SOX5表达量均下降,而1A型骨形态发生蛋白受体和1B型骨形态发生蛋白受体同时缺失可导致小鼠更为严重的全身性的软骨发育不良。骨形态发生蛋白信号通路主要与IHH/PTHrp通路相互协调参与软骨形态发生的调控,IHH作用于骨形态发生蛋白信号的上游,后者通过1A型骨形态发生蛋白受体在增生前软骨细胞核关节周围区域调节PTHrp的产生,从而起到控制软骨细胞分化的作用。但在其他组织发育中1A型骨形态发生蛋白受体和1B型骨形态发生蛋白受体又可有相反的作用,比如1A型骨形态发生蛋白受体和1B型骨形态发生蛋白受体在脊髓损伤后,神经胶质的增生有相反的作用[51]。 2.6 骨形态发生蛋白拮抗剂 骨形态发生蛋白信号通路受到多种调控,而哪些因素限制了成骨期间的骨形态发生蛋白活性?骨形态发生蛋白抑制剂主要包括与信号受体竞争的假受体,抑制性阻断细胞信号Smads、结合Smad1/5的胞内结合蛋白以及诱导泛素化因子和可降解Smads蛋白,见表1。 Smad6/Smad7是Smad的抑制物,他们通过阻断丝氨酸/苏氨酸激酶受体来阻断信号传导,主要表现为Smad6/Smad7抑制Smad1/5的磷酸化进而阻止与Smad4形成复合物[52]。抑制性Smads调节转导的Smads,并且抑制性Smads信号也受到调控,从而间接调控转导Smads。Smad 6受到STAM(AMSH)的SH3结构域和组蛋白脱乙酰化酶的调控。AMSH是一种信号转导适配体蛋白,AMSH与Smad 6结合并阻碍其抑制作用,从而调节Smad1的磷酸化和下游基因的转录[53]。Ski(Sloan-Kettering)反转录病毒是致癌基因,可作为骨形态发生蛋白、转化生长因子β和激活素信号传导的Smad依赖性共抑制剂[54-55]。Ski可以抑制非洲爪蟾和哺乳动物细胞中的骨形态发生蛋白信号传导和靶基因的活化,从而使得骨形态发生蛋白无法促进成骨分化。Ski通过与MH2蛋白结构域结合从而来阻止骨形态发生蛋白的信号转导。Ski在中枢神经系统和颅面结构的形态发生中起着重要作用,而缺失Ski的小鼠出现颅骨发育不全,颅骨畸形和下颌骨畸形等表型[55-56]。Ski基因由骨骼肌和骨骼表达,并且过度表达Ski的转基因小鼠骨质减少[56]。这也说明了骨形态发生蛋白对成骨细胞分化的影响在维持骨量和结构方面起着重要作用。Erb B-2(Tob)基因是PC3/BTG/Tob家族中的一员。是一种与Erb相互作用的345个氨基酸的蛋白质,属于一种受体型蛋白酪氨酸激酶。参与细胞的增殖和分化[57-58]。Tob通过MH2结构域结合,降低骨形态发生蛋白2的转录活性,Tob主要在成骨细胞中表达,骨形态发生蛋白2可以提高其转录活性,这意味着Tob对于骨形态发生蛋白2的调控是一种负反馈机制,突变Tob基因的小鼠发现其成骨细胞数量增加、骨形成增加这表明Tob像Ski一样抑制骨形成[59]。Smad泛素化调节因子1(Smurf)1和Smurf2是Smad特异性E3泛素连接酶,选择性与BMP受体激活相互作用的Smad蛋白结合并讲降解其活性。Smurf1和2触发Smads的泛素化和蛋白酶体降解,从而失活BMP的转导活性[60-61]。Smurf 1优先与Smad 1和5相互作用,灭活骨形态发生蛋白信号。 以上介绍的都是细胞内因子对骨形态发生蛋白的调节,接下来介绍限制骨形态发生蛋白作用的分泌多肽。Zimmerman等[62]发现Noggin通过结合骨形态发生蛋白,从而阻断骨形态发生蛋白与其受体结合。Noggin已被用作阻断骨形态发生蛋白功能的工具,因为它是骨形态发生蛋白活性的相对特异性抑制剂,并且似乎没有独立于骨形态发生蛋白结合的作用,Noggin以不同程度的亲和力结合骨形态发生蛋白2,4,5,6和7,生长分化因子5,生长分化因子6和Vg1,但不与转化生长因子β家族相结合[62-63]。在骨钙蛋白启动子控制下过表达Noggin蛋白,突变鼠出现骨量减少并易诱发病理性骨 折[64],表明Noggin直接或间接通过结合骨形态发生蛋白从而控制骨的正常发育。Chordin调控骨形态发生蛋白的作用方式与Noggin相似,阻止骨形态发生蛋白受体信号传导,并且不与转化生长因子β家族结合,Chordin突变的小鼠死于胚胎期,主要是由于内耳和外耳发育缺陷、咽、肛门心血管组织异常[65]。双突变noggin/chordin小鼠前脑缺失,中胚层的发育异常[65]。这表明Chordin和Noggin是对于胚胎期的正常发育是必需的。Follistatin最初被发现作为一种激活素结合蛋白,抑制激活素信号传导,但它也可以通过结合骨形态发生蛋白4来抑制骨形态发生蛋白4表达和信号传导[66-67]。Noggin/ chordin/Follistatin作为骨形态发生蛋白的拮抗配体,与骨形态发生蛋白在上游结合,骨形态发生蛋白经初始刺激后,Noggin/chordin/Follistatin能够迅速抑制骨形态发生蛋白途径,但是Zohar等[68]提出的模型中,外源骨形态发生蛋白拮抗配体的连续存在促进了大鼠颅骨细胞的软骨形成。因此对于Noggin/chordin/ Follistatin抑制骨形态发生蛋白的体内评估模型数据,依然需要进一步的实验证据来确定Noggin/chordin/ Follistatin广泛的适用性。 "

| [1] Luu HH, Song WX, Luo X, et al. Distinct roles of bone morphogenetic proteins in osteogenic differentiation of mesenchymal stem cells.J Orthop Res. 2007;25(5):665-677. [2] Abula K, Muneta T, Miyatake K, et al. Elimination of BMP7 from the developing limb mesenchyme leads to articular cartilage degeneration and synovial inflammation with increased age. FEBS letters.2015; 589: 1240-1248. [3] Razzouk S, Sarkis R. BMP-2: biological challenges to its clinical use. The New York state dental journal. 2012;78:37-39. [4] Smoljanovic T,Caric D,Bojanic I.RE: Clinical applications of bone morphogenetic proteins: current evidence. Kanakaris, NK; Giannoudis, PV; JSOA 17(3):133-46, 2008.J Surg Orthop Adv. 2010 Summer;19(2): 135-137; author reply 137. [5] Noel D, Gazit D, Bouquet C, et al. Short-term BMP-2 expression is sufficient for in vivo osteochondral differentiation of mesenchymal stem cells.Stem cells 2004;22:74-85. [6] Tsuji K, Bandyopadhyay A, Harfe BD, et al.BMP2 activity, although dispensable for bone formation, is required for the initiation of fracture healing.Nature genetics.2006;38:1424-1429. [7] Tan TY,Gonzaga-Jauregui C, Bhoj EJ, et al. Monoallelic BMP2 variants predicted to result in haploinsufficiency cause craniofacial, skeletal, and cardiac features overlapping those of 20p12 deletions. Am J Hum Genet. 2017 Dec 7;101(6):985-994. [8] Luo G, Hofmann C, Bronckers AL, et al. BMP-7 is an inducer of nephrogenesis, and is also required for eye development and skeletal patterning. Genes Dev. 1995 Nov 15;9(22):2808-2820. [9] Solloway MJ, Dudley AT, Bikoff EK,et al. Mice lacking Bmp6 function. Dev genet.1998;22:321-339. [10] Daluiski A, Engstrand T, Bahamonde ME, et al. Bone morphogenetic protein-3 is a negative regulator of bone density. Nature genet.2001; 27:84-88. [11] Celil AB,Hollinger JO,Campbell PG.Osx transcriptional regulation is mediated by additional pathways to BMP2/Smad signaling. J Cell Biochem. 2005 Jun 1;95(3):518-528. [12] Kang Q, Song WX, Luo Q, et al. A comprehensive analysis of the dual roles of BMPs in regulating adipogenic and osteogenic differentiation of mesenchymal progenitor cells.Stem cells Dev.2009;18:545-559. [13] Luu HH, Song WX, Luo X, et al. Distinct roles of bone morphogenetic proteins in osteogenic differentiation of mesenchymal stem cells. J Orthop Res.2007;25:665-677. [14] Hak DJ, Fitzpatrick D, Bishop JA, et al. Delayed union and nonunions: epidemiology, clinical issues, and financial aspects. Injury.2014;45 Suppl 2:S3-7. [15] Xavier JR,Thakur T,Desai P,et al.Bioactive nanoengineered hydrogels for bone tissue engineering: a growth-factor-free approach. ACS nano. 2015;9:3109-3118. [16] Bez M, Sheyn D, Tawackoli W, et al.In situ bone tissue engineering via ultrasound-mediated gene delivery to endogenous progenitor cells in mini-pigs. Sci Transl Med. 2017 May 17;9(390). pii: eaal3128. [17] Li C,Jiang C,Deng Y, et al.RhBMP-2 loaded 3D-printed mesoporous silica/calcium phosphate cement porous scaffolds with enhanced vascularization and osteogenesis properties. Sci Rep. 2017;7:41331.[18] Lee SJ, Lee D, Yoon TR, et al. Surface modification of 3D-printed porous scaffolds via mussel-inspired polydopamine and effective immobilization of rhBMP-2 to promote osteogenic differentiation for bone tissue engineering. Acta biomaterialia.2016;40:182-91. [19] Huang KH, Lin YH, Shie MY, et al. Effects of bone morphogenic protein-2 loaded on the 3D-printed MesoCS scaffolds.J Formos Med Assoc. 2018;117(10):879-887.[20] Chen TH,Ghayor C,Siegenthaler B,et al.Lattice microarchitecture for bone tissue engineering from calcium-phosphate compared to titanium. Tissue Eng Part A. 2018;24(19-20):1554-1561. [21] Grimsrud CD, Romano PR, D'Souza M, et al. BMP signaling stimulates chondrocyte maturation and the expression of Indian hedgehog. J Orthop Res. 2001;19(1):18-25.[22] De Luca F, Barnes KM, Uyeda JA, et al. Regulation of growth plate chondrogenesis by bone morphogenetic protein-2. Endocrinology. 2001;142:430-436. [23] Minina E, Kreschel C, Naski MC,et al. Interaction of FGF, Ihh/Pthlh, and BMP signaling integrates chondrocyte proliferation and hypertrophic differentiation. Dev cell. 2002;3:439-449. [24] Kobayashi T, Lyons KM, McMahon AP, et al. BMP signaling stimulates cellular differentiation at multiple steps during cartilage development. Proc Natl Acad Sci U S A. 2005 Dec 13;102(50):18023-18027. [25] Komatsu Y, Kaartinen V, Mishina Y. Cell cycle arrest in node cells governs ciliogenesis at the node to break left-right symmetry. Development.2011;138:3915-3920. [26] Hayashi M,Muneta T,Ju YJ,et al.Weekly intra-articular injections of bone morphogenetic protein-7 inhibits osteoarthritis progression. Arthritis Res Ther. 2008;10(5):R118. 16:03 2019-1-7[27] Sekiya I,Tang T,Hayashi M,et al.Periodic knee injections of BMP-7 delay cartilage degeneration induced by excessive running in rats. J Orthop Res. 2009;27(8):1088-1092. [28] Takahashi T, Muneta T, Tsuji K, et al. BMP-7 inhibits cartilage degeneration through suppression of inflammation in rat zymosan-induced arthritis. Cell Tissue Res.2011;344:321-332. [29] Ozeki N, Muneta T, Koga H, et al. Transplantation of Achilles tendon treated with bone morphogenetic protein 7 promotes meniscus regeneration in a rat model of massive meniscal defect. Arthritis Rheum. 2013;65(11):2876-2886. [30] Blaney Davidson EN, Vitters EL, et al. Inducible chondrocyte-specific overexpression of BMP2 in young mice results in severe aggravation of osteophyte formation in experimental OA without altering cartilage damage. Ann Rheum Dis. 2015;74(6):1257-1264. [31] Simson JA, Strehin IA, Lu Q, et al. An adhesive bone marrow scaffold and bone morphogenetic-2 protein carrier for cartilage tissue engineering.Biomacromolecules. 2013;14:637-643. [32] Duprez D,Bell EJ,Richardson MK,et al. Overexpression of BMP-2 and BMP-4 alters the size and shape of developing skeletal elements in the chick limb. Mech Dev. 1996;57(2):145-157. [33] Kutz WE, Wang LW, Dagoneau N, et al. Functional analysis of an ADAMTS10 signal peptide mutation in Weill-Marchesani syndrome demonstrates a long-range effect on secretion of the full-length enzyme.Human Mutat.2008;29:1425-1434. [34] Lee KS, Hong SH, Bae SC. Both the Smad and p38 MAPK pathways play a crucial role in Runx2 expression following induction by transforming growth factor-beta and bone morphogenetic protein. Oncogene.2002;21:7156-7163. [35] Afzal F, Pratap J, Ito K,et al.Smad function and intranuclear targeting share a Runx2 motif required for osteogenic lineage induction and BMP2 responsive transcription. J Cell Physiol. 2005;204(1):63-72. [36] Franceschi RT,Xiao G.Regulation of the osteoblast-specific transcription factor, Runx2: responsiveness to multiple signal transduction pathways. J Cell Biochem. 2003;88(3):446-454. [37] Wang M, Jin H, Tang D, et al. Smad1 plays an essential role in bone development and postnatal bone formation. Osteoarthritis Cartilage. 2011;19(6):751-762. [38] Keller B, Yang T, Chen Y,et al. Interaction of TGFbeta and BMP signaling pathways during chondrogenesis. PloS one.2011;6:e16421. [39] Retting KN,Song B,Yoon BS,et al. BMP canonical Smad signaling through Smad1 and Smad5 is required for endochondral bone formation.Development. 2009;136:1093-1104. [40] Shore EM, Xu M, Feldman GJ,et al.A recurrent mutation in the BMP type I receptor ACVR1 causes inherited and sporadic fibrodysplasia ossificans progressiva. Nature genetics.2006;38:525-527. [41] Shen Q, Little SC, Xu M, et al. The fibrodysplasia ossificans progressiva R206H ACVR1 mutation activates BMP-independent chondrogenesis and zebrafish embryo ventralization. J Clin Invest. 2009;119(11):3462-3472. [42] Medici D, Shore EM, Lounev VY, et al. Conversion of vascular endothelial cells into multipotent stem-like cells. Nature Med. 2010; 16:1400-1406. [43] Hatsell SJ, Idone V, Wolken DM, et al. ACVR1R206H receptor mutation causes fibrodysplasia ossificans progressiva by imparting responsiveness to activin A. Sci Transl Med. 2015;7(303):303ra137. [44] van Dinther M,Visser N,de Gorter DJ,et al.ALK2 R206H mutation linked to fibrodysplasia ossificans progressiva confers constitutive activity to the BMP type I receptor and sensitizes mesenchymal cells to BMP-induced osteoblast differentiation and bone formation. J Bone Miner Res. 2010;25(6):1208-1215. [45] Hao J, Ho JN, Lewis JA,et al. In vivo structure-activity relationship study of dorsomorphin analogues identifies selective VEGF and BMP inhibitors.ACS chemical biology 2010;5:245-253. [46] Yu PB, Deng DY, Lai CS, et al. BMP type I receptor inhibition reduces heterotopic [corrected] ossification. Nature medicine. 2008;14: 1363-1369. [47] Yang C, Yang L, Wan M, et al. Generation of a mouse model with expression of bone morphogenetic protein type II receptor lacking the cytoplasmic domain in osteoblasts. Ann N Y Acad Sci. 2010;1192: 286-291. [48] Mishina Y, Starbuck MW, Gentile MA, et al. Bone morphogenetic protein type IA receptor signaling regulates postnatal osteoblast function and bone remodeling. J Biol Chem. 2004;279(26): 27560-27566. [49] Yoon BS, Ovchinnikov DA, Yoshii I,et al. Bmpr1a and Bmpr1b have overlapping functions and are essential for chondrogenesis in vivo. Proc Natl Acad Sci U S A. 2005;102(14):5062-5067. [50] Zou H, Wieser R, Massague J, et al. Distinct roles of type I bone morphogenetic protein receptors in the formation and differentiation of cartilage. Genes Dev. 1997;11(17):2191-2203. [51] Sahni V, Mukhopadhyay A,Tysseling V,et al. BMPR1a and BMPR1b signaling exert opposing effects on gliosis after spinal cord injury. J Neurosci. 2010;30(5):1839-1855. [52] Miyazono K.Signal transduction by bone morphogenetic protein receptors: functional roles of Smad proteins. Bone.1999;25:91-93. [53] Itoh F, Asao H, Sugamura K, et al. Promoting bone morphogenetic protein signaling through negative regulation of inhibitory Smads. EMBO J. 2001;20(15):4132-4142. [54] Bai S,Shi X,Yang X,et al. Smad6 as a transcriptional corepressor. J Biol Chem. 2000;275(12):8267-8270. [55] Luo K,Stroschein SL,Wang W, et al.The Ski oncoprotein interacts with the Smad proteins to repress TGFbeta signaling. Genes Dev. 1999; 13(17):2196-2206. [56] Wang W,Mariani FV,Harland RM,et al. Ski represses bone morphogenic protein signaling in Xenopus and mammalian cells. Proc Natl Acad Sci U S A. 2000;97(26):14394-14399. [57] Lana DP, Leferovich JM, Kelly AM,et al. Selective expression of a ski transgene affects IIb fast muscles and skeletal structure. Dev Dyn. 1996;205(1):13-23. [58] Guehenneux F, Duret L, Callanan MB, et al. Cloning of the mouse BTG3 gene and definition of a new gene family (the BTG family) involved in the negative control of the cell cycle. Leukemia. 1997;11(3): 370-375. [59] Ajima R,Ikematsu N, Ohsugi M, et al. Cloning and characterization of the mouse tob2 gene.Gene 2000;253:215-220. [60] Yoshida Y,Tanaka S,Umemori H,et al.Negative regulation of BMP/Smad signaling by Tob in osteoblasts. Cell. 2000;103:1085-1097. [61] Zhu H, Kavsak P, Abdollah S, et al. A SMAD ubiquitin ligase targets the BMP pathway and affects embryonic pattern formation. Nature.1999; 400:687-693. [62] Zhang Y, Chang C, Gehling DJ,et al. Regulation of Smad degradation and activity by Smurf2, an E3 ubiquitin ligase. Proc Natl Acad Sci U S A. 2001;98(3):974-979. [63] Zimmerman LB, De Jesus-Escobar JM, Harland RM. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4.Cell. 1996;86:599-606. [64] Aspenberg P,Jeppsson C, Economides AN. The bone morphogenetic proteins antagonist Noggin inhibits membranous ossification. J Bone Miner Res. 2001;16(3):497-500. [65] Gazzerro E, Du Z, Devlin RD,et al. Noggin arrests stromal cell differentiation in vitro. Bone.2003;32:111-119. [66] Bachiller D,Klingensmith J,Kemp C,et al.The organizer factors Chordin and Noggin are required for mouse forebrain development. Nature. 2000;403:658-661. [67] Fainsod A, Deissler K, Yelin R,et al. The dorsalizing and neural inducing gene follistatin is an antagonist of BMP-4. Mech Dev. 1997; 63(1):39-50. [68] Zohar R,McCulloch CA,Sampath K,et al.Flow cytometric analysis of recombinant human osteogenic protein-1 (BMP-7) responsive subpopulations from fetal rat calvaria based on intracellular osteopontin content. Matrix Biol. 1998;16(6):295-306. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||