Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (4): 584-590.doi: 10.3969/j.issn.2095-4344.1041
Previous Articles Next Articles
Magnetic fields applied in the field of bone biolegy: confirmed and unconfirmed problems
Zhang Lingyu1, 2, Jiang Jingyi1, 2, Fan Yubo1, 2, Zheng Lisha1, 2
- 1School of Biological Science and Medical Engineering, Beihang University, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, Beijing 100083, China; 2Advanced Innovation Center for Biomedical Engineering of Beihang University, Beijing 100083, China
-
Online:2019-02-08Published:2019-02-08 -
Contact:Zheng Lisha, PhD, Associate professor, Master’s supervisor, School of Biological Science and Medical Engineering, Beihang University, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, Beijing 100083, China; Advanced Innovation Center for Biomedical Engineering of Beihang University, Beijing 100083, China -
About author:Zhang Lingyu, Master candidate, School of Biological Science and Medical Engineering, Beihang University, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, Beijing 100083, China; Advanced Innovation Center for Biomedical Engineering of Beihang University, Beijing 100083, China -
Supported by:the National Natural Science Foundation of China, No. 11572030, 11120101001 and 11421202 (to ZLS); the National Key Research & Development Program of Ministry of Science and Technology of China No. 2017YFC0108505 and 2017YFC0108500
CLC Number:
Cite this article
Zhang Lingyu, Jiang Jingyi, Fan Yubo, Zheng Lisha. Magnetic fields applied in the field of bone biolegy: confirmed and unconfirmed problems[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(4): 584-590.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
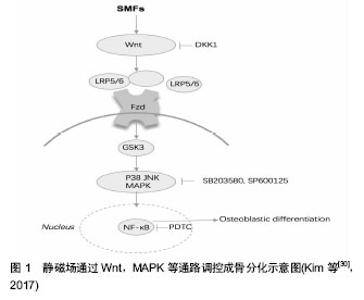
2.1 常用磁场 生物生存的环境存在着多种多样的磁场,如地磁、宇宙磁辐射,以及随着工业和科技的发展增加的各种家用电器、医疗设备、通讯设备等产生的磁场,它们对生物的行为活动存在潜在影响。在研究中,一般根据磁场强度和方向可分为静磁场和动磁场,静磁场又称为(稳)恒磁场,由永磁体或通有恒定电流的线圈产生,磁场的强度和方向都不随时间的变化而改变;动磁场一般由线圈中不断变化的电流产生,可按照磁感应强度和方向的不同,分为周期变化的交变磁场,非周期变化的脉冲电磁场(以及以固定频率旋转的旋转磁场。 按照强度大小,可分为超强磁场(>5 T),强磁场 (1-5 T),中等强度磁场(1 mT-1 T)以及弱磁场(<1 mT)。还根据频率可分为低频磁场、中频磁场和高频磁场。高强度、中高频的磁场对生物体的作用主要为热效应,中等强度和低频率磁场则主要表现为非热效应(即生物学效应),目前的研究以磁场的生物学效应为主。其中,脉冲电磁场和静磁场的应用最多,交变磁场和旋转磁场的研究相对较少,中等强度和低频磁场的效应较为热门。 2.2 磁场骨生物学效应的在体实验研究 磁场用于骨的修复治疗已有多年历史,实验证明磁场可通过促进成骨细胞、骨髓基质细胞增殖和分化,影响细胞膜、细胞形态、细胞外基质、基因表达等作用来诱导骨再生,加速骨愈合。近年来在动物模型实验中,复合磁场以及与材料等共同应用的方法逐渐成为研究热点。 2.2.1 骨折愈合/骨缺损 Hu等[3]建立了兔髌骨-髌韧带损伤模型,使用正弦交变磁场和静磁场组合磁场曝磁处理,显示新形成骨面积,骨密度和骨小梁厚度都显著高于对照组。除了单独在体外施加磁场,有研究者将磁性材料处理后植入骨缺损处,对骨愈合也具有促进作用。Kim等[4]将钕磁铁植入新西兰白兔的胫骨,增加了骨小梁数量和厚度,并使得细胞外基质相关基因和生长因子相关基因表达上调。在兔股骨缺损处埋置钕铁硼磁铁与胶原/羟基磷灰石混合支架,其促骨再生的作用优于非磁性支架,研究者认为静磁场与磁性支架的动态力学作用可能是促进骨再生的潜在因素[5]。不过单独应用磁性纳米颗粒材料可能阻碍骨再生,其对软骨形成并无作用,且在体内的影响和最终命运还不清楚,材料的生物相容性需进一步研究[6]。 多数研究证明,将外加磁场与体内植入磁性材料共同应用一般具有良好的效果。Yun等[7]建立小鼠颅骨缺陷模型,在缺损处植入磁性纳米颗粒-聚己内酯复合支架,同时施加静磁场,发现与磁性纳米颗粒联合应用可加强静磁场促骨形成的效应,其新生骨数量是仅植入聚己内酯支架组的2.7倍。聚乳酸-羟基乙酸/含铁的羟基磷灰石颗粒也可产生类似作用,研究者认为铁纳米粒子可在静磁场下形成微小磁场,具有超顺磁性,更有利于成骨细胞的黏附与增殖。除了静磁场,外加脉冲电磁场与磁柱共同应用也具有更好的促进骨修复能力。正弦低频电磁场促进骨缺损修复效果也优于只植入支架组。Calcagnotto等[8]的研究则显示自体骨移植和磁场作用对骨组织的修复有更好的效果。 此外,辅以药物治疗对磁场的促进骨愈合具有干预作用。周围等[9]建立大鼠胫骨骨折模型,施加中药治疗和电磁场(2 mT,2 Hz)治疗,发现大鼠血清骨形态发生蛋白2含量均显著高于对照组,应用中药的效果又优于磁场干预。Atalay等[10]发现磷酸二酯酶抑制剂己酮可可碱和电磁场[(1.5±0.2) mT,50 Hz]均可促进大鼠骨折愈合,但二者联合应用时则对骨折愈合并无积极作用。 2.2.2 骨质疏松/增加骨量 骨质疏松主要是由于骨吸收和骨形成发生了失衡,磁场可有效改善骨质疏松,增加骨量,其中最常用的磁场是脉冲电磁场。Li等[11]建立大鼠后肢失用性骨质疏松模型,分别予阿仑膦酸钠和脉冲电磁场(10 Hz,3.82 mT)处理,结果显示脉冲电磁场比阿仑膦酸钠能更好地改善骨的机械性能。Zhou等[12]也发现脉冲电磁场可增强伊班膦酸盐治疗骨质疏松的效果。脉冲电磁场还可阻止切除卵巢引起的腰椎骨和股骨小梁骨质量减少和骨小梁微结构恶化,不过Lei等[13]发现3 000-5 000 Hz的脉冲电磁场既能抑制骨形成也可抑制骨吸收,对骨质疏松无作用;Aksoy等[14]的实验采用的0.8 mT,7.3 Hz脉冲电磁场对骨质疏松大鼠骨量的恢复也无显著影响。此外,在骨质疏松早期应用脉冲电磁场治疗的效果要好于12周后才开始治疗[15]。 静磁场对骨质疏松也具有改善作用。Zhang等[16]用静磁场处理1型糖尿病大鼠,发现大鼠骨小梁和皮质骨的结构恶化和强度的降低被抑制,骨形态发生蛋白2和Runx2基因表达上调,血清骨钙素和骨小梁数目增加,这提示静磁场可能成为1型糖尿病相关骨质减少/骨质疏松症的有用的物理治疗手段。研究还发现,饮用/进食经静磁场处理的水也可显著提升大鼠骨矿物质密度,骨矿含量和骨抵抗力。 交变电磁场对增加骨密度、提升骨量也具有一定作用。15 Hz,1 mT的正弦交变电磁场可显著抑制甲亢引起的骨质流失和微结构恶化[17]。周建等[18]对SD大鼠给予正弦交变电磁场照射,发现50 Hz,0.1 mT的磁场可显著增加大鼠全身骨密度,改善骨小梁结构和数目, 90 min为最佳作用时间。 关于旋转磁场的成骨作用研究报道较少,Jing等[19]发现中等强度的旋转磁场中(0.38-0.60 T,7 Hz)对失用引起的大鼠股骨小梁和皮质骨微构造的恶化无明显影响,骨形成和骨吸收标志物表达均无明显变化,表明旋转磁场目前可能不是用于抑制废用性骨质减少/骨质疏松症的最佳方式。 除了用于治疗骨折不愈合、骨质疏松等常见疾病,磁场还可抑制炎症,促进软组织愈合,对骨关节炎症、肩袖撕裂、股骨头坏死和椎体融合等也具有改善作用,并吸引了越来越多的研究。另外,交变磁场诱导磁性阳离子脂质体产生的靶向高温可显著抑制小鼠骨肉瘤的增长及转移,这为相关疾病的临床治疗提供了新的思路。 2.3 磁场骨生物学效应的细胞层面研究 骨代谢是成骨和破骨作用的平衡过程,成骨细胞、破骨细胞、骨髓间充质干细胞等多种细胞与这一活动密切相关,此外,骨组织的重建与修复还需要软骨细胞、血管内皮细胞等周围组织细胞的支持。磁场对骨转换具有调节作用,探索应用磁场的最佳条件和效果具有重要的价值。 2.3.1 成骨细胞 许多研究表明,磁场可影响成骨细胞的增殖与分化等功能,而不同类型和参数的磁场可对成骨细胞产生不同的影响。Zhou等[20]发现50 Hz正弦交变电磁场可促进成骨细胞形成碱性磷酸酶、钙化结节等骨向分化标志,且1.8 mT和3.6 mT效果最为明显。脉冲电磁场也可提高成骨细胞的碱性磷酸酶活性,促进骨钙素等蛋白表达及矿化结节形成,并在4.1 mT达到峰值。Li等[21]将MC3T3-E1细胞暴露于400、2 800个脉冲的脉冲电磁场(0.5 Hz)中,发现 2 800次脉冲对促进成骨有抑制作用,400次脉冲对其则无影响。 Yang等[22]发现不同强度的静磁场对成骨细胞的诱导作用不一,同时铁水平和转铁蛋白受体1 mRNA表达也受到影响。这提示成骨细胞分化可以通过改变静磁场的强度来调节,并且铁可能参与该过程。 2.3.2 破骨细胞 破骨细胞的分化和功能与成骨细胞密切相关,骨溶解常伴随着破骨细胞形成,对于成骨作用和骨修复具有不利影响。脉冲电磁场和静磁场均可影响破骨细胞的分化,脉冲电磁场能够抑制破骨细胞增殖,加速成熟的破骨细胞凋亡,其作用效果与电磁刺激的频率或者强度有关。 75 Hz,2.5 mT的脉冲电磁场可抑制外周血单核细胞生成破骨细胞,促进骨再生的作用优于富血小板血 浆[23]。8 Hz,3.8 mT 的脉冲电磁场可显著抑制巨噬细胞集落刺激因子+核因子κB受体活化因子配体RANKL诱导破骨细胞生成,并降低核因子κB受体活化因子RANK mRNA表达的水平[24]。Zhang等[25]发现500 nT和0.2 T的静磁场可促进破骨细胞前体细胞高表达几乎所有的破骨细胞形成基因,使细胞向破骨方向分化和形成,而16 T则有抑制作用,这与其对成骨细胞的作用正好相反。 2.3.3 骨髓间充质干细胞 骨髓间充质干细胞可分化为成骨细胞、软骨细胞和脂肪细胞等多种细胞,是骨组织工程的种子细胞。磁场可以调节骨髓间充质干细胞的分化方向,增强成骨分化,抑制脂肪形成[26]。与成骨细胞类似,中等强度的静磁场(3,15,50 mT)可上调骨髓间充质干细胞中成骨向相关基因的表达,其中转录因子Osterix和Runx2上升水平最高,15 mT达到峰值[27]。 静磁场(0.25 T)和/或磁力介导的剪切力可显著上调人骨髓间充质干细胞软骨分化标志物Ⅱ型胶原纤维α1基因COL2A1和SOX9的基因表达,并促进硫酸化透明质酸和胶原的合成与分泌。磁场驱动的剪切力比静磁场作用效果更显著,二者联合应用的效果则更加突出[28]。脉冲电磁场(25/50 Hz,1.6 mT)对骨髓间充质干细胞 COL2和SOX9的基因表达无明显作用,转化生长因子- β1比单独的脉冲电磁场处理具有更大的影响,其与25 Hz 脉冲电磁场联合作用效果最强[29]。 骨组织的整合与修复常常伴随着软骨形成和血管再生,磁场可促进软骨细胞分化,诱导血管内皮细胞生发[7],进而加速骨组织的再生与修复。此外,静磁场对牙周膜细胞和成牙骨质细胞矿化也有促进作用[30]。磁场对骨肉瘤细胞作用的研究很少,新近报道表明脉冲电磁场可增强化疗药物对骨肉瘤细胞的杀伤作用,旋转磁场也能增强超顺磁性氧化铁颗粒诱导的骨肉瘤细胞凋亡,这为骨肿瘤的治疗提供了一条新的途径[31]。 总之,磁场可通过调节成骨细胞、破骨细胞和骨髓间充质干细胞等细胞的增殖与分化,发挥对骨代谢的影响。不同类型、参数的磁场对细胞可有不同影响,骨组织对低频脉冲电磁场可具有更强的敏感性,这可能与脉冲电磁场可引发更强的跨膜电位变化和Ca2+内流有关;多数研究证明静磁场的促成骨作用与其可诱导成骨细胞增殖、分化以及改变细胞骨架密切相关。四种类型的磁场都能影响多种骨细胞分泌骨生长因子,进而影响成骨,其对不同细胞发挥作用的详细参数和影响机制则还需进一步研究确定。 2.4 磁场骨生物学效应的机制研究 对磁场影响骨再生、骨重建的机制认识不多,目前发现以下几种信号通路可能参与这一过程。 2.4.1 经典Wnt信号通路 在骨的形成和平衡转换中,Wnt信号通路起着重要的调节作用,经典Wnt通路中Wnt分子通过与细胞膜上的frizzled受体和脂蛋白受体相关蛋白5/6共受体结合,使得胞内非磷酸化β-catenin蛋白增多,并进入细胞核内调节目的基因转录水平,进而促进成骨。研究显示,磁场可以通过激活Wnt/β-catenin信号通路阻止卵巢切除诱导的骨质流失和骨微结构和强度的恶化[32],15 Hz,2.4 mT和1.6 mT的脉冲电磁场均可显著上调去卵巢大鼠Wnt,LRP5和β-catenin mRNA水平,较高频率的电磁场也有类似作用[33-34]。Kim等[30]发现15 mT静磁场可上调人成骨细胞、牙周膜细胞和成牙骨质细胞Wnt1和Wnt3a的蛋白表达,并提升糖原合成酶激酶-3β的磷酸化水平和总β-catenin蛋白表达。Fathi等[35]发现电磁场与Zn2+共同作用可显著促进脂肪干细胞Wnt1,Wnt3a和β-catenin mRNA表达,静磁场与磁性颗粒的联用也可通过Wnt通路促进成骨[36],见图1。 2.4.2 RANK/RANKL/OPG信号通路 RANK/RANKL/ OPG信号通路对骨形成和骨吸收具有重要作用。RANK主要分布在破骨细胞表面,RANKL则由骨细胞和成骨细胞分泌,RANKL与RNK结合可激活破骨细胞内的信号转导途径,加速破骨细胞成熟和骨吸收等,OPG可与RANKL结合干扰这一过程。磁场可通过影响RANK、RANKL、OPG的表达促进骨形成[37]。Lei等[34]对切除卵巢的大鼠应用8周的脉冲电磁场治疗,可预防骨量减少并改善骨的结构和性能,RANKL和OPG mRNA表达显著提高,RANK mRNA表达降低,RANKL/OPG比值则未收到影响,显示磁场可能主要通过降低破骨细胞活动达到抑制骨吸收的作用。Calcagnotto等[13]使用的电磁场则提高了RANKL/OPG比值,不过对破骨相关基因的表达并无影响。"
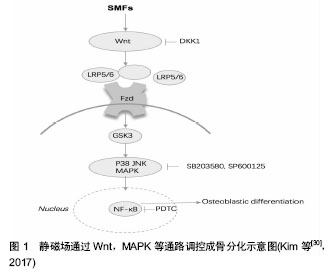
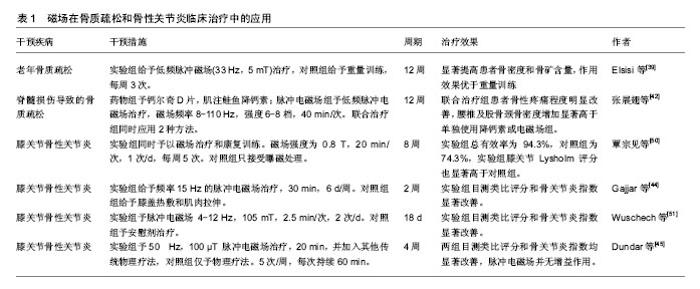
2.4.3 MAPK信号通路 MAPK通路在应力感应中扮演关键角色,MAPK通路被激活后可诱导成骨分化,对骨的代谢平衡起到调节作用。大量研究显示,磁场能够提高p38,c-Jun氨基末端激酶JNK和细胞外调节蛋白激酶ERK1/2的磷酸化水平,促进骨形成,不同条件的磁场激活不同的MAPK通路,加入MAPK抑制剂则会干扰这一作用[30,38]。Yun等[7]发现静磁场和磁性复合支架对促进P38,ERK1/2和JNK的活化具有协同作用。脉冲电磁场还可通过MAPK通路促进C2C12成肌细胞增殖,抑制软骨破 坏[29]。可见MAPK通路在电磁场诱导的成肌成骨作用中扮演不同角色,更详细的作用机制还有很大的研究空间。Wnt、MAPK及RANK/RANKL/OPG信号通路在骨代谢中的作用已有很多研究,磁场对骨平衡的影响与上述通路有密切联系。另外,还有一些重要的信号通路也参与了磁场介导的成骨作用,包括BMP-Smad信号通路、TGF-β通路以及RhoA/Rho应力感应通路。目前的研究表明磁场可通过调节这些通路上的标志物表达影响成骨与破骨,这为探明磁场的骨生物学效应提示了更多的研究方向。 2.5 临床应用最新进展 基于促进骨再生,改善骨代谢,增加骨量,抑制炎症,镇痛和促进软组织修复等作用,磁场在临床上已作为辅助治疗手段,用于多种骨科疾病的治疗,常见的有骨质疏松、骨性关节炎症等,并取得了一定的疗效,被证明是一种良好的无侵入性的物理治疗方法。近年来,磁场应用的方式逐渐扩展,具体条件参数和效果也获得了越来越多的临床验证,见表1。 2.5.1 骨质疏松 磁场对改善骨质疏松、增加骨量有促进作用。Elsisi等[39]对老年妇女进行低频脉冲磁场治疗,发现磁场可显著提高患者骨密度和骨矿含量,效果优于重量训练。低频脉冲磁场还可改善绝经后骨质疏松症患者骨代谢异常情况,并缓解疼痛[40]。Meszaros等[41]采用的静磁场则对骨质疏松患者并无积极作用,甚至低于假手术组,因此在治疗时应选用合适条件的磁场。 磁场与药物同时应用对骨质疏松有增益作用,张展翅等[42]将低频脉冲电磁场与鲑鱼降钙素联合用以治疗脊髓损伤导致的骨质疏松症,持续治疗12周后,联合治疗组患者骨性疼痛程度明显改善,腰椎及股骨颈骨密度增加显著高于单独使用降钙素或电磁场组,脉冲电磁场与钙尔奇D片、骨化三醇和阿仑膦酸钠联合应用也取得了类似效果[43]。 2.5.2 关节炎症 脉冲磁疗可以缓解炎症部位神经应激反应,从而减弱痛觉,并促进局部血液循环,加速炎症反应物的消除,将脉冲磁疗法与康复训练联合应用可显著提升骨关节炎临床治愈率,效果优于单一应用脉冲磁场。Gajjar等[44]对57例膝关节骨性关节炎患者应用脉冲电磁场治疗,骨关节炎指数和目测类比疼痛评分显著提升,日常活动能力明显提高。Dundar等[45]则发现脉冲电磁场并不能增强物理治疗对膝关节炎的效用,这可能与磁场作用时间较短、样本数目限制等有关。 2.5.3 骨折愈合 尹萍等[46]对70例骨折患者应用超短波电疗法与脉冲电磁疗法,可改善血液循环,减轻局部炎症,加速骨折愈合。Phillips等[47]通过大量临床数据证明组合磁场对治疗骨折愈合具有良好的有效性和安全性,2 370例患者中有75%达到骨折愈合。股骨骨折患者的愈合率为64.0%,腕骨/掌骨骨折患者的愈合率为89.7%,加权平均治愈时间为(4.9±1.0)个月。 2.5.4 牙齿种植、正畸 磁场在口腔领域有不少应用,Gujjalapudi等[48]尝试在种植牙后应用静磁场使骨整合加速,将圆形钕铁硼静磁体植入患者覆盖义齿中,结果发现磁性侧种植体稳定性明显大于非磁性侧,磁场与骨整合之间存在正相关关系。Jung等[49]证明脉冲电磁航可减少初始正畸牙齿移动引起的疼痛。 磁场刺激在骨质疏松、骨折、骨性炎症以及牙齿正畸等领域作为常辅助治疗手段,并取得了较好的临床效果,可有效地提升骨量,抑制炎症,减轻疼痛,缩短恢复时间,磁场与药物、运动等其他方法的综合使用可以产生促进作用。不过不同条件的磁场影响不一,且临床观察的时间有限,磁场对人体作用的远期效应还不清楚,因此应谨慎选择参数,并注意预防危害。"

| [1] Bassett CA, Pawluk RJ, Pilla AA. Acceleration of fracture repair by electromagnetic fields. a surgically noninvasive method. Ann N Y Acad Sci.1974;238:242-262.[2] Yang Y, Tao C, Zhao D, et al. Emf acts on rat bone marrow mesenchymal stem cells to promote differentiation to osteoblasts and to inhibit differentiation to adipocytes. Bioelectromagnetics. 2010;31(4): 277-285.[3] Hu J, Zhang T, Xu D, et al. Combined magnetic fields accelerate bone-tendon junction injury healing through osteogenesis. Scand J Med Sci Sports.2015;25(3):398-405.[4] Kim EC, Leesungbok R, Lee SW, et al. Effects of static magnetic fields on bone regeneration of implants in the rabbit: micro-ct, histologic, microarray, and real-time pcr analyses. Clin Oral Implants Res. 2017;28(4):396-405.[5] Russo A, Bianchi M, Sartori M, et al. Magnetic forces and magnetized biomaterials provide dynamic flux information during bone regeneration.J Mater Sci Mater Med.2016;27(3):51.[6] Panseri S, Russo A, Giavaresi G, et al. Innovative Magnetic Scaffolds for Orthopedic Tissue Engineering. J Biomed Mater Res A.2012;100(9):2278-2286.[7] Yun HM, Ahn SJ, Park KR, et al. Magnetic nanocomposite scaffolds combined with static magnetic field in the stimulation of osteoblastic differentiation and bone formation. Biomaterials.2016; 85:88-98.[8] Calcagnotto T, Schwengber M M B, De Antoni C C, et al. Magnetic field effects on bone repair after calcium phosphate cement implants: histometric and biochemistry evaluation. Ann Maxillofac Surg.2017;7(1): 18-24.[9] 周围,李月玮,蒋秋冬,等.续骨丹外敷疗法对大鼠胫骨缺损模型血清bmp-2的影响[J]. 基因组学与应用生物学, 2017,36(4):1336-1342.[10] Atalay Y, Gunes N, Guner MD, et al. Pentoxifylline and electromagnetic field improved bone fracture healing in rats. Drug Des Devel Ther.2015;9:5195-5201.[11] Li B, Bi J, Li W, et al. Effects of pulsed electromagnetic fields on histomorphometry and osteocalcin in disuse osteoporosis rats. Technol Health Care.2017;25(S1):13-20.[12] Zhou J, Liao Y, Xie H, et al. Effects of combined treatment with ibandronate and pulsed electromagnetic field on ovariectomy-induced osteoporosis in rats. Bioelectromagnetics. 2017;38(1):31-40.[13] Lei T, Li F, Liang Z, et al. Effects of four kinds of electromagnetic fields (emf) with different frequency spectrum bands on ovariectomized osteoporosis in mice. Sci Rep. 2017; 7(1):553.[14] Aksoy MÇ, Topal O, Özkavak HV, et al. Effects of pulsed electromagnetic field on mineral density, biomechanical properties, and metabolism of bone tissue in heparin-induced osteoporosis in male rats. Biomed Res Int.2017;28(6):2724-2729.[15] Zhou J, Liao Y, Zeng Y, et al. Effect of intervention initiation timing of pulsed electromagnetic field on ovariectomy-induced osteoporosis in rats. Bioelectromagnetics. 2017;38(6):456-465.[16] Zhang H, Gan L, Zhu X, et al. Moderate-intensity 4mt static magnetic fields prevent bone architectural deterioration and strength reduction by stimulating bone formation in streptozotocin-treated diabetic rats. Bone. 2018;107: 36-44.[17] Liu C, Zhang Y, Fu T, et al. Effects of electromagnetic fields on bone loss in hyperthyroidism rat model. Bioelectromagnetics. 2017,38(2):137-150.[18] 周建,马小妮,陈克明,等.电磁场不同处理时间对人脐带间充质干细胞增殖与分化的影响[J].解放军医药杂志, 2015,3:11-15+19.[19] Jing D, Cai J, Wu Y, et al. Moderate-intensity rotating magnetic fields do not affect bone quality and bone remodeling in hindlimb suspended rats. Plos One. 2014;9(7): e102956.[20] Zhou J, Ming LG, Ge BF, et al. Effects of 50 Hz sinusoidal electromagnetic fields of different intensities on proliferation, differentiation and mineralization potentials of rat osteoblasts. Bone.2011;49(4):753-761.[21] Li K, Ma S, Li Y, et al. Effects of pemf exposure at different pulses on osteogenesis of mc3t3-e1 cells. Arch Oral Biol. 2014;59(9): 921-927.[22] Yang J, Zhang J, Ding C, et al. Regulation of osteoblast differentiation and iron content in mc3t3-e1 cells by static magnetic field with different intensities. Biol Trace Elem Res. 2017. doi: 10.1007/s12011-017-1161-5. [23] Tschon M, Veronesi F, Contartese D, et al. Effects of pulsed electromagnetic fields and platelet rich plasma in preventing osteoclastogenesis in an in vitro model of osteolysis. J Cell Physiol. 2018;233(3): 2645-2656.[24] He J, Zhang Y, Chen J, et al. Effects of pulsed electromagnetic fields on the expression of nfatc1 and caii in mouse osteoclast-like cells. Aging Clin Exp Res. 2015;27(1): 13-19.[25] Zhang J, Meng X, Ding C, et al. Regulation of osteoclast differentiation by static magnetic fields. Electromagn Biol Med. 2017;36(1): 8-19.[26] Du L, Fan H, Miao H, et al. Extremely low frequency magnetic fields inhibit adipogenesis of human mesenchymal stem cells. Bioelectromagnetics.2014;35(7): 519-530.[27] Kim EC, Leesungbok R, Lee SW, et al. Effects of moderate intensity static magnetic fields on human bone marrow-derived mesenchymal stem cells. Bioelectromagnetics. 2015;36(4): 267-276.[28] Son B, Kim H D, Kim M, et al. Physical stimuli-induced chondrogenic differentiation of mesenchymal stem cells using magnetic nanoparticles. Adv Healthc Mater. 2015;4(9):1339-1347.[29] Kavand H, Haghighipour N, Zeynali B, et al. Extremely low frequency electromagnetic field in mesenchymal stem cells gene regulation: chondrogenic markers evaluation. Artif Organs. 2016; 40(10): 929-937.[30] Kim EC, Park J, Kwon IK, et al. Static magnetic fields promote osteoblastic/cementoblastic differentiation in osteoblasts, cementoblasts, and periodontal ligament cells. J Periodontal Implant Sci. 2017;47(5): 273-291.[31] Du S, Li J, Du C, et al. Overendocytosis of superparamagnetic iron oxide particles increases apoptosis and triggers autophagic cell death in human osteosarcoma cell under a spinning magnetic field. Oncotarget.2017;8(6): 9410-9424.[32] Zhou J, He H, Yang L, et al. Effects of pulsed electromagnetic fields on bone mass and wnt/beta-catenin signaling pathway in ovariectomized rats. Arch Med Res. 2012;43(4): 274-282.[33] Jing D, Li F, Jiang M, et al. Pulsed electromagnetic fields improve bone microstructure and strength in ovariectomized rats through a wnt/lrp5/beta-catenin signaling-associated mechanism. PLoS One. 2013;8(11): e79377.[34] Lei T, Liang Z, Li F, et al. Pulsed electromagnetic fields (pemf) attenuate changes in vertebral bone mass, architecture and strength in ovariectomized mice. Bone. 2018;108:10-19.[35] Fathi E, Farahzadi R. Enhancement of osteogenic differentiation of rat adipose tissue-derived mesenchymal stem cells by zinc sulphate under electromagnetic field via the pka, erk1/2 and wnt/beta-catenin signaling pathways. PLoS One.2017;12(3): e0173877.[36] Rotherham M, Henstock J R, Qutachi O, et al. Remote regulation of magnetic particle targeted wnt signaling for bone tissue engineering. Nanomedicine. 2018;14(1):173-184.[37] Zhou J, Chen S, Guo H, et al. Pulsed electromagnetic field stimulates osteoprotegerin and reduces rankl expression in ovariectomized rats. Rheumatol Int.2013;33(5): 1135-1141.[38] Yong Y, Ming Z D, Feng L, et al. Electromagnetic fields promote osteogenesis of rat mesenchymal stem cells through the pka and erk1/2 pathways. J Tissue Eng Regen Med. 2016;10(10): e537-e545.[39] Elsisi H F, Mousa G S, Mt E L. Electromagnetic field versus circuit weight training on bone mineral density in elderly women. Clin Interv Aging. 2015;10: 539-547.[40] 杜迅,郝建凤.低频脉冲磁场对绝经后骨质疏松症患者骨代谢标志物水平的影响[J].中国临床研究.2016;9: 1216-1218.[41] Meszaros S, Tabak AG, Horvath C, et al. Influence of local exposure to static magnetic field on pain perception and bone turnover of osteoporotic patients with vertebral deformity - a randomized controlled trial. Int J Radiat Biol.2013;89(10): 877-885.[42] 张展翅,李晓强,马隽,等.鲑鱼降钙素结合脉冲电磁场治疗脊髓损伤致骨质疏松症的临床效果[J].中国骨质疏松杂志, 2017(09): 1191-1194+1260.[43] 王鑫,白倩,王斌,等.运动、药物联合物理因子治疗对脊髓损伤后骨质疏松症疗效的观察[J].中国疗养医学, 2017,11: 1121-1124.[44] Gajjar B, Sheth M, Sharma S, et al. Effect of pulsed electromagnetic energy therapy on pain and function in subjects with knee osteoarthritis. Int J Med Sci Public Health. 2014;3(6): 1.[45] Dundar U, Asik G, Ulasli AM, et al. Assessment of pulsed electromagnetic field therapy with serum ykl-40 and ultrasonography in patients with knee osteoarthritis. Int J Rheum Dis.2016;19(3):287-293.[46] 尹萍.超短波电疗法联合脉冲磁疗治疗骨折效果研究[J].中国疗养医学,2018,1: 41-42.[47] Phillips M, Baumhauer J, Sprague S, et al. Use of combined magnetic field treatment for fracture nonunion. J Long Term Eff Med Implants.2016;26(3): 277-284.[48] Gujjalapudi M, Anam C, Mamidi P, et al. Effect of magnetic field on bone healing around endosseous implants -an in-vivo study. J Clin Diagn Res.2016;10(10): Zf01-zf04.[49] Jung JG, Park JH, Kim SC, et al. Effectiveness of pulsed electromagnetic field for pain caused by placement of initial orthodontic wire in female orthodontic patients: a preliminary single-blind randomized clinical trial. Am J Orthod Dentofacial Orthop.2017;152(5): 582-591.[50] 覃宗见.康复训练联合脉冲磁疗对膝关节骨性关节炎患者膝关节功能恢复的影响[J].医药前沿, 2017,7(14):386-387.[51] Wuschech H, Von Hehn U, Mikus E, et al. Effects of pemf on patients with osteoarthritis: results of a prospective, placebo-controlled, double-blind study. Bioelectromagnetics. 2015;36(8):576-585. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Chen Junyi, Wang Ning, Peng Chengfei, Zhu Lunjing, Duan Jiangtao, Wang Ye, Bei Chaoyong. Decalcified bone matrix and lentivirus-mediated silencing of P75 neurotrophin receptor transfected bone marrow mesenchymal stem cells to construct tissue-engineered bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 510-515. |
| [5] | Ma Zhijie, Li Jingyu, Cao Fang, Liu Rong, Zhao Dewei. Influencing factors and biological property of novel biomedical materials: porous silicon carbide coated with bioactive tantalum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 558-563. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [8] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [9] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [10] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [11] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [12] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [13] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [14] | Yang Junhui, Luo Jinli, Yuan Xiaoping. Effects of human growth hormone on proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3956-3961. |
| [15] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||