Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (26): 4229-4234.doi: 10.3969/j.issn.2095-4344.0945
Previous Articles Next Articles
Preparation and research progress of long-circulating and pH-sensitive liposomes containing cisplatin
Shi Wei1, Lu Ying2, Gong Rui2
- 1Department of Hepatobiliary and Pancreatic Surgery, 2Department of Pharmacy, Second Hospital of Tianjin Medical University, Tianjin 300211, China
-
Received:2018-07-10 -
About author:Shi Wei, Master, Attending physician, Department of Hepatobiliary and Pancreatic Surgery, Second Hospital of Tianjin Medical University, Tianjin 300211, China -
Supported by:the Scientific Foundation of Tianjin Municipal Health and Family Planning Committee, No. 2014KZ109
CLC Number:
Cite this article
Shi Wei, Lu Ying, Gong Rui. Preparation and research progress of long-circulating and pH-sensitive liposomes containing cisplatin[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(26): 4229-4234.
share this article

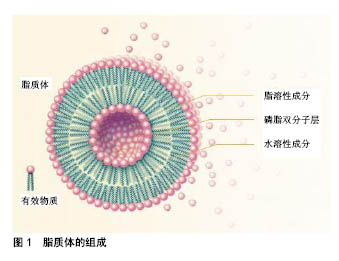
2.1 脂质体制备方案 脂质体制剂可采用多种方法制备,包括薄膜水化法、逆向蒸发法、乙醇注射法、冻融和冻干再水化法等。脂质体的许多特性,如大小和封装率,很大程度上取决于所采用的制备方法。优秀的制备方法应具备截留效率高、粒径分布窄的特性,并长期稳定[13]。逆向蒸发法以水相替代以往的水-混相溶剂,形成具有亲水率高的脂质体和脂相溶性物质。然而,在脂质体制制备中使用的有机溶剂必须被去除,因为它们可能对人体健康产生危害,并影响产品的稳定性。与以往常用的有机试剂相比,较高沸点的有机溶剂二乙醚,是一种更为合适的替代方法[14]。这些有机溶剂使用时需在防爆区(由于空气中含有溶剂蒸汽)中应用,且有机溶剂必须与溶液中的水相混溶。最后,需要考虑的另一点是溶剂的毒性,在可注射产品中,它们的毒性必须小于生物可接受的量[15]。脂质体的组成见图1。"
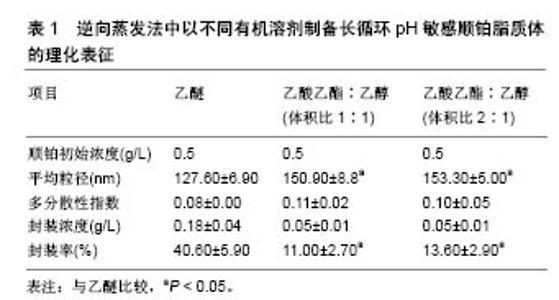
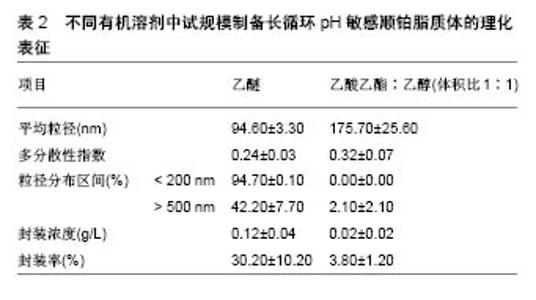
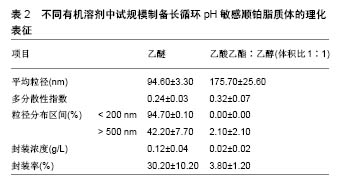
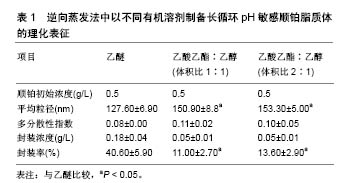
根据欧洲药品管理局(EMA,2 011)规定,溶剂被分为3类,第1类,避免使用溶剂;第2类,限制使用溶剂;第3类,低毒性的溶剂。乙醇(第3类)、乙酸乙酯(第3类)和氯仿(第2类)药剂制品的浓度限制为60 ppm,可用于各类药品的制备。实验室制备方法常用的有机溶剂包括乙酸乙酯、乙酸乙酯/乙醇混合物、异丙醚/氯仿[16]。在逆向蒸发法中,脂质体的中间体被冷冻干燥然后可控制的进行再水化。冷冻和干燥过程中会产生不同程度的压力,冷冻过程中压力变化最为剧烈,冷冻保护剂则可用来防止这种压力的损伤[17]。与冻干法相比,冻融过程短而快,可作为预实验试筛选冷冻干燥过程中冷冻保护剂的种类和浓度。此外,已证明冻结速率影响冻干法产物的物理稳定性[18]。 Desai等[19]采用逆向蒸发法制备脂质体,所用材料有顺铂、二硬脂酰磷脂酰乙醇胺聚乙二醇2000、二油酰磷脂酰乙醇胺、胆固醇琥珀酸单酯、氯化钠、葡萄糖、蔗糖、羟丙基β-环糊精,均为分析纯。首先用逆向蒸发法制备长循环pH敏感脂质体(空脂质体),用氯仿溶剂将二油酰磷脂酰乙醇胺,胆固醇琥珀酸单酯和二硬脂酰磷脂酰乙醇胺聚乙二醇2000按5.7∶3.8∶0.5的摩尔浓度混合,总的脂质浓度为40 mmol/L,将混合液置入圆底烧瓶,在低压下进行蒸发,获得一层薄的脂质膜。将这种脂质膜溶解在不同的有机溶剂(二乙醚或乙酸乙酯:乙醇混合物体积比为2∶1或1∶1)中,然后添加到生理盐水以制备pH敏感脂质体,或将顺铂(0.25,0.5,1.0,2.0 g/L)溶解于生理盐水以制备长循环pH敏感顺铂脂质体。水溶液和有机相的比例为1∶3(二乙醚为有机相)或1∶4(乙酸乙酯乙醇混合物为有机相)。所产生的混合物经快速涡旋搅拌,以产生水/油型乳剂。然后,有机溶剂挥发,形成脂质体。pH敏感脂质体和长循环pH敏感顺铂脂质体分散体分别通过0.4,0.2,0.1 mm聚碳酸酯膜过滤(各5次);150 000×g离心力,10 ℃超速离心 90 min,以消除未被结合的游离顺铂;去除上清液后,将长循环pH敏感顺铂脂质体颗粒加入等量的生理盐水中保存。 2.2 影响脂质体包封率及粒径的因素 2.2.1 顺铂浓度对脂质体包封率及粒径的影响 长循环pH敏感顺铂脂质体的给药方式是通过静脉注射,因此,它们的粒径大小和负载率决定了顺铂在疾病部位的有效治疗浓度高低。用乙醚作为溶剂,用不同初始浓度顺铂制备的所有长循环pH敏感顺铂脂质体脂质体粒径均为130 nm,且具有高度的单分散性[20]。通过研究证实,0.5 g/L初始质量浓度的顺铂,在长循环pH敏感顺铂脂质体制备中即可满足最大装载量,且不会有较多的游离顺铂被回收,是最佳制备质量浓度。而低于此质量浓度会导致装载量下降,降低疗效。 2.2.2 有机溶剂对脂质体包封率及粒径的影响 许多研究采用不同的有机溶剂制备脂质体,以提高产量。乙醚、乙酸乙酯、体积比1∶1的异丙醚/氯仿混合物、体积比1∶1的乙酸乙酯/乙醇混合物和体积比2∶1的乙酸乙酯/乙醇混合物,在逆向蒸发法方法的第一步被用来溶解脂质体,以制备长循环pH敏感顺铂脂质体。不同有机溶剂在溶解脂质膜和形成脂质体的能力上也各有差别。所有上述溶剂都能溶解脂质,逆向蒸发法的第一步主要目的是形成一种黏性凝胶,它负责脂质体的自发形成。这些脂质体的特性取决于乳液中脂质和水的分散。研究表明在蒸发过程中,体积比为1∶1的乙酸乙酯和异丙醚/氯仿混合物有机相会形成聚合物,而体积比为2∶1、 1∶1的乙酸乙酯/乙醇则可避免此类现象。因此根据它们的生物相容性特点,逆向蒸发法制备长循环pH敏感顺铂脂质体常选择这两种溶剂。与用乙醚制备的长循环pH敏感顺铂脂质体相比,用体积比为1∶1、2∶1乙酸乙酯/乙醇制备的长循环pH敏感顺铂脂质体粒径更大,而所有脂质体制剂都表现出类似的单分散性,但在0.50 g/L顺铂下乙醚制备的脂质体较前者封装率明显升高,且囊泡尺寸较小。原因是由于界面现象乙醚可拥有更高的表面区域;另一种原因是乙酸乙酯/乙醇混合物介电常数大于乙醚,有利于顺铂溶解,因而在顺铂封装中产生分隔,只有较低浓度的顺铂可被封装在脂质体水室中。 2.2.3 冻结速率及冷冻保护剂对脂质体粒径的影响 在冷冻干燥过程中,碳水化合物对脂质体的稳定作用源于脂质体与介质的结合,以及脂质体和脂质之间在赋形剂与脂类极性头之间氢键的形成,当双层稳定水被升华除去时,可形成稳定的膜。这些赋形剂可在冷冻时玻璃化,形成一个在干燥后分子流动性较低的囊泡周围保护性非晶型矩阵。这种玻璃结构避免了融合过程的发生,并防止了冰的形成,而冰的形成会导致囊泡大小的增加。冻结过程中低温保护剂对稳定机制的另一种解释是粒子隔离假说。冻融研究的原理是:在冷冻干燥过程中,如果赋形剂不能保护脂质体,它就不可能是一种有效的低温保护剂。另外,过程参数如冻结速率,直接影响冻干配方的稳定性。因而冻融法应在玻璃转换温度(Tg)以下的低温进行,以保证冻干脂质体形成及再分散过程没有脂质体聚集且制备更加容易。研究表明,只有缓慢冷却才能防止脂质体因冰晶生长而破裂和融合过程[21]。快速冷却后,在没有冷冻保护剂的冷冻样品中,可清楚地观察到大量团聚体且囊泡分布不均;然而在冷冻保护剂存在的情况下,与未冷冻样品相比,在快速冷却后可观察到粒径显著变小。值得注意的是,在快速或缓慢冷却后,使用冷冻保护剂pH敏感脂质体的平均直径是相似的。因此冷冻保护剂在脂质体制备中起到关键作用。研究证实,聚乙二醇2000脂类聚合物链可引起的空间斥力,所以聚乙二醇外壳可有效预防冷冻时囊泡的聚集[22]。采用海藻糖作为低温保护剂,可观察到在快速或慢速冷冻时,脂质体颗粒的形态及双层脂膜结构均无改变。与此相反,快速或慢速冷冻时,采用羟丙基-β-环糊精作为冷冻保护剂,可使得载体颗粒变得明显细长,说明在脂质双层和羟丙基-β-环糊精间存在较强的界面相互作用。采用葡萄糖作为低温保护剂时,可观察到在载体颗粒表面主要形成低聚糖薄膜。与双糖相比,低聚糖对脂质双层具有更大的压力,并诱导形态学改变。 2.3 中试规模逆向蒸发法制备脂质体 首先用如上所述的逆向蒸发法制备长循环pH敏感顺铂脂质体。以乙醚或体积比为1∶1的乙酸乙酯/乙醇作为有机相,顺铂质量浓度为0.5 g/L。Charcosset等[23]实验采用单级的高压均质器再循环模式来进行脂质体颗粒直径的校准,均化器压力调整为50 MPa,样品处理最小体积为110 mL,在室温下进行均质化,每一个周期都相当于样品总体积容积通过同质化室,一共循环9个周期,未被结合的游离顺铂,用密理博(Millipore)Pellicon XL切向流超滤膜系统去除,并连接到切向流过滤系统。 中试规模逆向蒸发法制备脂质体常用的有机相为体积比1∶1的乙酸乙酯/乙醇。尽管在实验室使用此法制备脂质体时,顺铂的封装率很低,但在长循环pH敏感顺铂脂质体试验生产中,通过改良实验条件后该溶剂的效率大幅增加。条件优化后,长循环pH敏感顺铂脂质体的生产规模可增加25倍。通过高压均质化方法对载体直径进行标定可发现,与体积比为1∶1的乙酸乙酯/乙醇有机溶剂相比,使用乙醚时的载体顺铂封装率增加,载体粒径变小,且两者制备的脂质体粒径差别不显著[24]。由于乙醚作为有机溶剂制备的脂质体属于均匀分散系,95%的载体粒径均< 200 nm。由于均匀分散系的长循环pH敏感顺铂脂质体更加适合静脉注射,因此近期认为乙醚作为有机相是生产脂质体的可行过程。 2.4 长循环pH敏感顺铂脂质体的表征 脂质体表征的判定是其包封率和平均泡囊直径。采用关联能谱法测定平均泡囊直径和多分散度指数,用石墨炉原子吸收光谱法定量分析铂类化合物,以计算脂质体的包封率。用透射电镜可对脂质体形态进行研究。脂质体被放置在碳涂层的铜网上,并以2%磷质酸溶液染色,染色样品可采用PHilips CM 120电子显微镜观测。一些实验观察到不同以有机溶剂采用逆向蒸发法制备的脂质体表征差异,见表1[25-26]。不同有机溶剂中试规模制备长循环pH敏感顺铂脂质体的理化表征,见表2。 2.5 长循环pH敏感顺铂脂质体在小鼠胰腺癌模型中的疗效和毒性研究 胰腺腺癌的特点是死亡率非常高。胰腺肿瘤治疗最相关的不利预后因素之一是其化学抗性,肿瘤组织的特征是致密的基质和较差的血管化,减少了抗肿瘤药物的输入[27]。除顺铂外,很少有化疗药物能有效治疗胰腺肿瘤,而纳米载体长循环pH敏感顺铂脂质体,在以往化疗方案难治性晚期胰腺肿瘤治疗中显示了令人鼓舞的结果[28-32]。长循环pH敏感顺铂脂质体可改变顺铂的药代动力学,减少毒性,提高药物的生物利用度,在晚期胰腺肿瘤治疗方面展示了极大的应用前景[33-36]。 用上述方法制备的长循环pH敏感顺铂脂质体纳米给药系统,包含二油酰磷脂酰乙醇胺、胆固醇琥珀酸单酯、二乙基磷脂酰乙醇胺聚乙二醇2000[37]。在酸性介质如肿瘤组织中,胆固醇琥珀酸单酯被质子化,使脂质体的结构不再稳定,释放顺铂。因此,药物可被明确地释放到肿瘤部位,其疗效会增加而毒性降低。实验表明在静脉注射长循环pH敏感顺铂脂质体后,顺铂在肿瘤组织中的浓度远高于游离顺铂[38-41]。此外与单纯顺铂相比,注射长循环pH敏感顺铂脂质体肾血分配系数较低,这表明肾的顺铂保留较少,因此肾毒性较低。其他研究报道显示,在健康和埃利希肿瘤小鼠体内静脉注射长循环pH敏感顺铂脂质体后,动物体质量减少和生存率明显提 高[42-46]。体内长循环pH敏感顺铂脂质体对胰腺肿瘤的疗效和毒性尚不清楚。因此,Gunji等[47]比较了长循环pH敏感顺铂脂质体和单纯顺铂对胰腺肿瘤小鼠模型的抗肿瘤作用和体内毒性。在小鼠胰腺癌模型中,小鼠被接种5×106的Capan-1胰腺癌细胞系悬液,在肿瘤达到 100 mm3大小时,给予不同剂量的顺铂、长循环pH敏感顺铂脂质体及空白pH敏感脂质体。肿瘤治疗效果的评价主要依赖于肿瘤大小变化、肿瘤病理切片坏死程度及肿瘤组织对99Tcm-HYNIC-[beta]Ala-bombesin的摄取进行评估。如用99Tcm标记的蛙皮素衍生物99Tcm-HYNIC- [beta]Ala-bombesin,可作为放射性示踪剂来评估治疗前后肿瘤的摄取[48-50]。而药物毒性评价则主要依赖于小鼠体质量的减轻程度及小鼠肝脏、脾脏、肾脏、小肠部位组织切片的病理变化。 实验表明,顺铂治疗的胰腺肿瘤小鼠显示出低于长循环pH敏感顺铂脂质体的放射性示踪剂肿瘤摄取,与功能肿瘤组织的减少量一致[51],且这种效果在长循环pH敏感顺铂脂质体16 mg/kg剂量下尤为明显。此外,组织病理学和组织形态学分析显示,肿瘤在顺铂和长循环pH敏感顺铂脂质体治疗后会出现坏死区,长循环pH敏感顺铂脂质体给药组坏死区域未明显大于顺铂组[52-53]。 "
| [1] Zeb A,Qureshi OS,Kim HS,et al. High payload itraconazole-incorporated lipid nanoparticles with modulated release property for oral and parenteral administration. J Pharm Pharmacol. 2017;69(8):955-966. [2] Qureshi OS,Kim HS, Zeb A,et al. Sustained release docetaxel- incorporated lipid nanoparticles with improved pHarmacokinetics for oral and parenteral administration. J Microencapsul. 2017;34(3): 250-261. [3] Battaglia L,Gallarate M,Peira E,et al. Solid lipid nanoparticles for potential doxorubicin delivery in glioblastoma treatment: preliminary in vitro studies. J Pharm Sci. 2014;103(7):2157-2165. [4] Kakkar D,Dumoga S,Kumar R,et al. PEGylated solid lipid nanoparticles: design, methotrexate loading and biological evaluation in animal models. Med Chem Commun. 2015;6(8):1452-1463. [5] Patel MN,Lakkadwala S,Majrad MS,et al. Characterization and evaluation of 5-fluorouracil-loaded solid lipid nanoparticles prepared via a temperature-modulated solidification technique. AAPS Pharm Sci Tech. 2014;15(6):1498-1508. [6] Lee WH,Loo CY,Traini D,et al. Nano-and micro-based inhaled drug delivery systems for targeting alveolar macropHages. Expert Opin Drug Deliv. 2015;12(6):1009-1026. [7] Giuberti CDS,Boratto FA,Degobert G,et al. Investigation of alternative organic solvents and methods for the preparation of long-circulating and pH-sensitive liposomes containing cisplatin. J Liposome Res. 2013;23(3):220-227. [8] Leite EA,Souza CM,Carvalho-Júnior AD,et al.Encapsulation of cisplatin in long-circulating and pH-sensitive liposomes improves its antitumor effect and reduces acute toxicity.Int J Nanomedicine. 2012; 7:5259-5269.[9] Giuberti CS,Reis ECO,Rocha TGR,et al. Study of the pilot production process of long-circulating and pH-sensitive liposomes containing cisplatin. J Liposome Res. 2011;21:60-69. [10] Biswas S, Kumari P, Lakhani PM, Ghosh B. Recent advances in polymeric micelles for anti-cancer drug delivery. Eur J PHarm Sci. 2016;83:184-202. [11] Gothwal A,Khan I,Gupta U.Polymeric micelles: recent advancements in the delivery of anticancer drugs. Pharm Res. 2016;33(1):18-39. [12] Ng CM,Loh HS,Muthoosamy K,et al. Conjugation of insulin onto the sidewalls of single-walled carbon nanotubes through functionalization and diimide-activated amidation. Int J Nanomedicine. 2016;11: 1607-1614. [13] Johnsson M, Edwards K. Liposomes, disks and spHerical micelles: aggregate structure in mixtures of gel pHase pHospHatidylcholines and poly(ethyleneglycol)-pHospHolipids. BiopHys J. 2003;85: 3839-3847. [14] Kasper JC, Friess W. The freezing step in lyopHilization: pHysico-chemical fundamentals, freezing methods and consequences on process performance and quality attributes of biopHarmaceuticals. Eur J Pharm Biopharm. 2011;78:248-263. [15] Leite EA,Lana AMQ, Junior ADC, et al.Acute toxicity study of cisplatin loaded long-circulating and pH-sensitive liposomes administered in mice. J Biomed Nanotechnol. 2012;8:229-239. [16] Prabhakar N, Zhang J,Desai D,et al. Stimuli-responsive hybrid nanocarriers developed by controllable integration of hyperbranched PEI with mesoporous silica nanoparticles for sustained intracellular siRNA delivery. Int J Nanomedicine. 2016;11:6591-6608. [17] Wagner A,Vorauer-Uhl K. Liposome technology for industrial purposes. J Drug Deliv. 2011;2011: 591325. [18] Wessman P, Edwards K, Mahlin D.Structural effects caused by spray- and freeze-drying of liposomes and bilayes disks. J PHarm Sci. 2010; 99:2032-2048. [19] Desai D, Zhang J, Sandholm J, et al. Lipid bilayer-gated mesoporous silica nanocarriers for tumor-targeted delivery of zoledronic acid in vivo. Mol Pharm. 2017;14(9):3218-3227. [20] Han N,Zhao Q,Wan L,et al. Hybrid lipid-capped mesoporous silica for stimuli-responsive drug release and overcoming multidrug resistance. ACS Appl Mater Interfaces. 2015;7(5):3342-3351. [21] Ganesanab P, Narayanasamya D. Lipid nanoparticles: Different preparation techniques, characterization, hurdles, and strategies for the production of solid lipid nanoparticles and nanostructured lipid carriers for oral drug delivery. Sustain Chem Pharm 2017;6:37-56. [22] Geszke-Moritz M, Moritz M.Solid lipid nanoparticles as attractive drug vehicles: Composition, properties and therapeutic strategies.Mater Sci Eng C Mater Biol Appl. 2016;68:982-994.[23] Charcosset C, Audrey J, Valour JP, et al. Preparation of liposomes at large scale using the ethanol injection method: Effect of scale-up and injection devices. Chem Eng Res Des. 2015;94(2):508-515. [24] Huang Z, Li X, Zhang T, et al. Progress involving new techniques for liposome preparation. Asian J Pharm Sci. 2014;9(4):176-182. [25] Wagner A,Vorauer-Uhl K,Katinger H.Liposomes produced in a pilot scale: production, purification and efficiency aspects.Eur J Pharm Biopharm. 2002;54(2):213-219. [26] Pham TT, Jaafar-Maalej C,Charcosset C,et al.Liposome and niosome preparation using a membrane contactor for scale-up.Colloids Surf B Biointerfaces. 2012;94:15-21. [27] Silva JO, Fernandes RS,Lopes SCA,et al. pH-Sensitive, Long-Circulating Liposomes as an Alternative Tool to Deliver Doxorubicin into Tumors: a Feasibility Animal Study. Molecular Imaging Biol.2016;18(6):1-7. [28] Carlesso FN, Araújo RS, Fuscaldi LL, et al. Preliminary data of the antipancreatic tumor efficacy and toxicity of long-circulating and pH-sensitive liposomes containing cisplatin. Nucl Med Commun. 2016;37(7):727-734. [29] Abuasal BS,Lucas C,Peyton B,et al.Enhancement of intestinal permeability utilizing solid lipid nanoparticles increases [gamma]-tocotrienol oral bioavailability. Lipids. 2012;5:461-469. [30] Andrade ME, Araujo RS,de Barros PA,et al. The role of immunomodulators on intestinal barrier homeostasis in experimental models. Clin Nutr.2015;34:1080-1087. [31] Araujo JG, Mota L, Leite EA, et al. Biodistribution and antitumoral effect of long-circulating and pH sensitive liposomal cisplatin administered in Ehrlich tumor-bearing mice. Exp Biol Med(Maywood). 2011;236:808-815. [32] Arifa RD,Madeira MF, de Paula TP,et al. Inflammasome activation is reactive oxygen species dependent and mediates irinotecan-induced mucositis through IL-1beta and IL-18 in mice. Am J Pathol. 2014;184: 2023-2034. [33] Arivarasu NA, Fatima S,Mahmood R. Effect of cisplatin on brush border membrane enzymes and anti-oxidant system of rat intestine. Life Sci. 2007;81:393-398. [34] Atista MA, Nicoli JR, Martins Fdos S,et al. Pretreatment with citrulline improves gut barrier after intestinal obstruction in mice. JPEN J Parenter Enteral Nutr. 2012;36:69-76. [35] Bearcroft CP, Domizio P, Mourad FH,et al. Cisplatin impairs fluid and electrolyte absorption in rat small intestine: a role for 5-hydroxytryptamine. Gut.1999;44:174-179. [36] Beutheu Youmba S , Belmonte L , Galas L , et al. Methotrexate modulates tight junctions through NF-kappaB, MEK, and JNK pathways. J Pediatr Gastroenterol Nutr. 2012;54:463-470. [37] Beutheu S,Ouelaa W,Guerin C, et al. Glutamine supplementation, but not combined glutamine and arginine supplementation, improves gut barrier function during chemotherapy-induced intestinal mucositis in rats. Clin Nutr. 2014;33:694-701. [38] Bodiga VL, Bodiga S,Surampudi S, et al. Effect of vitamin supplementation on cisplatin-induced intestinal epithelial cell apoptosis in Wistar/NIN rats. Nutrition. 2012;28:572-580. [39] Carvalho Junior AD,Vieira FP, Melo VJ, et al. Preparation and cytotoxicity of cisplatin-containing liposomes. Braz. J Med Biol Res. 2007;40:1149-1157. [40] de Carvalho Maroni L, de Oliveira Silveira AC, Leite EA, et al. Antitumor effectiveness and toxicity of cisplatin-loaded long-circulating and pH-sensitive liposomes against Ehrlich ascitic tumor. Exp Biol Med (Maywood). 2012;237:973-984. [41] Dobrovolskaia MA,McNeil SE.Immunological properties of engineered nanomaterials. Nat Nanotechnol. 2007;2:469-478. [42] Elsabahy M , Wooley KL. Cytokines as biomarkers of nanoparticle immunotoxicity. Chem Soc Rev. 2013;12:5552-5576. [43] Ferreira Ddos S,Lopes SC,Franco MS, et al. pH-sensitive liposomes for drug delivery in cancer treatment. Ther. Deliv. 2013;4:1099-1123. [44] Frankenberger M,Haussinger K,Ziegler-Heitbrock L. Liposomal methylprednisolone differentially regulates the expression of TNF and IL-10 in human alveolar macropHages. Int ImmunopHarmacol. 2015; 5:289-299. [45] Araújo RS, Silveira ALM, de Sales E Souza ÉL, et al. Intestinal toxicity evaluation of long-circulating and pH-sensitive liposomes loaded with cisplatin. Eur J Pharm Sci. 2017;106:142-151. [46] Monteiro LOF,Fernandes RS,Oda CMR,et al. Paclitaxel-loaded folate-coated long circulating and pH-sensitive liposomes as a potential drug delivery system: A biodistribution study. Biomed Pharmacother. 2018;97:489-495. [47] Gunji S,Obama K,Matsui M,et al. A novel drug delivery system of intraperitoneal chemotherapy for peritoneal carcinomatosis using gelatin microspHeres incorporating cisplatin.Surgery. 2013;154:991-999. [48] Duan Y,Wei L,Petryk J.Formulation, characterization and tissue distribution of a novel pH-sensitive long-circulating liposome-based theranostic suitable for molecular imaging and drug delivery. Int J Nanomedicine. 2016;11:5697-5708. [49] Leite EA,Giuberti Cdos S,Wainstein AJ, et al. Acute toxicity of long-circulating and pH-sensitive liposomes containing cisplatin in mice after intraperitoneal administration. Life Sci. 2009;8(19-20): 641-649. [50] Schillaci O, Corleto VD, Annibale B,et al. Single photon emission computed tomography procedure improves accuracy of somatostatin receptor scintigraphy in gastro-entero pancreatic tumours. Ital J Gastroenterol Hepatol.1999;31 Suppl 2:S186-189. [51] Li J,Sun K,Ni L,et al. Sodium selenosulfate at an innocuous dose markedly prevents cisplatin-induced gastrointestinal toxicity. Toxicol Appl PHarmacol. 2012;258:376-383. [52] Nose S,Wasa M,Tazuke Y,et al.Cisplatin upregulates glutamine transport in human intestinal epithelial cells: the protective mechanism of glutamine on intestinal mucosa after chemotherapy. JPEN J Parenter Enteral Nutr. 2010;34:530-537. [53] Kohli AG, Kieler-Ferguson HM, Chan D, et al. A robust and quantitative method for tracking liposome contents after intravenous administration.J Control Release. 2014;176:86-93. [54] Kohli AG, Kierstead PH, Venditto VJ, et al. Designer lipids for drug delivery: from heads to tails. J Control Release.2014;90:274-287. [55] Lajunen,T, Kontturi LS,Viitala L,et al. Indocyanine green-loaded liposomes for light-triggered drug release. Mol Pharm. 2016;13: 2095-2107. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||