Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (25): 4089-4094.doi: 10.3969/j.issn.2095-4344.0908
Previous Articles Next Articles
Wnt/beta-catenin signaling pathway regulates the differentiation of mesenchymal stem cells into epithelial cells in the treatment of chronic obstructive pulmonary disease
Zhou Xiang-xiang1, Yan Xing-yan2, Zhong Yu-lan1, Gan Xin1
- 1Department of Respiration, 2Department of Infection, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China
-
Revised:2018-05-16Online:2018-09-08Published:2018-09-08 -
Contact:Gan Xin, M.D., Chief physician, Department of Respiration, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China -
About author:Zhou Xiang-xiang, Master candidate, Department of Respiration, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China -
Supported by:the National Natural Science Foundation of China, No. 81660009; the Natural Science Foundation of Jiangxi Province, No. [2016]12
CLC Number:
Cite this article
Zhou Xiang-xiang, Yan Xing-yan, Zhong Yu-lan, Gan Xin. Wnt/beta-catenin signaling pathway regulates the differentiation of mesenchymal stem cells into epithelial cells in the treatment of chronic obstructive pulmonary disease[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 4089-4094.
share this article
2.1 间充质干细胞 间充质干细胞被誉为组织工程的种子细胞及基因载体[16],广泛存在于骨髓、胎盘和脐带等组织,其中骨髓间充质干细胞研究最多、最早。骨髓间充质干细胞的鉴定标准(国际细胞治疗协会制定)如下:①具有贴壁性,表现为贴壁生长,具有长期传代、扩增培养的特征;②具有相同的分化表型,如表达CD105、CD90及CD73,但CD11b、CD14、CD19、CD34、CD45、CD79和HLA-DR的表达为阴性;③具有多重分化潜能,如在体外诱导条件下,可以向脂肪细胞、成骨细胞及成软骨细胞分化。间充质干细胞起源于中胚层,在诱导条件下可分化为间质类细胞,如同样起源于中胚层的成骨细胞、软骨细胞、脂肪细胞。在特定条件下也可以跨胚层分化,如神经元细胞(外胚层)、胰岛细胞及肺上皮细胞(内皮细胞)等。正是因为间充质干细胞的强大分化能力,为干细胞体内移植进行细胞替代治疗及组织器官修复提供了无限的探索空间。 19世纪60年代末,首例造血干细胞移植的成功为血液系统疾病(如白血病、再生障碍性贫血等)患者带来福音。干细胞移植治疗成为各大领域的研究热点,均取得相应的成就。就呼吸系统而言,干细胞移植最初用于先天性肺发育不良等难治性肺疾病的研究居多,然后逐渐涉及难治性哮喘、慢性阻塞性肺病等慢性肺部疾病。肺部对间充质干细胞似乎有特殊的吸引力,经静脉途径进行间充质干细胞移植时,发现许多间充质干细胞被肺内毛细血管网拦截而滞留于肺脏,称为首过效应[17]。在肺毛细血管滞留后,间充质干细胞将逐步迁移至肺间质和肺泡,使肺内间充质干细胞数量增加。许多动物实验发现损伤肺组织的微环境可促进间充质干细胞向肺内“归巢”[18]。体内体外实验均表明,间充质干细胞移植对呼吸系统常见疾病均有一定疗效。Marinas-Pardo等[19]将间充质干细胞经静脉途径移植到哮喘小鼠肺组织内,发现间充质干细胞能调节气道内弹性组织的重建,对损伤的肺组织具有修复作用。在高氧诱导的支气管肺发育不良小鼠实验中,发现单次静脉注射间充质干细胞可以一定程度上逆转肺纤维化、降低肺动脉高压、减少肺血管损失,而长期经静脉进行间充质干细胞移植后,小鼠肺功能较前明显好转[20]。白冲等[21]研究团队进行干细胞移植治疗慢阻肺小鼠实验中,发现间充质干细胞迁徙至肺组织后逐渐分化为Ⅱ型肺泡上皮细胞、气道黏膜细胞及平滑肌细胞,并能改善慢阻肺的气道重塑情况。然而,间充质干细胞肺内移植工程依然面临在活体肺组织内存活时间短、分化为肺自身细胞的数量少等难题。例如,Mei等[22]发现在间充质干细胞经静脉途径注入脂多糖诱导的急性呼吸窘迫综合征小鼠体内,第3天只有小于8%的间充质干细胞保留在肺脏。同样,在间充质干细胞移植治疗急性肺损伤小鼠实验中,移植48 h后检测到间充质干细胞分化为肺上皮细胞的比例不足5%[23]。因此,如何提高间充质干细胞体内增殖能力及向肺泡上皮细胞的分化率成为亟待解决的难题。随着对干细胞移植工程的不懈研究,发现信号通路可以从分子水平调控间充质干细胞靶向修复,其中Wnt/β-catenin信号通路在间充质干细胞的生长、分化过程中起到的重要作用,有望解决间充质干细胞增殖率、分化率低的困境。 2.2 Wnt/β-catenin信号通路 wnt基因是由int1及wingless两个基因名词合并而来。Nusse等研究人员于1982年由乳腺癌病毒诱导的小鼠乳腺癌细胞中克隆出一种原癌基因,命名为int-1基因,后来证实int-1基因与果蝇遗传突变筛选中发现的wingless基因为同源基因,故二者统称为wnt基因。Wnt家族成员庞大,wnt基因可编码众多糖蛋白,分泌到细胞外发挥作用,其中有大量受体蛋白或协同受体,数量达15种以上[24]。间充质干细胞能表达多种Wnt配体,如Wnt2,Wnt4,Wnt5a,Wnt11,Wnt16,Wnt受体,Wnt抑制剂[25]。wnt基因调控的信号通路传导系统即为Wnt信号通路[26-27],包括经典与非经典Wnt通路,经典Wnt通路即Wnt/β-catenin信号通路,依赖β-catenin的转录活性发挥作用[28](图1)。非经典Wnt信号通路不依赖β-catenin发挥作用,包括平细胞极性通路(Wnt/PCP通路)及Wnt/Ca2+信号通路等[14]。这两类信号通路并非独立作用,而是相互作用的。例如,Wnt4通过非经典通路途径调节β-catenin的亚细胞定位[29],使其向细胞膜移动,导致入核的β-catenin数量减少,这将限制Wnt/β-catenin信号通路的转录活性发挥作用。目前为止,Wnt/β-catenin信号通路是Wnt信号中研究最清楚的通路。Wnt/β-catenin信号通路与非经典Wnt信号通路之间是否存在更多“串讲”(cross-talk)还需要进一步去探索。"
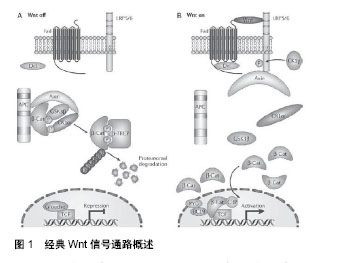

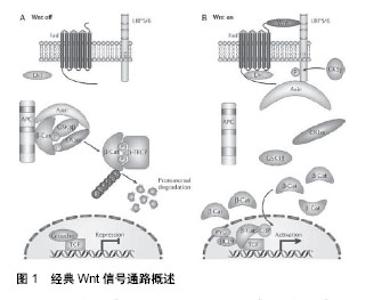
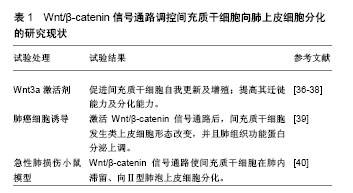
Wnt/β-catenin信号通路是高度保守的信号传导途径,不论在低等生物(如果蝇、家蚕等)或者高等哺乳动物,信号通路中成员的同源性保持一致。Wnt/β-catenin信号通路参与调控胚胎早期发育、成体组织的稳态维持、干细胞自我更新及分化等多种生物学过程[30-31]。Wnt/β-catenin信号通路包括Wnt糖蛋白、跨膜受体、胞质蛋白及核内转录因子等,信号通路途径如下:wnt基因编码Wnt配体→配体通过自分泌或旁分泌机制与细胞膜表面的糖卷曲蛋白(FZD)以及低密度脂蛋白受体相关蛋白(LRP5/6)结合形成复合物→复合物激活胞内含PDZ结构域的蓬乱蛋白(Dvl)并释放信号使β-catenin降解复合体失活→导致β-catenin在胞质内稳定表达并进入细胞核→β-catenin与核内的转录因子T细胞因子/淋巴样增强因子结合形成转录复合体(β-catenin/TCF/LEF转录复合体)→转录复合体特异性激活下游的靶基因,并使其发生转录[32-34]。当胞外无Wnt信号作用,胞内的β-catenin蛋白维持在低水平状态。由此可见,β-catenin的浓度及核内β-catenin/TCF/LEF转录复合体的形成是Wnt/β-catenin信号通路功能实现的关键步骤。胞内β-catenin浓度是依赖降解复合体与拮抗降解蛋白调节的。β-catenin降解复合体由糖原合成激酶(GSK-3β)、结肠腺癌息肉蛋白(APC)和轴蛋白(Axin)组成。拮抗降解蛋白类包括GSK-3β的结合蛋白(GSK -3βbinding protein,GBP)等。基因芯片等技术鉴定Wnt/β-catenin信号通路的靶基因库至少有500个成员[24,35],包括①细胞周期与细胞凋亡相关基因,如cyclin D1、c-myc;②关于生长因子的基因,如碱性成纤维因子等;③影响肿瘤发生发展的基因;④转录因子相关基因,如过氧化物酶增殖体激活受体(PPARs);⑤其他靶基因。近年来研究均表明Wnt/β-catenin信号通路在间充质干细胞定向分化过程中起着重要调节作用,为干细胞移植治疗呼吸系统疾病提供理论依据。然而,检索PubMed,万方数据库等国内外相关数据库,发现关于Wnt/β-catenin信号通路调节间充质干细胞定向分化为肺上皮细胞用于慢阻肺修复的研究报道十分罕见。 2.3 Wnt/β-catenin信号通路调控间充质干细胞向肺上皮分化的研究现状 Wnt3a是激活Wnt/β-catenin信号通路的关键分子,小剂量Wnt3a在体内外实验中均可促进间充质干细胞自我更新及增殖[36]。另外,Neth等[37]发现Wnt3a激活Wnt/β-catenin信号通路后,间充质干细胞向组织损伤部位迁徙能力及定向分化能力明显提高。还有动物实验发现,向慢阻肺鼠模型中静脉输注间充质干细胞,间充质干细胞也表现出肺内“归巢”现象,招募了更多外源性间充质干细胞至肺脏[38]。由此可见,间充质干细胞对慢阻肺损伤的肺组织同样存在亲和力,并且激活间充质干细胞的Wnt/β-catenin信号通路可以提升这种亲和力。与此同时,Popov等[39]进行体外共培养实验,发现肺癌细胞(A-549细胞)通过旁分泌机制激活间充质干细胞内Wnt/β-catenin信号通路后,间充质干细胞不仅形态上发生类上皮细胞样改变,而且检测到肺上皮细胞的结构蛋白及Ⅱ型肺泡细胞分泌的功能蛋白表达上调,这说明Wnt/β-catenin信号通路的激活可以促进间充质干细胞向肺上皮细胞分化。在博莱霉素介导的小鼠急性肺损伤实验中,Wnt/β-catenin信号通路促使间充质干细胞在肺内滞留、向Ⅱ型肺泡上皮细胞分化,从而显著提高了间充质干细胞对急性肺损伤的治疗效果[40],见表1。同样,很多动物实验发现Wnt/β-catenin信号通路对间充质干细胞的肺上皮细胞分化过程具有相似的调控作用[41-42]。由此可见,Wnt/β-catenin信号通路在间充质干细胞向肺上皮细胞分化过程中发挥重要作用,为Wnt/β-catenin信号通路调控间充质干细胞移植作为慢阻肺的“治本”治疗提供了理论依据。"
| [1] Baraldo S, Turato G, Saetta M. Pathophysiology of the small airways in chronic obstructive pulmonary disease. Respiration. 2012;84(2):89-97.[2] Gupta D, Agarwal R, Aggarwal AN, et al. Guidelines for diagnosis and management of chronic obstructive pulmonary disease: Joint ICS/NCCP (I) recommendations. Lung India. 2013;30(3):228-267.[3] Marçôa R, Rodrigues DM, Dias M, et al. Classification of Chronic Obstructive Pulmonary Disease (COPD) according to the new Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2017: Comparison with GOLD 2011. COPD. 2018;15(1):21-26.[4] Forey BA, Thornton AJ, Lee PN. Systematic review with meta-analysis of the epidemiological evidence relating smoking to COPD, chronic bronchitis and emphysema. BMC Pulm Med. 2011;11:36.[5] Angelis N, Porpodis K, Zarogoulidis P, et al. Airway inflammation in chronic obstructive pulmonary disease. J Thorac Dis. 2014;6 Suppl 1:S167-172.[6] Dang-Tan T, Ismaila A, Zhang S, et al. Clinical, humanistic, and economic burden of chronic obstructive pulmonary disease (COPD) in Canada: a systematic review. BMC Res Notes. 2015;8:464.[7] Ho TW, Tsai YJ, Ruan SY, et al. In-hospital and one-year mortality and their predictors in patients hospitalized for first-ever chronic obstructive pulmonary disease exacerbations: a nationwide population-based study. PLoS One. 2014;9(12):e114866.[8] Ogura-Tomomatsu H, Asano K, Tomomatsu K, et al. Predictors of osteoporosis and vertebral fractures in patients presenting with moderate-to-severe chronic obstructive lung disease. COPD. 2012; 9(4):332-337.[9] Feary JR, Rodrigues LC, Smith CJ, et al. Prevalence of major comorbidities in subjects with COPD and incidence of myocardial infarction and stroke: a comprehensive analysis using data from primary care. Thorax. 2010;65(11):956-962.[10] Wen F, Shen Y. Differential Features of COPD in China. BRN Rev. 2017;3:74-85.[11] Zhong N, Wang C, Yao W, et al. Prevalence of chronic obstructive pulmonary disease in China: a large, population-based survey. Am J Respir Crit Care Med. 2007;176(8):753-760.[12] Kearley J, Silver JS, Sanden C, et al. Cigarette smoke silences innate lymphoid cell function and facilitates an exacerbated type I interleukin-33-dependent response to infection. Immunity. 2015;42(3): 566-579.[13] Zong DD, Ouyang RY, Chen P. Epigenetic mechanisms in chronic obstructive pulmonary disease. Eur Rev Med Pharmacol Sci. 2015;19(5):844-856.[14] Shang J, Zhao J, Wu X, et al. Interleukin-33 promotes inflammatory cytokine production in chronic airway inflammation. Biochem Cell Biol. 2015;93(4):359-366.[15] Martínez-González I, Cruz MJ, Moreno R, et al. Human mesenchymal stem cells resolve airway inflammation, hyperreactivity, and histopathology in a mouse model of occupational asthma. Stem Cells Dev. 2014;23(19):2352-2363.[16] Huang K, Kang X, Wang X, et al. Conversion of bone marrow mesenchymal stem cells into type II alveolar epithelial cells reduces pulmonary fibrosis by decreasing oxidative stress in rats. Mol Med Rep. 2015;11(3):1685-1692.[17] Fischer UM, Harting MT, Jimenez F, et al. Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev. 2009;18(5):683-692.[18] Liang ZX, Sun JP, Wang P, et al. Bone marrow-derived mesenchymal stem cells protect rats from endotoxin-induced acute lung injury. Chin Med J (Engl). 2011;124(17):2715-2722.[19] Mariñas-Pardo L, Mirones I, Amor-Carro O, et al. Mesenchymal stem cells regulate airway contractile tissue remodeling in murine experimental asthma. Allergy. 2014;69(6):730-740.[20] Hansmann G, Fernandez-Gonzalez A, Aslam M, et al. Mesenchymal stem cell-mediated reversal of bronchopulmonary dysplasia and associated pulmonary hypertension. Pulm Circ. 2012;2(2):170-181.[21] 白冲,戈霞晖.间充质干细胞是否可用于哮喘的治疗[J].中国呼吸与危重监护杂志,2011,10(6):517-519.[22] Mei SH, McCarter SD, Deng Y, et al. Prevention of LPS-induced acute lung injury in mice by mesenchymal stem cells overexpressing angiopoietin 1. PLoS Med. 2007 Sep;4(9):e269.[23] Gupta N, Su X, Popov B, et al. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J Immunol. 2007;179(3):1855-1863.[24] 苏尚,吴畏. Wnt/β-catenin信号通路对靶基因转录的调控[J].中国科学:生命科学,2014,44(10):1029-1042.[25] Willert K, Nusse R. Wnt proteins. Cold Spring Harb Perspect Biol. 2012; 4(9):a007864.[26] Holstein TW. The evolution of the Wnt pathway. Cold Spring Harb Perspect Biol. 2012;4(7):a007922.[27] Niehrs C. The complex world of WNT receptor signalling. Nat Rev Mol Cell Biol. 2012;13(12):767-779.[28] 郑群,徐立红.经典Wnt信号通路中β-连环蛋白的影响[J].细胞生物学杂志,2009,31(2):183-190.[29] Bernard P, Fleming A, Lacombe A, et al. Wnt4 inhibits beta-catenin/TCF signalling by redirecting beta-catenin to the cell membrane. Biol Cell. 2008;100(3):167-177.[30] Mohammed MK, Shao C, Wang J, et al. Wnt/β-catenin signaling plays an ever-expanding role in stem cell self-renewal, tumorigenesis and cancer chemoresistance. Genes Dis. 2016;3(1):11-40. [31] Clevers H, Loh KM, Nusse R. Stem cell signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science. 2014;346(6205):1248012. [32] MacDonald BT, Tamai K, He X. Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell. 2009;17(1):9-26. [33] Kikuchi K, Niikura Y, Kitagawa K, et al. Dishevelled, a Wnt signalling component, is involved in mitotic progression in cooperation with Plk1. EMBO J. 2010;29(20):3470-3483.[34] Osolodkin DI, Palyulin VA, Zefirov NS. Glycogen synthase kinase 3 as an anticancer drug target: novel experimental findings and trends in the design of inhibitors. Curr Pharm Des. 2013;19(4):665-679.[35] Wisniewska MB, Nagalski A, Dabrowski M, et al. Novel β-catenin target genes identified in thalamic neurons encode modulators of neuronal excitability. BMC Genomics. 2012;13:635.[36] Holland JD, Klaus A, Garratt AN, et al. Wnt signaling in stem and cancer stem cells. Curr Opin Cell Biol. 2013;25(2):254-264.[37] Neth P, Ciccarella M, Egea V, et al. Wnt signaling regulates the invasion capacity of human mesenchymal stem cells. Stem Cells. 2006;24(8):1892-1903.[38] Zhang WG, He L, Shi XM, et al. Regulation of transplanted mesenchymal stem cells by the lung progenitor niche in rats with chronic obstructive pulmonary disease. Respir Res. 2014;15:33.[39] Popov BV, Serikov VB, Petrov NS, et al. Lung epithelial cells induce endodermal differentiation in mouse mesenchymal bone marrow stem cells by paracrine mechanism. Tissue Eng. 2007;13(10):2441-2450.[40] Wang C, Zhu H, Sun Z, et al. Inhibition of Wnt/β-catenin signaling promotes epithelial differentiation of mesenchymal stem cells and repairs bleomycin-induced lung injury. Am J Physiol Cell Physiol. 2014; 307(3):C234-244.[41] Sun Z, Gong X, Zhu H, et al. Inhibition of Wnt/β-catenin signaling promotes engraftment of mesenchymal stem cells to repair lung injury. J Cell Physiol. 2014;229(2):213-224.[42] Cai SX, Liu AE, Chen S, et al. Activation of Wnt/β-catenin signalling promotes mesenchymal stem cells to repair injured alveolar epithelium induced by lipopolysaccharide in mice. Stem Cell Research & Therapy. 2015;6:65.[43] Okamura M, Kudo H, Wakabayashi K, et al. COUP-TFII acts downstream of Wnt/beta-catenin signal to silence PPARgamma gene expression and repress adipogenesis. Proc Natl Acad Sci U S A. 2009; 106(14):5819-5824.[44] Hoverter NP, Waterman ML. A Wnt-fall for gene regulation: repression. Sci Signal. 2008;1(39):pe43.[45] Falconi G, Fabiani E, Fianchi L, et al. Impairment of PI3K/AKT and WNT/β-catenin pathways in bone marrow mesenchymal stem cells isolated from patients with myelodysplastic syndromes. Exp Hematol. 2016;44(1):75-83.e1-4.[46] Wang R, Ahmed J, Wang G, et al. Down-regulation of the canonical Wnt β-catenin pathway in the airway epithelium of healthy smokers and smokers with COPD. PLoS One. 2011;6(4):e14793. [47] Osiris Therapeutics reports interim data for COPD stem cell study. http://investor.osiris.com/release.cfm?Year=&Release Type=&DisplayPage=2[48] 48 Grigoryan T, Wend P, Klaus A, et al. Deciphering the function of canonical Wnt signals in development and disease: conditional loss- and gain-of-function mutations of beta-catenin in mice. Genes Dev. 2008;22(17):2308-2341. [49] 李春艳,张莹,卢瑶,等. β-连环素/TCF-4复合体在非小细胞肺癌的研究[J].中国现代医学杂志,2008,18(19):2809-2812.[50] Baarsma HA, Spanjer AI, Haitsma G, et al. Activation of WNT/β-catenin signaling in pulmonary fibroblasts by TGF-β? is increased in chronic obstructive pulmonary disease. PLoS One. 2011;6(9):e2545. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Zhou Wu, Wang Binping, Wang Yawen, Cheng Yanan, Huang Xieshan. Transforming growth factor beta combined with bone morphogenetic protein-2 induces the proliferation and differentiation of mouse MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3630-3635. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||