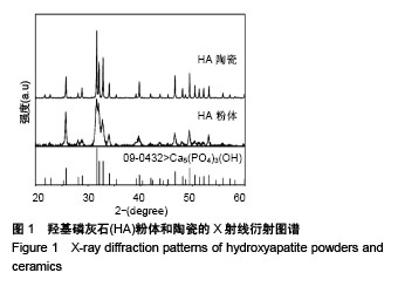
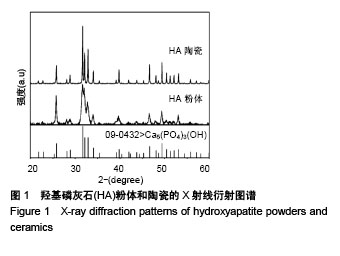
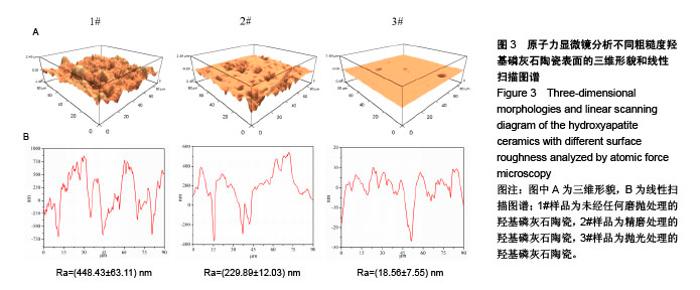
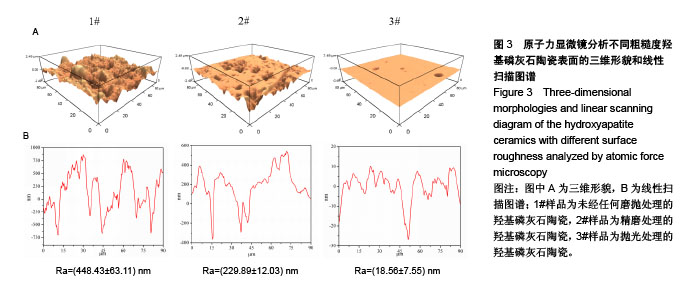
Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (18): 2903-2909.doi: 10.3969/j.issn.2095-4344.0883
Previous Articles Next Articles
Effect of nano-scaled surface roughness on the biological properties of hydroxyapatite ceramics
Liu Dan, Wu Yong-hao, Li Xiang-feng, Zhu Xiang-dong, Zhang Xing-dong
- Engineering Research Center in Biomaterials, Sichuan University, Chengdu 610064, Sichuan Province, China
-
Received:2018-05-21Online:2018-06-28Published:2018-06-28 -
Contact:Zhu Xiang-dong, Doctoral supervisor, Researcher, Engineering Research Center in Biomaterials, Sichuan University, Chengdu 610064, Sichuan Province, China -
About author:Liu Dan, Master candidate, Engineering Research Center in Biomaterials, Sichuan University, Chengdu 610064, Sichuan Province, China -
Supported by:the National Key Research and Development Program of China during the Thirteenth Five-Year Period, No. 2016YFC1102000, 2016YFC1102001; Key Technology Support Program of Sichuan Province, No. 2015SZ0026
CLC Number:
Cite this article
Liu Dan, Wu Yong-hao, Li Xiang-feng, Zhu Xiang-dong, Zhang Xing-dong. Effect of nano-scaled surface roughness on the biological properties of hydroxyapatite ceramics[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(18): 2903-2909.
share this article
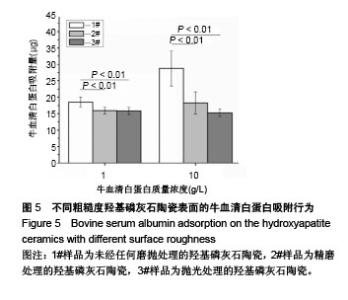
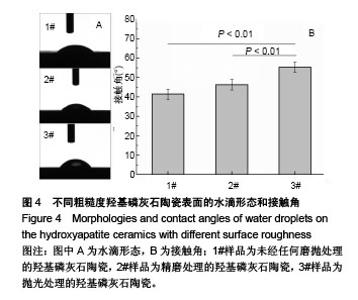
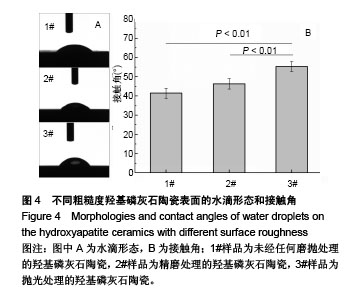
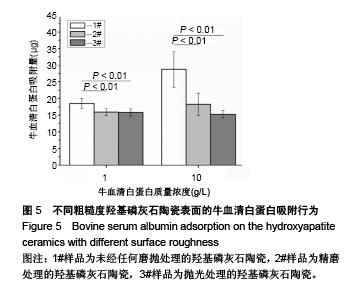
2.5 陶瓷表面的牛血清白蛋白吸附 图5为牛血清白蛋白在不同表面粗糙度的羟基磷灰石陶瓷样品表面的蛋白吸附量。从图中可以看出,表面粗糙度对羟基磷灰石陶瓷的蛋白吸附量有较大影响,高粗糙表面有利于材料吸附更多的牛血清白蛋白。1#样品具有最高的表面粗糙度,随着蛋白溶液的初始质量浓度从1.0 g/L增加到10.0 g/L,陶瓷表面吸附牛血清白蛋白总量显著增加;但随着表面粗糙度的降低,陶瓷表面吸附牛血清白蛋白总量受蛋白溶液初始质量浓度的影响变小,当蛋白溶液初始质量浓度从1.0 g/L增加到10.0 g/L时,2#和3#样品表面吸附的牛血清白蛋白总量没有明显变化,可能是因为在1.0 g/L蛋白溶液初始质量浓度下,这两种陶瓷表面的牛血清白蛋白吸附量就已趋于饱和。"
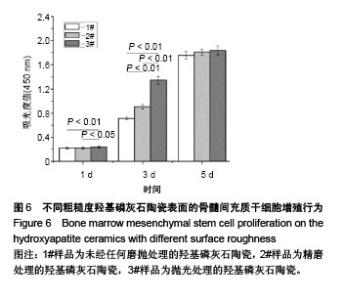
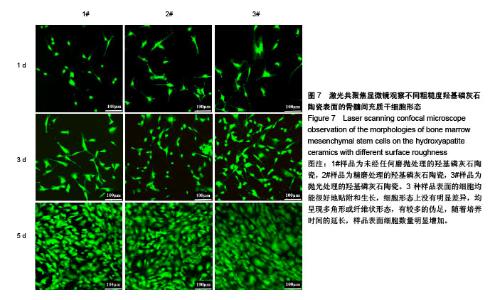
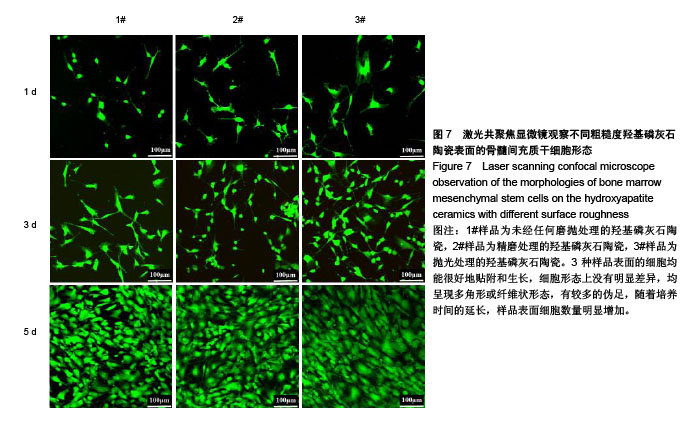
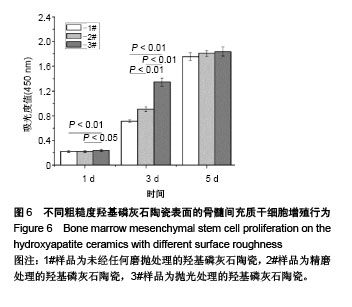
2.6 陶瓷表面细胞的增殖和生长 图6为CCK-8法检测不同粗糙度羟基磷灰石陶瓷表面骨髓间充质干细胞的增殖情况。从图中可以看出,3种不同粗糙度羟基磷灰石陶瓷表面均能够促进细胞增殖,细胞增殖数量随着培养时间的延长而显著增加,但同时也可以发现,粗糙度对陶瓷表面细胞增殖速率有一定的影响,在培养1 d时,表面粗糙度最低的3#样品表面细胞增殖数量高于1#、2#样品;在培养3 d时,表面粗糙度最低的3#样品表面细胞增殖数量最多,而表面粗糙度最高的1#样品表面细胞增殖数量最少;在培养5 d时,3种样品表面细胞增殖无显著差异,可能与细胞增殖已达到初步融合有关。 图7为激光共聚焦显微镜观察不同粗糙度羟基磷灰石陶瓷表面的骨髓间充质干细胞生长形态。从图中可以看出,3种样品表面的细胞均能很好地贴附和生长,细胞形态上没有明显差异,均呈现多角形或纤维状形态,有较多的伪足,随着培养时间的延长,样品表面细胞数量明显增加;培养到3 d时,表面粗糙度低的3#样品表面显示有更多的细胞贴附生长;培养到5 d时,所有样品表面均呈现细胞融合状态,与CCK-8细胞增殖分析结果呈现高度的一致性。"
| [1] Yang XY,Chen LH,Li Y,et al.Hierarchically porous materials: synthesis strategies and structure design.Chem Soc Rev. 2017;46:481-558.[2] Zadpoor AA.Bone tissue regeneration: the role of scaffold geometry. Biomater Sci-Uk. 2015;3:231-245.[3] Tang Z,Li X,Tan Y,et al.The material and biological characteristics of osteoinductive calcium phosphate ceramics.Regen Biomater.2018; 5:43-59.[4] Zhang K,Fan Y,Dunne N,et al.Effect of microporosity on scaffolds for bone tissue engineering. Regenerative Biomaterials.2018;5:115-124.[5] Hong YL,Fan HS,Li B,et al.Fabrication, biological effects, and medical applications of calcium phosphate nanoceramics.Mat Sci Eng R. 2010; 70:225-242.[6] Ranella A,Barberoglou M,Bakogianni S,et al.Tuning cell adhesion by controlling the roughness and wettability of 3D micro/nano silicon structures.Acta Biomater. 2010;6:2711-2720.[7] Rosales-Leal JI,Rodriguez-Valverde MA,Mazzaglia G,et al.Effect of roughness, wettability and morphology of engineered titanium surfaces on osteoblast-like cell adhesion.Colloid Surface A.2010;365:222-229.[8] Dee KC,Puleo DA,Bizios R.Protein-surface interactions. In: Dee KC, Puleo DA, Bizios R, editors. An introduction to tissue-biomaterial interactions.New York:John Wiley & Sons, 2002:37-52.[9] Wang J,Zhu Y,Wang M,et al.Fabrication and preliminary biological evaluation of a highly porous biphasic calcium phosphate scaffold with nano-hydroxyapatite surface coating.Ceram Int. 2018;44:1304-1311.[10] Bohner M,Loosli Y,Baroud G,et al.Commentary: Deciphering the link between architecture and biological response of a bone graft substitute. Acta Biomater.2011;7:478-484.[11] Best SM,Porter AE,Thian ES,et al.Bioceramics: Past, present and for the future.J Eur Ceram Soc.2008;28:1319-1327.[12] Dorozhkin SV.Nanodimensional and Nanocrystalline Apatites and Other Calcium Orthophosphates in Biomedical Engineering, Biology and Medicine.Materials. 2009;2:1975-2045.[13] Tripathi G,Basu B.A porous hydroxyapatite scaffold for bone tissue engineering: Physico-mechanical and biological evaluations.Ceram Int.2012;38:341-349.[14] Yang PP,Quan ZW,Li CX,et al.Bioactive, luminescent and mesoporous europium-doped hydroxyapatite as a drug carrier.Biomaterials. 2008; 29:4341-4347.[15] Civelek AC,Pacheco EM,Natarajan TK,et al.Quantitative Measurement of Vascularization and Vascular Ingrowth Rate of Coralline Hydroxyapatite Ocular Implant by Tc-99m Mdp Bone Imaging. Clin Nucl Med.1995;20:779-787.[16] Cowper TR.Hydroxyapatite Motility Implants in Ocular Prosthetics.J Prosthet Dent. 1995;73:267-273.[17] Bose S,Dasgupta S,Tarafder S,et al.Microwave-processed nanocrystalline hydroxyapatite: Simultaneous enhancement of mechanical and biological properties.Acta Biomater. 2010;6:3782-3790.[18] Lin KL,Chen L,Chang J.Fabrication of Dense Hydroxyapatite Nanobioceramics with Enhanced Mechanical Properties via Two-Step Sintering Process.Int J Appl Ceram Tec.2012;9:479-485.[19] Adeleke SA,Ramesh S,Bushroa AR,et al.The properties of hydroxyapatite ceramic coatings produced by plasma electrolytic oxidation. Ceram Int.2018;44:1802-1811.[20] Machado GC,Garcia-Tunon E,Bell RV,et al.Calcium phosphate substrates with emulsion-derived roughness: Processing, characterisation and interaction with human mesenchymal stem cells.J Eur Ceram Soc.2018;38:949-961.[21] Roy M,Fielding GA,Beyenal H,et al.Mechanical, In vitro Antimicrobial, and Biological Properties of Plasma-Sprayed Silver-Doped Hydroxyapatite Coating.Acs Appl Mater Inter. 2012;4:1341-1349.[22] Heimann RB.Plasma-Sprayed Hydroxylapatite-Based Coatings: Chemical, Mechanical, Microstructural,and Biomedical Properties.J Therm Spray Techn.2016;25:827-850.[23] Zhang XD,Yuan HP,de Groot K.Calcium phosphate biomaterials with intrinsic osteoinductivity. The 6th world biomaterials congress.Hawaii, USA,2000:15-20.[24] Tang XM,Huang K,Dai J,et al.Influences of surface treatments with abrasive paper and sand-blasting on surface morphology, hydrophilicity, mineralization and osteoblasts behaviors of n-CS/PK composite.Sci Rep.2017;7(1):568. [25] Fujibayashi S,Neo M,Kim HM,et al.Osteoinduction of porous bioactive titanium metal.Biomaterials.2004;25:443-450.[26] Del Fabbro M,Taschieri S,Canciani E,et al.Osseointegration of Titanium Implants With Different Rough Surfaces: A Histologic and Histomorphometric Study in an Adult Minipig Model.Implant Dent. 2017;26:357-366.[27] Lin KL,Xia LG,Gan JB,et al.Tailoring the Nanostructured Surfaces of Hydroxyapatite Bioceramics to Promote Protein Adsorption, Osteoblast Growth, and Osteogenic Differentiation.Acs Appl Mater Inter.2013;5:8008-8017.[28] Hayashi K,Inadome T,Tsumura H,et al.Effect of surface roughness of hydroxyapatite-coated titanium on the bone-implant interface shear strength.Biomaterials. 1994;15:1187-1191.[29] Puleo DA,Nanci A.Understanding and controlling the bone-implant interface.Biomaterials. 1999;20:2311-2321.[30] Zhu XD,Zhang HJ,Fan HS,et al.Effect of phase composition and microstructure of calcium phosphate ceramic particles on protein adsorption.Acta Biomater.2010;6:1536-1541.[31] Zhu XD,Fan HS,Xiao YM,et al.Effect of surface structure on protein adsorption to biphasic calcium-phosphate ceramics in vitro and in vivo.Acta Biomater.2009;5:1311-1318.[32] Zhu XD,Zhang HJ,Li DX,et al.Study on the enhanced protein adsorption of microwave sintered hydroxyapatite nanoceramic particles: Role of microstructure.J Biomed Mater Res B Appl Biomater.2012;100(2): 516-523.[33] Hennessy KM,Clem WC,Phipps MC,et al.The effect of RGD peptides on osseointegration of hydroxyapatite biomaterials.Biomaterials. 2008; 29:3075-3083.[34] Deligianni DD,Katsala N,Ladas S,et al.Effect of surface roughness of the titanium alloy Ti-6Al-4V on human bone marrow cell response and on protein adsorption.Biomaterials. 2001;22:1241-1251.[35] Deligianni DD,Katsala ND,Koutsoukos PG,et al.Effect of surface roughness of hydroxyapatite on human bone marrow cell adhesion, proliferation, differentiation and detachment strength. Biomaterials. 2001;22:87-96.[36] dos Santos EA,Farina M,Soares GA,et al.Chemical and topographical influence of hydroxyapatite and beta-tricalcium phosphate surfaces on human osteoblastic cell behavior.J Biomed Mater Res A.2009;89A:510-520. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [3] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [4] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [5] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [6] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [7] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [8] | Zhou Jihui, Yao Meng, Wang Yansong, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Influence of novel nanoscaffolds on biological behaviors of neural stem cells and the related gene expression [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 532-536. |
| [9] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [10] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [11] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [12] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [13] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [14] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [15] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||