Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (18): 2927-2932.doi: 10.3969/j.issn.2095-4344.0749
Previous Articles Next Articles
Effect of hyaluronic acid and its derivatives on osteoblast function: molecular mechanisms and clinical values
Di Jia-wei, Li Xiang, Xie Rui-min, Wang Yong-ping
- Department of Orthopedics, First Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China
-
Received:2018-02-06Online:2018-06-28Published:2018-06-28 -
Contact:Wang Yong-ping, M.D., Master’s supervisor, Department of Orthopedics, First Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China -
About author:Di Jia-wei, Department of Orthopedics, First Hospital of Lanzhou University, Lanzhou 730000, Gansu Province, China -
Supported by:the Scientific Research Plan Program of Lanzhou City, No. 2017-4-58
CLC Number:
Cite this article
Di Jia-wei, Li Xiang, Xie Rui-min, Wang Yong-ping. Effect of hyaluronic acid and its derivatives on osteoblast function: molecular mechanisms and clinical values[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(18): 2927-2932.
share this article
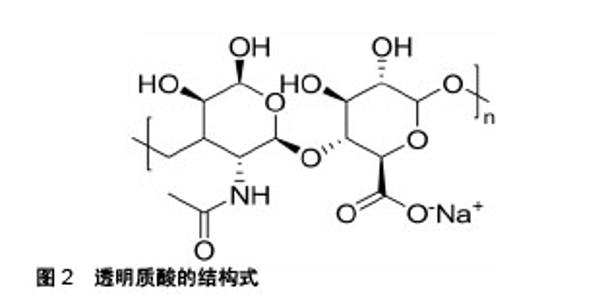
2.1 透明质酸的理化性质 透明质酸的分子式为(C14H20NNaO11)n(结构式见图2),是通过β-糖苷键将D-葡萄糖醛酸和N-乙酰-D-葡糖胺重复连接组成的线形多糖结构。其本身带有负电荷,存在于脊椎动物的结缔组织中,与结缔组织中其他糖胺聚糖不同,透明质酸合成于质膜而非内质网或高尔基体,故其一级结构不含肽,也是唯一没有与核心蛋白连接,没有硫酸化,也没有经过任何合成后修饰的糖胺聚糖。尽管它与其他糖胺聚糖一样由单一的多糖链组成,然而透明质酸的相对分子量通常达到数百万,同时沿轴线形成的次级氢键使其在空间上呈刚性的螺旋柱状结构和非极性相对疏水的面,柱的内侧存在大量的羟基有着强亲水性,这种结构赋予了透明质酸出色的流变性能、润滑性、渗透性和保水作用,当在水合状态下透明质酸产生了特殊的黏弹性,使透明质酸在医学领域有着巨大的发展潜力[14]。 2.2 透明质酸对成骨细胞功能的影响 透明质酸是一种无毒性、无免疫原性、无致炎性的生物材料,并具有良好的生物降解性和生物相容性,其独特的保水能力和渗透性对细胞的增殖分化具有重要意义,加上其特殊的结构,可以作为一种临时的细胞支架支持并促进成骨细胞生长,完成支架作用后逐渐降解被成骨细胞钙化形成的新生骨组织所替代[15-16]。Sadikoglu等[17]向过度扩张上下颌的大鼠颌缝区注射高分子量透明质酸(5 000 ku),组织形态学研究发现颌骨区和骨缝区成骨细胞比例均显著增加。Sá等[11]通过MTT和免疫荧光技术研究发现低浓度透明质酸(10,1,100 mg/L)对成骨细胞活性没有影响,而高浓度透明质酸(1 g/L)处理成骨细胞48 h后细胞活性降低;Von Kossa染色可见100 mg/L透明质酸显著增加了成骨细胞矿化成骨结节的面积,RT-PCR显示这可能与透明质酸干预后,Ⅰ型和Ⅲ型胶原蛋白,骨钙蛋白和骨形态发生蛋白2和4的mRNA表达增加有关。Kim等[18]向比格犬拔牙后的牙槽窝内注射1%的透明质酸,组织形态学结果显示牙槽窝内填充矿化骨量增加且周围成骨细胞较多;Masako等[19]通过MTS发现透明质酸对细胞增殖几乎没有影响,但茜素红染色显示透明质酸可以诱导成骨细胞分化形成矿化结节,RT-PCR同样提示这可能与骨钙素的mRNA表达增加有关。Zhao等[20]研究发现随着透明质酸分子量的增加,成骨细胞所形成的钙结节数量增加。Neunzehn等[21]通过动力学氧气测量发现透明质酸干预的细胞活性较高,免疫组化染色也显示其Ⅰ型胶原蛋白、骨连结蛋白表达明显增多。Mladenovic等[22]使用白细胞介素1干预成骨细胞制作了炎症模型,透明质酸保护后发现,由于白细胞介素1刺激产生炎性反应而升高的基质金属蛋白酶3、基质金属蛋白酶13、ADAMTS-4、ADAMTS-5、RANKL、CD44和细胞间黏附分子1基因表达降低。Boeckel等[23]同样通过MTT发现透明质酸对成骨细胞活力的影响没有显著性意义。但申德山等[24]的研究结果显示透明质酸在骨组织形成方面无明显差异。 2.3 化学改性的透明质酸对成骨细胞功能的影响 透明质酸在水体系中不具有持久的机械完整性,为了获得基于透明质酸且具有机械性能和所需降解速率的水凝胶,同时保持其生物学功能,势必需要对透明质酸进行一定的化学修饰和共价交联,改变透明质酸的分子质量,获得具有新的生物活性和功能性的透明质酸交联衍生物[25]。制备原理就是将透明质酸分子的羟基、羧基和乙酰氨基作为化学交联的位点,通过化学交联剂的相应官能团与之反应,使透明质酸分子相互交联。通过化学改性制备的透明质酸衍生物与天然透明质酸相比表现出更强的机械性能,且不易酶促降解。针对透明质酸应用于组织工程的交联改性,合成的透明质酸水凝胶的化学物质应该是化学选择性的,在生理条件下不产生任何有毒的副产物也不导致透明质酸的降解[26]。在这个背景下,许多研究者对透明质酸进行了各种化学改性,并应用于成骨细胞进行试验。 Cui等[27]使用3,3'-二硫代二丙酸通过腙键交联合成的3,3'-二硫代二丙酸酰肼修饰的透明质酸和交联剂聚乙二醇二乙酰丙酸酯反应合成了命名为HA-PEG的水凝胶,通过活细胞/死细胞染色实验和MTT发现,HA-PEG具有良好的生物相容性,能够支持MC3T3-E1细胞的附着和增殖;使用腙键交联和光交联反应合成了聚(Nε-丙烯酰基-L-赖氨酸)/透明质酸互穿网络,通过MTT和苏木精-伊红染色发现,其有效提高了生物相容性,允许细胞和组织浸润并维持细胞活力,促进增殖。Jing等[28]使用戊烯酸酐和乙醇通过硫醇-烯反应功能化透明质酸,合成了戊烯酸改性的透明质酸,又通过光诱导的硫醇-烯反应的得到了透明质酸水凝胶,结果发现透明质酸水凝胶有利于成骨细胞的扩散,EdU增殖实验和茜素红染色也显示,其对于成骨细胞的增殖和矿化也有积极意义。Wu等[29]使用高分子量透明质酸与高碘酸钠反应合成HA-CHO、中等分子量透明质酸与己二酰肼反应合成HA-ADH,最后使用两步法合成了HA-PolyP。使用钙黄绿素、茜红素染色和碘化丙锭的活/死细胞染色发现成骨细胞在HA-PolyP凝胶网络生长良好,钙积累增多,碱性磷酸酶的表达量也更多。Mermerkaya等[30]使用了商品化的HYALONECTHA®进行试验,该产品是一种生物可吸收的针织网格,由透明质酸酯化后的苄基酯HYAFF组成,研究首次应用放射性核素显像技术证明了骨缺损位点的成骨细胞活性明显增加。Picke等[31]使用臭氧分解高分子量透明质酸制备了低分子量透明质酸,又通过SO3-DMF硫酸化低分子量透明质酸合成了高硫酸化透明质酸衍生物,研究高糖环境下高硫酸化透明质酸衍生物对成骨细胞的影响。体外实验结果发现,无论糖浓度如何,成骨细胞骨形态发生蛋白2、骨桥蛋白的基因表达均升高,而RANKL mRNA表达降低,RANKL/骨桥蛋白表达比率显著降低,这提示高硫酸化透明质酸衍生物是用于改善糖尿病状况下骨缺损再生的潜在生物材料。 2.4 基于透明质酸的人造细胞外基质对成骨细胞功能的影响 细胞外基质是细胞微环境的重要组成部分,它与各种生长因子一起,在调节增殖黏附、成熟分化方面起着重要作用,动物组织的细胞外基质由蛋白质和多糖构成,主要部分由糖胺聚糖组成,而硫酸化的糖胺聚糖对于骨形成是十分重要的,它们大多数可以与参与成骨细胞谱系调节的生长因子结合,所以不言而喻,作为糖胺聚糖重要成分之一的透明质酸自然也在这其中起着至关重要的作用。许多研究者基于组织工程的仿生方法,以体内微环境为镜像,以透明质酸为核心材料,制作了一系列应用于成骨细胞的新型功能性人造细胞外基质。Mathews等[32]使用涂覆包含各种糖胺聚糖的组织培养板培养细胞,PCR结果发现与成骨细胞分化相关的基因,例如Osterix,骨钙素,骨桥蛋白和Ⅰ型胶原蛋白α1均表现出显着的上调,然而通过横向比较发现,含透明质酸的组织培养板为成骨细胞分化和骨基质合成提供了最有利的条件。Hempel等[33]首先制备了硫酸化透明质酸衍生物sHA1Δ6S,又与胶原蛋白混合在组织培养板上作为细胞生长的人造细胞外基质,结果显示人造细胞外基质明显提高了组织非特异性碱性磷酸酶的活性和钙沉积量,且在早期成骨细胞中最为突出。Salbach-Hirsch等[34]通过控制热降解天然高分子量透明质酸制备了低分子量透明质酸,又通过硫酸化透明质酸和软骨素并复胶原蛋白制造了人造胶原蛋白涂层,PCR结果显示碱性磷酸酶,骨保护素显著上调,局部微环境内Ca2+浓度更高,矿化钙沉积也更多,在骨重建过程中该涂层有着良好的表现。该团队为进一步证明该人造细胞外基质的临床应用价值,就其分子机制通过信号生物学做出了进一步分析,结果表明,涂层中高度硫酸化的糖胺聚糖可能以浓度依赖性和硫酸依赖性的方式通过与调节成骨细胞的主要信号通路Wnt上的硬化蛋白结合,从而干扰硬化蛋白/LRP5/6复合物形成,以实现控制骨稳态,促进骨重建的生物学特性[35]。 2.5 基于透明质酸的纳米材料对成骨细胞功能的影响 以上研究所使用的透明质酸水凝胶由聚阴离子链相互连接形成,属于大体积凝胶。如果将交联网络缩小则可获得纳米凝胶材料。由于这种材料的稳定性,低细胞毒性,尺寸可调性,高空隙率,大表面积和防止酶降解等特点,对于成骨细胞增殖分化具有更积极的意义,也能作为理想的药物递送载体。现已有各种研究采用多种加工技术来生产基于透明质酸的纳米材料。 Sujana等[36]通过静电纺丝技术制备了与天然细胞外基质相近的生物相容性聚(L-乳酸)-聚(ε-己内酯)-丝素-羟基磷灰石-透明质酸纳米纤维支架,通过MTS检测表明生长于该材料上的成骨细胞增殖率明显增加,场发射扫描电子显微镜也观察到了细胞矿化增多,这与检测到的碱性磷酸酶,骨钙素表达上调有着密切关系。Jensen等[37]使用透明质酸、β-磷酸三钙和聚己内酯制成了HT-PCL微纳米支架,比色法测定Ca2+发现该材料能促进成骨分化和Ca2+沉积,流式细胞术和电镜扫描也表明HT-PCL支架能够支持细胞增殖和细胞迁移,使细胞在该材料中均匀分散。RT-PCR提示与成骨细胞成熟分化相关的骨钙素,骨形态发生蛋白2,骨桥蛋白基因表达明显上调。Hu等[38]使用透明质酸,纳米级羟基磷灰石,壳聚糖和硫酸软骨素组成了仿生磷酸钙矿化有机-无机杂化支架,结果证明该支架材料具有良好的生物相容性,并且所添加的成分能够促进成骨细胞的增殖和分化。Rammal等[39]利用透明质酸,磷酸钙和低分子量壳聚糖合成了复合材料。共聚焦荧光图像显示导致早期成骨样表型的蛋白骨桥蛋白,骨钙素表达增加,ELISA表明骨桥蛋白,血管内皮生长因子和骨形态发生蛋白2等骨组织调节因子的表达上调,除此之外,研究惊喜的发现该材料有着良好的细菌抗黏附性,是潜在的组织工程支架材料。 2.6 透明质酸复合材料结合生长因子或药物对成骨细胞的影响 由于透明质酸及其衍生物具有良好的生物相容性及可降解性,很多研究将基于透明质酸的复合材料与各种生长因子或药物包被,构成缓释载体作用于成骨细胞。透明质酸经过适当交联后分子链增长,相对分子质量增大,形成“超分子笼”,从而实现生长因子或药物的递送与缓释。 骨形态发生蛋白2是转化生长因子β超家族成员,具有促进成骨细胞成熟分化,参与骨软骨生长发育和重建的重要功能,可以加速骨缺损修复。Nath等[40]将透明质酸与壳聚糖交联形成聚电解质复合物支架,又将骨形态发生蛋白2固定于支架中,MTT检测表明成骨细胞在该支架上活性高,增殖性和黏附性好,且在骨形态发生蛋白2的存在下,与骨基质合成及矿化相关的碱性磷酸酶和骨桥蛋白的mRNA表达是显著升高的。Zhang等[41]使用透明质酸,阿仑膦酸钠和骨形态发生蛋白2制备了一种天然多糖衍生的纳米复合材料,体外实验结果显示,纳米复合材料中的成骨细胞具有较高的细胞迁移能力,碱性磷酸酶活性高,与成骨细胞矿化相关的基因Runx2,Osterix等表达上调,有效促进了成骨细胞分化和矿化结节的形成。Chen等[42]通过链霉亲和素共轭蛋白官能化透明质酸(透明质酸-链霉亲和素共轭蛋白)和生物素末端星形聚乙二醇缀合制作了纳米纤维水凝胶,并将骨形态发生蛋白2一同包封在材料内培养成骨细胞,结果显示该材料对成骨细胞活性和分化具有优异作用。与骨形态发生蛋白2同属一个家族的骨形态发生蛋白7也是对于成骨细胞具有重要作用的生长因子,Jung等[43]将透明质酸-明胶水凝胶涂布在多通道双相磷酸钙颗粒上,制备聚(乳酸-乙醇酸共聚物)微球体并加载骨形态发生蛋白7后用聚多巴胺修饰,开发出新型杂化材料复合体系,体外研究表明其能促进成骨细胞增殖和维持其高活性状态。Hamlet等[44]使用透明质酸水凝胶包裹骨形态发生蛋白7和成骨细胞,涂布在以医用级聚己内酯为材料的3D打印支架上,以评估成骨细胞的矿化潜力。荧光染色显示包裹在透明质酸水凝胶中的成骨细胞活力没有受到影响,结果表明了该材料具有良好的生物相容性,在体外能为成骨细胞提供良好的增殖环境。血小板是血液中一种重要细胞成分,含有一系列必要的生长因子,在Son等[45]的工作中,使用了透明质酸等天然聚合物对富含血小板的血浆进行封固,并将其装载在双相磷酸钙支架中。共聚焦显微图像显示支架上成骨细胞活性较好,黏附率高且细胞增殖率增加。纤连蛋白是血浆中含量较高的蛋白质,所以在假体手术植入物置入后有大量富集。此外,纤连蛋白在肉芽组织形成期间可以提供用于细胞附着的基质,在愈合过程中起着关键作用。Galli等[46]使用抗纤连蛋白适体包裹聚乙二醇二丙烯酸酯/硫醇化透明质酸水凝胶,并在水凝胶上培养成骨细胞,通过荧光显微镜观察到,水凝胶上的成骨细胞活性更高,且增殖率和迁移程度明显提升,说明透明质酸水凝胶负载抗纤连蛋白适体显著促进了成骨细胞的黏附和定植。骨膜素是高表达于骨膜的基质蛋白,与愈合功能相关。另外,其又进一步将透明质酸和聚乙二醇用水重构得到硫醇化水凝胶,并将骨膜素混合加载在材料中,结果发现该材料可维持细胞活性,促进细胞黏附[47]。透明质酸复合材料对药物的递送作用和缓释作用也同样引人关注。Sauerova等[48]测试了对细胞有害的阳离子表面活性剂乙氧羰基新霉素和十六烷基三甲基溴化铵与透明质酸形成胶体复合物后用于药物递送的潜在可能性,结果发现两种复合物均能降低表面活性剂单独存在时对的成骨细胞的毒性,且透明质酸分子量决定着“透明质酸-细胞”的相互作用,并指导药物递送与细胞运输功能,这可能与成骨细胞表面CD44受体(细胞表面主要的透明质酸受体)有关。Bang等[49]使用1,4-丁二醇二缩水甘油醚与透明质酸,并将2种对于骨再生具有积极作用的药物二甲基羟乙基甘氨酸和硼酸钠装载其中,体外实验结果表明,成骨细胞在该材料中显示了高增殖率,高碱性磷酸酶活性,良好的成骨反应以及HIF-1α和Runx2表达水平的上调,可应用于骨再生领域。"
| [1] Laurent TC, Fraser JR. Hyaluronan. FASEB J. 1992;6(7):2397-2404.[2] Meyer K, Palmer JW. The polysaccharide of the vitreous humor. J Biol Chem. 1934;107(3):629-634.[3] Furlan S, La Penna G, Perico A, et al. Hyaluronan chain conformation and dynamics. Carbohydr Res. 2005;340(5):959-970.[4] Yoneda M, Shimizu S, Nishi Y, et al. Hyaluronic acid-dependent change in the extracellular matrix of mouse dermal fibroblasts that is conducive to cell proliferation. J Cell Sci. 1988;90 (2):275.[5] 拉希德,刘世清.透明质酸对体外培养大鼠退变关节软骨细胞的影响[J].武汉大学学报(医学版),2007,28(2):177-180.[6] Turley EA. Hyaluronan and cell locomotion. Cancer Metastasis Rev. 1992;11(1):21.[7] Suzuki Y, Yamaguchi T. Effects of hyaluronic acid on macrophage phagocytosis and active oxygen release. Agents Actions. 1993;38(1): 32-37.[8] Lisignoli G, Grassi F, Zini N, et al. Anti‐Fas–induced apoptosis in chondrocytes reduced by hyaluronan: Evidence for CD44 and CD54 (intercellular adhesion molecule 1) involvement. Arthritis Rheumatol. 2001;44(8):1800-1807.[9] Noble PW. Hyaluronan and its catabolic products in tissue injury and repair. Matrix Biol. 2002;21(1):25-29.[10] Patel SR, Malhotra A, White DP, et al. The mechanism of action for hyaluronic acid treatment in the osteoarthritic knee: A systematic review. BMC Musculoskelet Disord. 2015;16(1):321.[11] Sá MA, Ribeiro HJ, Valverde TM, et al. Single-walled carbon nanotubes functionalized with sodium hyaluronate enhance bone mineralization. Braz J Med Biol Res. 2016;49(2): e4888.[12] Khorshidi HR, Kasraianfard A, Derakhshanfar A, et al. Evaluation of the effectiveness of sodium hyaluronate, sesame oil, honey, and silver nanoparticles in preventing postoperative surgical adhesion formation. An experimental study. Acta Cir Bras. 2017;32(8):626.[13] Mendes RM, Silva GA, Lima MF, et al. Sodium hyaluronate accelerates the healing process in tooth sockets of rats. Arch Oral Biol. 2008;53(12):1155-1162.[14] Fraser J, Laurent T, Laurent U. Hyaluronan: its nature, distribution, functions and turnover. J Intern Med. 1997;242(1):27-33.[15] 蔺栋鹏,吕瑾茹,赵天一,等.透明质酸、TGF-β1对下颌骨髁突软骨增殖分化的影响[J].实用口腔医学杂志, 2017,33(5):584-588.[16] 陈明伟,范彬彬,卢华定.新型透明质酸改性的CS-g-PEI/pDNA的构建及介导转染软骨细胞的体外研究[J].中国医药导报, 2017,14(21):4-9.[17] Sadikoglu TB, Nalbantgil D, Ulkur F, et al. Effect of hyaluronic acid on bone formation in the expanded interpremaxillary suture in rats. Orthod Craniofac Res. 2016;19(3):154-161.[18] Kim JJ, Song HY, Ben AH, et al. Hyaluronic acid improves bone formation in extraction sockets with chronic pathology: a pilot study in dogs. J Periodontol. 2016;87(7):790-795.[19] Fujioka-Kobayashi M, Schaller B, Kobayashi E, et al. Hyaluronic acid gel-based scaffolds as potential carrier for growth factors: an in vitro bioassay on its osteogenic potential. J Clin Med. 2016;5(12):E112.[20] Zhao N, Wang X, Qin L, et al. Effect of molecular weight and concentration of hyaluronan on cell proliferation and osteogenic differentiation in vitro. Biochem Biophys Res Commun. 2015;465(3): 569-574.[21] Neunzehn J, Szuwart T, Wiesmann HP. Eggshells as natural calcium carbonate source in combination with hyaluronan as beneficial additives for bone graft materials, an in vitro study. Head Face Med. 2015;11:12.[22] Mladenovic Z, Saurel AS, Berenbaum F, et al. Potential role of hyaluronic acid on bone in osteoarthritis: matrix metalloproteinases, aggrecanases, and RANKL expression are partially prevented by hyaluronic acid in interleukin 1-stimulated osteoblasts. J Rheumatol. 2014;41(5):945-954.[23] Boeckel DG, Shinkai RS, Grossi ML, et al. In vitro evaluation of cytotoxicity of hyaluronic acid as an extracellular matrix on OFCOL II cells by the MTT assay. Oral Surg Oral Med Oral Pathol Oral Radiol. 2014;117(6):e423-428.[24] 申德山,刘亚蕊,张清彬,等.牙周膜干细胞移植促进兔牙槽突裂成骨修复研究[J].中国实用口腔科杂志,2013,2:96-99.[25] 聂素云.改性透明质酸薄膜的体外释药性能研究[J].中国药学杂志, 2012, 47(19): 1558-1560.[26] 岩晓丽,冯玉杰,卢伟,等.胶原/氧化透明质酸复合水凝胶支架的制备与表征.中国实用医药, 2011,6(29):3-5.[27] Cui N, Qian J, Xu W, et al. Preparation, characterization, and biocompatibility evaluation of poly(Nvarepsilon-acryloyl-L-lysine)/ hyaluronic acid interpenetrating network hydrogels. Carbohydr Polym. 2016,136:1017-1026.[28] Jing J, Fournier A, Szarpak-Jankowska A, et al. Type, density, and presentation of grafted adhesion peptides on polysaccharide-based hydrogels control preosteoblast behavior and differentiation. Biomacromolecules. 2015;16(3):715-722.[29] Wu AT, Aoki T, Sakoda M, et al. Enhancing osteogenic differentiation of MC3T3-E1 cells by immobilizing inorganic polyphosphate onto hyaluronic acid hydrogel. Biomacromolecules. 2015;16(1):166-173.[30] Mermerkaya MU, Doral MN, Karaaslan F, et al. Scintigraphic evaluation of the osteoblastic activity of rabbit tibial defects after HYAFF11 membrane application. J Orthop Surg Res. 2016;11(1):57.[31] Picke AK, Salbach-Hirsch J, Hintze V, et al. Sulfated hyaluronan improves bone regeneration of diabetic rats by binding sclerostin and enhancing osteoblast function. Biomaterials. 2016;96:11-23.[32] Mathews S, Mathew SA, Gupta PK, et al. Glycosaminoglycans enhance osteoblast differentiation of bone marrow derived human mesenchymal stem cells. J Tissue Eng Regen Med. 2014;8(2):143-152.[33] Hempel U, Preissler C, Vogel S, et al. Artificial extracellular matrices with oversulfated glycosaminoglycan derivatives promote the differentiation of osteoblast-precursor cells and premature osteoblasts. Biomed Res Int. 2014;2014:938368.[34] Salbach-Hirsch J, Ziegler N, Thiele S, et al. Sulfated glycosaminoglycans support osteoblast functions and concurrently suppress osteoclasts. J Cell Biochem. 2014;115(6):1101-1111.[35] Salbach-Hirsch J, Samsonov SA, Hintze V, et al. Structural and functional insights into sclerostin-glycosaminoglycan interactions in bone. Biomaterials. 2015;67:335-345.[36] Sujana A, Venugopal JR, Velmurugan B, et al. Hydroxyapatite- intertwined hybrid nanofibres for the mineralization of osteoblasts. J Tissue Eng Regen Med. 2017;11(6):1853-1864.[37] Jensen J, Kraft DC, Lysdahl H, et al. Functionalization of polycaprolactone scaffolds with hyaluronic acid and beta-TCP facilitates migration and osteogenic differentiation of human dental pulp stem cells in vitro. Tissue Eng Part A. 2015;21(3-4):729-739.[38] Hu Y, Chen J, Fan T, et al. Biomimetic mineralized hierarchical hybrid scaffolds based on in situ synthesis of nano-hydroxyapatite/ chitosan/chondroitin sulfate/hyaluronic acid for bone tissue engineering. Colloids Surf B Biointerfaces. 2017;157:93-100.[39] Rammal H, Dubus M, Aubert L, et al. Bioinspired nanofeatured substrates: suitable environment for bone regeneration. ACS Appl Mater Interfaces. 2017;9(14):12791-12801.[40] Nath SD, Abueva C, Kim B, et al. Chitosan-hyaluronic acid polyelectrolyte complex scaffold crosslinked with genipin for immobilization and controlled release of BMP-2. Carbohydr Polym. 2015;115:160-169.[41] Zhang Y, Hu Y, Luo Z, et al. Simultaneous delivery of BMP-2 factor and anti-osteoporotic drugs using hyaluronan-assembled nanocomposite for synergistic regulation on the behaviors of osteoblasts and osteoclasts in vitro. J Biomater Sci Polym Ed. 2015;26(5):290-310.[42] Chen H, Xing X, Jia Y, et al. Nano-Fibrous Biopolymer Hydrogels via Biological Conjugation for Osteogenesis. J Nanosci Nanotechnol. 2016;16(6):5562-5568.[43] Jung A, Makkar P, Amirian J, et al. A novel hybrid multichannel biphasic calcium phosphate granule-based composite scaffold for cartilage tissue regeneration. J Biomater Appl. 2017:885328217741757.[44] Hamlet SM, Vaquette C, Shah A, et al. 3-Dimensional functionalized polycaprolactone-hyaluronic acid hydrogel constructs for bone tissue engineering. J Clin Periodontol. 2017;44(4):428-437.[45] Son SR, Sarkar SK, Nguyen-Thuy BL, et al. Platelet-rich plasma encapsulation in hyaluronic acid/gelatin-BCP hydrogel for growth factor delivery in BCP sponge scaffold for bone regeneration. J Biomater Appl.2015;29(7):988-1002.[46] Galli C, Parisi L, Piergianni M, Smerieri A, Passeri G, Guizzardi S, et al. Improved scaffold biocompatibility through anti-Fibronectin aptamer functionalization. Acta Biomater. 2016;42:147-156.[47] Galli C, Piergianni M, Piemontese M, et al. Periostin improves cell adhesion to implantable biomaterials and osteoblastic differentiation on implant titanium surfaces in a topography-dependent fashion. J Biomed Mater Res A. 2014;102(11):3855-3861.[48] Sauerova P, Pilgrova T, Pekar M, et al. Hyaluronic acid in complexes with surfactants: The efficient tool for reduction of the cytotoxic effect of surfactants on human cell types. Int J Biol Macromol. 2017;103: 1276-1284.[49] Bang S, Das D, Yu J, et al. Evaluation of MC3T3 cells proliferation and drug release study from sodium hyaluronate-1,4-butanediol diglycidyl ether patterned gel. Nanomaterials (Basel). 2017;7(10): E328. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||