Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (18): 2933-2938.doi: 10.3969/j.issn.2095-4344.0855
Previous Articles Next Articles
Zoledronic acid promotes dental implant osseointegration in osteoporosis
Huang Xiao-lin, Yao Chao, Cheng Yu-ting, Li Fang, Wang Yong, Liao Jian
- School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2018-03-22Online:2018-06-28Published:2018-06-28 -
Contact:Liao Jian, M.D., Associate professor, Associate chief physician, Master’s supervisor, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Huang Xiao-lin, Master candidate, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 8166040135; the Science and Technology Foundation of Guizhou Province, No. [2016]1124
CLC Number:
Cite this article
Huang Xiao-lin, Yao Chao, Cheng Yu-ting, Li Fang, Wang Yong, Liao Jian. Zoledronic acid promotes dental implant osseointegration in osteoporosis[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(18): 2933-2938.
share this article
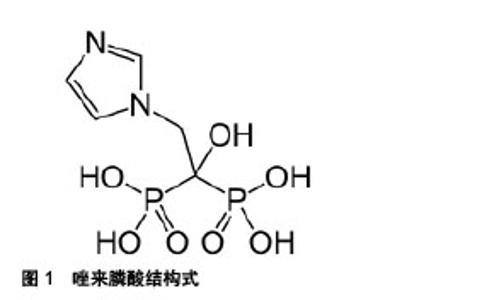
2.1.1 唑来膦酸对破骨细胞的影响 唑来膦酸通过复杂的机制抑制骨吸收,而主要作用机制旨在细胞水平[6]。作为一类强有力的破骨细胞抑制剂,唑来膦酸主要作用于破骨细胞,它的促成骨效应主要归因于它能抑制破骨细胞的生成和骨吸收功能[9]。唑来膦酸咪唑侧链上含2个氮原子(图1),与骨质中的羟磷灰石有高度亲和力[10],能优先被转运到暴露的骨矿化物部位,牢固地吸附在骨小梁表面,与骨骼结合并沉积于骨组织中,在局部骨表面维持较高浓度,直接干扰破骨细胞在骨面附着,干扰破骨细胞从骨基质接受骨吸收信号,抑制破骨细胞对骨小梁的溶解和破坏,增加骨密度;其也能被正在侵蚀骨组织的破骨细胞摄取,通过吞噬作用随骨基质进入细胞内,改变破骨细胞的超微结构,抑制其更新,降低生存期;还能通过阻断甲羟戊酸等途径,抑制法尼基焦磷酸合成酶,阻碍合成破骨细胞结构的相关蛋白如小GTPase蛋白的异戊二烯化,使细胞丧失褶皱缘,扰乱肌动蛋白换的形成并阻碍质子泵分泌H+的功能,促进细胞凋亡[11-12]。"

另有证据证明,唑来膦酸抑制破骨前体细胞分化为破骨细胞,激活破骨细胞抑制因子,减少骨组织表面破骨细胞的活动,下调基质金属蛋白酶,抑制骨转换[13],并且Abe等[14]的研究发现,破骨前体细胞暴露于唑来膦酸的反应和成熟破骨细胞相似。此外,破骨细胞的黏附性是发挥骨吸收的基本条件,也是细胞迁移及发挥功能的调节因素,对其发挥骨趋化作用有着重要影响。破骨前体细胞分化为成熟破骨细胞前应该先迁移至骨吸收靶点,而有课题组通过破骨前体细胞的体外研究发现,一定浓度的唑来膦酸可抑制破骨细胞黏附性和迁移性,使附壁破骨细胞数目减少,主动穿过小孔发生迁移活动的破骨细胞数目也明显减少[15]。他们还发现在最低有效浓度 1×10-6 mol/L下,唑来膦酸抑制破骨细胞的能力已明显增强,而更高浓度唑来膦酸对破骨细胞抑制作用的增强已趋于平缓,这就打破了很多研究者认为它剂量越大,对破骨细胞的抑制作用越强的理论[16-17],也解释了为什么临床应用大剂量唑来膦酸反而没有达到预期疗效。然而,唑来膦酸对破骨细胞的具体调节机制尚存在争论[18]。 2.1.2 唑来膦酸对成骨细胞的影响 唑来膦酸作用于破骨细胞调节的骨损伤疾病中,其抗吸收作用却和它对成骨细胞的影响密不可分,甚至有证据表明,双膦酸盐类药物很大一部分是通过调控成骨细胞来发挥抗吸收功能[19]。有研究表明唑来膦酸可能通过对成骨细胞的间接作用来抑制骨吸收,于此同时,还可刺激成骨前体细胞的形成,影响成骨细胞功能,如调控成骨细胞增殖、分化、细胞外基质蛋白的合成[20],具有潜在的促进早期成骨能力[21]。此外,成骨细胞分泌核转录因子NF-κB受体激动剂配体、巨噬细胞集落刺激因子和骨保护素等特异性分子,而唑来膦酸能调控成骨细胞分泌和表达相关细胞因子和生长因子[22-24]。 Greiner等[25]体外研究唑来膦酸对人成骨样细胞的影响,集中于研究介导破骨细胞分化的合成因子核转录因子 NF-κB受体激动剂配体、骨保护素的表达。他们将不同浓度唑来膦酸涂层于聚外消旋乳酸种植体上,对照组是未涂层的种植体、聚外消旋乳酸涂层的种植体和单纯不同浓度的唑来膦酸。原代人类成骨细胞培养144 h后检测前胶原Ⅰ型合成物、可溶性核转录因子NF-κB受体激动剂配体、骨保护素的表达。研究结果显示,涂层于种植体的唑来膦酸浓度达到50 μmol/L时前胶原Ⅰ型合成物含量最高,10 μmol/L唑来膦酸涂层于种植体则骨保护素表达明显上调,而不同浓度唑来膦酸涂层于种植体时,可溶性核转录因子NF-κB受体激动剂配体表达都下调。总之,暴露于特定浓度唑来膦酸的种植体有利于成骨细胞的分泌和蛋白合成,对其增殖无影响。可溶性核转录因子NF-κB受体激动剂配体、骨保护素的产量改变可能抑制破骨吸收,其局部抗吸收效应可能在种植体与周围骨结合中有明显意义。 2.1.3 唑来膦酸对成骨细胞、破骨细胞共培养的影响 近年来,学者们已证实局部应用唑来膦酸能够安全、有效的直接作用于植入物周围的破骨细胞和成骨细胞[26],有效促进植入体周围新骨形成,表明唑来膦酸的抗吸收活动和其对破骨细胞和成骨细胞的影响有关,但它对破骨细胞、成骨细胞单独培养的效应在二者共培养中是否重现,是否两种细胞共同作用影响种植体与周围骨结合也是个值得探讨的问题。 Greiner等[27]认为原代成骨细胞与破骨样细胞共培养,可能有助于深入了解双膦酸盐类药物对这些细胞及细胞与细胞直接相互作用的影响,他们的另一项实验通过成骨细胞、破骨细胞体外共培养,将负载不同浓度唑来膦酸的聚外消旋乳酸涂层种植体置于共培养的培养板中,检测细胞数、Ⅰ型胶原合成、骨保护素分泌、可溶性核转录因子NF-κB受体激动剂配体的表达、TRAP+细胞数及骨吸收陷窝数目,研究唑来膦酸对成骨细胞、破骨细胞体外共培养的影响。结果显示,负载于聚外消旋乳酸涂层种植体的唑来膦酸达到50 μmol/L时,对共培养的成骨细胞和破骨细胞活力没有影响,其中 10 μmol/L 浓度的唑来膦酸具有最大的促进骨保护素、Ⅰ型胶原合成作用,表明暴露于特定浓度唑来膦酸的种植体有利于成骨细胞分化和蛋白合成;成骨细胞数目以剂量依赖性方式减少,但其骨吸收活动和可溶性核转录因子 NF-κB受体激动剂配体的分泌却无明显降低,此研究可能对减少种植体松动有很大作用。 2.2 单独应用唑来膦酸对种植体骨结合的影响 全身应用唑来膦酸对种植体骨结合具有积极作用[28]。Dikicier等[29]通过动物实验已证实在雌激素缺乏鼠中,全身应用唑来膦酸可提高种植体与周围骨结合。众所周知,长期应用糖皮质激素会导致骨质疏松,巴西学者Carvas等[30]就应用糖皮质激素建立骨质疏松兔模型,发现糖皮质激素的使用会诱导骨损失,不利于钛种植体的骨结合状态;而采用皮下单剂量注射唑来膦酸的全身应用对骨质丧失的钛种植体骨结合起到增强作用,可扭转糖皮质激素对钛种植体骨结合的不利影响。因此在临床上,唑来膦酸可改善长期使用糖皮质激素患者的骨损失状态,获得种植义齿修复治疗的机会,提高生活质量。为了确定以上唑来膦酸的效应在大动物身上是否同样产生,Huja等[31]在两三岁雄犬身上进行实验,将唑来膦酸静脉注射后,再将2枚微型植体植入单侧上下颌骨,体内连续荧光骨标记,结果示种植体支持骨中以增强骨转化为特征的愈合反应减缓,但未完全消除,而种植体支持骨的骨改建明显增加。Dundar等[8]试图评价局部和全身应用唑来膦酸对两种不同表面处理钛种植体(钛表面陶瓷粉浆喷砂处理Tio-CB和喷砂酸蚀处理SLA)骨结合的影响。他们将12只新西兰白鼠分为6组,即无唑来膦酸处理的对照组:TiO-CB组和SLA组,含2 mg唑来膦酸的1 mL生理盐水注射入植窝内的局部处理组:TiO-CB-LZA组和SLA-LZA组,以及0.1 mg/kg唑来膦酸在种植体植入期间单次注射的全身用药组:TiO-CB-SZA组和SLA-SZA组,一段时间后,记录种植体和骨接触长度与总的种植体表面长度比值作为骨结合率,最终发现在他们用到的这两种表面处理方式中,局部应用唑来膦酸在增加骨结合率上相比对照组有更好的效果,而全身应用又比局部应用效果更佳。 在一定时间内唑来膦酸的扩散有助于维持迷你植体稳定性,因为种植初期,植体周围有大量骨小梁。Ying等[34]进一步通过实验推测这种效应很可能部分是由骨髓中颌骨坏死诱导的糖基化末端产物过生产受抑制所致。Miettinen等[35]也通过局部注射唑来膦酸研究其对种植体周围骨组织和骨丧失诱导的膜内骨再生刺激的影响,同样证实了上述现象,发现局部应用唑来膦酸4周后种植体周围骨量相比对照组增加。因此唑来膦酸可通过抑制种植体植入初期骨吸收抑制损伤诱导的髓内种植体周围骨组织丧失,增加种植体初期稳定性。Friedl等[36]研究单独应用4 mg唑来膦酸浸泡处理是否可预防股骨头坏死患者的种植体松动,对50例进行髋关节成形术患者种植时分别进行唑来膦酸、盐缓冲液处理,手术前后进行拍片、骨转化生化参数及Harris髋等级记录,结果表明单独应用唑来膦酸浸泡有希望提高种植体初期稳定性。由于上下颌骨可行性限制,多数研究者在解剖上可行的股骨上成功检测出骨结合状况,而Blazsek等[37]创建了一种独特的多功能体内实验性种植模型,来研究长期唑来膦酸处理下骨新生状况,该模型便于将唑来膦酸的全身和或局部应用效应进行比较。他们具体的操作方法是将表面多孔的钛种植体植入大鼠松软的尾椎骨,创造了一种促使钛种植体与骨结合的组织环境,并全身应用唑来膦酸,最后在不同条件下用生物物理方法检测反映种植体骨结合状况的量化骨新生刺激效应;结果发现相比于无唑来膦酸处理的对照组,唑来膦酸处理下骨结合增长了36%,并且新生骨很稳固;在骨再生的第6周新骨量增长了3.1倍,他们通过此项研究肯定了唑来膦酸在恢复骨损失和促进骨结合上发挥的积极作用。 2.3 唑来膦酸与其他药物联合应用对种植体骨结合的影响 许多研究表明唑来膦酸可抑制种植体周围骨吸收,最近几年的研究证实其可促进种植体周围骨形成,而唑来膦酸与其他药物联合应用时效果更佳,适应于不同身体状况患者的需求。Gao等[38]研究发现,局部应用唑来膦酸或碱性成纤维细胞生长因子(bFGF)都可提高骨小梁厚度,提高卵巢切除鼠的种植体稳定性;而二者联合治疗时唑来膦酸抑制骨吸收,碱性成纤维细胞生长因子刺激骨形成,对骨质疏松动物模型的种植体周围骨形成有明显改善。Li等[39]则在卵巢切除鼠胫骨中植入种植体,检测甲状旁腺激素和唑来膦酸单独或联合用药对骨质疏松骨愈合的影响。结果显示,甲状旁腺激素和唑来膦酸在加强种植体稳定性上有叠加效应,另外表明甲状旁腺激素的组织代谢效应强大,而同时应用时,在种植体周围骨愈合期间该效应不会因为唑来膦酸的加入而减弱。 作为种植体基体的材料有多种,通过长期的科学研究和临床实践发现,单一材料往往很难满足复杂的生理环境需求;而唑来膦酸的局部应用也需要借助各种基体材料,如羟基磷灰石、β-磷酸三钙和纤维蛋白原矩阵等作为缓释介质,涂层中的这些物质可作为载体使唑来膦酸持续释放。将双膦酸盐类药物直接应用于金属植入体周围的研究较少,复合生物材料的研制和应用才是目前研究的热点。如果把唑来膦酸负载于其他不同基体材料上也看作一种联合处理的话,则这样的复合方式非常多,比如已有研究结果表明,羟基磷灰石和唑来膦酸联合对种植体周围骨有加强的效应[40]。Kettenberger等[41]也通过RAW264.7细胞体外实验证实了这点,发现唑来膦酸对鼠破骨前体细胞的抑制效应通过将其涂层于纳米粒羟基磷灰石上得到加强,此后他们又在鼠绝经后的骨质疏松模型中进行体内实验,制备透明质酸水凝胶,然后将复合唑来膦酸的纳米粒羟基磷灰石和单独的纳米粒羟基磷灰石都与凝胶混成一体,随后将该凝胶注入鼠腓骨骨节上植入微小植体前钻的螺纹洞内;结果发现小鼠体内的水凝胶竟然快速矿化形成颗粒,作为新骨形成的支架。结合于纳米粒羟基磷灰石中的唑来膦酸释放,进一步抑制矿化水凝胶的减少及种植体周围骨吸收,并且和非负载的唑来膦酸同样有效。透明质酸结合含唑来膦酸的纳米粒羟基磷灰石,由于其诱导快速矿化和抑制骨吸收的双重效应,可以说是一种多功能且有潜力的骨修复材料。Li等[42]创建一种负载唑来膦酸可降解的镁种植体,该种植体可在体外持续地释放镁降解产物和唑来膦酸。同样建立骨质疏松模型,在腓骨骨折处植入髓内种植体,发现相比于传统的不锈钢植体材料,镁种植体可通过促进骨痂形成,增强骨质疏松型骨折修复。而唑来膦酸和镁联合作用可发挥二者的协同效应,进一步增加骨形成率和骨痂大小,明显提升骨质并抑制破骨细胞和骨改建。然而,少数研究认为联合用药并不能使唑来膦酸起到协同作用,如de Oliveira等[7]研究唑来膦酸和地塞米松联合处理对植入动物模型的钛种植体骨结合的影响中发现,单独唑来膦酸组、唑来膦酸联合地塞米松组和注射生理盐水组在种植体骨结合中无统计学差异;并且与注射生理盐水组相比,唑来膦酸联合或不联合地塞米松处理在种植体植入14-28 d后的骨改建活动都明显减少,因此唑来膦酸不干涉种植体骨结合。 2.4 唑来膦酸应用的一种严重并发症——BRONJ 2003年报道了不常见却严重的双膦酸盐治疗并发症BRONJ,从此学者们对该并发症的研究如火如荼地进行,双膦酸盐治疗则开始成为牙种植的的一个限制或排除因素。学者们主要通过回顾性研究和Meta分析来评估其风险状况,大多数研究者发现双膦酸盐可导致颌骨坏死(代表药物及抗骨溶解效力如表1)[43-45],认为应用双膦酸盐(包括唑来膦酸)之前、期间或之后禁止种植,也有部分作者展现骨质疏松并静注双膦酸盐患者经BRONJ相邻区域外科种植成功的案例,说明双膦酸盐治疗患者进行牙种植可获得较好的早期骨结合,不会使种植成功率下降[46-48];或者BRONJ愈合后,种植修复是可行的。甚至有研究者认为骨质疏松不会导致种植失败,口服双膦酸盐和BRONJ无关[49],提示双膦酸盐治疗可作为一种安全种植的治疗模式。长期应用双膦酸盐可否进行牙种植一直是个争论的问题,其实,是否可植入种植体取决于患者的个人风险状态,风险评估必须建立在个体身上。至少在现有证据中,接受单剂量静脉注射唑来膦酸治疗骨质疏松不是牙种植的绝对禁忌证[50],并且可充分利用局部应用该药比全身应用更安全可靠这点优势,避免此并发症。"

| [1] Dursun CK,Dursun E,Eratalay K,et al.Effect of Porous Titanium Granules on Bone Regeneration and Primary Stability in Maxillary Sinus: A Human Clinical, Histomorphometric, and Microcomputed Tomography Analyses.J Craniofac Surg.2016;27(2):391-397.[2] Dursun E,Keceli HG,Uysa Sl,et al.Management of Limited Vertical Bone Height in the Posterior Mandible: Short Dental Implants Versus Nerve Lateralization With Standard Length Implants.J Craniofac Surg. 2016;27(3):578-585.[3] 闫怡轩.种植体表面生物活性涂层的研究现状及进展[J].全科口腔医学杂志(电子版),2017,4(12):10-12.[4] Ma T,Ge XY,Hao KY,et al.Simple 3,4-Dihydroxy-L-Phenylalanine Surface Modification Enhances Titanium Implant Osseointegration in Ovariectomized Rats.Sci Rep.2017;7(1):17849.[5] 邢帅,高延征,高坤,等.双膦酸盐类药物治疗原发性骨质疏松症研究进展[J].风湿病与关节炎,2016, 5(3):70-73.[6] Marin-Fernandez AB,Garcia Medina B,Aguilar-Salvatierra A,et al.Jaw osteonecrosis management around a dental implant inserted 2 years before starting treatment with zoledronic acid.J Clin Exp Dent. 2015; 7(3):e444-446.[7] de Oliveira MA,Asahi DA,Silveira CA,et al.The effects of zoledronic acid and dexamethasone on osseointegration of endosseous implants: histological and histomorphometrical evaluation in rats. Clin Oral Implants Res.2015;26(4):e17-21.[8] Dundar S,Yaman F,Gecor O,et al.Effects of Local and Systemic Zoledronic Acid Application on Titanium Implant Osseointegration: An Experimental Study Conducted on Two Surface Types. J Craniofac Surg.2017;28(4):935-938.[9] 阴露佳,林珏杉,温黎明,等.唑来膦酸对破骨细胞分化中钙调蛋白、钙调磷酸酶表达的影响[J].南京医科大学学报(自然科学版), 2016,61(2): 171-175.[10] Zhu M,Zhu Y,Ni B,et al.Mesoporous silica nanoparticles/hydroxyapatite composite coated implants to locally inhibit osteoclastic activity.ACS Appl Mater Interfaces.2014;6(8):5456-5466.[11] Tai TW,Chen CY,Su FC,et al.Reactive oxygen species are required for zoledronic acid-induced apoptosis in osteoclast precursors and mature osteoclast-like cells.Sci Rep. 2017;7:44245.[12] 庞雪,刘英,曹积弘,等.唑来膦酸治疗绝经后骨质疏松症的一过性反应的观察与护理[J].中国骨质疏松杂志, 2013,19(11): 1187-1189.[13] Ardine M,Generali D,Donadio M,et al.Could the long-term persistence of low serum calcium levels and high serum parathyroid hormone levels during bisphosphonate treatment predispose metastatic breast cancer patients to undergo osteonecrosis of the jaw? Ann Oncol. 2006; 17(8):1336-1337.[14] Abe K,Yoshimura Y,Deyama Y,et al.Effects of bisphosphonates on osteoclastogenesis in RAW264.7 cells.Int J Mol Med. 2012;29(6): 1007-1015.[15] 李鹏飞,杨虎,贾楠,等.基于唑来膦酸对破骨细胞抑制特征的实验研究[J].生物医学工程学杂志,2017,34(1): 78-82.[16] Murphy CM,Schindeler A,Gleeson JP,et al.A collagen-hydroxyapatite scaffold allows for binding and co-delivery of recombinant bone morphogenetic proteins and bisphosphonates.Acta Biomater. 2014; 10(5):2250-2258.[17] Dedes PG,Kanakis I,Gialeli C,et al.Preclinical evaluation of zoledronate using an in vitro mimetic cellular model for breast cancer metastatic bone disease.Biochim Biophys Acta. 2013;1830(6): 3625-3634.[18] 吴骁伟,李少晗,曹文娟.唑来膦酸影响单个核细胞向破骨细胞分化及RANK表达的研究[J].中华全科医学, 2016,14(10):1644-1646.[19] Maruotti N,Corrado A,Neve A,et al.Bisphosphonates: effects on osteoblast.Eur J Clin Pharmacol.2012;68(7):1013-1018.[20] Reinholz GG,Getz B,Pederson L,et al.Bisphosphonates directly regulate cell proliferation, differentiation, and gene expression in human osteoblasts.Cancer Res. 2000;60(21):6001-6007.[21] Giuliani N,Pedrazzoni M,Negri G,et al.Bisphosphonates stimulate formation of osteoblast precursors and mineralized nodules in murine and human bone marrow cultures in vitro and promote early osteoblastogenesis in young and aged mice in vivo.Bone. 1998; 22(5):455-461.[22] Furlaneto FA,Nunes NL,Oliveira Filho IL,et al.Effects of locally administered tiludronic acid on experimental periodontitis in rats.J Periodontol.2014;85(9):1291-1301.[23] Dalle Carbonare L,Mottes M,Malerba G,et al.Enhanced Osteogenic Differentiation in Zoledronate-Treated Osteoporotic Patients.Int J Mol Sci.2017;18(6):1261.[24] Soto SA,Barbará AC.Bisphosphonates: Pharmacology and Clinical Approach to Their Use in Equine Osteoarticular Diseases.J Equine Vet Sci.2014;34(6):727-737.[25] Greiner S,Kadow-Romacker A,Lubberstedt M,et al.The effect of zoledronic acid incorporated in a poly(D,L-lactide) implant coating on osteoblasts in vitro.J Biomed Mater Res A. 2007;80(4):769-775.[26] Gao Y,Zou S,Liu X,et al.The effect of surface immobilized bisphosphonates on the fixation of hydroxyapatite-coated titanium implants in ovariectomized rats. Biomaterials. 2009;30(9):1790-1796.[27] Greiner S,Kadow-Romacker A,Schmidmaier G,et al.Cocultures of osteoblasts and osteoclasts are influenced by local application of zoledronic acid incorporated in a poly(D,L-lactide) implant coating. J Biomed Mater Res A. 2009;91(1):288-295.[28] Ayan M,Dolanmaz D,Mihmanli A,et al.The effect of systemically administrated zoledronic acid on the osseointegration of dental implants.Oral Dis. 2012;18(8):802-808.[29] Dikicier E,Karacayli U,Dikicier S,et al.Effect of systemic administered zoledronic acid on osseointegration of a titanium implant in ovariectomized rats.J Craniomaxillofac Surg. 2014;42(7): 1106-1111.[30] Carvas JS,Pereira RM,Caparbo VF,et al.A single dose of zoledronic acid reverses the deleterious effects of glucocorticoids on titanium implant osseointegration. Osteoporos Int.2010; 21(10):1723-1729.[31] Huja SS,Kaya B,Mo X, et al.Effect of zoledronic acid on bone healing subsequent to mini-implant insertion.Angle Orthod. 2011;81(3): 363-369.[32] Stadlinger B,Korn P,Todtmann N,et al.Osseointegration of biochemically modified implants in an osteoporosis rodent model.Eur Cell Mater.2013;25:326-340;discussion 339-340.[33] Cuairan C,Campbell PM,Kontogiorgos E,et al.Local application of zoledronate enhances miniscrew implant stability in dogs.Am J Orthod Dentofacial Orthop. 2014;145(6):737-749.[34] Ying G,Bo L,Yanjun J,et al.Effect of a local, one time, low-dose injection of zoledronic acid on titanium implant osseointegration in ovariectomized rats.Arch Med Sci. 2016;12(5):941-949.[35] Miettinen SS,Jaatinen J,Pelttari A,et al.Effect of locally administered zoledronic acid on injury-induced intramembranous bone regeneration and osseointegration of a titanium implant in rats. J Orthop Sci. 2009; 14(4):431-436.[36] Friedl G,Radl R,Stihsen C,et al.The effect of a single infusion of zoledronic acid on early implant migration in total hip arthroplasty. A randomized, double-blind, controlled trial.J Bone Joint Surg Am. 2009; 91(2):274-281.[37] Blazsek J,Dobo Nagy C,Blazsek I,et al.Aminobisphosphonate stimulates bone regeneration and enforces consolidation of titanium implant into a new rat caudal vertebrae model.Pathol Oncol Res. 2009; 15(4):567-577.[38] Gao Y,Luo E,Hu J,et al.Effect of combined local treatment with zoledronic acid and basic fibroblast growth factor on implant fixation in ovariectomized rats.Bone. 2009;44(2):225-232.[39] Li YF,Li XD,Bao CY,et al.Promotion of peri-implant bone healing by systemically administered parathyroid hormone (1-34) and zoledronic acid adsorbed onto the implant surface. Osteoporos Int. 2013;24(3): 1063-1071.[40] Peter B,Pioletti DP,Laib S,et al.Calcium phosphate drug delivery system: influence of local zoledronate release on bone implant osteointegration.Bone.2005;36(1):52-60.[41] Kettenberger U,Luginbuehl V,Procter P,et al.In vitro and in vivo investigation of bisphosphonate-loaded hydroxyapatite particles for peri-implant bone augmentation. J Tissue Eng Regen Med. 2017;11(7): 1974-1985.[42] Li G,Zhang L,Wang L,et al.Dual modulation of bone formation and resorption with zoledronic acid-loaded biodegradable magnesium alloy implants improves osteoporotic fracture healing: An in vitro and in vivo study.Acta Biomater. 2018;65:486-500.[43] Rugani P,Kirnbauer B,Acham S,et al.Implant Placement Adjacent to Successfully Treated Bisphosphonate-Related Osteonecrosis of the Jaw(BRONJ).J Oral Implantol. 2015;41:377-381.[44] Marínfernández AB,García MB,Aguilarsalvatierra A,et al.Jaw osteonecrosis management around a dental implant inserted 2 years before starting treatment with zoledronic acid.J Clin Exp Dent. 2015; 7(3):e444.[45] Miniello TG,Araújo JP,Lopes RN,et al.Osteonecrosis related to once-yearly zoledronic acid treatment in an osteoporotic patient after dental implant.Braz Dent J.2015;26(1):86-88.[46] Ata-Ali J, Ata-Ali F,Penarrocha-Oltra D,et al.What is the impact of bisphosphonate therapy upon dental implant survival? A systematic review and meta-analysis.Clin Oral Implants Res.2016; 27(2):e38-46.[47] Kim JW,Baik J,Jeon JH.Dental implant treatment after healing of bisphosphonate-related osteonecrosis of the jaw (BRONJ) in the same region: a case report.J Korean Assoc Oral Maxillofac Surg. 2016;42(3): 157-161.[48] Siebert T,Jurkovic R,Statelova D,et al.Immediate Implant Placement in a Patient With Osteoporosis Undergoing Bisphosphonate Therapy: 1-Year Preliminary Prospective Study.J Oral Implantol. 2015;41: 360-365.[49] Al-Sabbagh M,Robinson FG,Romanos G,et al.Osteoporosis and bisphosphonate-related osteonecrosis in a dental school implant patient population.Implant Dent.2015;24(3):328-332.[50] Mattheos N,Caldwell P,Petcu EB,et al.Dental implant placement with bone augmentation in a patient who received intravenous bisphosphonate treatment for osteoporosis.J Can Dent Assoc. 2013; 79:d2. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Yang Caihui, Liu Qicheng, Dong Ming, Wang Lina, Zuo Meina, Lu Ying, Niu Weidong. Serine/threonine protein kinases can promote bone destruction in mouse models of chronic periapical periodontitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3654-3659. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||