Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (26): 4247-4252.doi: 10.3969/j.issn.2095-4344.0733
Previous Articles Next Articles
Application of controlled-release nanoparticles in chemotherapy of bone tumors
Ma Hong-yun1, 2, Zhuang Xin-ming1, Kao Yan-bin1, Wang Zhen-yu1, Qu Zhi-gang1, Song Qing-xu1, Xu Wei-guo2, Liu Yi1
- 1Department of Spine, First Hospital of Jilin University, Changchun 130021, Jilin Province, China; 2Key Laboratory of Polymer Ecomaterials, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Changchun 130022, Jilin Province, China
-
Received:2018-02-09 -
Contact:Liu Yi, Professor, Doctoral supervisor, Department of Spine, First Hospital of Jilin University, Changchun 130021, Jilin Province, China -
About author:Ma Hong-yun, Master candidate, Department of Spine, First Hospital of Jilin University, Changchun 130021, Jilin Province, China; Key Laboratory of Polymer Ecomaterials, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Changchun 130022, Jilin Province, China -
Supported by:the National Natural Science Foundation of China, No. 51603204
CLC Number:
Cite this article
Ma Hong-yun, Zhuang Xin-ming, Kao Yan-bin, Wang Zhen-yu, Qu Zhi-gang, Song Qing-xu, Xu Wei-guo, Liu Yi. Application of controlled-release nanoparticles in chemotherapy of bone tumors[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(26): 4247-4252.
share this article
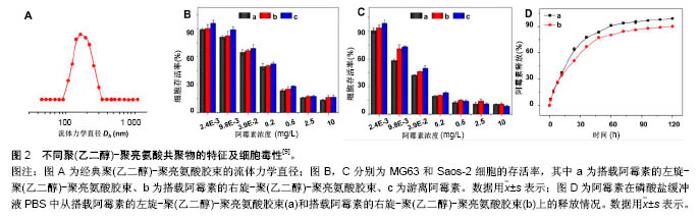
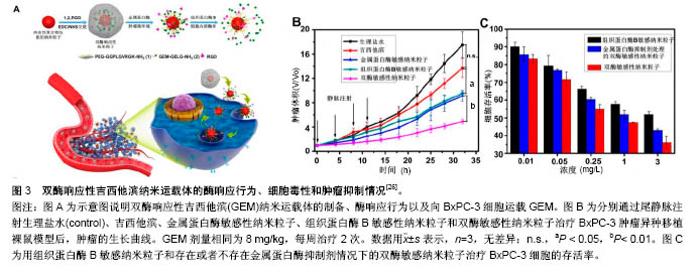
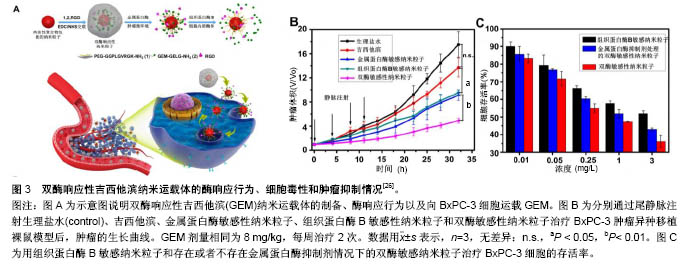
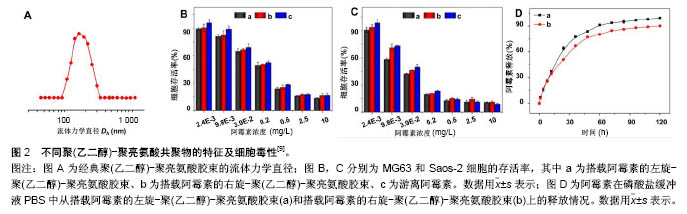
2.1 被动靶向纳米粒子应用于骨肿瘤化疗 被动靶向制剂主要通过肿瘤组织自身的生理特性,实现药物在肿瘤组织中被动、靶向聚集。一方面,由于肿瘤组织内皮细胞间隙增大、外膜细胞缺陷,与正常组织相比具有较高的通透性,另一方面,由于肿瘤组织缺乏淋巴回流,所以纳米粒子渗出液可以选择性的聚集到肿瘤组织中,增加局部药物浓度。这种实体瘤的高通透性和滞留效应,使得某些尺寸的纳米药物更趋向于聚集在肿瘤组织中,被称为高渗透长滞留效应[7]。正是这种高渗透长滞留效应,使纳米药物具有被动靶向性。被动靶向制剂根据作用机制的不同可分为非响应性纳米粒子和刺激响应性纳米粒子。 2.1.1 非响应性纳米粒子 纳米抗肿瘤药物载体:阿霉素、顺铂、紫杉醇、甲氨蝶呤、异环磷酰胺等是骨肿瘤治疗中最常见的化疗药物。Ding等[8-9]的小组通过亮氨酸氮羧基环内酸酐的开环聚合合成聚(乙二醇)-聚亮氨酸二嵌段或三嵌段共聚物。阿霉素通过纳米沉淀技术装入胶束。共聚物可以自发地在pH 7.4的PBS中自组装成流体力学直径为(179±7.8) nm的胶束(图2A)。装载阿霉素的胶束可以通过内吞作用被有效地吸收,并且可以实现阿霉素药物的有效释放(图2D),在MG63和Saos-2两种人骨肉瘤细胞系中展示了良好的抗骨肉瘤效率(图2B,C)。Ding等[9]认为基于三嵌段介导的手性多肽胶束PEG-PLUC共聚物,在减少的不良反应的同时,具有增强的化疗功效的作用,在骨肉瘤的治疗中显示了巨大的潜力。 此外,纳米技术在通过药物改性提高抗肿瘤效率方面发挥了重要作用。紫杉醇由于水溶性差、渗透性低,常常引起严重的过敏反应,因此基于紫杉醇的治疗方案往往难以完全发挥效力,临床应用常受到限制。纳米粒子和胶束作为紫杉醇运载体,可显著提高其水溶性。Gustafson等[10]设计了一种聚乙二醇化聚磷酸酯胶束(Micelles)和壳交联的克内德尔纳米胶束(Shell cross-linked knedel-like micelles,SCKs)用来交付紫杉醇。SCKs具有较高的载药浓度10%,这使得水中悬浮紫杉醇浓度高于4.8 g/L,相比于游离紫杉醇水溶环境 下< 2.0 mg/L的浓度,显著提高了疏水药物紫杉醇的水溶性。与此同时,研究者利用紫杉醇搭载胶束和紫杉醇®的药物制剂研究其对2个骨肉瘤细胞系(CCH-OS-O和SJSA)的细胞毒性,结果发现搭载紫杉醇的SCKs具有较高的细胞毒性,这主要归因于其缓慢的药物释放。SCKs在肺中滞留的时间接近胶束滞留时间的2倍,分别约为8 d和4 d,证明交联能够降低纳米粒子从肺等组织中释放的速度。因此,纳米抗肿瘤药物载体通过肺部直接给药,持续缓慢释放化疗药物,在骨肉瘤肺转移的治疗上有着巨大潜力。 光动力治疗纳米粒子:光动力疗法是用光敏药物和激光活化治疗肿瘤的一种新方法。光动力疗法的主要机制在于通过特定波长的光照射,使选择性聚集在肿瘤部位的光敏药物将能量传递给周围的氧,生成高活性的单态氧。单态氧能与附近的生物大分子发生氧化反应,产生细胞毒性进而杀伤肿瘤细胞[11-12]。 在一个研究中,研究者通过绿色合成途径制备了小尺寸、具有良好光热转换效率的三叶草样铂纳米粒子[13]。这种纳米粒子对正常细胞毒性很小,但在近红外光照射下可以有效杀死肿瘤细胞。在体内PC9骨转移模型中,这些纳米粒子可以有效抑制肿瘤生长,防止骨质溶解破坏。这项研究为骨转移性肿瘤的治疗提供了新的思路和技术支持,拓宽了骨肿瘤治疗方案的选择。此外,光动力疗法在骨肉瘤的治疗中也展示出了良好的治疗效果。Sotgiu等[14]设计了一种三组分系统,包括间充质干细胞、光敏剂内消旋-四(4-磺酰苯基)卟啉(Meso-tetrakis (4-sulfonato-phenyl) porphyrin,TPPS)和载体核壳荧光聚(甲基丙烯酸甲酯)纳米粒子(Fluorescent core-shell PMMA nanoparticles,FNP)。TPPS@FNP在45 mg/L浓度时可以被间充质干细胞有效吸收,进而形成三组分系统TPPS@FNPs-MSC。研究证实这种生物共载系统作为被动靶向交付策略可以有效诱导人类骨肉瘤细胞死亡。 2.1.2 刺激响应性纳米粒子 随着癌症生物学和癌症微环境研究的不断深入,人们发现肿瘤和正常组织在代谢、pH值、氧含量、血管等方面存在许多生理差异。基于这种生理差异,研究者设计了可以针对肿瘤微环境实现药物刺激响应性释放的多种聚合物纳米粒子[15-16]。对于骨肿瘤,这些刺激响应性纳米聚合物载体主要集中于pH-响应、酶响应和还原响应等方面。 pH-响应性纳米粒子:众所周知,正常组织(pH 7.4)和肿瘤组织(pH 6.5-7.2)的pH值有轻微的差异[17-19]。肿瘤的快速增长导致肿瘤局部缺乏营养和氧气,使得肿瘤部位酸性代谢物积累,引起pH值降低[20]。此外,细胞内某些组件(如内涵体和溶酶体)pH值可能会更低(pH 4.5-6.5)。因此,pH响应性纳米粒子抗肿瘤系统具有广泛应用前景。 异环磷酰胺是一种广谱、细胞周期非特异性抗肿瘤药物,不仅能显著改善骨肉瘤患者的无事件生存和总生存率,还能增加良性组织学反应率[21]。Chen等[22]设计、制备了一种搭载异环磷酰胺的pH响应性聚乳酸葡聚糖纳米粒子(PD/IFS),粒子尺寸为(124±3.45) nm,能够显著抑制MG63和SaOS-2骨肉瘤细胞生长。具体来说,它可以在水性介质中形成聚乳酸-葡聚糖自组装聚合物胶束,并且具有很好的聚合物分散指数,指数为0.124。其次,异环磷酰胺可以被有效地搭载到纳米颗粒中,这些负载药物的核壳纳米载体可以在pH 7.4环境中持续释放药物,并且在pH 5.0的条件下显著加速水解释放。体外MG63和Saos-2抗癌活性的分析证实了PD/IFS纳米颗粒与游离异环磷酰胺相比,具有更高抗肿瘤活性。这项研究表明纳米粒子包载抗肿瘤药物能够靶向针对肿瘤酸性环境发挥作用,有效增加治疗功效,有望成为骨肿瘤最具前景的治疗方法。 还原响应性系统:由于细胞内和细胞外谷胱甘肽含量存在明显差异,而肿瘤细胞内谷胱甘肽浓度又是正常细胞含量的7-10倍[23]。基于这一差异,还原响应性纳米颗粒载体被成功开发。Tomás's[24]研究小组设计开发了一种以胱氨酸作为交联剂、通过细乳液聚合法制备的还原响应性纳米凝胶(Alginate/cystamine,AG/Cys)。阿霉素可以通过强静电相互作用搭载到纳米凝胶上面。他们通过荧光显微镜观察阿霉素的细胞内吞过程,发现搭载阿霉素的纳米凝胶除了存在于细胞质中,还出现在CAL-72细胞(骨肉瘤细胞系)细胞核中。与游离阿霉素相比,他们发现搭载阿霉素的纳米凝胶在体外具有更高的抗肿瘤效率[24]。 酶响应性制剂:酶响应性纳米制剂主要依赖脂类或者短肽的裂解实现其功能[25]。由于在类似癌症等的病理条件下,某些特定的酶(如蛋白酶、磷脂酶、糖苷酶)表达会发生变化,因此研究者们利用这一特点来实现在特定生物靶点酶介导的药物控制释放[15]。Parak等[26]设计了一种双酶响应的纳米载体搭载吉西他滨用以治疗胰腺癌。胰腺癌晚期多会发生骨转移,恶性度极高,难以治愈。双酶响应性吉西他滨纳米载体主要将金属蛋白酶9可拆式聚乙二醇挂钩,组织蛋白酶B可裂解吉西他滨和靶向集团环状RGD(CycloRGD,精氨酸-甘氨酸-天冬氨酸序列(Arg-Gly-Asp,简称RGD序列))装配到硒化镉/硫化锌纳米粒子来实现(图3A)。聚乙二醇修饰的吉西他滨纳米载体,可以避免药物非特异性的相互作用,并且延长血液半衰期。当纳米粒子通过高渗透长滞留效应聚集到肿瘤时,肿瘤组织过表达的金属蛋白酶9会使聚乙二醇裂解、暴露RGD,促进细胞内吞。一旦纳米粒子内吞进入肿瘤细胞后,溶酶体中增高的组织蛋白酶B会进一步促进吉西他滨的释放。通过探究这种双酶响应的纳米载体的作用机制,他们发现该纳米载体无论是体内还是体外都具有很好的抗肿瘤效果(图3B,C)。 2.2 主动靶向制剂应用于骨肿瘤化疗 源于癌症治疗使药物最大程度聚集于患病组织并减小非靶向不良反应的迫切需要,癌症靶向治疗被认为是目前骨肿瘤治疗中最有发展前景的治疗方法之一。主动靶向纳米粒子可以选择性地结合细胞表面分子、受体、新生血管相关配体以及肿瘤组织细胞外基质,增加纳米粒子和细胞之间的亲和力,提高药物的内化,减小化疗药物的系统毒性[27-28]。 2.2.1 阿仑膦酸钠骨靶向 阿仑膦酸钠是一种氨基二膦酸盐骨代谢调节剂,与骨内羟磷灰石有强亲和力。近年来随着阿仑膦酸钠研究和应用的不断深入,大量研究发现并证明了阿仑膦酸钠的骨靶向作用,特别是其在聚合物纳米制药方面的应用展示了巨大潜力。Choi等[29]设计了一种阿仑膦酸钠共轭的纳米钻石(Alendronate- conjugated nanodiamonds, Alen-NDs),研究它们与骨矿物质羟基磷灰石的亲和力。通过不同细胞组的内吞实验,证明了Alen-NDs对成骨细胞的细胞特异性。此外,他们通过尾静脉注射Alen-NDs来研究其在小鼠体内的作用机制,发现Alen-NDs可以很好地积累到骨骼组织中,展现出对碱性磷酸酶良好的协同机制。 随着阿仑膦酸钠骨靶向机制研究的不断成熟,研究者们根据其靶向性,制备了一系列的抗骨肿瘤主动靶向纳米制剂。其中,较为典型的研究是Morton等[30]利用逐层装配技术制备了组织特异性药物载体,用于治疗原发性骨肉瘤。具体来说,就是通过阿仑膦酸侧链官能化,来表面修饰水改性聚丙烯酸载药纳米粒子聚合物。研究表明搭载阿霉素的纳米粒子可以有效地聚集在143B骨肉瘤异种移植小鼠模型肿瘤部位,减轻肿瘤负荷,延长小鼠的生存时间。更重要的是,该研究证明了这些功能化纳米粒子通过骨靶向治疗侵袭性转移癌(如乳腺癌和肺癌)的功效,因此研究者认为该类靶向纳米粒子非常有研究前途。 2.2.2 极迟抗原4(Very late antigen-4,VLA-4)靶向 多发性骨髓瘤作为一种常见的浆细胞源性恶性疾病,可原发于骨组织并引起一系列并发症,包括广泛的骨破坏、肾衰竭、贫血和高钾血症等,很难被治愈[31-32]。为了提高多发性骨髓瘤靶向治疗的功效,Pan和同事[33]提出了一个Sn2脂肪酶不稳定磷脂前体药物结合、接触易化、呈递释放的新概念。他们合成设计的药物可以靶向于b-HLHZip转录因子c-Myc(MYC是一种强大的原癌基因,可以激活和促进骨髓瘤的发展[34])。在Pan等[33]的研究中,索引化合物(10058-F4)被合成并改性为Sn2前药形式:c-Myc-inhibitor-1前体药物(MI1-PD)。MI1-PD在细胞毒性试验中,可以显著增加对人H929和U266和小鼠5TGM1骨髓瘤细胞的药物效力。他们通过尾静脉注射VLA-4靶向的MI1-PD纳米粒子治疗弥漫性骨髓瘤的原位小鼠模型,发现MI1-PD可显著降低肿瘤负荷,增加荷瘤小鼠存活率。在另外一项研究中,Ashley等[35]设计了VLA-4靶向的卡非佐米(Carfilzomib, CFZ)脂质体纳米颗粒(VLA-4 targeted liposomal carfilzomib nanoparticles,TNP [CFZ]),它可以有效靶向杀伤VLA-4表达的多发性骨髓瘤细胞。CFZ脂质体纳米颗粒(Non-targeted liposomal carfilzomib nanoparticles, NP [CFZ])主要通过被动负载技术,搭载CFZ进入脂质体来制备。与游离CFZ相比,NP [CFZ]和TNP [CFZ]在体外可以增加对骨髓瘤的细胞毒性、诱导凋亡,在体内则可以显着抑制骨髓瘤生长、减少全身毒性。这些研究表明,无论是第一代脂质体CFZ纳米颗粒,还是Sn2脂肪酶不稳定的磷脂前药与接触促进药物交付技术结合应用时,都可以有效治疗多发性骨髓瘤并改善患者预后。 2.2.3 其他 骨肿瘤细胞表面有各种各样的受体,主要包括血管内皮生长因子、胰岛素样生长因子、肝配蛋白A型受体2、CD44等[36-37]。尽管靶向特定受体可以使肿瘤组织中的药物分布大大提高,但是这些靶向受体并不单单过表达于靶点病变细胞,有些正常组织细胞也会表达这些受体,进而吸收纳米粒子引起细胞毒性。因此,Levine等[38]设计了一种双配体系统,进而确定双靶向受体,这可以显著提高纳米粒子对肿瘤组织的靶向选择性。虽然目前许多骨肿瘤特异的分子受体已经被确定,但应用靶向集团功能化的纳米粒子治疗骨肿瘤的研究依然处于初级阶段。利用主动靶向集团修饰纳米粒子治疗骨肿瘤非常有应用前景。 "
| [1] Mundy GR. Metastasis to bone: Causes, consequences and therapeutic opportunities. Nat Rev Cancer. 2002;2(8):584-593.[2] Kumar N, Gupta B. Global incidence of primary malignant bone tumors. Curr Orthop Pract. 2016;27(5):530-534.[3] Redondo A, Bague S, Bernabeu D, et al. Malignant bone tumors (other than Ewing's): clinical practice guidelines for diagnosis, treatment and follow-up by Spanish Group for Research on Sarcomas (GEIS). Cancer Chemother Pharmacol. 2017;80(6):1113-1131.[4] Saracino R, Luciano R, Battafarano G, et al. Nanoparticles-Based Treatment for Bone Metastasis. Curr Drug Targets. 2016;17(3): 303-310.[5] He X, Cao H, Wang H, et al. Inflammatory Monocytes Loading Protease-Sensitive Nanoparticles Enable Lung Metastasis Targeting and Intelligent Drug Release for Anti-Metastasis Therapy. Nano Lett. 2017;17(9):5546-5554.[6] Zhao K, Li D, Xu W, et al. Targeted hydroxyethyl starch prodrug for inhibiting the growth and metastasis of prostate cancer. Biomaterials. 2017;116:82-94.[7] Maeda H. Polymer therapeutics and the EPR effect. J Drug Target. 2017;25(9-10):781-785.[8] Li C, Xu W, Ding J, et al. Micellization of Antineoplastic Agent to Significantly Upregulate Efficacy and Security. Macromol Biosci. 2015; 15(3):328-341[9] Ding J, Li C, Zhang Y, et al. Chirality-mediated polypeptide micelles for regulated drug delivery. Acta Biomater. 2015;11:346-55[10] Zhang FW, Zhang SY, Pollack SF, et al. Improving Paclitaxel Delivery: In Vitro and In Vivo Characterization of PEGylated Polyphosphoester- Based Nanocarriers. J Am Chem Soc. 2015;137(5):2056-2066[11] Abrahamse H, Kruger CA, Kadanyo S, et al. Nanoparticles for Advanced Photodynamic Therapy of Cancer. Photomed Laser Surg. 2017;35(11):581-588.[12] Dang J, He H, Chen D, et al. Manipulating tumor hypoxia toward enhanced photodynamic therapy (PDT). Biomater Sci. 2017;5(8): 1500-1511.[13] Wang C, Cai X, Zhang J, et al. Trifolium-like Platinum Nanoparticle- Mediated Photothermal Therapy Inhibits Tumor Growth and Osteolysis in a Bone Metastasis Model. Small.2015;11(17):2080-2086.[14] Duchi S, Sotgiu G, Lucarelli E, et al. Mesenchymal stem cells as delivery vehicle of porphyrin loaded nanoparticles: Effective photoinduced in vitro killing of osteosarcoma. J Control Release.2013;168(2):225-237.[15] Mura S, Nicolas J, Couvreur P. Stimuli-responsive nanocarriers for drug delivery. Nat Mater. 2013;12(11):991-1003.[16] Li X, Zhao Y, Liu P, et al. Senegenin Inhibits Hypoxia/ Reoxygenation-Induced Neuronal Apoptosis by Upregulating RhoGDIα. Mol Neurobiol. 2014;52(3):1561-1571.[17] Chen J, Ding J, Zhang Y, et al. Polyion complex micelles with gradient pH-sensitivity for adjustable intracellular drug delivery. Polym Chem. 2015;6(3):397-405.[18] Xu W, Ding J, Xiao C, et al. Versatile preparation of intracellular-acidity-sensitive oxime-linked polysaccharide-doxorubicin conjugate for malignancy therapeutic. Biomaterials. 2015;54:72-86.[19] Sun D, Ding J, Xiao C, et al. Preclinical Evaluation of Antitumor Activity of Acid-Sensitive PEGylated Doxorubicin. ACS Appl Mater Interfaces. 2014;6(23):21202-21214.[20] Feron O. Pyruvate into lactate and back: From the Warburg effect to symbiotic energy fuel exchange in cancer cells. Radiother Oncol. 2009;92(3):329-333.[21] Fan XL, Cai GP, Zhu LL, et al. Efficacy and safety of ifosfamide-based chemotherapy for osteosarcoma: a meta-analysis. Drug Des Dev Ther. 2015;9:5925-5932.[22] Chen B, Yang JZ, Wang LF, et al. Ifosfamide-loaded poly (lactic-co- glycolic acid) PLGA-dextran polymeric nanoparticles to improve the antitumor efficacy in Osteosarcoma. BMC Cancer. 2015;15:752.[23] Aluri S,Janib SM, Mackay JA. Environmentally responsive peptides as anticancer drug carriers. Adv Drug Deliv Rev. 2009;61(11):940-52.[24] Maciel D, Figueira P, Xiao S, et al. Redox-responsive alginate nanogels with enhanced anticancer cytotoxicity. Biomacromolecules. 2013;14(9):3140-3146.[25] Fleige E, Quadir MA, Haag R. Stimuli-responsive polymeric nanocarriers for the controlled transport of active compounds: Concepts and applications. Adv Drug Deliv Rev. 2012;64(9):866-884.[26] Han HJ, Valdeperez D, Jin Q, et al. Dual Enzymatic Reaction-Assisted Gemcitabine Delivery Systems for Programmed Pancreatic Cancer Therapy. Acs Nano. 2017;11(2):1281-1291.[27] Mu CF, Shen JL, Liang J, et al. Targeted drug delivery for tumor therapy inside the bone marrow. Biomaterials. 2018;155:191-202.[28] Chae YK, Pan AP, Davis AA, et al. Path toward Precision Oncology: Review of Targeted Therapy Studies and Tools to Aid in Defining "Actionability" of a Molecular Lesion and Patient Management Support. Mol Cancer Ther. 2017;16(12):2645-2655.[29] Ryu TK, Kang RH, Jeong KY, et al. Bone-targeted delivery of nanodiamond-based drug carriers conjugated with alendronate for potential osteoporosis treatment. J Control Release. 2016;232: 152-160.[30] Morton SW, Shah NJ, Quadir MA, et al. Osteotropic therapy via targeted layer-by-layer nanoparticles. Adv Healthc Mater. 2014;3(6): 867-75.[31] Mitsiades CS, Mitsiades N, Munshi NC, et al. Focus on multiple myeloma. Cancer Cell. 2004;6(5):439-444.[32] Wu WC, Fernández-Seara M, Detre JA, et al. A theoretical and experimental investigation of the tagging efficiency of pseudocontinuous arterial spin labeling. Magn Reson Med. 2007; 58(5):1020-1027.[33] Pan D, Pham CTN, Weilbaecher KN, et al. Contact-facilitated drug delivery with Sn2 lipase labile prodrugs optimize targeted lipid nanoparticle drug delivery. Wires Nanomed Nanobi. 2016;8(1):85-106.[34] Kuehl WM. Mouse models can predict cancer therapy. Blood. 2012; 120(2):238-240.[35] Ashley JD, Stefanick JF, Schroeder VA, et al. Liposomal carfilzomib nanoparticles effectively target multiple myeloma cells and demonstrate enhanced efficacy in vivo. J Control Release. 2014;196:113-121.[36] Hassan SE, Bekarev M, Kim MY, et al. Cell surface receptor expression patterns in osteosarcoma. Cancer. 2012;118(3):740-749.[37] Van Driel M, Van Leeuwen J. Cancer and bone: A complex complex. Arch Biochem Biophys. 2014;561:159-166.[38] Levine RM, Kokkoli E. Dual-ligand alpha(5)beta(1) and alpha(6)beta(4) integrin targeting enhances gene delivery and selectivity to cancer cells. J Control Release. 2017;251:24-36.[39] Rejinold NS, Baby T, Chennazhi KP, et al. Multi Drug Loaded Thermo-Responsive Fibrinogen-graft-Poly(N-vinyl Caprolactam) Nanogels for Breast Cancer Drug Delivery. J Biomed Nanotechnol. 2015;11(3):392-402.[40] Kolter M, Ott M, Hauer C, et al. Nanotoxicity of poly(n-butylcyano- acrylate) nanoparticles at the blood–brain barrier, in human whole blood and in vivo. J Control Release. 2015;197:165-179.[41] Oh SS, Lee BF, Leibfarth FA, et al. Synthetic aptamer-polymer hybrid constructs for programmed drug delivery into specific target cells. J Am Chem Soc. 2014;136(42):15010-15015.[42] Kanapathipillai M, Brock A, Ingber DE. Nanoparticle targeting of anti-cancer drugs that alter intracellular signaling or influence the tumor microenvironment. Adv Drug Deliv Rev. 2014;79-80:107-118.[43] Wicki A, Witzigmann D, Balasubramanian V, et al. Nanomedicine in cancer therapy: Challenges, opportunities, and clinical applications. J Control Release. 2015;200:138-157.[44] Lorscheidt S, Lamprecht A. Safety assessment of nanoparticles for drug delivery by means of classic in vitro assays and beyond. Expert Opin Drug Deliv. 2016;13(11):1545-1558.[45] Yang Q, Parker CL, Mccallen JD, et al. Addressing challenges of heterogeneous tumor treatment through bispecific protein-mediated pretargeted drug delivery. J Control Release. 2015;220(Pt B):715-726.[46] Cheng Z, Al Zaki A, Hui JZ, et al. Multifunctional nanoparticles: cost versus benefit of adding targeting and imaging capabilities. Science. 2012;338(6109):903-910.[47] Li CY, Zhang YJ, Chen GC, et al. Engineered Multifunctional Nanomedicine for Simultaneous Stereotactic Chemotherapy and Inhibited Osteolysis in an Orthotopic Model of Bone Metastasis. Adv Mater. 2017;29(13).[48] Wang YT, Yang J, Liu HM, et al. Osteotropic peptide-mediated bone targeting for photothermal treatment of bone tumors. Biomaterials. 2017;114:97-105.[49] Tan YF, Chandrasekharan P, Maity D, et al. Multimodal tumor imaging by iron oxides and quantum dots formulated in poly (lactic acid)-D- alpha-tocopheryl polyethylene glycol 1000 succinate nanoparticles. Biomaterials. 2011;32(11):2969-2978. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||