Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (34): 5526-5533.doi: 10.3969/j.issn.2095-4344.0638
Previous Articles Next Articles
Application of degradable implants in fracture fixation
Li Junwei1, 2, Du Chengfei1, 2, Yuchi Chenxi1, 2, Zhang Chunqiu1, 2
- 1Tianjin Key Laboratory for Advanced Mechatronic System Design and Intelligent Control, School of Mechanical Engineering, Tianjin University of Technology, Tianjin 300384, China; 2National Demonstration Center for Experimental Mechanical and Electrical Engineering Education, Tianjin University of Technology, Tianjin 300384, China
-
Received:2018-06-28Online:2018-12-08Published:2018-12-08 -
Contact:Du Chengfei, Lecturer, Tianjin Key Laboratory for Advanced Mechatronic System Design and Intelligent Control, School of Mechanical Engineering, Tianjin University of Technology, Tianjin 300384, China; National Demonstration Center for Experimental Mechanical and Electrical Engineering Education, Tianjin University of Technology, Tianjin 300384, China -
About author:Li Junwei, Master candidate, Tianjin Key Laboratory for Advanced Mechatronic System Design and Intelligent Control, School of Mechanical Engineering, Tianjin University of Technology, Tianjin 300384, China; National Demonstration Center for Experimental Mechanical and Electrical Engineering Education, Tianjin University of Technology, Tianjin 300384, China -
Supported by:the National Natural Science Foundation of China, No. 11432016, 11602172
CLC Number:
Cite this article
Li Junwei, Du Chengfei, Yuchi Chenxi, Zhang Chunqiu. Application of degradable implants in fracture fixation[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(34): 5526-5533.
share this article

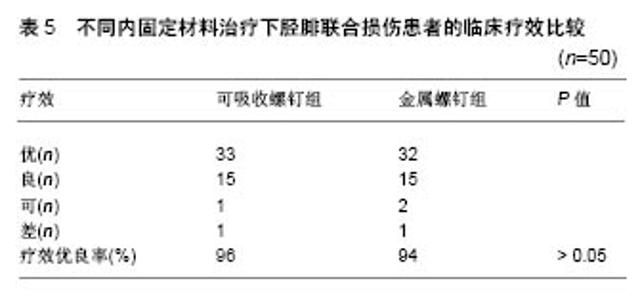
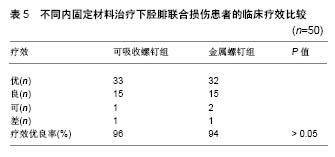
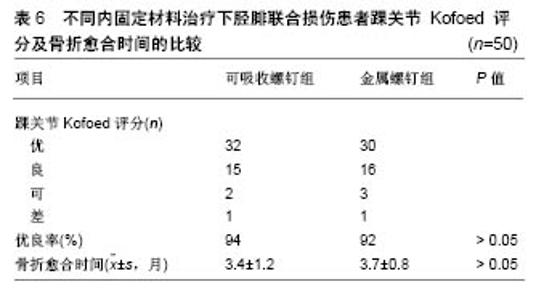
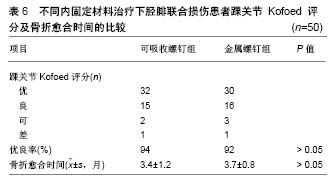
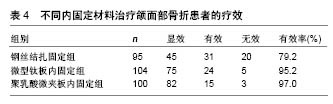
2.1 不同材料的可降解植入物 2.1.1 聚乳酸类植入物 聚乳酸类骨科植入物由于具有良好的机械性能、物理性能、可降解特性及与人骨相近的弹性模量,避免了应力遮挡和二次手术取出内植入物的风险,因而常被作为内植入物应用到临床骨折的治疗中。常见的聚乳酸类骨科植入物有聚乳酸、内消旋乳酸和左旋聚乳酸等。 聚乳酸:聚乳酸作为骨科内植入物应用于临床已有30余年,能够很好地治疗松质骨骨折,同时聚乳酸也是最早作为骨、软骨组织工程支架的材料[8-9]。曾飞跃等[10]对比了不同材料内植入物固定颌面部骨折的术后疗效,由于聚乳酸微夹板具有可降解特性,能够减少应力遮挡,使得骨早期承受生理性应力,促进骨折的愈合,术后采用聚乳酸微夹板治疗的效果优于其他两组(表4)。对患有下胫腓联合损伤的患者,采用聚乳酸螺钉进行治疗,术后聚乳酸螺钉能够提供足够的疲劳强度和失效强度来修复下胫腓联合损伤[11]。赵学寨等[12]分别采用聚乳酸螺钉和钛合金拉力螺钉对踝关节骨折患者进行治疗,术后两组治疗效果及愈合时间无显著差别(表5,6),采用聚乳酸螺钉治疗的患者,前期螺钉具有较高的机械强度,伴随螺钉的降解,应力逐渐转移到骨组织,从而促进骨折愈合与重建,避免了应力遮挡的发生。"
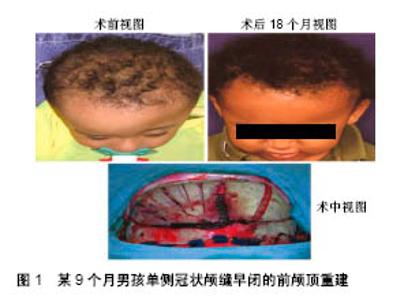
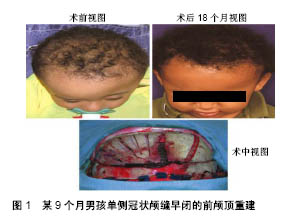
内消旋乳酸:采用内消旋乳酸与DL-聚乳酸螺钉、接骨板治疗下颌骨骨折,均可在满足生物力学的原则上促进骨愈合,然而DL-聚乳酸的结晶度高,降解时间长,容易引发异物反应;内消旋乳酸的亲水性较好,具有较高的机械强度,降低了降解时间,提高了骨折固定的安全性。此外,采用内消旋乳酸固定的下颌骨,术后8周新生骨小梁已将骨折断面之间的缝隙充满[13]。采用内消旋乳酸植入物进行固定下颌骨骨折,能够提供与钛螺钉固定下颌骨同等的固定强度,术后患者恢复良好[14-15]。此外,临床也报道了采用内消旋乳酸板和螺钉对281例面部骨折患者进行治疗,术后16例患者产生并发症,总体治疗效果良好[16]。Tiihonen等[17]尝试采用内消旋乳酸植入物对软组织缺陷及骨质缺损患者进行治疗,虽然内消旋乳酸植入物能够为患者减轻疼痛,但不能使患者恢复到术前状态,术后患者恢复效果较差,因而对于患有软组织缺陷及骨质缺损患者不宜采用内消旋乳酸植入物进行治疗。 左旋聚乳酸:1966年左旋聚乳酸缝合线第1次作为内植入物应用到动物体内,发现左旋聚乳酸缝合线无毒且可逐渐降解[18]。Eppley等[19]回顾性分析了采用左旋聚乳酸-聚乙交酯植入物进行颅面重建患者的临床疗效,术后植入物对患处起到稳定的固定,在降解过程中引发的异物反应很低,患者恢复良好,见图1。对于前交叉韧带损伤患者,由于软骨组织、滑膜及关节腔内韧带分泌的基质金属蛋白酶变化促进了关节液中基质金属蛋白酶2的升高,阻碍了前交叉韧带自身愈合[20]。前交叉韧带损伤,一旦未及时治疗可能导致膝关节过早退变[21]。动物实验表明,采用左旋聚乳酸/羟基磷灰石/α-Fe2O3与左旋聚乳酸-聚乙二醇/羟基磷灰石螺钉进行前交叉韧带重建,未出现移植物断裂及固定装置失效,提供了足够的力学性能[22]。前交叉韧带重建后骨隧道扩大会导致手术的复杂化。Arama等[23]分别采用左旋聚乳酸-羟基磷灰石螺钉与钛螺钉进行前交叉韧带重建,术后二者提供的力学性能无统计学差异,但采用左旋聚乳酸-羟基磷灰石螺钉治疗的患者,不仅降低了手术并发症发生概率,而且未引起骨隧道扩大。Robinson等[24]进一步证明采用左旋聚乳酸-羟基磷灰石螺钉进行前交叉韧带重建,能有效地降低骨隧道扩大发生的概率。此外,前交叉韧带重建过程中最薄弱的一个环节,为移植物在胫骨处的固定。采用左旋聚乳酸-羟基磷灰石螺钉对移植物进行固定,术后螺钉对移植物起到了稳定固定作用[25]。踝关节由距骨、胫骨下端和腓骨下端组成,其损伤在骨科非常常见,每年10万骨折患者中约有187例为踝关节骨折[26]。Rangdal等[27]对采用左旋聚乳酸螺钉治疗踝关节骨折患者进行随访,术后14周1例患者出现局部疼痛和肿胀,服用抗生素后并无大碍,其余患者恢复良好。临床也报道了采用左旋聚乳酸螺钉修复半月板损伤,可有效降低神经血管损伤风险[28]。通过前路减压和左旋聚乳酸螺钉进行骨移植固定可有效治疗颈椎病,术后植入物与相邻椎骨融合良好,螺钉按预期的时间降解且并发症较少[29]。Wendelstein等[30]对34例采用左旋聚乳酸螺钉矫正小脚趾畸形患者进行了为期3年的随访,术后84.6%的患者愈合良好,3例患者术后出现脚趾排列不齐,2例患者畸形复发。"
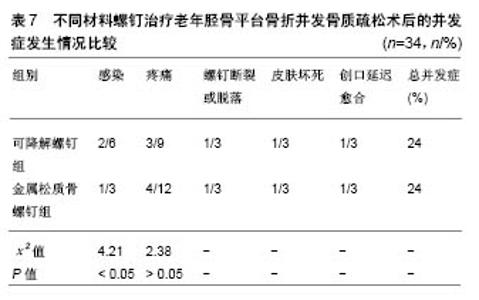
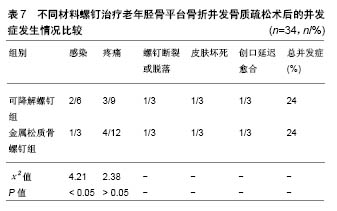
其他聚合物:采用可降解Lacto Sorb(由聚羟基乙酸与左旋聚乳酸按18∶82摩尔比例制成)螺钉对剥脱性骨软骨炎患者进行治疗,术后螺钉为患处提供了稳定的固定,螺钉在降解过程中未引发患处炎症反应[31]。Park等[32]采用聚(乳酸-乙醇酸)板和螺钉对兔下颌骨进行治疗,所有骨折部位在术后8,10周均有新骨生成,无严重炎症发生。王金华等[33]回顾性分析了可降解INION(由左旋聚乳酸、内消旋聚乳酸、聚乙醇酸、三亚甲基碳酸酯等合成)螺钉与金属螺钉治疗老年胫骨平台骨折并发骨质疏松的临床疗效及并发症发生情况(观察组采用可降解螺钉内固定+外固定治疗,对照组采用金属松质骨螺钉内固定+外固定治疗),术后采用可降解螺钉治疗的患者骨折的愈合时间及骨折线消失时间均小于采用金属螺钉治疗组,且膝关节活动范围大于金属螺钉治疗组,但在治疗阶段需要对骨折部位预防感染(表7)。"

2.1.2 镁合金植入物 镁合金作为骨科内植入物具有如下优点:镁是人体内必需的元素(含量21-25 g),约53%在骨骼中,27%在肌肉,19%在软组织,0.5%在红细胞,0.3%在血清;镁可促进新骨形成及骨组织的代谢;镁的弹性模量接近于人骨,避免了应力遮挡效应;镁合金在体内具有良好的生物相容性、骨传导性和可降解特性,其可降解特性避免了二次手术取出内植入物;镁在自然界的含量较丰富。因而镁合金植入物常被用于治疗骨折患者。常用的镁合金植入物材料主要为高纯度镁合金、MgCa0.8合金、MgYREZr合金及AZ系列镁合金等。 高纯度镁合金:高纯度镁合金避免了第二相杂质所产生的微电偶腐蚀,因而具有无毒、低降解率等优点。浸渍实验、细胞毒性及生物活性测试表明,高纯度镁螺钉在体外表现出均匀腐蚀行为,可提高细胞活性、骨组织的碱性磷酸酶活性及与成骨分化相关基因(如碱性磷酸酶、骨桥蛋白、人骨髓间充质干细胞RUNX2等信使核糖核酸)的表达[34]。动物体内实验表明,高纯度镁螺钉显示出了良好的骨整合性能,增加了骨量和骨密度,不但不会影响骨折部位的愈合,而且可促进新骨形成[34-37],使骨折愈合之后达到和正常骨接近的抗弯性能[36]。高纯度镁还能通过刺激骨形成蛋白2和血管内皮生长因子的表达,促进前交叉韧带重建[38]。此外,临床上也报道了高纯度镁螺钉固定后的带血管蒂骨移植,可明显改善骨股头坏死患者髋关节功能,降低骨瓣产生位移的概率[39]。 MgCa0.8合金:将MgCa0.8螺钉植入兔胫骨,术后螺钉在体内表现出良好的耐受性以及生物相容性,但其降解速率过快,可能导致其力学性能不足(图2)[40]。Thomann等[41]将采用MgF2进行表面涂层处理的MgCa0.8植入到新西兰白兔胫骨骨髓腔,术后植入物在兔体内耐受性良好且能有效减缓其降解速率,与未涂层植入物相比,6个月后涂层植入物的强度高于未涂层植入物的强度。此外,Adam等[42]通过动物实验进一步证明MgCa0.8作为内植入物应用到动物体内无毒性。"


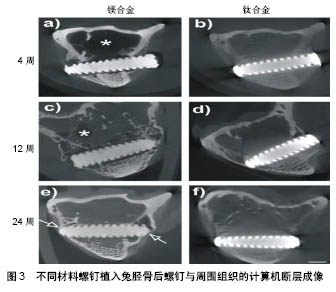
MgYREZr合金:MgYREZr合金由于RE元素的存在,组织均匀,经过大塑性变形可获得尺寸<1 mm的晶粒,具有较高的屈服强度、抗拉强度及延伸率等力学性能;MgYREZr可稳定腐蚀层,对MgYREZr合金进行合理的热处理可降低其降解速率[43]。2013年Syntellix公司采用MgYREZr螺钉治疗拇指外翻,不仅取得了良好的治疗效果,同时MgYREZr合金成为世界上第1个获得认证的可降解镁合金骨内植入产品,可在欧盟范围内进行商业销售[44]。Diekmann等[45]分别将MgYREZr螺钉和钛合金螺钉植入兔胫骨进行前交叉韧带重建实验,采用MgYREZr螺钉治疗的兔,术后24周观察到螺钉与肌腱形成了良好的骨结合,临床上未出现肌腱断裂或松动,组织学评价显示无炎症发生或肌腱坏死症状(图3)。"
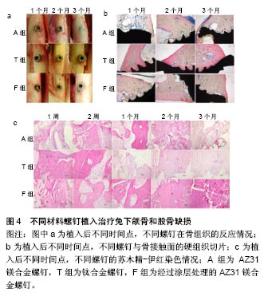
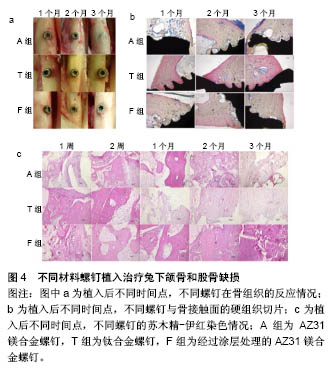
然而临床报道了对患有急性腕舟骨骨折的患者,采用MgYREZr压紧螺钉进行治疗,术后部分患者出现严重的骨溶解和囊肿现象,且术后6个月之内所有患者骨折部位均未得到稳定的固定,因而目前对于此类患者不宜采用MgYREZr压紧螺钉进行治疗[46]。此外,控制MgYREZr合金的降解速率以适应不同的临床需求,仍然是其面临的主要问题。 AZ系列镁合金:Witte等[47]对两种镁合金(AZ91D,LAE442)植入物进行体内和体外实验,发现植入物在体内的腐蚀速率比在体外的腐蚀速率低4个数量级,因而由体外获得的镁合金植入物的降解速率不能用来预测其在体内的降解速率。将AZ31镁合金螺钉植入羊髋骨,术后3个月取出螺钉,在螺钉表面发现了新骨形成,且在羊的泌尿、免疫及循环系统未发现不利影响[48]。滕继平等[49]通过实验对比了AZ31B镁合金肋骨髓内钉与左旋聚乳酸肋骨髓内钉在肋骨骨折治疗中的生物力学性能,三点弯曲实验表明镁合金髓内钉的测试结果更接近正常标本组;扭转强度实验表明镁合金髓内钉与正常标本无显著性差异;拉伸实验表明,镁合金髓内钉的抗拔出力及抗拉强度均优于左旋聚乳酸髓内钉。动物实验结果表明,采用氟化物(主要成分MgO和MgF2)对AZ31B镁合金螺钉表面进行涂层处理,将其植入兔下颌骨和股骨(A组为未涂层的AZ31B螺钉,T组为钛螺钉,F组为涂层处理的AZ31B螺钉),术后3个月,经过涂层处理的螺钉逐渐被新生骨组织包裹,无明显炎症反应,见图4[50]。"
| [1] Kim BS,Mooney DJ.Development of biocompatible synthetic extracellular matrices for tissue engineering.Trends Biotechnol. 1998;16(5):224-230.[2] Gefen A.Computational simulations of stress shielding and bone resorption around existing and computer-designed orthopaedic screws.Med Biol Eng Comput.2002;40(3):311-322. [3] Viljanen J,Kinnunen J,Bondestam S,et al.Bone changes after experimental osteotomies fixed with absorbable self-reinforced poly-l-lactide screws or metallic screws studied by plain radiographs, quantitative computed tomography and magnetic resonance imaging.Biomater.1995;16(17):1353-1358.[4] Waris E,Konttinen YT,Ashammakhi N,et al.Bioabsorbable fixation devices in trauma and bone surgery: current clinical standing. Expert Rev Med Devices.2004;1(2):229-240.[5] Hughes TB.Bioabsorbable implants in the treatment of hand fractures: an update.Clin Orthop Relat R.2006;445(445):169-174.[6] Gristina AG.Biomaterial-centered infection: microbial adhesion versus tissue integration. Science. 1987;237(4822):1588-1595.[7] Litsky AS.Clinical reviews: Bioabsorbable implants for orthopedic fracture fixation. Journal App Biomater. 2010;4(1):109-111.[8] Kim WS,Vacanti CA,Upton J,et al.Bone defect repair with tissue-engineered cartilage. Plast Reconstr Surg. 1994;94(5): 580-584.[9] 曹谊林,商庆新.软骨、骨组织工程的现状与趋势[J].中华创伤杂志, 2001, 17(1):7-9.[10] 曾飞跃,陈慧娟.不同材料在颌面部骨折内固定术中的临床应用比较研究[J].中国临床医生杂志, 2016,44(9):85-86. [11] Thordarson DB,Hedman TP,Gross D,et al.Biomechanical evaluation of polylactide absorbable screws used for syndesmosis injury repair.Foot Int.1997;18(10):622-627.[12] 赵学寨,李海军,孟彩云,等.可吸收螺钉与金属螺钉内固定修复踝关节骨折:生物相容性及踝关节功能比较[J].中国组织工程研究, 2016, 20(31): 4687-4692.[13] 陈宇轩,吕春堂,周树夏,等.可吸收聚乳酸接骨板螺钉行下颌骨骨折内固定的实验研究[J].解放军医药杂志,2002,14(1):5-8.[14] Lee HB,Oh JS,Kim SG,et al.Comparison of titanium and biodegradable miniplates for fixation of mandibular fractures.J Oral Maxil Surg.2010; 68(9):2065-2069.[15] Leno MB,Liu SY,Chen CT,et al.Comparison of functional outcomes and patient-reported satisfaction between titanium and absorbable plates and screws for fixation of mandibular fractures: a one-year prospective study.J Cranio Maxill Surg. 2017;45(5): 704-709.[16] Bell RB,Kindsfater CS.The use of biodegradable plates and screws to stabilize facial fractures.J Oral Maxil Surg. 2006;64(1): 31-39.[17] Tiihonen R,Honkanen PB,Belt EA,et al.The mean seven years' results of the use of poly-L/D-lactic acid (PLDLA) interposition implant and bone packing in revision metacarpophalangeal arthroplasty: a prospective cohort study.Scand J Surg. 2012; 101(4):265-270.[18] Kulkarni RK,Pani KC,Neuman C,et al.Polylactic acid for surgical implants.Arch Surg-Chicago.1966;93(5):839-843.[19] Eppley BL,Morales L,Wood R,et al.Resorbable PLLA-PGA plate and screw fixation in pediatric craniofacial surgery: clinical experience in 1883 patients.Plasti Recons Surg.2004; 114(4): 850-856.[20] Tang Z,Yang L,Wang Y,et al.Contributions of different intraarticular tissues to the acute phase elevation of synovial fluid MMP-2 following rat ACL rupture.J Orthop Res.2009; 27(2):243-248.[21] Papalia R,Vasta S,D'Adamio S,et al.Metallic or bioabsorbable interference screw for graft fixation in anterior cruciate ligament (ACL) reconstruction? Br Med Bull. 2013;109(1):19-29.[22] 杨坤,朱伟民,黄江鸿,等.两种改性可吸收界面螺钉重建前交叉韧带的生物力学研究[J].实用骨科杂志,2016,22(6):518-521.[23] Arama Y,Salmon LJ,Sri-Ram K,et al.Bioabsorbable Versus Titanium Screws in Anterior Cruciate Ligament Reconstruction Using Hamstring Autograft: A Prospective, Blinded, Randomized Controlled Trial With 5-Year Follow-up.Am J Sports Med. 2015; 43(8):1893.[24] Robinson J,Huber C,Jaraj P,et al.Reduced bone tunnel enlargement post hamstring ACL reconstruction with poly-L-lactic acid/hydroxyapatite bioabsorbable screws.Knee.2006;13(2): 127-131.[25] Patkowski M,Królikowska A,Reichert P.Comparison of Bioabsorbable Interference Screws Composed of Poly-l-lactic Acid and Hydroxyapatite (PLLA-HA) to WasherLoc Tibial Fixation in Patients After Anterior Cruciate Ligament Reconstruction of the Knee Joint. Polim Med. 2016;46(1):53.[26] Court-Brown CM,Mcbirnie J,Wilson G.Adult ankle fractures--an increasing problem?Acta Orthop Scand.1998;69(1):43-47. [27] Rangdal S,Singh D,Joshi N,et al.Functional outcome of ankle fracture patients treated with biodegradable implants.Foot Ankle Surg.2012; 18(3):153-156.[28] Bohnsack M,Börner C,Schmolke S,et al.Clinical results of arthroscopic meniscal repair using biodegradable screws. Knee Surg Sports Traumatol Arthrosc.2003;11(6):379-383. [29] Zhao B,Qiu X,Dong W,et al.Application of bioabsorbable screw fixation for anterior cervical decompression and bone grafting. Clinics(Sao Paulo).2016;71(6):320-324.[30] Wendelstein JA,Goger P,Bock P,et al.Bioabsorbable fixation screw for proximal interphalangeal arthrodesis of lesser toe deformities.Foot Ankle Int.2017;38(9):1020-1025.[31] Larsen MW,Pietrzak WS,Delee JC.Fixation of osteochondritis dissecans lesions using poly(l-lactic acid)/ poly(glycolic acid) copolymer bioabsorbable screws.Am J Sport Med.2005; 33(1): 68-76.[32] Park S,Kim JH,Kim IH,et al.Evaluation of poly(lactic-co-glycolic acid) plate and screw system for bone fixation.J Craniofac Surg. 2013;24(3): 1021-1025.[33] 王金华,徐世华,冷干生,等.可吸收螺钉治疗34例老年胫骨平台骨折并发骨质疏松的效果及其对疼痛和并发症的影响[J].上海医药, 2016, 37(17):34-37.[34] Han P,Cheng P,Zhang S,et al.In vitro and in vivo studies on the degradation of high-purity Mg (99.99wt.%) screw with femoral intracondylar fractured rabbit model. Biomater.2015;64:57-69.[35] Chaya A,Yoshizawa S,Verdelis K,et al. Fracture healing using degradable magnesium fixation plates and screws.J Oral Maxillof Surg.2014;73(2):295-305.[36] Chaya A,Yoshizawa S,Verdelis K,et al.In vivo study of magnesium plate and screw degradation and bone fracture healing.Acta Biomater.2015;18:262-269.[37] Yang JX,Cui FZ,Lee IS,et al.In vivo biocompatibility and degradation behavior of Mg alloy coated by calcium phosphate in a rabbit model.J Biomater Appl. 2012;27(2):153-164.[38] Cheng P,Han P,Zhao C,et al.High-purity magnesium interference screws promote fibrocartilaginous entheses regeneration in the anterior cruciate ligament reconstruction rabbit model via accumulation of BMP-2 and VEGF. Biomater. 2016;81:14-26.[39] Zhao D,Huang S,Lu F,et al.Vascularized bone grafting fixed by biodegradable magnesium screw for treating osteonecrosis of the femoral head.Biomater. 2015;81(1):84-92. [40] Erdmann N,Angrisani N,Reifenrath J,et al.Biomechanical testing and degradation analysis of MgCa0.8 alloy screws: a comparative in vivo study in rabbits.Acta Biomater.2011; 7(3):1421-1428.[41] Thomann M,Krause C,Angrisani N,et al.Influence of a magnesium‐fluoride coating of magnesium‐based implants (MgCa0. 8) on degradation in a rabbit model.J Biomed Mater Res A.2010;93(4): 1609-1619.[42] Adam R,Orban H,Plopeanu E,et al.Results of In Vivo Biological Tests Performed on a Mg-0.8Ca Alloy.Key Eng Mat. 2017;745: 50-61.[43] 谭丽丽,陈军修,于晓明,等.生物可降解MgYREZr合金的研究进展[J].金属学报,2017,53(10):1207-1214.[44] Windhagen H,Radtke K,Weizbauer A,et al.Biodegradable magnesium-based screw clinically equivalent to titanium screw in hallux valgus surgery: short term results of the first prospective, randomized, controlled clinical pilot study. Biomed Eng Online. 2013;12(1):62.[45] Diekmann J,Bauer S,Weizbauer A,et al.Examination of a biodegradable magnesium screw for the reconstruction of the anterior cruciate ligament: A pilot in vivo study in rabbits.Mater Sci Eng.2016;59:1100-1109.[46] Meier R,Panzica M.First results with a resorbable MgYREZr compression screw in unstable scaphoid fractures show extensive bone cysts.Handchir Mikrochir Plast Chir. 2017;49(1):37-41.[47] Witte F,Fischer J,Nellesen J,et al.In vitro and in vivo corrosion measurements of magnesium alloys.Biomater. 2006;27(7): 1013-1018.[48] Duygulu O,Kaya RA,Oktay G,et al.Investigation on the Potential of Magnesium Alloy AZ31 as a Bone Implant.Mater Sci Forum. 2007; 546:421-424.[49] 滕继平,杨志胤,程佑爽,等.一种镁合金可吸收肋骨髓内钉的生物力学性能[J].中国组织工程研究, 2013,17(48):8388-8393.[50] Sun W,Zhang G,Tan L,et al.The fluoride coated AZ31B magnesium alloy improves corrosion resistance and stimulates bone formation in rabbit model.Mater Sci Eng. 2016;63:506-511.[51] Yan T,Tan L,Xiong D,et al.Fluoride treatment and in vitro corrosion behavior of an AZ31B magnesium alloy.Mat Sci Eng C. 2010; 30(5):740-748.[52] Yu W,Zhao H,Ding Z,et al.In vitro and in vivo evaluation of MgF2 coated AZ31 magnesium alloy porous scaffolds for bone regeneration. Colloid Surface B.2017;149:330-340.[53] Robinson J,Huber C,Jaraj P,et al.Reduced bone tunnel enlargement post hamstring ACL reconstruction with poly- l -lactic acid/hydroxyapatite bioabsorbable screws.Knee.2006; 13(2): 127-131. [54] Martinek V,Friederich NF.Tibial and pretibial cyst formation after anterior cruciate ligament reconstruction with bioabsorbable interference screw fixation.Arthroscopy.1999; 15(3):317-320.[55] Appelt A,Baier M.Recurrent locking of knee joint caused by intraarticular migration of bioabsorbable tibial interference screw after arthroscopic ACL reconstruction[J]. Knee Surg Sport Tr A. 2007;15(4):378-380. [56] Peltoniemi HH,Tulamo RM,Pihlajamäki HK,et al.Consolidation of craniotomy lines after resorbable polylactide and titanium plating: A comparative experimental study in sheep. Plast Reconstr Surg. 1998;101(1):123-133.[57] Weiler A,Hoffmann RFG,Stähelin AC,et al.Biodegradable implants in sports medicine: the biological base.Arthroscopy. 2000;16(3): 305-321.[58] Tang Z,Yang L,Wang Y,et al.Contributions of different intraarticular tissues to the acute phase elevation of synovial fluid MMP-2 following rat ACL rupture.J Orthop Res.2009; 27(2):243-248.[59] Chen Y,Xu Z,Smith C,et al.Recent advances on the development of magnesium alloys for biodegradable implants.Acta Biomater. 2014;10(11):4561-4573.[60] Argo D,Pekguleryuz M,Labelle P,et al.Process parameters and diecasting of Noranda's AJ 52 high temperature Mg-Al-Sr alloy[C]//Magnesium Technology 2002 as held at the 2002 TMS Annual Meeting.2002:87-93.[61] Li H,Pang S,Liu Y,et al.Biodegradable Mg–Zn–Ca–Sr bulk metallic glasses with enhanced corrosion performance for biomedical applications.Mater Design.2015;67:9-19.[62] Seiler HG,Sigel H,Sigel A.Handbook on toxicity of inorganic compounds.Anal Chim Acta.1988; 237.[63] Wu W,Petrini L,Gastaldi D,et al.Finite element shape optimization for biodegradable magnesium alloy stents. Ann Biomed Eng. 2010;38(9):2829-2840. [64] Debusschere N,Segers P,Dubruel P,et al.A finite element strategy to investigate the free expansion behaviour of a biodegradable polymeric stent.J Biomech. 2015;48(10):2012-2018.[65] Debusschere N,Segers P,Dubruel P,et al.A computational framework to model degradation of biocorrodible metal stents using an implicit finite element solver.Ann Biomed Eng. 2016; 44(2):382-390. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||