Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (29): 4698-4705.doi: 10.3969/j.issn.2095-4344.0629
Previous Articles Next Articles
The roles of integrins and E-cadherin in the reprogramming and stemness maintenance of induced pluripotent stem cells
Hu Ruo-wen, Chen Xiao-fang
- Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China
-
Revised:2018-05-24Online:2018-10-18Published:2018-10-18 -
Contact:Chen Xiao-fang, PhD, Associate professor, Master’s supervisor, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China -
About author:Hu Ruo-wen, Master candidate, Key Laboratory for Biomechanics and Mechanobiology of Ministry of Education, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China -
Supported by:the National Natural Science Foundation of China, No. 81301334
CLC Number:
Cite this article
Hu Ruo-wen, Chen Xiao-fang. The roles of integrins and E-cadherin in the reprogramming and stemness maintenance of induced pluripotent stem cells[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(29): 4698-4705.
share this article
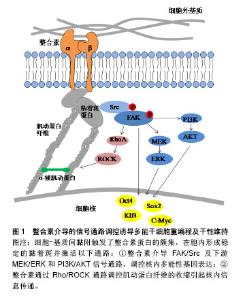
2.1 纳入文献基本情况 整合素和E-钙黏素作为参与iPSCs重编程及干性维持的重要细胞黏附分子,会在感知外界来源的机械力下发生构象变化而被活化,将微环境机械信号传递到细胞质内,然后在胞质中转换为生化信号,进而激活下游一系列信号通路,与维持多能性的关键基因相互作用,影响iPSCs的产生和维持[15]。另一方面,整合素和E-钙黏素均可以与质膜下的细胞骨架相连,通过肌动蛋白纤维的收缩传导机械力信号来影响iPSCs的功能和命运,细胞骨架参与细胞黏附分子的信号通路的重要性也越来越突出。因此,文章从以下3个方面探讨细胞黏附分子在iPSCs重编程及干性维持中的作用:①整合素介导的信号通路与重编程;②E-钙黏素介导的信号通路与重编程;③细胞骨架参与整合素和E-钙黏素的作用。 2.2 整合素介导的信号通路与重编程 整合素是一类介导细胞-基质间黏附的细胞表面受体,由α和β两个亚基形成异二聚体。整合素作为桥梁连接了细胞外基质-整合素-细胞骨架整个信号通路[8]。整合素在没有与细胞外基质结合时是分散在细胞表面的,且不与肌动蛋白细胞骨架相连,一旦与细胞外基质中的配体结合被激活,则通过其胞内域与细胞骨架发生联系,并发生簇集,形成黏着斑(focal adhesion,FA),一方面激活下游信号通路,另一方面与肌动蛋白骨架直接连接,见图1。"
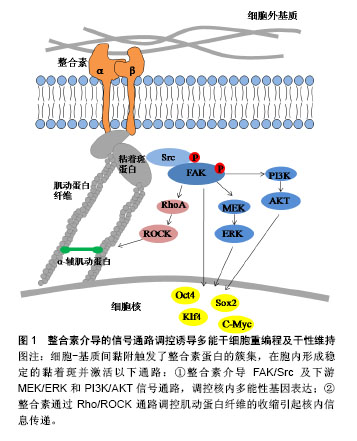
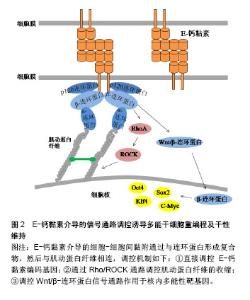
2.2.1 整合素介导的FAK/Src通路 整合素的2种重要下游信号传导介质是黏着斑激酶(focal adhesion kinase,FAK)和Src家族激酶(Src family kinase,SFK)。当整合素被激活时,FAK在397位酪氨酸上进行自磷酸化,为Src创建一个磷酸化结合位点,同时也使FAK中的其他酪氨酸残基磷酸化,从而到达激酶活性最大化,引起一系列级联放大反应,调节细胞功能[16]。Hayashi等[10]和Villa-Diaz等[17]先后证实了整合素激活的FAK信号通路对小鼠和人的胚胎干细胞多能性维持均有抑制作用,反之,FAK信号的失活可促进其自我更新和多能性维持。Villa-Diaz等[18]在最近的研究中还发现了人多能干细胞中的一种新的信号通路,整合素α6β1作为层粘连蛋白α5的受体可以抑制FAK的自磷酸化,最终促进了人多能干细胞的自我更新和多能性转录因子的表达。该实验结果提供了一种层粘连蛋白α5重编程人多能干细胞的新方法。与FAK作用相同的还有SFK,抑制SFK活性可维持人胚胎干细胞的多能性状态,反之激活SFK可以启动分化过程[19]。Staerk等[20]证明了SFK通路的激活会阻止细胞重编程,而SFK抑制剂也被证明可以作为Sox2转录因子的化学替代物来进行重编程实验研究。 细胞外基质的物理性质,如基质的表面形貌、软硬度都会通过影响黏着斑的形成进而影响下游信号通路的活性[4]。Jeon等[21]以加工了纳米结构图案的聚二甲基硅氧烷作为基底材料培养小鼠胚胎干细胞,他们发现这种粗糙的纳米结构促进了小鼠胚胎干细胞的自我更新,可有效维持干细胞的未分化状态。随着这种结构粗糙度的增加,FAK活性被显著削弱,并且由于基质与细胞的相互作用增强了对抑制干细胞分化有重要作用的白血病抑制因子的表达。类似的研究也开始在临床展开,诱导心肌细胞对于心脏疾病的治疗具有重要意义,但是诱导心肌细胞的重编程效率一直很低。Yoo等[22]发现通过纳米图案修饰培养基底,可以下调FAK活性,相较于平基底显著提高了将成纤维细胞诱导重编程为心肌细胞的效率。 当FAK/Src信号事件得以起始,下游一系列信号通路将被启动,如MEK/ERK,PI3K/AKT和BMP/SMAD通路,也均被报道过用来协调干细胞的干性维持和参与体细胞重编程。Watt[8]在综述整合素下游信号分子表达及功能时提出,被FAK激活的MEK/ERK级联反应对干细胞分化具有重要的调节作用,激活ERK能够启动胚胎干细胞向分化细胞类型转变,抑制ERK则能使细胞维持自我更新状态。使用MEK/ERK抑制剂PD0325901和GSK3抑制剂CHIR99021(2 inhibitor,2i)联合处理可促进iPSCs的产生,用于体细胞重编程[23-26]。Higuchi等[3]希望利用这种信号传导机制,模拟细胞外基质作用,使得无饲养层细胞和特殊基质的干细胞培养成为可能,最终他们用一种解整合素和2i的组合试剂成功建立了小鼠iPSCs培养体系。这种新型简单的培养方式提示可以通过更多的信号通路作用开发iPSCs重编程技术。 2.2.2 整合素对细胞骨架的调控 区别于上述的信号事件,由于黏着斑是锚定在细胞骨架上的,因此可直接调控肌动蛋白纤维的组装来影响细胞行为。除了直接传导机械力信号,整合素与胞外基质分子的结合还可以通过调控Rho GTP酶家族成员的活性,来控制肌动蛋白纤维的组装和收缩。已知的调控过程如下:以Rho GTP酶家族为中心的信号通路被激活,Rho是Rho GTP酶家族中最主要的成员,Rho分为RhoA、B、C三类。Rho相关蛋白激酶(Rho-associated protein kinase,ROCK)和形成素相关蛋白(mammalian diaphanous protein,mDia)是RhoA的直接效应体,它们分别可以促进肌动-肌球蛋白收缩和非肌球蛋白Ⅱ(non-myosin Ⅱ,NMⅡ)驱使下肌动蛋白纤维的组装[27]。研究表明,正常范围内的NMⅡ活性对于人胚胎干细胞的多能性维持是不可或缺的[28],但是NMⅡ的过度活化会导致小鼠和人多能干细胞具有分化倾向及过低的细胞存活率[29-30],如Walker等[30]使用NMⅡ ATPase抑制剂抑制NMⅡ活性,证明其可以提高人iPSCs的克隆密度和悬浮培养条件下的细胞存活率,而且提高了多能性因子Oct3/4和Nanog的表达,可用于人胚胎干细胞和人iPSCs的体外培养。另外,Rosowski等[31]在培养人胚胎干细胞中发现,相较于细胞群体中心,群体边缘位置的细胞会表达出显著的肌动蛋白组装、更大的肌球蛋白活性和更强的牵引力,重要的是这部分细胞将先于中心部分细胞发生分化,在外周产生出一个“分化带”,这也预示着可以通过控制Rho及其下游反应来抑制干细胞分化。Higuchi等[32]发现在软的聚丙烯酰胺凝胶上进行重编程,可以下调ROCK2的表达,使得肌动蛋白纤维装配紊乱,最终胚胎干细胞标志基因Oct4和Nanog的表达水平显著提高,重编程被促进。与之一致的是,一种ROCK抑制剂Y-27632也被发现可显著促进人皮肤成纤维细胞的重编程[33]。显然,通过这些最后集中到调节肌动蛋白纤维组装上的方法,可以定向调控干细胞行为。 综上,整合素可以通过FAK/Src信号通路和对细胞骨架的调控作用参与iPSCs重编程的命运转变。利用这2种机制,可以更好的进行iPSCs重编程方法的开发,如使用抑制剂下调FAK/Src及其下游一系列信号通路可促进iPSCs的重编程;在Rho通路上调控肌动蛋白纤维的组装与收缩能够改变干细胞命运;由于整合素介导细胞-基质间黏附的特性,直接改变细胞与细胞外基质界面间的物理信号也可能有效响应上述2种机制。 2.3 E-钙黏素介导的信号通路与重编程 2.3.1 E-钙黏素编码基因的高表达 在哺乳动物细胞中,细胞间的连接大多是由钙黏素的同亲型结合形成的,通过钙黏素可以将相邻细胞的细胞膜与肌动蛋白纤维连接使细胞彼此黏着。由于干细胞聚团生长的特性,作为介导干细胞间黏附的主要分子,E-钙黏素对于干细胞的存活和自我更新至关重要。人胚胎干细胞在被消化成单个细胞后具有较差的克隆形成能力[9],而E-钙黏素的表达上调可以显著提高其单细胞存活率和克隆形成率[34];同时,E-钙黏素也是重编程早期间质-上皮转化过程中的决定性因子[14,35-36]。Choi等[37]发现软的水凝胶基底可以使E-钙黏素表达上调,激活细胞从间充质向上皮型的转化,从而提高了多能性标志基因Oct4,Sox2的表达,有效进行了iPSCs重编程。通过基因敲除,siRNA和蛋白合成阻断的方法抑制E-钙黏素的表达,iPSCs则无法产生[14,35-36,38],而多种化学物质可以通过上调E-钙黏素或者使E-钙黏素过表达来促进重编程过程,显着增强重编程效率[36]。Redmer等[38]还发现通过反转录病毒将E-钙黏素外源转入,甚至可以代替Oct4因子完成iPSCs的重编程。这些研究结果表明,直接作用于上调E-钙黏素表达的方法可以有效促进iPSCs重编程及干性维持。 2.3.2 E-钙黏素对细胞骨架的调控 与整合素一样,钙黏素也需要通过特殊蛋白结构与胞内细胞骨架相连。E-钙黏素胞内结构域与肌动蛋白骨架之间机械传导链中的主要组分是连环蛋白,目前已发现的连环蛋白包括α-连环蛋白,β-连环蛋白,γ-连环蛋白和p120连环蛋白。β-连环蛋白与E-钙黏素的胞内结构域结合,形成E-钙黏素/β-连环蛋白复合物,而α-连环蛋白则调控这个复合物与肌动蛋白纤维的动态连接[9]。这个机械传导链在形成细胞间黏附连接的同时,也调控了肌动蛋白纤维的组装和肌动-肌球蛋白收缩[39]。Rho GTP酶家族成员对细胞骨架的调控作用在E-钙黏素介导的信号通路中也被证实,见图2。目前有研究报道,Rho和ROCK信号因子的活性与E-钙黏素复合物存在关联[40-41],并且E-钙黏素/β-连环蛋白复合物可参与协调Rho活化剂和抑制剂的平衡[39]。Li等[28]发现NMⅡ可以促进p120连环蛋白结合并稳定E-钙黏素,加强细胞间黏附,从而促进人胚胎干细胞克隆形成、多能性维持和存活率的提高。因此,与整合素参与干细胞重编程一致的是,E-钙黏素也可以通过Rho/ROCK作用于细胞骨架,再通过细胞骨架的收缩调控干细胞命运。"
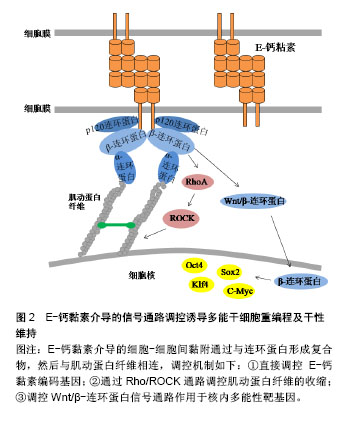
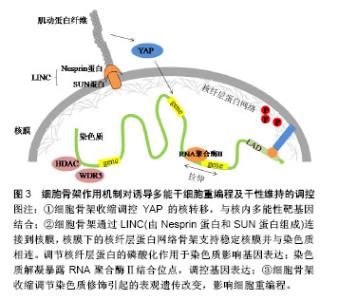
2.3.3 β-连环蛋白对多能性相关基因的调控 Wnt/β-连环蛋白信号通路是一种可以诱导干细胞多能性的重要信号通路。当Wnt蛋白高表达时,其可以阻断β-连环蛋白的降解复合体,从而使胞内β-连环蛋白积累。一方面可以与E-钙黏素形成复合物,连接细胞骨架,介导细胞间黏附作用。另一方面,游离的β-连环蛋白会进入细胞核内,作为辅助转录因子,启动与干细胞多能性相关的靶基因表达[42],如细胞周期蛋白D1(Cyclin D1,CCND1)作为β-连环蛋白靶基因的作用已被证实[43]。Zhao等发现CCND1的过表达可以诱导表皮细胞重编程为干细胞样细胞。他们将CCND1基因转入表皮细胞,发现细胞形态开始从细胞核较小的扁平状细胞变为细胞核较大的圆形细胞,表现出聚集成团生长并有长期增殖能力,同时伴随多种多能性基因的表达。值得一提的是,当诱导细胞植入实验动物模型的伤口时,伤口愈合加快。此实验结果不仅开发出一种有效的重编程方法,还应用到了可能的临床治疗中。Yamazaki等[44]揭示了一种核骨架对β-连环蛋白的定位作用,他们通过人为促进核肌动蛋白的积累,发现这种定位作用可以增强β-连环蛋白与下游靶基因的结合,使得Wnt/β-连环蛋白信号通路中诱导干细胞多能性的基因表达。 此外,还有一些研究表明E-钙黏素可以被N-钙黏素替代以维持细胞间黏附作用和干细胞多能性[45-46]。用E-钙黏素/α-连环蛋白嵌合蛋白去模拟β-连环蛋白的功能,也足以维持小鼠诱导多能干细胞的多能性[47]。细胞间黏附是干性状态稳定的前提,并没有一个特定的“钙黏素密码”来唯一决定iPSCs的生成。在研究E-钙黏素介导的细胞间黏附参与重编程的机制时,研究者们从一开始把E-钙黏素当做必不可少的调控因子,到通过调控Rho/ROCK信号通路作用于细胞骨架,调控Wnt/β-连环蛋白信号通路直接作用于多能性靶基因,寻找到了更多高效的iPSCs重编程方法,也为开发iPSCs临床治疗打下基础。 2.4 细胞骨架参与整合素和E-钙黏素的作用 细胞骨架是真核细胞中的动态蛋白纤维网络结构,肌动蛋白纤维的组装与去组装可以调节多种细胞行为。由以上可知,整合素和E-钙黏素都可以通过特殊分子结构与细胞骨架网络相连,而细胞骨架网络可以与细胞核的支架系统相互作用,将信息传递至细胞核内的核仁、染色质和DNA[48]。最近在重编程机制的研究中,细胞骨架的潜在作用越来越突出。Sakurai等[49]研究表明,肌动蛋白的聚合作用是发生细胞重编程的障碍,他们通过抑制活性调节因子TESK1和LIMK2来抑制丝切蛋白的磷酸化。丝切蛋白是一种重要的肌动蛋白结合蛋白,其去磷酸化作用可使肌动蛋白单体从肌动蛋白丝的顶端解离,从而使肌动蛋白纤维以不稳定状态存在,该方法最终促进了间质-上皮转化过程,并提高了重编程效率。 为了深入理解细胞骨架对于干细胞命运的调控过程,以开发促进iPSCs重编程的技术,还需要探索细胞骨架将信息传递至细胞核内的机制,通过这些机制使细胞骨架的组装和收缩可以产生基因水平、蛋白质水平和表观遗传学水平上的变化,并最终影响细胞重编程,见图3。"
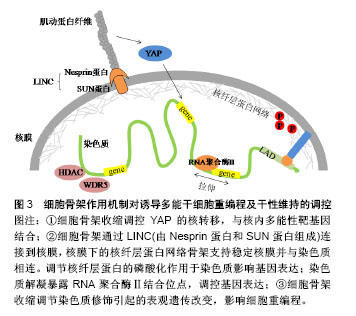

2.4.1 细胞骨架收缩调控YAP的核转移 SirioDupont的研究报告指出,在刚性细胞外基质和细胞形变激活的机械信号传导链中,细胞骨架的收缩促使Yes相关蛋白(Yes-associated protein,YAP)向细胞核转位[50],最终对胚胎干细胞的自我更新产生重要影响。YAP结合靶点的全基因组分析显示,YAP直接与许多胚胎干细胞多能性基因的启动子结合并刺激它们的表达[12]。敲低YAP会导致胚胎干细胞的多能性丧失,而YAP过表达会抑制胚胎干细胞分化[12,51],增强小鼠iPSCs的重编程效率。 2.4.2 细胞骨架与染色质的连接 核骨架-细胞骨架复合体蛋白(linker of nucleoskeleton and cytoskeleton,LINC)可以物理性的连接细胞骨架与核膜,包含核纤层在内的多组分核内支架可以稳定核膜并将核膜与染色质偶联[52]。通过核膜、细胞骨架可以最终与染色质连接,细胞骨架上产生的机械力进而传递到核内并影响基因表达[15]。以下2种传导机制先后被证实:①核纤层位于内层核膜与染色质之间,核纤层蛋白向外与内层核膜上的蛋白结合,向内与染色质的特定区段结合,与核纤层直接相连的染色质结构称为核纤层相关结构域(lamina associateddomains,LAD),LAD多为异染色质区,包括核纤层蛋白A/C在内的几种核蛋白具有机械敏感性,并且当受到细胞骨架传递的力时可以发生形变以改变它们的磷酸化状态[53-54]。有关研究表明,核纤层蛋白A/C可以将外周异染色质锚定在核膜上,其磷酸化可以促进细胞核中的蛋白合成和降解的平衡,这个过程反过来能调节异染色质定位、基因表达、细胞分化和可能的细胞重编程[55-56];②细胞骨架上的机械力可以直接拉伸染色质,暴露RNA聚合酶的结合位点,如经历脉冲力的细胞在5 s内就表现出显著的染色质解凝[57]。最近,Wang团队通过一种包被磁珠,利用三维磁粒扭转细胞测量仪给细胞表面施加局部压力,在活细胞中显像了染色质变形,以及压力诱导的绿色荧光蛋白标记基因发生转录,其机制为施加给整合素的局部压力从拉紧的肌动蛋白骨架处传送至LINC复合物,随后通过核纤层与染色质的相互作用可以直接拉伸染色质,上调转录。反之,当破坏肌动蛋白纤维或抑制肌球蛋白收缩可以下调机械力诱导的转录[58]。这些实验结果表明力学信号不需要依赖细胞质内的生物化学信号通道就可以直接诱导基因表达,这同时也为iPSCs重编程效率的提高打开了新思路。 2.4.3 细胞骨架收缩调控表观遗传修饰 细胞骨架的收缩还可以调节染色质修饰酶的表达、活性和核分布,进而引起表观遗传水平上的变化[59-60]。Jain等[60]证明肌动-肌球蛋白的收缩扰动会导致组蛋白脱乙酰激酶3(HDAC3)从细胞质到细胞核的再分配,进而使得赖氨酸9处的组蛋白H3乙酰化减少(AcH3K9)。细胞骨架的组装和收缩引起的染色质修饰改变已被证明会影响干细胞重编程过程[11,61]。Downing等[11]的研究中,由微基底形貌引起的细胞骨架重构抑制了细胞组蛋白脱乙酰激酶2(HDAC2)的活性,并上调了组蛋白H3甲基转移酶亚单位WDR5的表达,进而使组蛋白H3的乙酰化(AcH3)修饰以及H3上赖氨酸4的二甲基化(H3K4me2)和三甲基化(H3K4me3)修饰均显著增强,最终促进了细胞重编程。Yoo等[62]制备的具有纳米沟槽的聚氨酯丙烯酸酯表面能够促进组蛋白H3K4me3修饰,最终有效提高转录因子Ascl1、Pitx3、Nurr1和Lmx1a介导的成纤维细胞向多巴胺神经元细胞的重编程。研究者们也开始利用表观遗传水平上的调控开发新的重编程方法。组蛋白分子伴侣可以调节组蛋白代谢,是染色质修饰的重要调控因子。Syed等[63]首次报道组蛋白分子伴侣Aprataxin PNK-like factor (APLF)的下调可以促进活性组蛋白H3K4me2标记在多能性基因Nanog和Klf4的启动子上,从而增强了iPSCs重编程的效率。 综上,细胞骨架作为整合素和E-钙黏素介导的细胞黏附中的重要结构,以及其本身在多种细胞活动中所处的重要地位,使得其对iPSCs重编程及干性维持的调控具有一定的复杂性。从细胞骨架的结构角度,直接抑制肌动蛋白纤维的聚合可以有效促进重编程过程。细胞骨架将机械力信号传递至核内的机制又可分为以下3个方面:①细胞骨架定位YAP与多能性基因结合;②通过LINC复合物和核纤层建立细胞骨架与染色质的直接连接;③调节染色质修饰酶,引起表观遗传水平上的变化。相信通过对这些机制的进一步清晰探索,将会获得体外重编程技术和再生医学研究的一次技术革新。 文章引用的重要文献见表1。"

| [1] Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126(4):663-676.[2] Kimbrel EA, Lanza R. Current status of pluripotent stem cells: moving the first therapies to the clinic. Nat Rev Drug Discov. 2015;14(10):681-692.[3] Higuchi S, Yoshina S, Mitani S. Inhibition of the integrin signal constitutes a mouse iPS cell niche. Dev Growth Differ. 2016; 58(7):586-599.[4] Murphy WL, McDevitt TC, Engler AJ. Materials as stem cell regulators. Nat Mater. 2014;13(6):547-557.[5] Chowdhury F, Li Y, Poh YC, et al. Soft substrates promote homogeneous self-renewal of embryonic stem cells via downregulating cell-matrix tractions. PLoS One. 2010;5(12): e15655.[6] Yue XS, Fujishiro M, Nishioka C, et al. Feeder cells support the culture of induced pluripotent stem cells even after chemical fixation. PLoS One. 2012;7(3):e32707.[7] Hsu YC, Fuchs E. A family business: stem cell progeny join the niche to regulate homeostasis. Nat Rev Mol Cell Biol. 2012;13(2):103-114.[8] Watt FM. Role of integrins in regulating epidermal adhesion, growth and differentiation. EMBO J. 2002;21(15):3919-3926.[9] Leckband DE, de Rooij J. Cadherin adhesion and mechanotransduction. Annu Rev Cell Dev Biol. 2014;30: 291-315.[10] Hayashi Y, Furue MK, Okamoto T, et al. Integrins regulate mouse embryonic stem cell self-renewal. Stem Cells. 2007; 25(12):3005-3015.[11] Downing TL, Soto J, Morez C, et al. Biophysical regulation of epigenetic state and cell reprogramming. Nat Mater. 2013; 12(12):1154-1162.[12] Lian I, Kim J, Okazawa H, et al. The role of YAP transcription coactivator in regulating stem cell self-renewal and differentiation. Genes Dev. 2010;24(11):1106-1118.[13] Pieters T, van Roy F. Role of cell-cell adhesion complexes in embryonic stem cell biology. J Cell Sci. 2014;127(Pt 12): 2603-2613.[14] Samavarchi-Tehrani P, Golipour A, David L, et al. Functional genomics reveals a BMP-driven mesenchymal-to-epithelial transition in the initiation of somatic cell reprogramming. Cell Stem Cell. 2010;7(1):64-77.[15] Crowder SW, Leonardo V, Whittaker T, et al. Material Cues as Potent Regulators of Epigenetics and Stem Cell Function. Cell Stem Cell. 2016;18(1):39-52.[16] Mitra SK, Schlaepfer DD. Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr Opin Cell Biol. 2006;18(5):516-523.[17] Villa-Diaz LG, Ross AM, Lahann J, et al. Concise review: The evolution of human pluripotent stem cell culture: from feeder cells to synthetic coatings. Stem Cells. 2013;31(1):1-7.[18] Villa-Diaz LG, Kim JK, Laperle A, et al. Inhibition of Focal Adhesion Kinase Signaling by Integrin α6β1 Supports Human Pluripotent Stem Cell Self-Renewal. Stem Cells. 2016;34(7): 1753-1764.[19] Zhang X, Simerly C, Hartnett C, et al. Src-family tyrosine kinase activities are essential for differentiation of human embryonic stem cells. Stem Cell Res. 2014;13(3 Pt A):379-389.[20] Staerk J, Lyssiotis CA, Medeiro LA, et al. Pan-Src family kinase inhibitors replace Sox2 during the direct reprogramming of somatic cells. Angew Chem Int Ed Engl. 2011;50(25):5734-5736.[21] Jeon K, Oh HJ, Lim H, et al. Self-renewal of embryonic stem cells through culture on nanopattern polydimethylsiloxane substrate. Biomaterials. 2012;33(21):5206-5220.[22] Yoo J, Chang Y, Kim H, et al. Efficient Direct Lineage Reprogramming of Fibroblasts into Induced Cardiomyocytes Using Nanotopographical Cues. J Biomed Nanotechnol. 2017;13(3):269-279.[23] Ying QL, Wray J, Nichols J, et al. The ground state of embryonic stem cell self-renewal. Nature. 2008;453(7194): 519-523.[24] Silva J, Barrandon O, Nichols J, et al. Promotion of reprogramming to ground state pluripotency by signal inhibition. PLoS Biol. 2008;6(10):e253.[25] Burdon T, Stracey C, Chambers I, et al. Suppression of SHP-2 and ERK signalling promotes self-renewal of mouse embryonic stem cells. Dev Biol. 1999;210(1):30-43.[26] Li W, Wei W, Zhu S, et al. Generation of rat and human induced pluripotent stem cells by combining genetic reprogramming and chemical inhibitors. Cell Stem Cell. 2009;4(1):16-19.[27] Chen W, Shao Y, Li X, et al. Nanotopographical Surfaces for Stem Cell Fate Control: Engineering Mechanobiology from the Bottom. Nano Today. 2014;9(6):759-784.[28] Li D, Zhou J, Wang L, et al. Integrated biochemical and mechanical signals regulate multifaceted human embryonic stem cell functions. J Cell Biol. 2010;191(3):631-644.[29] Chen G, Hou Z, Gulbranson DR, et al. Actin-myosin contractility is responsible for the reduced viability of dissociated human embryonic stem cells. Cell Stem Cell. 2010;7(2):240-248.[30] Walker A, Su H, Conti MA, et al. Non-muscle myosin II regulates survival threshold of pluripotent stem cells. Nat Commun. 2010;1:71.[31] Rosowski KA, Mertz AF, Norcross S, et al. Edges of human embryonic stem cell colonies display distinct mechanical properties and differentiation potential. Sci Rep. 2015;5: 14218. [32] Higuchi S, Watanabe TM, Kawauchi K, et al. Culturing of mouse and human cells on soft substrates promote the expression of stem cell markers. J Biosci Bioeng. 2014; 117(6):749-755.[33] Lai WH, Ho JC, Lee YK, et al. ROCK inhibition facilitates the generation of human-induced pluripotent stem cells in a defined, feeder-, and serum-free system. Cell Reprogram. 2010;12(6):641-653.[34] Li L, Wang BH, Wang S, et al. Individual cell movement, asymmetric colony expansion, rho-associated kinase, and E-cadherin impact the clonogenicity of human embryonic stem cells. Biophys J. 2010;98(11):2442-2451.[35] Li R, Liang J, Ni S, et al. A mesenchymal-to-epithelial transition initiates and is required for the nuclear reprogramming of mouse fibroblasts. Cell Stem Cell. 2010; 7(1):51-63.[36] Chen T, Yuan D, Wei B, et al. E-cadherin-mediated cell-cell contact is critical for induced pluripotent stem cell generation. Stem Cells. 2010;28(8):1315-1325.[37] Choi B, Park KS, Kim JH, et al. Stiffness of Hydrogels Regulates Cellular Reprogramming Efficiency Through Mesenchymal-to-Epithelial Transition and Stemness Markers. Macromol Biosci. 2016;16(2):199-206.[38] Redmer T, Diecke S, Grigoryan T, et al. E-cadherin is crucial for embryonic stem cell pluripotency and can replace OCT4 during somatic cell reprogramming. EMBO Rep. 2011;12(7): 720-726.[39] Lecuit T, Yap AS. E-cadherin junctions as active mechanical integrators in tissue dynamics. Nat Cell Biol. 2015;17(5): 533-539.[40] Yonemura S, Hirao-Minakuchi K, Nishimura Y. Rho localization in cells and tissues. Exp Cell Res. 2004;295(2): 300-314.[41] Smith AL, Dohn MR, Brown MV, et al. Association of Rho-associated protein kinase 1 with E-cadherin complexes is mediated by p120-catenin. Mol Biol Cell. 2012;23(1):99-110.[42] Bhukhai K, Suksen K, Bhummaphan N, et al. A phytoestrogen diarylheptanoid mediates estrogen receptor/Akt/glycogen synthase kinase 3β protein-dependent activation of the Wnt/β-catenin signaling pathway. J Biol Chem. 2012;287(43): 36168-36178.[43] Zhao A, Yang L, Ma K, et al. Overexpression of cyclin D1 induces the reprogramming of differentiated epidermal cells into stem cell-like cells. Cell Cycle. 2016;15(5):644-653.[44] Yamazaki S, Yamamoto K, de Lanerolle P, et al. Nuclear F-actin enhances the transcriptional activity of β-catenin by increasing its nuclear localization and binding to chromatin. Histochem Cell Biol. 2016;145(4):389-399.[45] Hawkins K, Mohamet L, Ritson S, et al. E-cadherin and, in its absence, N-cadherin promotes Nanog expression in mouse embryonic stem cells via STAT3 phosphorylation. Stem Cells. 2012;30(9):1842-1851.[46] Bedzhov I, Alotaibi H, Basilicata MF, et al. Adhesion, but not a specific cadherin code, is indispensable for ES cell and induced pluripotency. Stem Cell Res. 2013;11(3):1250-1263.[47] del Valle I, Rudloff S, Carles A, et al. E-cadherin is required for the proper activation of the Lifr/Gp130 signaling pathway in mouse embryonic stem cells. Development. 2013;140(8): 1684-1492.[48] Wang N, Tytell JD, Ingber DE. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nat Rev Mol Cell Biol. 2009;10(1):75-82.[49] Sakurai K, Talukdar I, Patil VS, et al. Kinome-wide functional analysis highlights the role of cytoskeletal remodeling in somatic cell reprogramming. Cell Stem Cell. 2014;14(4): 523-534.[50] Dupont S, Morsut L, Aragona M, et al. Role of YAP/TAZ in mechanotransduction. Nature. 2011;474(7350):179-183.[51] Tamm C, Böwer N, Annerén C. Regulation of mouse embryonic stem cell self-renewal by a Yes-YAP-TEAD2 signaling pathway downstream of LIF. J Cell Sci. 2011;124(Pt 7):1136-1144.[52] Ungricht R, Kutay U. Mechanisms and functions of nuclear envelope remodelling. Nat Rev Mol Cell Biol. 2017;18(4): 229-245.[53] Ihalainen TO, Aires L, Herzog FA, et al. Differential basal-to-apical accessibility of lamin A/C epitopes in the nuclear lamina regulated by changes in cytoskeletal tension. Nat Mater. 2015;14(12):1252-1261.[54] Swift J, Ivanovska IL, Buxboim A, et al. Nuclear lamin-A scales with tissue stiffness and enhances matrix-directed differentiation. Science. 2013;341(6149):1240104.[55] Solovei I, Wang AS, Thanisch K, et al. LBR and lamin A/C sequentially tether peripheral heterochromatin and inversely regulate differentiation. Cell. 2013;152(3):584-598.[56] Zuo B, Yang J, Wang F, et al. Influences of lamin A levels on induction of pluripotent stem cells. Biol Open. 2012;1(11): 1118-1127.[57] Iyer KV, Pulford S, Mogilner A, et al. Mechanical activation of cells induces chromatin remodeling preceding MKL nuclear transport. Biophys J. 2012;103(7):1416-1428.[58] Tajik A, Zhang Y, Wei F, et al. Transcription upregulation via force-induced direct stretching of chromatin. Nat Mater. 2016; 15(12):1287-1296.[59] Heo SJ, Thorpe SD, Driscoll TP, et al. Biophysical Regulation of Chromatin Architecture Instills a Mechanical Memory in Mesenchymal Stem Cells. Sci Rep. 2015;5:16895.[60] Jain N, Iyer KV, Kumar A, et al. Cell geometric constraints induce modular gene-expression patterns via redistribution of HDAC3 regulated by actomyosin contractility. Proc Natl Acad Sci U S A. 2013;110(28):11349-11354.[61] Kulangara K, Adler AF, Wang H, et al. The effect of substrate topography on direct reprogramming of fibroblasts to induced neurons. Biomaterials. 2014;35(20):5327-5336.[62] Yoo J, Noh M, Kim H, et al. Nanogrooved substrate promotes direct lineage reprogramming of fibroblasts to functional induced dopaminergic neurons. Biomaterials. 2015;45:36-45.[63] Syed KM, Joseph S, Mukherjee A, et al. Histone chaperone APLF regulates induction of pluripotency in murine fibroblasts. J Cell Sci. 2016;129(24):4576-4591. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||