Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (31): 5027-5033.doi: 10.3969/j.issn.2095-4344.0566
Previous Articles Next Articles
Application of three-dimensional printing metal implants in orthopedics
Zhao Min-chao1, Huang Yan1, Yuan Wei-jian2, Chi Yu-fei3, Li Zu-hao2, Wu Han1
- 1Department of Orthopedics, China-Japan Union Hospital of Jilin University, Changchun 130033, Jilin Province, China; 2Department of Orthopedics, the Second Hospital of Jilin University, Changchun 130041, Jilin Province, China; 3Henan University of Science and Technology, Luoyang 471000, Henan Province, China
-
Online:2018-11-08Published:2018-11-08 -
Contact:Wu Han, Chief physician, Professor, Department of Orthopedics, China-Japan Union Hospital of Jilin University, Changchun 130033, Jilin Province, China Corresponding author: Li Zu-hao, Master candidate, Department of Orthopedics, the Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Zhao Min-chao, Master candidate, Department of Orthopedics, China-Japan Union Hospital of Jilin University, Changchun 130033, Jilin Province, China -
Supported by:the International Cooperation Project of Ministry of Science, No. 2014DFG52510; the “Double-Ten Engineering” Major Science and Technology Research and Development Project of Jilin Provincial Science and Technology Supporting Program, No. 20130201005GX
CLC Number:
Cite this article
Zhao Min-chao, Huang Yan, Yuan Wei-jian, Chi Yu-fei, Li Zu-hao, Wu Han. Application of three-dimensional printing metal implants in orthopedics [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(31): 5027-5033.
share this article

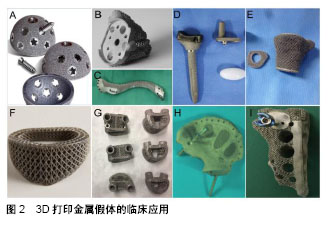
2.1 临床应用 近年来,3D打印技术在骨科中得到了广泛应用。对于一些特殊的患者,如骨肿瘤切除术后和严重骨缺损的翻修手术,外科医生必须延长切口或增加植骨量以保证植入物的稳定性和匹配性。延长切口会加重机体损伤,不利于恢复。自体移植是治疗骨缺损的金标准,但其应用受限于供区并发症、恢复时间长、成本高和手术造成的组织损伤大等诸多因素[15-17]。此外,异种移植物也有免疫排斥和感染的风险[18-19]。3D打印技术可以模拟手术前的情况,并设计个体化的假体用于植入。该部分总结了相关病例,探讨3D打印在临床的应用前景。 在3D打印关节方面,由于近年来人工髋关节翻修术的增多,复杂的髋臼缺损甚至骨盆不连续变得更为常见,3D打印髋臼假体在翻修手术中得到了极大的关注,且3D打印髋臼假体已获准上市(图2A)。多个研究结果表明,3D打印髋臼假体可显著改善Harris髋关节评分,恢复髋关节中心,减少手术并发症,尤其是严重的缺损[20-22]。3D打印全肩关节假体尚未见报道,但肩关节周围部位,如关节盂、锁骨、肩胛骨等已有随访2年以上的病例。Stoffelen等[23]研制出一种3D打印关节盂用于肩关节置换术后严重的关节盂缺损。多孔结构良好地填充了骨缺损,并且部件的附加螺纹孔允许转换成反向构型。该病例在2.5年随访中获得了良好的功能评分(图2B)。Fan等[24]应用3D打印技术治疗位于锁骨尤文肉瘤(图2C)。多孔结构减少了弹性模量,相邻结构被不可吸收缝线固定到假体的小孔上,术后获得了良好的美国骨肿瘤学会评分,无术后并发症产生。吉林大学第二医院王金成教授团队报道了多例应用3D打印金属假体质量建肿瘤切除术的关节功能,具有仿生结构的Co-Cr-Mo合金假体完美填充了桡骨远端巨细胞瘤切除术后产生的骨缺损,关节面通过表面拟合,以确保腕关节运动轨迹的流畅(图2D),平均11个月的随访提示结果满意[12]。此外该团队还为膝关节巨细胞瘤切除后的骨缺损制备了3D打印钛合金胫骨端微孔垫块,为侧副韧带、髌韧带和骨质提供附着点和长入点(图2E),使非限制型膝关节假体替代限制型膝关节肿瘤假体、重建胫骨上端成为可能[13]。 在脊柱方面,北医三院刘忠军教授团队与爱康宜城公司合力设计的3D打印椎体已获准上市。他们针对上颈椎(C2)尤文氏肉瘤进行了3D打印定制钛合金椎体的重建,并且优化了内植物的显微结构,获得了生物力学稳定性、骨愈合的孔隙率、支柱尺寸之间超微结构的平衡。术后CT提示骨性融合良好,未见植入物下沉或移位[25]。西安交通大学第二附属医院贺西京教授团队也已将3D打印的寰枢椎钢板及人工寰枢椎应用于临床。此外,澳大利亚Mobbs教授团队也证明3D打印金属内植物在脊柱肿瘤切除术后重建中获得满意效果(图2F,G)[26-27]。 由于自身结构的复杂不规则、过大的承载负荷、病变缺损的广泛以及深部具有重要组织结构,使得骨盆肿瘤切除术后重建一直是巨大的挑战[28-29],传统的重建方法无法达到满意的结果[30]。3D打印技术在骨盆肿瘤切除后的缺损重建中具有显著地优势,减少了手术创伤、实现了早期下地,精准的个体化匹配获得良好的功能恢复。3D打印金属假体在骶骨骨肉瘤半骶骨切除术后重建(图2H)[31]、骶骨脊索瘤术后重建(图2I)中均获得了满意结果[32]。 总的来说,3D打印金属内植物临床应用的中短期随访表明该技术可使假体与骨缺损精确匹配,实现早期稳定性,内植物与宿主骨结合牢固,假体松动等并发症少见,获得了较满意的效果。 "
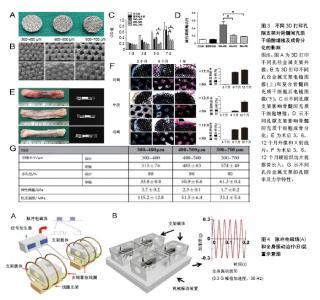
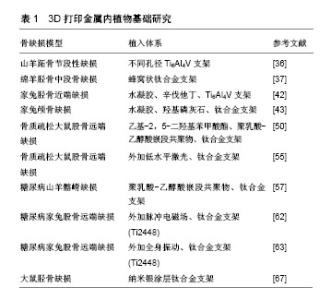
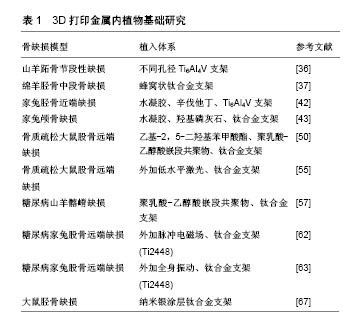
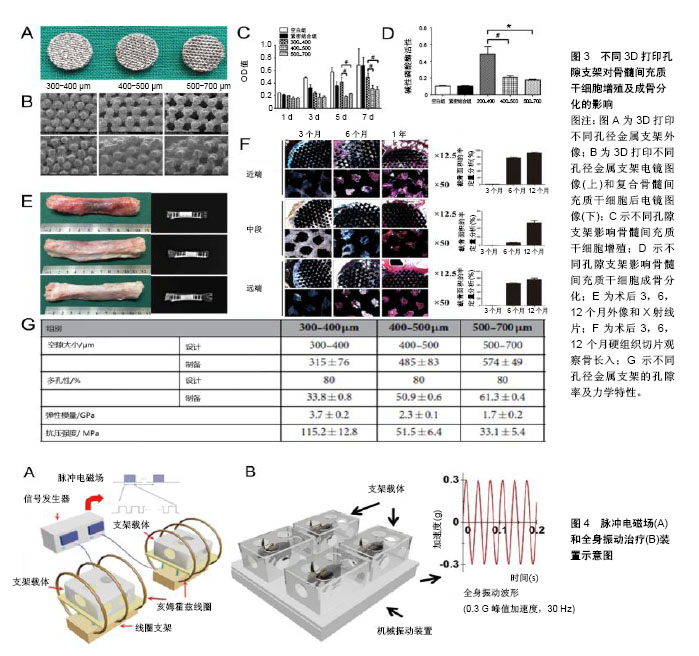
2.2.1 正常状态下骨缺损 3D打印金属支架孔径是影响骨长入的一个重要因素,通过优化孔径可以促进骨整合。以往研究表明,骨长入的最佳孔径范围为100-500 μm[34]。另外有研究推荐孔径大于300 μm的支架可以更好地促进新骨形成和毛细血管生成[35]。Li等[36]利用3D打印技术制备了3种不同孔径的钛合金支架(300-400,400-500,500-700 μm),体外与骨髓间充质干细胞共培养发现孔径300-400 μm的支架更利于骨髓间充质干细胞增殖和成骨分化,在山羊体内3 cm长的节段性缺损中植入不同规格孔径的支架,可见孔径为300-400 μm的支架有明显的骨长入。除了孔径直径,孔隙形状的改变也可以改善微孔支架的力学和成骨性能。Pobloth等[37]采用蜂窝状结构的设计使应力屏蔽最小化,同时确保避免了机械失效,这种优化了孔隙形状的钛网很好的促进了羊胫骨中段骨缺损的愈合。同时通过计算机有限元设计与优化也应用于改善支架性能[38]。 在孔隙支架中封装有利于骨再生的药物,并使其在局部组织中缓释,是促进骨生长愈合一种有潜力的研究方向。辛伐他汀作为一种常见的血酯调节剂,局部给药可促进骨形成和骨缺损愈合[39-40]。水凝胶作为细胞载体或者药物缓释体系都可以明显促进缺损组织再生[41]。3D打印多孔Ti6Al4V支架填充负载辛伐他汀的泊洛沙姆407水凝胶植入家兔胫骨近端骨缺损,术后第8周0.1 mg和0.5 mg辛伐他丁组支架内骨体积对比对照组分别为16.09% 和31.35%,且有显著的骨密度提高,结果证明该复合支架可显著改善骨整合、促进骨长入与新生血管形成[42]。复合明胶凝胶和纳米羟基磷灰石的多孔钛合金支架可明显促进成骨细胞前体MC3TC-E1细胞的增殖黏附和成骨分化,在骨缺损模型中明胶与羟基磷灰石按1∶1混合的支架可以明显加速颅骨缺损的愈合,成骨相关基因和蛋白的表达也明显增加[43]。与此相似Kumar等[44]制备了一种由海藻酸钠、明胶和纳米羟基磷灰石组成的水凝胶搭载成骨细胞,在3D打印的多孔结构中观察到存在细胞外基质样水凝胶有利于 成骨细胞的黏附、增殖、成骨能力,相关研究结果见图3。 2.2.2 病理状态下骨缺损 在一些病理状态下,如骨质疏松、糖尿病、感染等,假体植入通常会导致骨愈合时间延长、再生骨质量差、易发生再骨折、植入物下沉或松动,以及长期卧床导致的一些并发症,如坠积性肺炎和压疮 等[45-48]。尽管3D打印金属支架较传统支架更易促进骨长入,但是对3D打印金属内植物的优化,将进一步促进支架-宿主骨整合,获得最佳效果。 骨质疏松症是一种以低骨量为特征的疾病,最常见的是由于骨吸收速度增加与骨形成速度不相匹配。骨质疏松性骨缺损中不平衡骨重建是造成内植物失败的主要原因,骨质与内植物结合不牢固易造成植入物松动,导致假体周围骨折[49]。乙基-2,5-二羟基苯甲酸酯作为一种促进骨形成和抑制骨吸收的药物,被聚乳酸-乙醇酸嵌段共聚物封装后以涂层形式存在于钛合金空隙支架表面。复合支架植入骨质疏松大鼠股骨缺损后第4,8和12周微CT检测显示支架周围骨吸收减少,骨形成增加。因此,证实在骨质疏松模型中,这种具有药物缓释作用的3D打印钛合金支架可使骨愈合增加,内植物稳定性增强[50]。一些抗骨质疏松药物常被添加到支架材料中以促进骨质疏松性骨缺损的愈合。掺杂锶(Sr)和双磷酸盐的泡沫铁支架可以促进骨质疏松性骨折的愈合[51]。低水平激光治疗对骨丢失严重、骨代谢紊乱影响骨重建的患者可以有效刺激骨生长[52]。此外,低水平激光治疗能够加速新骨形成,从而增加成骨细胞活性,加快血管化和胶原纤维的分布,提高抗菌和免疫效果[53]。低水平激光治疗的其他积极作用还包括在内植物周围进行的血流刺激、改善植入物与骨表面之间的接触以及促进更快的骨成熟[54]。在骨质疏松大鼠股骨缺损处植入钛合金支架之后体外给予低水平激光治疗可以明显加速钛合金支架内的骨修复,且在非骨质疏松大鼠缺损中也得到了相似结果[55]。 糖尿病相关的骨内植入物有较高的失败风险,需要有效的方法来改善骨植入物界面的骨整合[56]。具有聚乳酸-乙醇酸嵌段共聚物涂层的3D打印钛合金支架对糖尿病坏境中的成骨细胞具有保护作用,其降解产物乳酸促进糖尿病骨缺损中的血管形成[57]。糖尿病的存在与骨质疏松的发生关系密切[58],外加脉冲电磁场的特异性波形抑制骨质疏松动物的骨丢失,并促进成骨细胞的活性和功能[59-60]。在健康家兔股骨缺损处植入3D打印的多孔Ti6Al4V支架后给予脉冲电磁场治疗对促进骨再生的能力已被证明(图4A)[61]。但脉冲电磁场能否促进病理状态下的骨缺损修复还有待进一步研究。Cai等[62]在1型糖尿病家兔股骨缺损处植入钛合金支架后1周开始进行为期8周的体外脉冲电磁场治疗。与对照组相比,脉冲电磁场治疗通过激活成功相关的Wnt/β-catenin信号通路而不影响破骨相关的RANKL/RANK通路,明显促进了糖尿病骨缺损的骨整合。此外,对同样是股骨缺损处植入相同成分钛合金支架的糖尿病家兔给以全身振动治疗同样可以激活Wnt/β-catenin通路,从而促进1型糖尿病兔骨代谢、骨组织学和多孔钛种植体骨整合(图4B)[63]。 假体感染仍然是骨科术后的一个严重并发症,感染的骨缺损中由于系统性抗生素治疗没有达到特定的作用位点而迁延不愈[64]。研究者用许多方法改良内植物表面从而达到抑制细菌黏附和生长的目的。纳米银可有效预防细菌黏附、生物膜形成和后续的假体感染[65-66]。将纳米银涂层的3D打印钛合金支架植入大鼠胫骨缺损中,骨-内植物界面的纳米级元素图谱和透射电子显微镜显示银主要存在于骨组织中,并与硫共定位,很好的验证了纳米银在体内缓释,在不影响成骨的同时具有抗菌的潜力[67]。"
| [1] Holzapfel BM, Pilge H, Toepfer A, et al. Proximal tibial replacement and alloplastic reconstruction of the extensor mechanism after bone tumor resection. Oper Orthop Traumatol. 2012;24(3):247-262. [2] Papagelopoulos PJ, Savvidou OD, Koutsouradis P, et al. three-dimensional technologies in orthopedics. Orthopedics. 2018;41(1):12-20. [3] Lee SW, Kim HG, Ham MJ, et al. Custom implant for reconstruction of mandibular continuity defect. J Oral Maxillofac Surg. 2018;76(6):1370-1376. [4] Mohan Raj R, Priya P, Raj V. Gentam-loaded ceramic-biopolymer dual layer coatings on the Ti with improved bioactive and corrosion resistance properties for orthopedic applications. J Mech Behav Biomed Mater. 2018;82:299-309. [5] Heller M, Bauer HK, Goetze E, et al. Applications of patient- specific 3D printing in medicine. Int J Comput Dent. 2016;19(4): 323-339. [6] Gross BC, Erkal JL, Lockwood SY, et al. Evaluation of 3D printing and its potential impact on biotechnology and the chemical sciences. Anal Chem. 2014;86(7):3240-3253. [7] Derby B. Printing and prototyping of tissues and scaffolds. Science. 2012;338(6109):921-926. [8] Farzadi A, Waran V, Solati-Hashjin M, et al. Effect of layer printing delay on mechanical properties and dimensional accuracy of 3D printed porous prototypes in bone tissue engineering. Ceram Int. 2015;41(7):8320-8330. [9] Le Cann S, Galland A, Rosa B, et al. Does surface roughness influence the primary stability of acetabular cups? A numerical and experimental biomechanical evaluation. Med Eng Phys. 2014; 36(9):1185-1190. [10] Chikarakara E, Fitzpatrick P, Moore E, et al. In vitro fibroblast and pre-osteoblastic cellular responses on laser surface modified Ti-6Al-4V. Biomed Mater. 2015;10(1):015007. [11] Chang B, Song W, Han TX, et al. Influence of pore size of porous titanium fabricated by vacuum diffusion bonding of titanium meshes on cell penetration and bone ingrowth. Acta Biomater. 2016;33:311-321. [12] Han Q, Qin YG, Zou Y, et al. Novel exploration of 3D printed wrist arthroplasty to solve the severe and complicated bone defect of wrist. Rapid Prototyping J. 2017;23(3):465-473. [13] Luo W, Huang L, Liu H, et al. Customized knee prosthesis in treatment of giant cell tumors of the proximal tibia: application of 3-dimensional printing technology in surgical design. Med Sci Monit. 2017;23:1691-1700. [14] Variola F, Brunski JB, Orsini G, et al. Nanoscale surface modifications of medically relevant metals: state-of-the art and perspectives. Nanoscale. 2011;3(2):335-353. [15] Fahmy-Garcia S, van Driel M, Witte-Buoma J, et al. NELL-1, HMGB1, and CCN2 enhance migration and vasculogenesis, but not osteogenic differentiation compared to BMP2. Tissue Engineering Part A. 2018;24(3-4):207-218. [16] Boriani F, Fazio N, Fotia C, et al. A novel technique for decellularization of allogenic nerves and in vivo study of their use for peripheral nerve reconstruction. J Biomed Mater Res A. 2017;105(8):2228-2240. [17] Prosthesi Campana V, Milano G, Pagano E, et al. Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med. 2014;25(10):2445-2461. [18] Hosny HAH, El-Bakoury A, Fekry H, et al. Mid-term results of graft augmentations ii cage and impacted allograft bone in revision hip arthroplasty. J Arthroplasty. 2018;33(5):1487-1493. [19] Waern JM, Yuan Q, Rudrich U, et al. Ectopic expression of murine CD47 minimizes macrophage rejection of human hepatocyte xenografts in immunodeficient mice. Hepatology. 2012;56(4): 1479-1488. [20] Li H, Qu X, Mao Y, et al. Custom Acetabular cages offer stable fixation and improved hip scores for revision tha with severe bone defects. Clin Orthop Relat Res. 2016;474(3):731-740. [21] Li HW, Wang L, Mao YQ, et al. Revision of complex acetabular defects using cages with the aid of rapid prototyping. J Arthroplasty. 2013;28(10):1770-1775. [22] Berasi CC, Berend KR, Adams JB, et al. Are custom triflange acetabular components effective for reconstruction of catastrophic bone loss? Clin Orthop Relat R. 2015;473(2):528-535. [23] Stoffelen DV, Eraly K, Debeer P. The use of 3D printing technology in reconstruction of a severe glenoid defect: a case report with 2.5 years of follow-up. J Shoulder Elb Surg. 2015; 24(8):E218-E222. [24] Fan HB, Fu J, Li XD, et al. Implantation of customized 3-D printed titanium prosthesis in limb salvage surgery: a case series and review of the literature. World J Surg Oncol. 2015;13:308. [25] Xu N, Wei F, Liu X, et al. Reconstruction of the upper cervical spine using a personalized 3d-printed vertebral body in an adolescent with ewing sarcoma. Spine. 2016;41(1):E50-E54. [26] Phan K, Sgro A, Maharaj MM, et al. Application of a 3D custom printed patient specific spinal implant for C1/2 arthrodesis. J Spine Surg. 2016;2(4):314-318. [27] Choy WJ, Mobbs RJ, Wilcox B, et al. Reconstruction of the thoracic spine using a personalized 3D-printed vertebral body in an adolescent with a T9 primary bone tumour: case report. World Neurosurg. 2017;105:1032. e13-1032. e17. [28] Jaiswal PK, Aston WJS, Grimer RJ, et al. Peri-acetabular resection and endoprosthetic reconstruction for tumours of the acetabulum. J Bone Joint Surg Br. 2008;90B(9):1222-1227. [29] Zoccali C, Skoch J, Patel A, et al. The surgical neurovascular anatomy relating to partial and complete sacral and sacroiliac resections: a cadaveric, anatomic study. Eur Spine J. 2015; 24(5):1109-1113. [30] Puchner SE, Funovics PT, Bohler C, et al. Oncological and surgical outcome after treatment of pelvic sarcomas. Plos One. 2017;12(2):e0172203. [31] Kim D, Lim JY, Shim KW, et al. Sacral Reconstruction with a 3D-Printed Implant after Hemisacrectomy in a Patient with Sacral Osteosarcoma: 1-Year Follow-Up Result. Yonsei Med J. 2017; 58(2):453-457. [32] Wei R, Guo W, Ji T, et al. One-step reconstruction with a 3D-printed, custom-made prosthesis after total en bloc sacrectomy: a technical note. Eur Spine J. 2017;26(7):1902-1909. [33] Yamamoto K, Yamaguchi S, Matsushita T, et al. Histologic evaluation of bone regeneration using titanium mesh prepared by selective laser melting technique. J Hard Tissue Biol. 2017;26(3): 257-260. [34] Mishra S, Tate MLK. Effect of lacunocanalicular architecture on hydraulic conductance in bone tissue: Implications for bone health and evolution. Anat Rec Part A. 2003;273A(2):752-762. [35] Kujala S, Ryhanen J, Danilov A, et al. Effect of porosity on the osteointegration and bone ingrowth of a weight-bearing nickel-titanium bone graft substitute. Biomaterials. 2003;24(25): 4691-4697. [36] Li GY, Wang L, Pan W, et al. In vitro and in vivo study of additive manufactured porous Ti6Al4V scaffolds for repairing bone defects. Sci Rep. 2016;6:34072. [37] Pobloth AM, Checa S, Razi H, et al. Mechanobiologically optimized 3D titanium-mesh scaffolds enhance bone regeneration in critical segmental defects in sheep. Sci Transl Med. 2018; 10(423). pii: eaam8828. [38] Luo DM, Rong QG, Chen Q. Finite-element design and optimization of a three-dimensional tetrahedral porous titanium scaffold for the reconstruction of mandibular defects. Med Eng Phys. 2017;47:176-183. [39] Zhao SF, Wen F, He FM, et al. In vitro and in vivo evaluation of the osteogenic ability of implant surfaces with a local delivery of simvastatin. Int J Oral Max Impl. 2014;29(1):211-220. [40] Tan J, Yang N, Fu X, et al. Single-dose local simvastatin injection improves implant fixation via increased angiogenesis and bone formation in an ovariectomized rat model. Med Sci Monitor. 2015; 21:1428-1439. [41] Liu H, Wang C, Li C, et al. A functional chitosan-based hydrogel as a wound dressing and drug delivery system in the treatment of wound healing. RSC Advances. 2018;8(14):7533-7549. [42] Liu H, Li W, Liu C, et al. Incorporating simvastatin/poloxamer 407 hydrogel into 3D-printed porous Ti6Al4V scaffolds for the promotion of angiogenesis, osseointegration and bone ingrowth. Biofabrication. 2016;8(4):045012. [43] Yin B, Xue BJ, Wu ZH, et al. A novel hybrid 3D-printed titanium scaffold for osteogenesis in a rabbit calvarial defect model. Am J Transl Res. 2018;10(2):474-482. [44] Kumar A, Nune KC, Misra RDK. Design and biological functionality of a novel hybrid Ti-6Al-4V/hydrogel system for reconstruction of bone defects. J Tissue Eng Regen Med. 2018; 12(4):1133-1144. [45] Kondiah PJ, Choonara YE, Kondiah PP, et al. A review of injectable polymeric hydrogel systems for application in bone tissue engineering. Molecules. 2016;21(11):E1580. [46] Sui BD, Hu CH, Liu AQ, et al. Stem cell-based bone regeneration in diseased microenvironments: Challenges and solutions. Biomaterials. 2017. [47] Cotts KG, Cifu AS. Treatment of osteoporosis. JAMA. 2018; 319(10):1040-1041.[48] Fujiwara S, Zhao X, Teoh C, et al. Disease burden of fractures among patients with osteoporosis in Japan: health-related quality of life, work productivity and activity impairment, healthcare resource utilization, and economic costs. J Bone Miner Metab. 2018. [49] Cheng N, Dai J, Cheng XR, et al. Porous CaP/silk composite scaffolds to repair femur defects in an osteoporotic model. J Mater Sci Mater Med. 2013;24(8):1963-1975. [50] Kwon BJ, Seon GM, Lee MH, et al. Locally delivered ethyl-2, 5-dihydroxybenzoate using 3d printed bone implant for promotion of bone regeneration in a osteoporotic animal model. Eur Cells Mater. 2018;35:1-12. [51] Ray S, Thormann U, Eichelroth M, et al. Strontium and bisphosphonate coated iron foam scaffolds for osteoporotic fracture defect healing. Biomaterials. 2018;157:1-16.[52] Khadra M. The effect of low level laser irradiation on implant-tissue interaction. In vivo and in vitro studies. Swed Dent J Suppl. 2005;(172):1-63. [53] Fukuhara E, Goto T, Matayoshi T, et al. Optimal low-energy laser irradiation causes temporal G2/M arrest on rat calvarial osteoblasts. Calcif Tissue Int. 2006;79(6):443-450. [54] Boldrini C, de Almeida JM, Fernandes LA, et al. Biomechanical effect of one session of low-level laser on the bone-titanium implant interface. Lasers Med Sci. 2013;28(1):349-352. [55] de Vasconcellos LMR, Barbara MAM, Rovai ED, et al. Titanium scaffold osteogenesis in healthy and osteoporotic rats is improved by the use of low-level laser therapy (GaAlAs). Lasers Med Sci. 2016;31(5):899-905. [56] Ormianer Z, Block J, Matalon S, et al. The effect of moderately controlled type 2 diabetes on dental implant survival and peri-implant bone loss: a long-term retrospective study. Int J Oral Maxillofac Implants. 2018;33(2):389-394. [57] Hu XF, Feng YF, Xiang G, et al. Lactic acid of PLGA coating promotes angiogenesis on the interface between porous titanium and diabetic bone. J Mater Chem B. 2018;6(15):2274-2288. [58] Weber DR, Schwartz G. Epidemiology of skeletal health in type 1 diabetes. Curr Osteoporos Rep. 2016;14(6):327-336. [59] Zhai MM, Jing D, Tong SC, et al. Pulsed electromagnetic fields promote in vitro osteoblastogenesis through a Wnt/-catenin signaling-associated mechanism. Bioelectromagnetics. 2016; 37(3):152-162. [60] Jing D, Shen GH, Huang JH, et al. Circadian rhythm affects the preventive role of pulsed electromagnetic fields on ovariectomy- induced osteoporosis in rats. Bone. 2010;46(2):487-495. [61] Jing D, Zhai MM, Tong SC, et al. Pulsed electromagnetic fields promote osteogenesis and osseointegration of porous titanium implants in bone defect repair through a Wnt/beta-catenin signaling-associated mechanism. Sci Rep. 2016;6:32045. [62] Cai J, Li W, Sun T, et al. Pulsed electromagnetic fields preserve bone architecture and mechanical properties and stimulate porous implant osseointegration by promoting bone anabolism in type 1 diabetic rabbits. Osteoporos Int. 2018;29(5):1177-1191. [63] Jing D, Yan ZD, Cai J, et al. Low-1 level mechanical vibration improves bone microstructure, tissue mechanical properties and porous titanium implant osseointegration by promoting anabolic response in type 1 diabetic rabbits. Bone. 2018, 106:11-21. [64] Stolzing A, Jones E, McGonagle D, et al. Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mech Ageing Dev. 2008;129(3): 163-173. [65] Schacht VJ, Neumann LV, Sandhi SK, et al. Effects of silver nanoparticles on microbial growth dynamics. J Appl Microbiol. 2013;114(1):25-35. [66] Devlin-Mullin A, Todd NM, Golrokhi Z, et al. Atomic layer deposition of a silver nanolayer on advanced titanium orthopedic implants inhibits bacterial colonization and supports vascularized de novo bone ingrowth. Advanced Healthcare Materials. 2017. [67] Geng H, Poologasundarampillai G, Todd N, et al. Biotransformation of silver released from nanoparticle coated titanium implants revealed in regenerating bone. Acs Appl Mater Inter. 2017;9(25):21169-21180. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [5] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Liu Jianyou, Jia Zhongwei, Niu Jiawei, Cao Xinjie, Zhang Dong, Wei Jie. A new method for measuring the anteversion angle of the femoral neck by constructing the three-dimensional digital model of the femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(24): 3779-3783. |
| [14] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [15] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||