Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (17): 2769-2774.doi: 10.3969/j.issn.2095-4344.0497
Previous Articles Next Articles
Embryonic stem cells and construction of a genetic disease model
Qiu Jia-hui, Tan Ji-chun
- Second Department of Reproduction, Reproductive Center of Shengjing Hospital, China Medical University, Shenyang 110022, Liaoning Province, China
-
Revised:2018-01-15Online:2018-06-18Published:2018-06-18 -
Contact:Tan Ji-chun, M.D., Doctoral supervisor, Second Department of Reproduction, Reproductive Center of Shengjing Hospital, China Medical University, Shenyang 110022, Liaoning Province, China -
About author:Qiu Jia-hui, Master candidate, Second Department of Reproduction, Reproductive Center of Shengjing Hospital, China Medical University, Shenyang 110022, Liaoning Province, China -
Supported by:the National High-Technology Research and Development Plan of China (863 Program) in 2015, No. 2015AA020407
CLC Number:
Cite this article
Qiu Jia-hui, Tan Ji-chun. Embryonic stem cells and construction of a genetic disease model[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(17): 2769-2774.
share this article
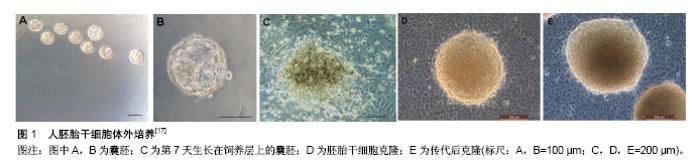
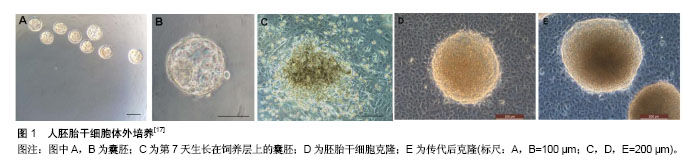
2.1 胚胎干细胞 2.1.1 胚胎干细胞的特征 胚胎干细胞是由哺乳类动物着床前囊胚内细胞团在体外特定条件下培养和扩增所获得的一种多能性干细胞。早在20世纪80年代初Evans等[1]就成功地从小鼠囊胚内分离出了小鼠胚胎干细胞。Martin等[2]也几乎同时建立了小鼠胚胎干细胞系。随后研究者又分离出其他哺乳动物的胚胎干细胞。而人胚胎干细胞的分离却没预想中顺利,1994年Bongso等[3]从人囊胚的内细胞团分离出了人胚胎干细胞,但是很遗憾并没有建系成功。直到1998年,研究者才从体外受精5 d的人囊胚中成功分离出世界首株人胚胎干细胞[4]。胚胎干细胞具有两大基本特征:无限的自我更新能力和分化的多能性。在体外培养过程中,胚胎干细胞可以长期进行自我复制,并保留其多向分化潜能。目前,胚胎干细胞主要应用于早期胚胎发育的研究、再生医学、疾病模型的构建等。 2.1.2 胚胎干细胞的获取与培养 随着技术的发展,目前已有成百上千的人胚胎干细胞系被建立。人胚胎干细胞来源于囊胚期的内细胞团,其获取主要有两种方式:免疫手术法、机械分离法。免疫手术法中使用了动物源性的产物,容易引起动物源性的污染,导致获取的人胚胎干细胞在后续应用中受限。机械手术法有效地避免了该问题,更适合有临床用途的人胚胎干细胞的培养[5]。此外,激光打孔法也被应用到内细胞团的分离上[6]。 人胚胎干细胞的体外培养过程既要保持胚胎干细胞自我更新能力又要维持其多能性,因而人胚胎干细胞的培养需要特殊的培养液及饲养层支持。常常需要在培养液中加入一些可以抑制胚胎干细胞分化的因子,如白细胞抑制因子。研究表明饲养层也可分泌特殊的生长因子以维持胚胎干细胞特征[7-11]。去除这些可以抑制胚胎干细胞分化的因子,通过体外悬浮培养,胚胎干细胞可分化为包含三胚层组织结构的拟胚体[12]。1998年第1例人胚胎干细胞的建系选择了小鼠胚胎成纤维细胞作为饲养层[4]。该种饲养层一直被沿用至今,但是为了排除动物源性的干扰,在人胚胎干细胞的培养中(图1),人们也开始尝试利用人源性的细胞作为饲养层,如输卵管上皮细胞、胎儿肌肉细胞、骨髓、羊膜上皮、人皮肤成纤维细胞、胎盘成纤维细胞等[13-17]。近年来,研究者也开始尝试无饲养层培养方法,通过在培养瓶中加入适量的matrigel,并在培养基加入一定的生长因子,经实验证实该无饲养层培养条件能使胚胎干细胞在多次传代后维持其形态及多能性,并具有稳定的核型,能够形成胚体分化成内胚层、外胚层、中胚层[18]。胚胎干细胞的体外培养是一个十分复杂且精细的过程,早期的一些研究中体外培养的胚胎干细胞只具有胚内发育潜能。2017年1月美国加州大学伯克利分校何琳教授研究团队发表在《Science》的一篇研究论文报道中通过移除一种名叫miR-34a的微RNA,成功使老鼠胚胎干细胞表现出类似受精卵的发育特性,能够分化成胚胎组织和胚胎外组织[19]。而2017年4月《Cell》上发表的一篇文章的研究结果突破了人胚胎干细胞体外培养的局限性,在该研究中邓宏魁等通过筛选建立了一种被称为LCDM的培养体系,这个培养体系可诱导产生具有胚内和胚外发育潜能的小鼠和人干细胞系,该团队将其称为扩展多能干细胞[20]。最新的一项研究表明胚胎干细胞的潜能性可能与pramel7蛋白的表达相关,该蛋白通过介导DNA的甲基化而决定胚胎干细胞的分化状态,因而体外培养过程种可通过影响pramel7的表达而使体外培养的胚胎干细胞具有“全能性”[21-22]。"
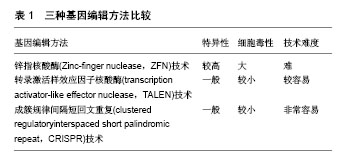
2.1.3 胚胎干细胞的细胞学鉴定 通过对胚胎干细胞的基本形态、生长特点、核型、多能性以及表面标记等特点进行鉴定。通常包括以下几个方面:①细胞的来源;②细胞在体外培养过程中能维持稳定增殖而不分化的状态;③体外长期培养后细胞核型分析;④体内或体外多向分化潜能;⑤OCT-4、NANOG、SOX2等基因表达情况[23-24]。目前鉴定胚胎干细胞的常用生物学方法包括碱性磷酸酶染色实验、细胞免疫荧光、流式细胞技术、拟胚体实验、体外畸胎瘤形成实验、核型分析等。 2.2 利用人胚胎干细胞构建遗传病的意义 遗传病是以遗传物质变化为基本特征的疾病,具有先天性、终身性和家族性的特点。中国是遗传病的高发国家,有1/4-1/5的人患有各种类型的遗传病,每年出生的先天残疾儿总数高达80万-120万,占每年出生总人口的4%-6%。遗传病严重降低了患儿的生活质量,给家庭及社会造成严重的经济、精神负担。目前已发现的7 000种罕见病中,有80%是由遗传变异引起的,严重威胁人类的健康。根据遗传物质在细胞不同层次的改变,可将其分为染色体病、单基因病、多基因病、体细胞遗传病。目前,遗传病的研究手段主要有细胞模型、动物模型的构建以及分子水平研究等。由于小鼠基因编辑的易操作性,与体外细胞模型相比,小鼠模型可获得更全面的信息,小鼠模型的应用较为广泛。但是由于物种特异性,人和小鼠在发育、遗传、生理上存在很大差异[25]。这导致通过基因编辑构建的人类遗传病小鼠模型有时无法产生与人类相同的表型。如具有囊性纤维化变异的小鼠在出现呼吸症状前常死于肠梗阻,基因编辑所获得的小鼠Lesch-Nyham综合征模型常不出现和人类相似的症状[26]。非整倍体常导致人类流产,而有单个X染色体的小鼠却有可能存活[27]。同时,Perel等[28]也对动物实验的有效性提出质疑,其对比了同种治疗方法在临床试验和动物实验中的治疗效果,发现在一些疾病上两者的疗效并不一致。另一方面,研究中所用的细胞模型通常是取材于患者的体细胞,部分疾病因发病特点和伦理学限制,导致携带某一疾病表型的原代细胞来源受限,且常存在不易分离和无法长期进行体外培养等问题,因而很难构建理想的细胞模型。1998年Thomson等人成功分离出人胚胎干细胞,为克服上述疾病模型存在的问题提供了新策略。 2.3 利用人胚胎干细胞构建遗传病模型的方法 2003年Zwaka等[29]首次发表了对人胚胎干细胞进行同源基因重组操作的文章。随后,Urbach等[30]利用人胚胎干细胞构建了Lesch-Nyham综合征的疾病模型。自此人们开始通过对人胚胎干细胞进行核基因组编辑构建各种类型的携带遗传病致病基因的胚胎干细胞系。但由于受到基因打靶技术的限制,人胚胎干细胞的基因编辑效率不高。目前,常用的基因编辑手段有锌指核酸酶技术、转录激活样效应因子核酸酶技术及成簇规律间隔短回文重复技术。3种方法的普及应用,显著提高了胚胎干细胞的基因编辑效率,为利用胚胎干细胞构建遗传疾病细胞模型提供更坚实的基础。另一方面,近年来随着人类辅助生殖技术的应用,利用产前基因诊断(PGD)和产前基因筛查(PGS)可获得含有特定基因变异的人胚胎干细胞,该类人胚胎干细胞取自废弃的胚胎,成功地回避了伦理学争议,因此也被广泛应用于人类遗传病模型的构建。目前已经利用PGD/PGS技术建立了多种遗传病的细胞系,如马凡氏综合征、脆性X染色体综合征、良性假肥大型肌营养不良、乙型血友病、囊性纤维化、肌强直性营养不良等[31-35]。除上述两种方法外,2014年Tachibana等[36]通过将体细胞核移植入去核的卵母细胞成功地建立了一种多能干细胞,该类干细胞与胚胎干细胞具有相似的性质,理论上也可被应用于遗传病模型的建立。携带遗传疾病致病基因的胚胎干细胞在体外可分化成各种类型的成体细胞,进而模拟遗传病的发生发展,以期探究遗传病的发生机制特点等,寻找疾病治愈的新方法。 2.3.1 常用基因编辑技术 锌指核酸酶(Zinc-finger nuclease,ZFN)技术:锌指核酸酶是第1代人工合成的限制性内切酶,由锌指DNA结合域与限制性内切酶的DNA切割域组成。研究者可根据不同的目的基因设计特异性的锌指核酸酶,该技术既可用于导入目的基因,也可用于敲出目的基因。锌指核酸酶技术简单易行,具有较高的特异性和基因编辑效率。但同时,锌指核酸酶技术也存在许多不足之处,最显著的问题就是其在基因剪切过程有较高的脱靶效应,导致核酸内切酶并没有对目的基因进行识别和切割,最终可能引起DNA的错配和序列的改变[37]。 转录激活样效应因子核酸酶(transcription activator-like effector nuclease,TALEN)技术:转录激活样效应因子核酸酶被认为是第2代人工合成的限制性内切酶,与锌指核酸酶相似,该类嵌合核酸酶由两部分组成,即1个可编码的序列特异性DNA结合模块与1个非特异性的DNA切割结构域。通过诱导DNA双链断裂来刺激容易出错的非同源末端连接或在特定基因所在的位置进行同源定向修复。一项研究中运用锌指核酸酶技术和转录激活样效应因子核酸酶技术分别作用在相同的基因位点CCR5上,相比于锌指核酸酶技术,转录激活样效应因子核酸酶技术在CCR5相似的作用位点CCR2上有较低脱靶效应[38]。转录激活样效应因子核酸酶技术的缺点在于其分子质量较大,使其在运送到细胞体内产生一定麻烦,因而限制了它的应用。 成簇规律间隔短回文重复(clustered regulatoryinterspaced short palindromic repeat,CRISPR)技术:该系统起初是在细菌的天然免疫系统内发现的,其主要功能是抵抗入侵的病毒及外源DNA,能使其特异性降解,从而起到抗病毒的作用[39- 40]。CRISPR/Cas系统由CRISPR序列原件与cas基因家族组成。目前,共有3种类型的CRISPR/Cas系统,2型系统是目前研究应用最多的,也就是常用的CRISPR/Cas9系统。与锌指核酸酶、转录激活样效应因子核酸酶不同的是,它能够完成RNA导向的DNA识别及编辑。该技术靶向精确、脱靶率低、细胞毒性低、价格更有优势。但是也存在特异性不高,容易产生随机毒性等缺点。 3种基因编辑方法比较,见表1。"
| [1] Evans MJ, Kaufman MH. Establishment in culture of pluripotential cells from mouse embryos. Nature. 1981;292 (5819):154-156.[2] Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A. 1981;78(12):7634-7638.[3] Bongso A, Fong CY, Ng SC, et al. Isolation and culture of inner cell mass cells from human blastocysts. Hum Reprod. 1994;9(11):2110-2117.[4] Thomson JA, Itskovitz-Eldor J, Shapiro SS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145-1147.[5] Ström S, Inzunza J, Grinnemo KH, et al. Mechanical isolation of the inner cell mass is effective in derivation of new human embryonic stem cell lines. Hum Reprod. 2007;22(12): 3051-3058.[6] Terstegge S, Rath BH, Laufenberg I, et al. Laser-assisted selection and passaging of human pluripotent stem cell colonies. J Biotechnol. 2009;143(3):224-230.[7] Lim JW, Bodnar A. Proteome analysis of conditioned medium from mouse embryonic fibroblast feeder layers which support the growth of human embryonic stem cells. Proteomics. 2002; 2(9):1187-1203.[8] Xu RH, Peck RM, Li DS, et al. Basic FGF and suppression of BMP signaling sustain undifferentiated proliferation of human ES cells. Nat Methods. 2005;2(3):185-190.[9] Cai J, Chen J, Liu Y, et al. Assessing self-renewal and differentiation in human embryonic stem cell lines. Stem Cells. 2006;24(3):516-530.[10] Chin AC, Fong WJ, Goh LT, et al. Identification of proteins from feeder conditioned medium that support human embryonic stem cells. J Biotechnol. 2007;130(3):320-328.[11] Park Y, Kim JH, Lee SJ, et al. Human feeder cells can support the undifferentiated growth of human and mouse embryonic stem cells using their own basic fibroblast growth factors. Stem Cells Dev. 2011;20(11):1901-1910.[12] Itskovitz-Eldor J, Schuldiner M, Karsenti D, et al. Differentiation of human embryonic stem cells into embryoid bodies compromising the three embryonic germ layers. Mol Med. 2000;6(2):88-95.[13] Amit M, Margulets V, Segev H, et al. Human feeder layers for human embryonic stem cells. Biol Reprod. 2003;68(6): 2150-2156.[14] Cheng L, Hammond H, Ye Z, et al. Human adult marrow cells support prolonged expansion of human embryonic stem cells in culture. Stem Cells. 2003;21(2):131-142.[15] Genbacev O, Krtolica A, Zdravkovic T, et al. Serum-free derivation of human embryonic stem cell lines on human placental fibroblast feeders. Fertil Steril. 2005;83(5): 1517-1529.[16] Unger C, Felldin U, Nordenskjöld A, et al. Derivation of human skin fibroblast lines for feeder cells of human embryonic stem cells. Curr Protoc Stem Cell Biol. 2008;Chapter 1:Unit 1C.7.[17] Lai D, Wang Y, Sun J, et al. Derivation and characterization of human embryonic stem cells on human amnion epithelial cells. Sci Rep. 2015;5:10014.[18] Narkilahti S, Rajala K, Pihlajamäki H, et al. Monitoring and analysis of dynamic growth of human embryonic stem cells: comparison of automated instrumentation and conventional culturing methods. Biomed Eng Online. 2007;6:11.[19] Choi YJ, Lin CP, Risso D, et al. Deficiency of microRNA miR-34a expands cell fate potential in pluripotent stem cells. Science. 2017;355(6325): eaag1927.[20] Yang Y, Liu B, Xu J, et al. Derivation of Pluripotent Stem Cells with In Vivo Embryonic and Extraembryonic Potency. Cell. 2017;169(2):243-257.[21] Graf U, Casanova EA, Wyck S, et al. Corrigendum: Pramel7 mediates ground-state pluripotency through proteasomal-epigenetic combined pathways. Nat Cell Biol. 2017;19(8):1003.[22] Graf U, Casanova EA, Wyck S, et al. Pramel7 mediates ground-state pluripotency through proteasomal-epigenetic combined pathways. Nat Cell Biol. 2017;19(7):763-773.[23] Wang Z, Oron E, Nelson B, et al. Distinct lineage specification roles for NANOG, OCT4, and SOX2 in human embryonic stem cells. Cell Stem Cell. 2012;10(4):440-454.[24] Silva J, Nichols J, Theunissen TW, et al. Nanog is the gateway to the pluripotent ground state. Cell. 2009;138(4): 722-737.[25] Mouse Genome Sequencing Consortium1, Waterston RH, Lindblad-Toh K, et al. Initial sequencing and comparative analysis of the mouse genome. Nature. 2002;420(6915): 520-562.[26] Wilson JM. Animal models of human disease for gene therapy. J Clin Invest. 1996;97(5):1138-1141.[27] Saenger P. Turner's syndrome. N Engl J Med. 1996;335(23): 1749-1754.[28] Perel P, Roberts I, Sena E, et al. Comparison of treatment effects between animal experiments and clinical trials: systematic review. BMJ. 2007;334(7586):197.[29] Zwaka TP, Thomson JA. Homologous recombination in human embryonic stem cells. Nat Biotechnol. 2003;21(3): 319-321.[30] Urbach A, Schuldiner M, Benvenisty N. Modeling for Lesch-Nyhan disease by gene targeting in human embryonic stem cells. Stem Cells. 2004;22(4):635-641.[31] Pickering SJ, Minger SL, Patel M, et al. Generation of a human embryonic stem cell line encoding the cystic fibrosis mutation deltaF508, using preimplantation genetic diagnosis. Reprod Biomed Online. 2005;10(3):390-397.[32] Mateizel I, De Temmerman N, et al. Derivation of human embryonic stem cell lines from embryos obtained after IVF and after PGD for monogenic disorders. Hum Reprod. 2006; 21(2):503-511.[33] Avitzour M, Mor-Shaked H, Yanovsky-Dagan S, et al. FMR1 epigenetic silencing commonly occurs in undifferentiated fragile X-affected embryonic stem cells. Stem Cell Reports. 2014;3(5):699-706.[34] Yang Q, Zhou X, Zhou H, et al. Human embryonic stem cells derived from abnormal blastocyst donated by Marfan syndrome patient. Stem Cell Res. 2015;15(3):640-642.[35] Hmadcha A, Aguilera Y, Lozano-Arana MD, et al. Derivation of HVR1, HVR2 and HVR3 human embryonic stem cell lines from IVF embryos after preimplantation genetic diagnosis (PGD) for monogenic disorder. Stem Cell Res. 2016;16(3): 635-639.[36] Tachibana M, Amato P, Sparman M, et al. Human embryonic stem cells derived by somatic cell nuclear transfer. Cell. 2013;153(6):1228-1238.[37] Kim JS, Lee HJ, Carroll D. Genome editing with modularly assembled zinc-finger nucleases. Nat Methods. 2010;7(2):91.[38] Mussolino C, Morbitzer R, Lütge F, et al. A novel TALE nuclease scaffold enables high genome editing activity in combination with low toxicity. Nucleic Acids Res. 2011;39(21): 9283-9293.[39] Barrangou R, Fremaux C, Deveau H, et al. CRISPR provides acquired resistance against viruses in prokaryotes. Science. 2007;315(5819):1709-1712.[40] Haft DH, Selengut J, Mongodin EF, et al. A guild of 45 CRISPR-associated (Cas) protein families and multiple CRISPR/Cas subtypes exist in prokaryotic genomes. PLoS Comput Biol. 2005;1(6):e60.[41] Eiges R, Urbach A, Malcov M, et al. Developmental study of fragile X syndrome using human embryonic stem cells derived from preimplantation genetically diagnosed embryos. Cell Stem Cell. 2007;1(5):568-577.[42] Urbach A, Benvenisty N. Studying early lethality of 45,XO (Turner's syndrome) embryos using human embryonic stem cells. PLoS One. 2009;4(1):e4175.[43] Yu J, Vodyanik MA, Smuga-Otto K, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318(5858):1917-1920. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||