Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (17): 2625-2630.doi: 10.3969/j.issn.2095-4344.0484
Roles of Rev-erb alphya and Ror alpha in osteoblastogenesis of mouse bone marrow mesenchymal stem cells
Lin Fu-wei1, Xu Xiao-mei1, Cui Yan2, Xie Yi-jia1, Zhao Qing3
- 1Orofacial Reconstruction and Regeneration Laboratory, Department of Orthodontics, the Affiliated Stomatological Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China; 2School of Public Health, Southwest Medical University, Luzhou 646000, Sichuan Province, China; 3State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, Department of Orthodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Revised:2017-12-07Online:2018-06-18Published:2018-06-18 -
Contact:Zhao Qing, M.D., Associate professor, State Key Laboratory of Oral Diseases, National Clinical Research Center for Oral Diseases, Department of Orthodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Lin Fu-wei, Master, Physician, Orofacial Reconstruction and Regeneration Laboratory, Department of Orthodontics, the Affiliated Stomatological Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China -
Supported by:the Fund of Luzhou Municipal Department of Science and Technology, No. 2015-S-50(2/3); the Youth Foundation of Southwest Medical University, No. 2014QN-052, the Funded Project by the Affiliated Stomatological Hospital of Southwest Medical University, No. 201402
CLC Number:
Cite this article
Lin Fu-wei, Xu Xiao-mei, Cui Yan, Xie Yi-jia, Zhao Qing. Roles of Rev-erb alphya and Ror alpha in osteoblastogenesis of mouse bone marrow mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(17): 2625-2630.
share this article
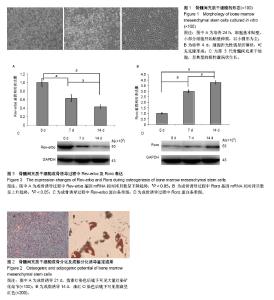
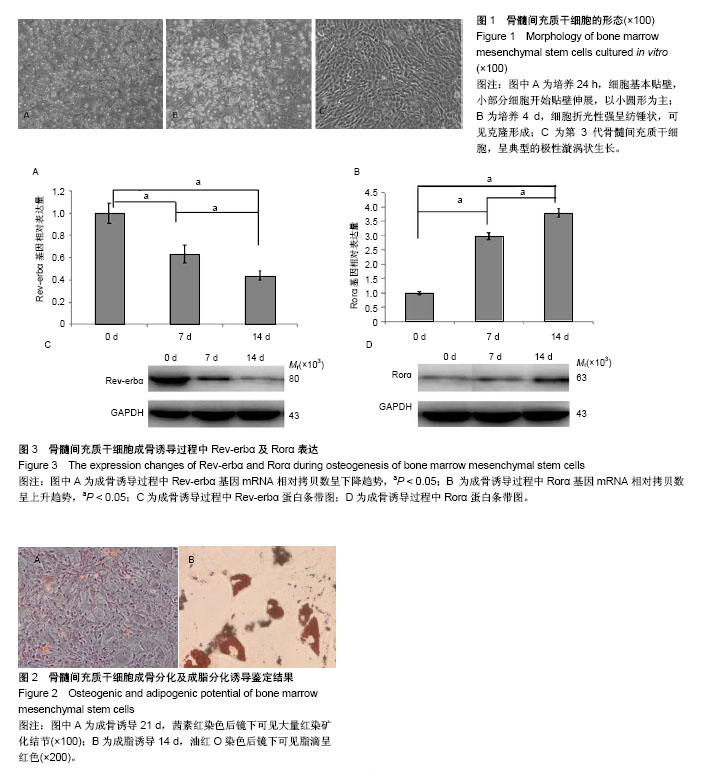
2.1 骨髓间充质干细胞形态 骨髓细胞刚接种时,悬浮于培养基中呈高亮圆形,大小不一,细胞核不能辨认。24 h后大部分细胞贴壁,形态不一,以小圆形为主,部分细胞贴壁伸展,呈泪滴状。4 d左右培养瓶细胞贴附面可见形态相对均一的折光性较强纺锤形细胞,部分贴壁细胞呈克隆样生长。7 d左右细胞呈现多种形态,细胞呈漩涡状排列,克隆交界处出现重叠、融合,细胞融合达80%。传代接种的骨髓间充质干细胞在24-72 h快速增殖,为细胞增殖高峰期,细胞逐渐呈漩涡状、网状或辐射状排列,胞浆丰富,核大,呈椭圆形,5 d左右即可达到80%融合。传代后的细胞体积均较前变大,形态上更加趋于一致,增殖快,胞核明显,呈放射状向周边生长,即典型的漩涡样生长,见图1。 2.2 骨髓间充质干细胞成骨诱导及茜素红染色鉴定结果 经成骨诱导液持续培养后,细胞呈复层生长,由梭形变为立方形,细胞体积增大。细胞外基质分泌增多,部分细胞重叠生长。21 d后茜素红染色可见大量红色矿化结节(图2A)。 2.3 骨髓间充质干细胞成脂诱导及油红O染色鉴定结果 经成脂诱导液培养至第7天左右,细胞形态由梭形变为圆形,体积增大,细胞胞浆内含有大量脂滴。至14 d时大量的间充质干细胞转变为脂肪细胞。油红O染色可见胞浆内有较多橙红色折光性强的脂滴(图2B)。 2.4 骨髓间充质干细胞成骨诱导后Rev-erbα及Rorα表达的变化 在骨髓间充质干细胞成骨诱导的第0,7,14天分别行Real Time PCR检测Rev-erbα及Rorα基因mRNA水平,Western Blot检测Rev-erbα及Rorα蛋白表达水平。 PCR结果显示:Rev-erbα基因mRNA水平呈下降趋势,见图3A。Rorα基因mRNA表达水平呈上升趋势,见图3B。 Western Blot结果显示:Rev-erbα蛋白表达呈下降趋势,见图3C。而Rorα蛋白表达呈上升趋势,见图3D。"
| [1] Bianco P, Riminucci M, Gronthos S, et al. Bone marrow stromal stem cells: nature, biology, and potential applications. Stem Cells. 2001;19(3):180-192.[2] Tang Z, Wei J, Yu Y, et al. γ-Secretase inhibitor reverts the Notch signaling attenuation of osteogenic differentiation in aged bone marrow mesenchymal stem cells. Cell Biol Int. 2016;40(4):439-447.[3] Zhou S, Greenberger JS, Epperly MW, et al. Age-related intrinsic changes in human bone-marrow-derived mesenchymal stem cells and their differentiation to osteoblasts. Aging Cell. 2008;7(3):335-343.[4] Tan J, Xu X, Tong Z, et al. Decreased osteogenesis of adult mesenchymal stem cells by reactive oxygen species under cyclic stretch: a possible mechanism of age related osteoporosis. Bone Res. 2015;3:15003.[5] Liu W, Qi M, Konermann A, et al. The p53/miR-17/Smurf1 pathway mediates skeletal deformities in an age-related model via inhibiting the function of mesenchymal stem cells. Aging (Albany NY). 2015;7(3):205-218.[6] Wang C, Wang J, Li J, et al. KDM5A controls bone morphogenic protein 2-induced osteogenic differentiation of bone mesenchymal stem cells during osteoporosis. Cell Death Dis. 2016;7(8):e2335.[7] Owen TA, Aronow MS, Barone LM, et al. Pleiotropic effects of vitamin D on osteoblast gene expression are related to the proliferative and differentiated state of the bone cell phenotype: dependency upon basal levels of gene expression, duration of exposure, and bone matrix competency in normal rat osteoblast cultures. Endocrinology. 1991;128(3):1496-1504.[8] Oldberg A, Jirskog-Hed B, Axelsson S, et al. Regulation of bone sialoprotein mRNA by steroid hormones. J Cell Biol. 1989;109(6 Pt 1):3183-3186.[9] Smith EP, Boyd J, Frank GR, et al. Estrogen resistance caused by a mutation in the estrogen-receptor gene in a man. N Engl J Med. 1994;331(16):1056-1061.[10] Miyamoto S, Cooper L, Watanabe K, et al. Role of retinoic acid-related orphan receptor-alpha in differentiation of human mesenchymal stem cells along with osteoblastic lineage. Pathobiology. 2010;77(1):28-37.[11] Meyer T, Kneissel M, Mariani J, et al. In vitro and in vivo evidence for orphan nuclear receptor RORalpha function in bone metabolism. Proc Natl Acad Sci U S A. 2000;97(16): 9197-9202.[12] Kojetin DJ, Burris TP. REV-ERB and ROR nuclear receptors as drug targets. Nat Rev Drug Discov. 2014;13(3):197-216.[13] Welch RD, Billon C, Valfort AC, et al. Pharmacological inhibition of REV-ERB stimulates differentiation, inhibits turnover and reduces fibrosis in dystrophic muscle. Sci Rep. 2017;7(1):17142.[14] Samsa WE, Vasanji A, Midura RJ, et al. Deficiency of circadian clock protein BMAL1 in mice results in a low bone mass phenotype. Bone. 2016;84:194-203.[15] Suyama K, Silagi ES, Choi H, et al. Circadian factors BMAL1 and RORα control HIF-1α transcriptional activity in nucleus pulposus cells: implications in maintenance of intervertebral disc health. Oncotarget. 2016;7(17):23056-23071.[16] Yang W, Kang X, Liu J, et al. Clock Gene Bmal1 Modulates Human Cartilage Gene Expression by Crosstalk With Sirt1. Endocrinology. 2016;157(8):3096-3107.[17] Lin F, Chen Y, Li X, et al. Over-expression of circadian clock gene Bmal1 affects proliferation and the canonical Wnt pathway in NIH-3T3 cells. Cell Biochem Funct. 2013;31(2): 166-172.[18] Friedenstein AJ, Deriglasova UF, Kulagina NN, et al. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp Hematol. 1974;2(2):83-92.[19] Lennon DP, Haynesworth SE, Bruder SP, et al. Human and animal mesenchymal progenitor cells from bone marrow: identification of serum for optimal selection and proliferation. In Vitro Cell Dev Biol. 1996;32:602-611.[20] Zheng YH, Xiong W, Su K, et al. Multilineage differentiation of human bone marrow mesenchymal stem cells in vitro and in vivo. Exp Ther Med. 2013;5(6):1576-1580.[21] Samsonraj RM, Raghunath M, Nurcombe V, et al. Concise Review: Multifaceted Characterization of Human Mesenchymal Stem Cells for Use in Regenerative Medicine. Stem Cells Transl Med. 2017;6(12):2173-2185.[22] Castro-Manrreza ME, Montesinos JJ. Immunoregulation by mesenchymal stem cells: biological aspects and clinical applications. J Immunol Res. 2015;2015:394917.[23] Castro-Manrreza ME, Mayani H, Monroy-García A, et al. Human mesenchymal stromal cells from adult and neonatal sources: a comparative in vitro analysis of their immunosuppressive properties against T cells. Stem Cells Dev. 2014;23(11):1217-1232.[24] Ma S, Xie N, Li W, et al. Immunobiology of mesenchymal stem cells. Cell Death Differ. 2014;21(2):216-225. [25] Javazon EH, Colter DC, Schwarz EJ, et al. Rat marrow stromal cells are more sensitive to plating density and expand more rapidly from single-cell-derived colonies than human marrow stromal cells. Stem Cells. 2001;19(3):219-225.[26] Volk SW, Diefenderfer DL, Christopher SA, et al. Effects of osteogenic inducers on cultures of canine mesenchymal stem cells. Am J Vet Res. 2005;66(10):1729-1737.[27] Bellantuono I, Aldahmash A, Kassem M. Aging of marrow stromal (skeletal) stem cells and their contribution to age-related bone loss. Biochimica et Biophysica Acta. 2009;1792(4): 364-370.[28] Colter DC, Class R, DiGirolamo CM, et al. Rapid expansion of recycling stem cells in cultures of plastic-adherent cells from human bone marrow. Proc Natl Acad Sci U S A. 2000;97(7): 3213-3218.[29] Mortezaee K, Pasbakhsh P, Ragerdi Kashani I, et al. Melatonin Pretreatment Enhances the Homing of Bone Marrow-derived Mesenchymal Stem Cells Following Transplantation in a Rat Model of Liver Fibrosis. Iran Biomed J. 2016;20(4):207-216.[30] 张荣耀,毕晓娟,马艳,等. 全骨髓法培养 C57 小鼠骨髓间充质干细胞的生物学特性[J].中国组织工程研究,2014,18(1):45-50. [31] Khanabdali R, Saadat A, Fazilah M, et al. Promoting effect of small molecules in cardiomyogenic and neurogenic differentiation of rat bone marrow-derived mesenchymal stem cells. Drug Des Devel Ther. 2015;10:81-91. [32] Bugge A, Feng D, Everett LJ, et al. Rev-erbα and Rev-erbβ coordinately protect the circadian clock and normal metabolic function. Genes Dev. 2012;26(7):657-667. [33] Turek FW, Joshu C, Kohsaka A, et al. Obesity and metabolic syndrome in circadian Clock mutant mice. Science. 2005; 308(5724):1043-1045.[34] Duez H, van der Veen JN, Duhem C, et al. Regulation of bile acid synthesis by the nuclear receptor Rev-erbalpha. Gastroenterology. 2008;135(2):689-698.[35] Galli C, Passeri G, Macaluso GM. Osteocytes and WNT: the mechanical control of bone formation. J Dent Res. 2010;89(4): 331-343.[36] Thudi NK, Martin CK, Murahari S, et al. Dickkopf-1 (DKK-1) stimulated prostate cancer growth and metastasis and inhibited bone formation in osteoblastic bone metastases. Prostate. 2011;71(6):615-625.[37] Kim JH, Liu X, Wang J, et al. Wnt signaling in bone formation and its therapeutic potential for bone diseases. Ther Adv Musculoskelet Dis. 2013;5(1):13-31.[38] Wang Y, Li YP, Paulson C, et al. Wnt and the Wnt signaling pathway in bone development and disease. Front Biosci (Landmark Ed). 2014;19:379-407.[39] Hosoi T. Cytokines in bone diseases. Wnt signal and excessive bone formation. Clin Calcium. 2010;20:1526-1531.[40] Caronia G, Wilcoxon J, Feldman P, et al. Bone morphogenetic protein signaling in the developing telencephalon controls formation of the hippocampal dentate gyrus and modifies fear-related behavior. J Neurosci. 2010;30:6291-6301.[41] Schulze J, Seitz S, Saito H, et al. Negative regulation of bone formation by the transmembrane Wnt antagonist Kremen-2. PLoS One. 2010;5:e10309.[42] Boland GM, Perkins G, Hall DJ, et al. Wnt 3a promotes proliferation and suppresses osteogenic differentiation of adult human mesenchymal stem cells.J Cell Biochem. 2004; 93(6):1210-1230.[43] Gaur T, Lengner CJ, Hovhannisyan H, et al. Canonical WNT signaling promotes osteogenesis by directly stimulating Runx2 gene expression. J Biol Chem. 2005;280(39): 33132-33140. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [14] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||