Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (9): 1319-1324.doi: 10.3969/j.issn.2095-4344.0459
Previous Articles Next Articles
A comparative study on the secretion of various cytokines by human adipose-derived stem cells at different passages
Wang Qian, Tang Wen-yan, Wang Zhao-yan, Yang Yin-xiang, Luan Zuo
- Department of Pediatrics, Navy General Hospital of PLA, Beijing 100048, China
-
Revised:2017-08-26Online:2018-03-28Published:2018-04-03 -
Contact:Luan Zuo, Chief physician, Department of Pediatrics, Navy General Hospital of PLA, Beijing 100048, China -
About author:Wang Qian, Department of Pediatrics, Navy General Hospital of PLA, Beijing 100048, China -
Supported by:the Military Logistics Research Projects of PLA, No. CHJ14C022
CLC Number:
Cite this article
Wang Qian, Tang Wen-yan, Wang Zhao-yan, Yang Yin-xiang, Luan Zuo. A comparative study on the secretion of various cytokines by human adipose-derived stem cells at different passages[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(9): 1319-1324.
share this article
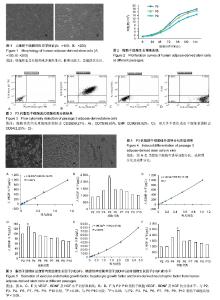
2.1 脂肪干细胞的形态学特征 原代培养24 h后可见细胞贴壁,可见有少量细胞贴壁生长,呈短小梭形,培养48 h首次细胞换液,随着培养时间的延长,呈长梭形生长,3 d后细胞融合达80%-90%传代后细胞增殖速度明显加快,四五天即长满瓶底,细胞形态呈长梭形、多角形或不规则形态,核质比大,呈漩涡状生长,见图1。细胞可稳定传代至第10代。 2.2 脂肪干细胞的生长增殖情况 第3,5,9代脂肪干细胞的生长增殖情况,保持了良好的生长趋势,见图2。P3,P5和P9脂肪干细胞的生长增殖能力均差异无显著性意义(P > 0.05)。 2.3 脂肪干细胞鉴定情况 流式细胞仪检测脂肪干细胞的细胞表面分子,其表达阳性细胞比例分别为CD29 (99.21%)、CD73(99.65%)和CD90(99.92%),而几乎不表达造血干细胞表面标志CD34(2.25%),见图3。 2.4 脂肪干细胞的分化潜能鉴定 取生长状态良好的第3代脂肪干细胞,加入成脂诱导完全培养基进行诱导第7天时可见少许细胞表面出现葡萄串珠样脂滴形成大小不一,到14 d时明显增多,21 d可见大部分细胞表面均有葡萄串珠样脂滴形成。使用油红O染色可见大量红色大小不一的圆形脂滴,边界清楚,位于细胞的一侧,呈葡萄串珠样脂滴,见图4A。由大约2.5×105个细胞离心后形成的细胞团在离心管中进行软骨分化诱导液诱导,诱导分化两三周后,进行甲苯胺蓝染色液浸染后可见有部分细胞被染成蓝色,且细胞中有许多小孔具有软骨特征,见图4B。成骨诱导14 d以后用冯库萨试剂盒染色可见黑褐色颗粒状钙节的形成,见图4C。 2.5 脂肪干细胞分泌血管内皮细胞生长因子,脑源性神经营养因子和肝细胞生长因子水平变化 酶联免疫吸附实验显示,随着代数的逐渐增加,脂肪干细胞分泌的血管内皮细胞生长因子和肝细胞生长因子水平逐渐增加。当细胞代数为P5时,血管内皮细胞生长因子的分泌水平最高,与其他代数相比差异有显著性意义(P < 0.05),此后,其分泌水平逐渐下降,见图5A,B。而P3代脂肪干细胞分泌的脑源性神经营养因子最高,其与P2和P4分泌水平相比,差异无显著性意义(P > 0.05),而与P5-P10分泌水平相比,差异有显著性意义(P < 0.05),见图5C,D。肝细胞生长因子的分泌水平于P5最高,且与其他代数相比,差异有显著性意义(P < 0.05),图5E,F。"
| [1] Hsiao ST, Lokmic Z, Peshavariya H, et al. Hypoxic Conditioning Enhances the Angiogenic Paracrine Activity of Human Adipose-Derived Stem Cells. Stem Cells Dev. 2013; 22(10):1614-1623.[2] Kocan B, Maziarz A, Tabarkiewicz J, et al. Trophic Activity and Phenotype of Adipose Tissue-Derived Mesenchymal Stem Cells as a Background of Their Regenerative Potential. Stem Cells Int. 2017;2017:1653254.[3] Rehman J, Traktuev D, Li J, et al. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation. 2004;109(10):1292-1298.[4] Tsuji W, Rubin JP, Marra KG. Adipose-derived stem cells: Implications in tissue regeneration. World J Stem Cells. 2014;6(3):312-321.[5] Yoshimura K, Suqa H, Eto H. Adipose-derived stem/progenitor cells: roles in adipose tissue remodeling and potential use for soft tissue augmentation. Regen Med. 2009;4(2):265-273.[6] Lee SH, Jin SY, Song JS, et al. Paracrine effects of adipose-derived stem cells on keratinocytes and dermal fibroblasts. Ann Dermatol. 2012;24(2):136-143.[7] Ivanova-Todorova E, Bochev I, Dimitrov R, et al. Conditioned medium from adipose tissue-derived mesenchymal stem cells induces CD4+FOXP3+ cells and increases IL-10 secretion. J Biomed Biotechnol. 2012;2012:295167.[8] Kilroy GE, Foster SJ, Wu X, et al. Cytokine profile of human Adipose-derived stem cells: expression of angiogenic, hematopoietic, and pro-inflammatory factors. J Cell Physiol. 2007;212(3):702-709.[9] Lee HC, An SG, Lee HW, et al. Safety and effect of adipose tissue-derived stem cell implantation in patients with critical limb ischemia: a pilot study. Circ J. 2012;76:1750-1760.[10] Lee EY, Xia Y, Kim WS, et al. Hypoxia-enhanced wound-healing function of adipose-derived stem cells: increase in stem cell proliferation and up-regulation of VEGF and bFGF. Wound Repair Regen. 2009;17:540-547. [11] Lee SC, Jeong HJ, Lee SK, et al. Hypoxic Conditioned Medium From Human Adipose-Derived Stem Cells Promotes Mouse Liver Regeneration Through JAK/STAT3 Signaling. Stem Cells Transl Med. 2016;5(6):816-825.[12] Rehman J, Traktuev D, Li J, et al. Secretion of angiogenic and antiapoptotic factors by human adipose stromal cells. Circulation. 2004;109:1292-1298.[13] Zhou L, Xia J, Qiu X, et al. In vitro evaluation of endothelial progenitor cells from adipose tissue as potential angiogenic cell sources for bladder angiogenesis. PLoS One. 2015;10(2): e0117644.[14] Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211-228.[15] Gronthos S, Franklin DM, Leddy HA, et al. Surface protein characterization of human adipose tissue-deprived stromal cells. J Cell Physiol. 2001;189(1):54-63.[16] Quaini F, Cigola E, Lagrasta C, et al. End-stage cardiac failure in humans is coupled with the induction of proliferating cell nuclear antigen and nuclear mitotic division in ventricular myocytes. Circ Res. 1994;75:1050-1063.[17] Suzuki E, Fujita D, Takahashi M, et al. Adipose tissue-derived stem cells as a therapeutic tool for cardiovascular disease. World J Cardiol. 2015;7(8):454-465.[18] Balducci L, Blasi A, Saldarelli M, et al. Immortalization of human adipose-derived stromal cells: production of cell lines with high growth rate, mesenchymal marker expression and capability to secrete high levels of angiogenic factors. Stem Cell Res Ther. 2014;5(3):63. [19] Cao JQ, Liang YY, Li YQ,et al.Adipose-derived stem cells enhance myogenic differentiation in the mdx mouse model of muscular dystrophy via paracrine signaling.Neural Regen Res. 2016;11(10):1638-1643.[20] Gatalica Z, Snyder C, Maney T, et al. Programmed cell death 1(PD-1) and its ligand (PD-L1) in common cancers and their correlation with molecular cancer type. Cancer Epidemiol Biomarkers Prev. 2014;23(12):2965-2970. [21] Goel HL, Mercurio AM. VEGF targets the tumour cell. Nat Rev Cancer. Nat Rev Cancer. 2013;13(12):871-882.[22] Lipsky RH, Marini AM. Brain-derived neurotrophic factor in neuronal survival and behavior-related plasticity. Ann N Y Acad Sci. 2007;1122:130-143.[23] Bernd P. The role of neurotrophins during early development. Gene Expr. 2008;14(4):241-250.[24] ]Binder DK, Scharfman HE. Brain-derived neurotrophic factor. Growth Factors. 2004;22(3):123-131.[25] Castren E. Neurotrophins as mediators of drug effects on mood, addiction, and neuroprotection. Mol Neurobiol. 2004; 29(3):289-302.[26] Cattaneo E, Zuccato C, Tartari M. Normal huntingtin function: an alternative approach to Huntington’s disease. Nat Rev Neurosci. 2005;6(12):919-930.[27] Murer MG, Yan Q, Raisman-Vozari R. Brain-derived neurotrophic factor in the control human brain, and in Alzheimer’s disease and Parkinson’s disease. Prog Neurobiol. 2001;63(1):71-124.[28] Russo-Neustadt AA, Chen MJ. Brain-derived neurotrophic factor and antidepressant activity. Curr Pharm Des. 2005; 11(12):1495-1510.[29] Mira A, Morello V, Céspedes MV, et al. Stroma-derived HGF drives metabolic adaptation of colorectal cancer to angiogenesis inhibitors. Oncotarget. 2017;8(24): 38193-38213. [30] Yang Y, Chen QH, Liu AR, et al. Synergism of MSC-secreted HGF and VEGF in stabilising endothelial barrier function upon lipopolysaccharide stimulation via the Rac1 pathway. Stem Cell Res Ther. 2015;6:250. [31] Gallo S, Sala V, Gatti S, et al. HGF/Met Axis in Heart Function and Cardioprotection. Biomedicines. 2014;2(4):247-262. |
| [1] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1602-1608. |
| [2] | Liu Xiaogang, Li Tian, Zhang Duo. Effect and mechanism of the effective components of Chinese medicine on promoting the differentiation of bone marrow mesenchymal stem cells into chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 121-126. |
| [3] | Wei Shan, Li Qin, Su Yi. Research hotspots and application potential of stem cells from human exfoliated deciduous teeth [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 127-131. |
| [4] | Shuai Zhiqin, Chen Jiameng, Liu Taotao, Hu Anling, Li Lisheng, Yu Limei, Xu Shangfu. Research status and problems of stem cells and their derived exosomes for prevention and treatment of vascular restenosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 138-144. |
| [5] | Li Zhongkang, Zheng Jiahua, Tian Yanpeng, Huang Xianghua. Latest progress and mechanisms of mesenchymal stem cells on premature ovarian failure [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 145-152. |
| [6] | Wang Shuyun, Xie Junhui, Yu Xuefeng. Effect and mechanism of mesenchymal stem cells in the treatment of diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 153-158. |
| [7] | Lin Miaoyuan, Li Yuwan, Liu Yi, Chen Bei, Zhang Li. Research hotspots and application value of tissue-engineered skin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 159-166. |
| [8] | Song Huifang, Tan Jiayin, Kang Yi, Li Bin, Bi Zhifei, Long Nü, Xia Zhongnian, Guo Rui. Hypoxic pretreatment enhances the protective effect of aged human bone marrow mesenchymal stem cells conditioned medium against H9C2 oxidative stress damage [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 1-6. |
| [9] | Mei Yunyun, Zhang Jianjun, Wang Dong. Hyperbaric oxygen combined with NgR gene silencing bone marrow mesenchymal stem cells transplantation for spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 12-19. |
| [10] | Xie Xingqi, Hu Wei, Tu Guanjun. Bone marrow mesenchymal stem cells-derived exosomes combined with chondroitinase ABC for treating spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 20-26. |
| [11] | Zhang Xuelei, Luo Gan, Yu Shenghui, Gu Zuchao, Peng Xu, He Xueling, Liu Yan, Zhang Xiaomei. Acellular nerve scaffold combined with bone marrow mesenchymal stem cells and platelet-rich gel for femoral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 27-32. |
| [12] | Zhao Laihe, Xia Bing, Ma Teng, Gao Jianbo, Li Shengyou, Gao Xue, Zheng Yi, Hu Guangwen, Luo Zhuojing, Huang Jinghui. Extracellular matrix of Schwann-like cells induced by bone marrow mesenchymal stem cells promotes axonal regeneration after peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 33-39. |
| [13] | Mai Liping, He Guodong, Chen Shaoxian, Zhu Jiening, Hou Xinghua, Zhang Mengzhen, Li Xiaohong. Expression of aldehyde dehydrogenase 3B1 during human bone marrow mesenchymal stem cells senescence [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 40-44. |
| [14] | Yang Tengyun, Li Yanlin, Liu Dejian, Wang Guoliang, Zheng Zhujun. Chondrogenic differentiation of peripheral blood-derived mesenchymal stem cells induced by transforming growth factor beta 3: a dose-effect relationship [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 45-51. |
| [15] | Meng Defeng, Li Changzai, Wu Chuntao. Effects of LINC02532 targeting miR-145 on proliferation, migration, invasion and apoptosis of pancreatic cancer stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 52-58. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||