Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (5): 669-674.doi: 10.3969/j.issn.2095-4344.0433
Previous Articles Next Articles
Icariin with different concentrations promotes osteogenic differentiation of human bone marrow mesenchymal stem cells
Wu Xi, Peng Rui
- College of Acupuncture and Orthopedics, Hubei University of Chinese Medicine, Wuhan 430061, Hubei Province, China
-
Revised:2017-09-25Online:2018-02-18Published:2018-02-18 -
Contact:Peng Rui, M.D., Chief physician, Professor, College of Acupuncture and Orthopedics, Hubei University of Chinese Medicine, Wuhan 430061, Hubei Province, China -
About author:Wu Xi, Studying for doctorate, Attending physician, College of Acupuncture and Orthopedics, Hubei University of Chinese Medicine, Wuhan 430061, Hubei Province, China
CLC Number:
Cite this article
Wu Xi, Peng Rui. Icariin with different concentrations promotes osteogenic differentiation of human bone marrow mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(5): 669-674.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
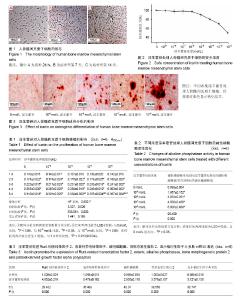
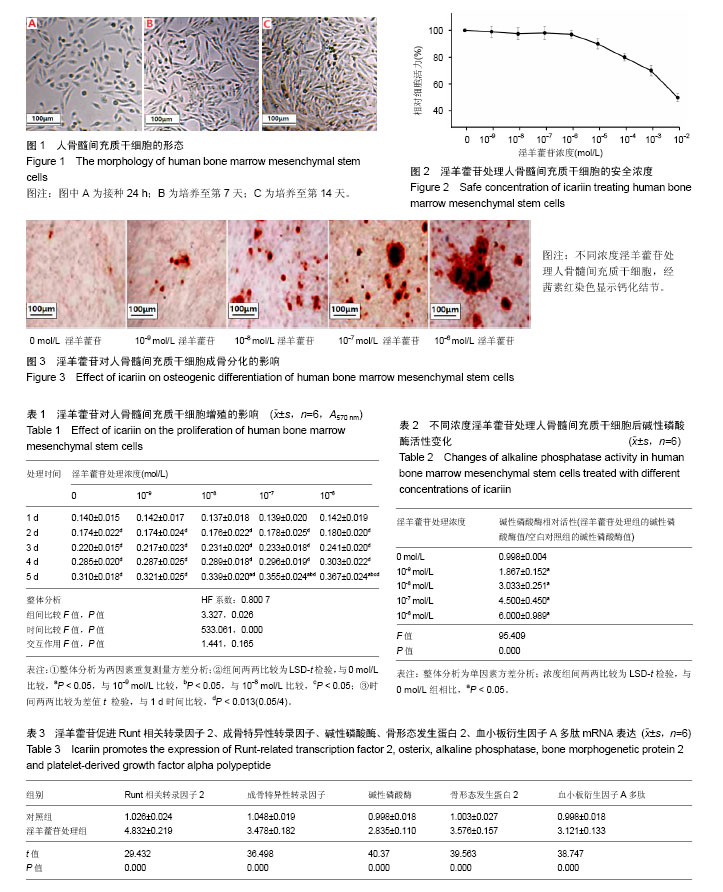
2.1 人骨髓间充质干细胞的形态学及生长特征 镜下观察发现,接种后24 h内可见少量细胞贴壁,呈短小棒状(图1A);第3,4天可见贴壁细胞数量增多,形成大小不同、散在分布的细胞集落,相比于贴壁初期,细胞呈细长梭形,伪足增长;第7天集落迅速增加、明显增多,细胞多呈梭形,排列紧密(图1B)。第14天细胞间相互紧密贴附生长,可达90%融合,呈漩涡状排列(图1C)。传代时,细胞经消化重新接种后4 h开始贴壁,初期呈圆形,逐渐伸展呈细小短棒状,最终完全舒展,恢复成梭形。传代后分裂增生速度明显加快,5-7 d传代1次,细胞均匀分布,不再以集落的方式生长,融合后再次呈典型漩涡状生长。 2.2 淫羊藿苷处理人骨髓间充质干细胞的合理浓度选择 以MTT方法来确定淫羊藿苷的合理工作浓度范围,将细胞以5×103/孔的密度接种96孔板,贴壁后,分别使用一系列梯度浓度(从10-9至10-2 mol/L)的淫羊藿苷处理细胞24 h,根据吸光度值绘制细胞相对活力曲线,结果发现淫羊藿苷在10-9 mol/L至10-6 mol/L范围内,人骨髓间充质干细胞存活均超过90%,即淫羊藿苷浓度在10-9 mol/L至10-6 mol/L范围内时,对人骨髓间充质干细胞的活力基本无影响(F=10.167,P=0.689),见图2,故后续实验均采用这几个浓度的淫羊藿苷处理细胞。 2.3 淫羊藿苷对人骨髓间充质干细胞增殖的影响 细胞以5×103/孔的密度接种于96孔板,贴壁后,换成无血清培养液培养24 h,使细胞周期同步化,选择0,10-9,10-8,10-7,10-6 mol/L的淫羊藿苷来处理人骨髓间充质干细胞5 d,观察其对细胞增殖能力的影响。如表1所示,随着时间延长,人骨髓间充质干细胞吸光度值逐渐升高,即细胞生长状态良好(P < 0.013);此外,淫羊藿苷对人骨髓间充质干细胞有轻度促增殖作用,但差异无显著性意义(P > 0.05)。 2.4 淫羊藿苷对人骨髓间充质干细胞成骨分化的影响 如表2所示,人骨髓间充质干细胞用含不同浓度淫羊藿苷的诱导分化培养基诱导后,与空白对照组相比,碱性磷酸酶活性升高,且碱性磷酸酶相对活性(淫羊藿苷组/空白对照组)随着淫羊藿苷处理浓度的加大而逐渐增强:10-9 mol/L淫羊藿苷处理组碱性磷酸酶活性是空白对照组的1.867倍,10-8 mol/L淫羊藿苷处理使碱性磷酸酶活性升高到3.033倍,10-7 mol/L淫羊藿苷处理使碱性磷酸酶活性升高到4.500倍,10-6 mol/L淫羊藿苷处理使碱性磷酸酶活性升高到6.000倍。 此外还观察了不同浓度淫羊藿苷处理组的矿化结节数(图3),茜素红染色结果表明:淫羊藿苷处理人骨髓间充质干细胞21 d后,细胞聚集生长、细胞外钙盐沉积,并形成点状的钙化结节,结节数目随着淫羊藿苷处理浓度的增加而增加,空白对照组有少量钙化结节,表明淫羊藿苷具有诱导人骨髓间充质干细胞骨向分化的作用。 2.5 淫羊藿苷上调Runt相关转录因子2、成骨特异性转录因子、碱性磷酸酶、骨形态发生蛋白2、血小板衍生因子Α多肽表达 当细胞在基础培养基中达到80%融合后,用成骨诱导分化培养基联合10-6 mol/L淫羊藿苷诱导培养9 d,提取RNA,荧光定量PCR检测相关基因表达水平,结果发现,与空白对照组比较,Runt相关转录因子2、成骨特异性转录因子、碱性磷酸酶、骨形态发生蛋白2、血小板衍生因子A多肽表达水平均有所增加(表3)。"
| [1] Mäkinen TJ, Abolghasemian M, Watts E, et al. Management of massive acetabular bone defects in revision arthroplasty of the hip using a reconstruction cage and porous metal augment. Bone Joint J. 2017;99-B(5):607-613. [2] Reddy BR, Sudhakar J, Rajesh N, et al. Comparative clinical and radiographic evaluation of mineralized cancellous bone allograft (puros®) and autogenous bone in the treatment of human periodontal intraosseous defects: 6-months follow-up study. J Int Soc Prev Community Dent. 2016;6(Suppl 3):S248-S253.[3] Naito K, Sugiyama Y, Obata H, et al. Screw Fixation and Autogenous Bone Graft for an Irreducible Distal Ulna Fracture Associated with Distal Radius Fracture. J Hand Surg Asian Pac Vol. 2017;22(2):236-239.[4] Wongwitwichot P, Kaewsrichan J. Osteogenic differentiation of mesenchymal stem cells is impaired by bone morphogenetic protein 7. Adv Med Sci. 2017;62(2):266-272.[5] 谢婷婷,杨乃龙,徐丽丽,等.尿酸影响人骨髓间充质干细胞成骨分化过程中BMP-2表达[J].现代生物医学进展,2015,15(12):2251-2256.[6] Blázquez-Prunera A, Díez JM, Gajardo R, et al. Human mesenchymal stem cells maintain their phenotype, multipotentiality, and genetic stability when cultured using a defined xeno-free human plasma fraction. Stem Cell Res Ther. 2017;8(1):103.[7] Moll CW, Schmiedinger T, Moll MA, et al. Extracellular matrix mimicking scaffold promotes osteogenic stem cell differentiation: A new approach in osteoporosis research. Biomed Mater Eng. 2017; 28(2):87-103.[8] Um S, Kim HY, Lee JH, et al. TSG-6 secreted by mesenchymal stem cells suppresses immune reactions influenced by BMP-2 through p38 and MEK mitogen-activated protein kinase pathway. Cell Tissue Res. 2017;368(3):551-561.[9] Zhang Y, Weng S, Yin J, Vitamin K, et al. Vitamin K2 promotes mesenchymal stem cell differentiation by inhibiting miR?133a expression. Mol Med Rep. 2017;15(5):2473-2480.[10] Wu T, Shu T, Kang L, et al. Icaritin, a novel plant-derived osteoinductive agent, enhances the osteogenic differentiation of human bone marrow- and human adipose tissue-derived mesenchymal stem cells. Int J Mol Med. 2017;39(4):984-992.[11] Kim DR, Lee JE, Shim KJ, et al. Effects of herbal Epimedium on the improvement of bone metabolic disorder through the induction of osteogenic differentiation from bone marrow-derived mesenchymal stem cells. Mol Med Rep. 2017;15(1):125-130.[12] Telang NT, Li G, Katdare M, et al. The nutritional herb Epimedium grandiflorum inhibits the growth in a model for the Luminal A molecular subtype of breast cancer. Oncol Lett. 2017;13(4): 2477-2482.[13] Han YY, Song MY, Hwang MS, et al. Epimedium koreanum Nakai and its main constituent icariin suppress lipid accumulation during adipocyte differentiation of 3T3-L1 preadipocytes. Chin J Nat Med. 2016;14(9):671-676.[14] Huang W, Khaldun AB, Lv H, et al. Isolation and functional characterization of a R2R3-MYB regulator of the anthocyanin biosynthetic pathway from Epimedium sagittatum. Plant Cell Rep. 2016;35(4):883-894.[15] Xie JP, Xiang JM, Zhu ZL. Determination of Five Major 8-Prenylflavones in Leaves of Epimedium by Solid-Phase Extraction Coupled with Capillary Electrophoresis. J Chromatogr Sci. 2016; 54(4):664-669.[16] 成魁,陈克明,葛宝丰,等.淫羊藿苷与金雀异黄酮骨保护作用的比较研究[J].中国药理学通报,2014,30(9):1315-1319.[17] Li X, Hou R, Yue C, et al. The Selenylation Modification of Epimedium Polysaccharide and Isatis Root Polysaccharide and the Immune-enhancing Activity Comparison of Their Modifiers. Biol Trace Elem Res. 2016;171(1):224-234.[18] Burim RA, Sendyk DI, Hernandes LS, et al. Repair of Critical Calvarias Defects With Systemic Epimedium sagittatum Extract. J Craniofac Surg. 2016;27(3):799-804.[19] Saha MK, Agrawal P, Saha SG, et al. Evaluation of Correlation between Salivary Calcium, Alkaline Phosphatase and Osteoporosis- A Prospective, Comparative and Observational Study. J Clin Diagn Res. 2017;11(3):ZC63-ZC66.[20] Sartor O, Coleman RE, Nilsson S, et al. An exploratory analysis of alkaline phosphatase, lactate dehydrogenase, and prostate-specific antigen dynamics in the phase 3 ALSYMPCA trial with radium-223. Ann Oncol. 2017;28(5):1090-1097.[21] Pettengill M, Matute JD, Tresenriter M, et al. Human alkaline phosphatase dephosphorylates microbial products and is elevated in preterm neonates with a history of late-onset sepsis. PLoS One. 2017;12(4):e0175936.[22] Wannhoff A, Rauber C, Friedrich K, et al. Von Willebrand factor and alkaline phosphatase predict re-transplantation-free survival after the first liver transplantation. United European Gastroenterol J. 2017; 5(1):86-93.[23] Oladipo OO, DeCrescenzo AJ, Marquez CP, et al. Increased Alkaline Phosphatase in a Child. Clin Chem. 2017;63(6):1174-1175.[24] Sun L, Yan ZH, Yang XT, et al. Osteogenic Ability Detection of Human Bone Morphogenetic Protein-2 Gene-activated Nano Bone Putty by Reusable Double-Cavity Bone Harvest Chamber. Orthop Surg. 2017;9(1):123-128.[25] Song R, Fullerton DA, Ao L, et al. An epigenetic regulatory loop controls pro-osteogenic activation by TGF-β1 or bone morphogenetic protein 2 in human aortic valve interstitial cells. J Biol Chem. 2017;292(21):8657-8666.[26] MacIsaac ZM, Henderson SE, Shakir S, et al. Biomechanical Integrity in Craniofacial Surgery: Calvarial Reconstruction in Favorable and Infected Defects with Bone Morphogenetic Protein 2. Plast Reconstr Surg. 2017;139(5):1141-1150.[27] Aksel H, Huang GT. Combined Effects of Vascular Endothelial Growth Factor and Bone Morphogenetic Protein 2 on Odonto/Osteogenic Differentiation of Human Dental Pulp Stem Cells In Vitro. J Endod. 2017;43(6):930-935.[28] Hindoyan K, Tilan J, Buser Z, et al. A Retrospective Analysis of Complications Associated With Bone Morphogenetic Protein 2 in Anterior Lumbar Interbody Fusion. Global Spine J. 2017;7(2): 148-153.[29] Li P, Li Y, Zhou AH, et al. Association Study of a Proliferation-inducing Ligand, Spermatogenesis Associated 8, Platelet-derived Growth Factor Receptor-alpha, and POLB Polymorphisms with Systemic Lupus Erythematosus in Chinese Han Population. Chin Med J (Engl). 2016;129(17):2085-2090.[30] Stegmann C, Hochdorfer D, Lieber D, et al. A derivative of platelet-derived growth factor receptor alpha binds to the trimer of human cytomegalovirus and inhibits entry into fibroblasts and endothelial cells. PLoS Pathog. 2017;13(4):e1006273.[31] Feng R, Feng L, Yuan Z, et al. Icariin protects against glucocorticoid-induced osteoporosis in vitro and prevents glucocorticoid-induced osteocyte apoptosis in vivo. Cell Biochem Biophys. 2013;67(1):189-197.[32] Lee JH, Kim K, Kim NR, et al. The complete chloroplast genome of a medicinal plant Epimedium koreanum Nakai (Berberidaceae). Mitochondrial DNA A DNA Mapp Seq Anal. 2016;27(6):4342-4343.[33] 方晔,邹斌,赵劲民,等.α-玉米赤霉醇在雌激素缺乏所致骨质疏松症中的研究进展[J].广西医科大学学报,2014,31(5):870-872.[34] 赵艳威,李宗旻,宋光明,等.葛根素促进人成骨样MG-63细胞分化的分子机制研究[J].中草药,2014,45(4):536-540.[35] Wan Y, Zhuo N, Li Y, et al. Autophagy promotes osteogenic differentiation of human bone marrow mesenchymal stem cell derived from osteoporotic vertebrae. Biochem Biophys Res Commun. 2017;488(1):46-52.[36] 李亘,贺韬,张超,等.瘦素促进骨髓间充质干细胞成骨分化并增加内植物-骨界面骨生成[J].上海医学,2016 39(3):156-159,194.[37] Gu R, Lei B, Jiang C, et al. Glucocorticoid-Induced Leucine Zipper Suppresses ICAM-1 and MCP-1 Expression by Dephosphorylation of NF-κB p65 in Retinal Endothelial Cells. Invest Ophthalmol Vis Sci. 2017;58(1):631-641.[38] Li A, Xia X, Yeh J, et al. PDGF-AA promotes osteogenic differentiation and migration of mesenchymal stem cell by down-regulating PDGFRα and derepressing BMP-Smad1/5/8 signaling. PLoS One. 2014;9(12):e113785.[39] 代志鹏,许伟华,杨述华,等.人骨髓间充质干细胞的生物学特性及成骨诱导分化的研究[J].中国矫形外科杂志,2014,22(15):1402-1407. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [14] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||