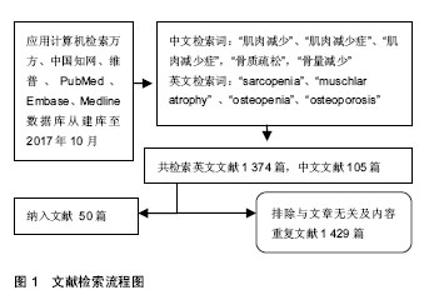
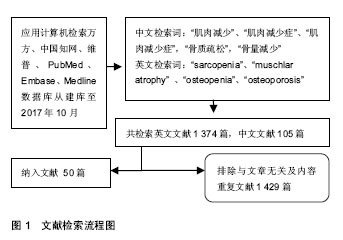
Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (32): 5222-5227.doi: 10.3969/j.issn.2095-4344.0380
Previous Articles Next Articles
Association of sarcopenia with osteoporosis: research and application
Ma Jian-xiong1, 2, 3, Kuang Ming-jie1, 2, He Wei-wei1, 2, Chen Heng-ting1, 2, Wang Ying1, 2, Sun Lei1, 2, Lu Bin1, 2, Tian Ai-xian1, 2, Wang Yan1, 2, Dong Ben-chao1, 2, Bo Hao-hao1, 2, Cui Yu-hong3, Ma Xin-long1, 2
- 1Orthopedic Institute of Tianjin Hospital, Tianjin University, Tianjin 300211, China; 2Tianjin Orthopedic Institute, Tianjin 300211, China; 3School of Mechanical Engineering, Tianjin University, Tianjin 300072, China
-
Received:2018-04-20Online:2018-11-18Published:2018-11-18 -
Contact:Ma Xin-long, Master, Professor, Chief physician, Orthopedic Institute of Tianjin Hospital, Tianjin University, Tianjin 300211, China; Tianjin Orthopedic Institute, Tianjin 300211, China -
About author:Ma Jian-xiong, PhD, Associate researcher, Orthopedic Institute of Tianjin Hospital, Tianjin University, Tianjin 300211, China; Tianjin Orthopedic Institute, Tianjin 300211, China; School of Mechanical Engineering, Tianjin University, Tianjin 300072, China -
Supported by:the National Natural Science Foundation of China, No. 11772226, 1572154, 1571723 and 1673994; a grant from the Science and Technology Research & Development Project of Tianjin Health Bureau, No. 2014KY31
CLC Number:
Cite this article
Ma Jian-xiong1, 2, 3, Kuang Ming-jie1, 2, He Wei-wei1, 2, Chen Heng-ting1, 2, Wang Ying1, 2, Sun Lei1, 2, Lu Bin1, 2, Tian Ai-xian1, 2, Wang Yan1, 2, Dong Ben-chao1, 2, Bo Hao-hao1, 2, Cui Yu-hong3, Ma Xin-long1, 2. Association of sarcopenia with osteoporosis: research and application[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(32): 5222-5227.
share this article
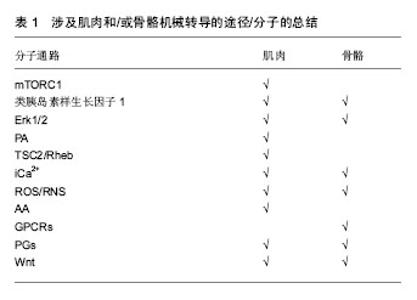
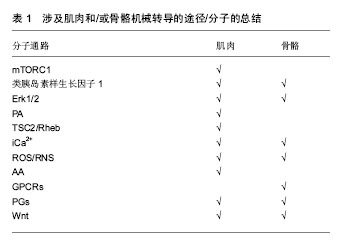
2.2 肌肉与骨骼的相互联系 肌肉骨骼均起源于中胚层,有着共同的间质前体,因此肌肉和骨骼会受到某些相同的遗传因素的调控;二者在运动功能上紧密关联,肌肉是骨与骨连接的纽带,与骨的生长和发育密切相关。肌肉和骨骼作为运动系统的两大重要组成部分,共同受机体多种因素的调节[10]。在生长发育过程中肌量与骨量密切相关,骨量和肌力的增加几乎同步进行,人体生长期时二者均增加,青壮年时期步入停滞期,最后进入减少期[11]。在一项对双胞胎的研究中,发现体质量和骨密度之间的关系被是受遗传因素的影响,而脂肪量和骨密度之间的关系不受遗传因素影响[12]。骨和肌肉间不仅在解剖上联系紧密,而且在功能和发育上密不可分。 2.3 肌肉与骨的相互调控 2.3.1 肌肉对骨的调控 1960年前,认为骨的生理模式主要是刺激-效应细胞-健康/疾病,决定人体骨骼强度的主要是非机械性因素对成骨细胞和破骨细胞这两个效应细胞的作用,进而发生成骨和骨吸收;显然,这种模式不仅忽视了机械性因素对骨质和骨强度的作用,也未重视组织和器官对骨骼的影响。认识到该模式的缺陷与不足,犹他大学医学院组织研究者们对骨生理模式进行研讨并改进,创立犹他骨生理模式[13]。使骨组织变形的外力作用中,主要由肌肉收缩产生[14];当骨应变超过骨重建阈值时,骨就会进入保留模式使骨吸收和形成平衡,骨得以保留;而当其低于骨重建阈值时,比如肌肉减少症患者,骨就会进入废用模式,骨的吸收多于形成,使骨量减少。也就是说,骨骼所承受的肌肉力学刺激对其发育和维持有着重要的作用。有文献对涉及肌肉和/或骨骼机械转导的分子通路进行总结,通过表1可以看出骨骼与肌肉这两种组织在力学传递方面有许多相似之处[15]。"

人体运动时,以肌肉收缩产生的力为主的应力系统开始对骨产生影响,在保证骨骼的健康方面有着重要的作用。Qin等[16]通过对脊柱损伤患者进行电刺激诱导肌肉收缩,进而减少骨质丢失的研究,提出肌肉收缩可减少破骨细胞生成,促进成骨细胞增殖,其机制可能与血清Ⅰ型胶原蛋白c端端肽、核因子kB活性受体的量相关。与之相对应的是Edwards等[17]发现,如若习惯性的运动缺失,像卧床、瘫痪等则会引起快速大量的骨矿质的丢失。 骨骼肌不仅通过力学作用对骨骼产生影响,还能通过化学作用对骨骼发挥作用。化学作用主要是指肌肉产生的化学物质可能通过旁分泌或内分泌机制作用于成骨(前体)细胞破骨细胞或骨细胞,促进成骨和/或抑制破骨。MyoD和Myf5基因敲除小鼠由于缺乏骨骼肌,小鼠胚胎在母体子宫内无自主活动能力,出生后难以存活,骨骼表现为矿化不良,且新生骨中的破骨细胞数量增多,提示肌肉可能促进骨骼发育的作用[18]。由肌肉产生的骨诱导因子和FAM5C是重要的分泌型骨形成因子,肌肉产生的其他内分泌因子包括类胰岛素1号增长因子、白细胞介素6、白细胞介素7、白细胞介素15、脑源性神经营养因子和成纤维细胞生长因子2等均可能影响骨代谢[19]。肌肉运动后产生的鸢尾素则可能通过Wnt-catenin 通路促进成骨细胞分化和RNKL/RNK途径抑制破骨细胞形成[20]。 2.3.2 骨骼对肌肉的调节 骨骼系统具有调节肌肉的作用,如成骨不全的患者表现为肌肉萎缩。成骨细胞或骨细胞分泌的因子,如骨钙素、类胰岛素1号增长因子、硬骨素、成纤维细胞生长因子23、前列腺素E2等,均可能对肌肉有调节作用[14]。骨钙素对肌肉会产生同化作用,骨骼中产生的骨钙素可能通过GPR6A/AMPK/ mTOR/S6激酶途径调节肌量和功能[21]。骨钙素还可能影响糖脂和能量代谢进而影响肌肉功能。异性因子羧基化骨钙素可部分修复功能受损的肌肉[21]。类胰岛素1号增长因子不仅可由肌细胞产生,骨细胞和成骨细胞也可生成,它可激活骨骼肌PI3K/Akt 通路[22]。在敲除骨细胞类胰岛素1号增长因子的小鼠中,类胰岛素1号增长因子的mRNA表达在骨骼和肌肉中分别中减少了65%和59%,虽然其在肌肉中的减少并不是关键性的,肌纤维形态并未改变,但是细胞核的大小和数量却发生改 变[23]。经典的Wnt信号通路的激活是肌肉分化所必需,骨细胞分泌的硬骨素和Ihh等对Wnt信号通路有调控作用,表明骨细胞可能远程调节肌细胞的分化[24]。骨细胞和成骨细胞分泌的成纤维细胞因子23具有抑制肾小管重吸收磷和降低1α羟化酶活性的作用,可导致低磷血症和活性维生素D水平过低,由此会影响肌肉的代谢和功能[25]。此外,骨骼细胞中的特异性间隙连接蛋白Connexin43可直接参与肌肉生长和功能的调控[26]。但究竟骨骼对肌肉的影响是如何通过机械应力和生物因子共同协调发挥作用的,仍有待深入研究。 2.4 肌肉与骨骼的激素调节 2.4.1 维生素D 又称为抗佝偻病维生素,属于脂溶性维生素。它对骨盐代谢的调节及保持肌肉和骨骼的正常功能有重要作用。血清维生素D缺乏对老年人来说是一个常见的健康问题,它可以导致身体活动能力变弱、增加跌倒的概率、同时也使骨折风险增大[27]。人骨骼肌细胞表面有活性维生素D受体,其受体的类型对骨骼肌的收缩力和骨盐密度有重要影响[28]。25-(OH)D是维生素D在体内最主要的循环形式,常作为反映体内维生素D营养状况的指标。有研究发现,25-(OH)D浓度≥20 μg/L的人群下肢骨骼肌肉功能和力量好于25-(OH)D浓度小于20 μg/L的患者[28]。在来自29个国家的多中心试验中也发现,绝经后患有骨质疏松症的女性(1.3%亚裔),血清25-(OH)D水平和骨密度关系密切[29]。 2.4.2 睾酮 睾酮又称睾丸素,由男性的睾丸或女性的卵巢分泌,肾上腺也可分泌少量睾酮。有研究发现性腺功能减退的青年及老年男性睾酮水平降低,继而骨量减少、骨质疏松,骨折发生率也因此明显增加[30]。对前列腺癌患者的研究表明,雄激素阻断治疗可以抑制睾酮产生,因此可控制肿瘤的生长,但却导致骨质疏松并增加骨折的发生[31]。此外,也有研究报道,雄激素阻断也可导致肌量的丢失、肌力减弱、身体活动能力衰退[32]。LeBlanc等[33]通过队列研究,最终测定了1 183名老年男性的性激素水平、身体成分和身体活动能力,发现若体内内源性睾酮含量较高,则老年人肌量的丢失会减少、下肢功能的减退会减缓。 2.4.3 糖皮质激素 糖皮质激素影响着肌肉的代谢与生长。Hallangerås等[34]在研究中,给小鼠连续7 d注射皮质醇,每天检测小鼠体质量和肌肉含量,发现实验组小鼠肌肉萎缩,尿中3-甲基组氨酸含量升高,与此相一致的是存在肌蛋白降解,并且发现泛素化mRNA,泛素化蛋白酶体C8亚单位,接合酶E214k等泛素化组分均升高,推测糖皮质激素可能通过泛素化途径来影响肌肉的代谢。并且有研究表示,注射米非司酮的大鼠可以阻断脓毒症引起的肌肉量的升高和肌纤维蛋白质分解[35]。同时糖皮质激素对骨组织细胞的分化、增殖与凋亡发挥着至关重要的作用。在生理浓度下,糖皮质激素调节成骨细胞的分化,刺激成骨细胞分泌Wnt信号通路相关蛋白,来激活经典Wnt信号通路,上调成骨细胞分化相关因子,使多能干细胞成骨分 化[36]。而外源性的糖皮质激素则主要会导致骨质疏松的发生。即使小剂量糖皮质激素也会对骨组织造成影响,当每日泼尼松龙的治疗剂量>7.5 mg 则可以引起骨质显著流失[37]。其具体机制虽不清楚,但有研究发现糖皮质激素可抑制成骨细胞分化和功能并且诱导其凋亡,进而抑制骨组织发育[38]。高水平的糖皮质激素通过糖原合成酶激酶3β泛素化功能和蛋白酶β-catenin 蛋白等可溶性拮抗因子致使间充质干细胞成骨分化下降,同时大剂量糖皮质激素也增加了过氧化物增殖物激活受体,促进间充质干细胞的成脂分化[39]。另一方面,成骨分化重要调节因子骨生成蛋白2,糖皮质激素抑制其下游信号,干预成骨分化。糖皮质激素致骨质疏松与骨组织细胞的凋亡机制相关,糖皮质激素还可以中断成骨细胞周期亦或是通过诱导的促凋亡激酶Pyk2和JNK的活化,诱导细胞凋亡[40]。而对于破骨细胞,糖皮质激素诱导的其数量以及活性的上升[41]。综上,糖皮质激素可通过抑制成骨细胞的生长周期,甚至诱导其凋亡来减少骨的生成,同时增加破骨细胞活性,加快骨量减少。 除了上述的维生素D、睾酮与皮质激素外,肌肉与骨骼的生长代谢还受着许多激素的共同调节,如生长激素、雌激素等。当体内这些激素的水平发生改变,骨骼和肌肉均可表现出相应症状,但由于骨骼和肌肉对于同一激素的敏感性并不相同,其中一者可能更早地表现出相应症状,这就为临床的早期诊断提供了机会,阻断骨骼和肌肉病变的进程,或是尽早预防、诊治另一者的病变。 2.5 肌肉减少症和骨质疏松相关性 肌肉减少症和骨质疏松症是增龄过程中两种较常见的病症,常同时出现。越来越多的证据表明肌肉减少症和骨质疏松症之间有很多相似点,比如:对某些激素分泌减少的反应、炎性因子的活跃性增加[14]。 多数大样本横断面研究显示,女性骨骼肌量与骨密度呈正相关,肌肉含量下降是骨质疏松症的重要危险因素。Miyakoshi等[42]调查40-89岁的2 400名日本女性,发现肌肉减少症在骨密度正常、骨量减少、骨质疏松的受试者中的检出率分别为10.4%,16.8%和20.4%,从而得出肌肉减少症和骨质减少以及骨质疏松症存在显著的相关性的结论。在对313名髋骨骨折的女性肌肉状况和骨质疏松状况的检测中发现,肌肉减少症患者为58%,骨质疏松者占74%,试验数据表明肌肉减少症与骨质疏松呈明显正相关关系[43]。 肌肉减少症和骨质疏松症具有相关性不仅存在于女性中,男性也不例外。Go等[44]调查了韩国1 397名老年男性,通过检测受试者四肢肌肉量,以及腰椎、股骨和股骨颈的骨密度,发现肌肉减少症和骨密度的减低存在相关。Pereira等[45]通过使用双能X射线吸收仪并以2010年欧洲老年肌肉减少症工作组对肌肉减少症的定义标准诊断肌肉减少症,对198名60岁以上男性的骨密度和是否患有肌肉减少症进行评估,发现肌肉减少症前期和肌肉减少症对骨密度减少存在影响。Verschueren等[46]为了探索肌肉减少症和骨质疏松症之间的关联性,检测了679个40-79岁的欧洲男性,发现患有肌肉减少症的男性较无肌肉减少症男性更易患骨质疏松症。国内有关肌肉减少症和骨密度相关性的研究报道较少。帅波等[47]研究武汉地区中老年男性肌肉减少症与骨密度的相关性,发现武汉地区中老年男性肌肉减少症与低骨密度和骨质疏松症密切相关。 2.6 肌肉减少症和骨折的相关性 骨折严重影响患者身心健康,并给家庭和社会带来沉重负担。肌肉减少症与骨质疏松的存在密不可分,而引起的骨密度减低显然会增加骨折风险。此外,肌肉减少症主要是Ⅱ型肌肉纤维的减少,并伴有运动神经元数量减少,显然,这些都影响肌肉含量及功能,从而引起步速减慢身体摇摆性增加,导致机体平衡能力减弱,因而增加跌倒风险,而跌倒往往是骨折的直接诱因[48-49]。同时,越来越多的国内外临床研究证实,肌肉减少症不仅与低骨密度密切关联,也是骨折的重要危险因素[50]。"

| [1] Rier HN, Jager A, Meinardi MC, et al.Severe sarcopenia might be associated with a decline of physical independence in older patients undergoing chemotherapeutic treatment.Support Care Cancer. 2017. doi: 10.1007/s00520-017-4018-8.[2] Fielding RA, Vellas B, Evans WJ, et al. Sarcopenia: an undiagnosed condition in older adults. Current consensus definition: prevalence, etiology, and consequences. International working group on sarcopenia. J Am Med Dir Assoc.2011;12(4):249-256.[3] Scherbakov N, Doehner W. Searching for a relevant definition of sarcopenia: results from the cross-sectional EPIDOS study. J Cachexia Sarcopenia Muscle.2016;7(1): 100-101.[4] Cheng Q, Zhu X, Zhang X, et al. A cross-sectional study of loss of muscle mass corresponding to sarcopenia in healthy Chinese men and women: reference values, prevalence, and association with bone mass. J Bone Miner Metab.2014;32(1): 78-88.[5] Peterson SJ, Braunschweig CA. Prevalence of Sarcopenia and Associated Outcomes in the Clinical Setting.Nutr Clin Pract.2016; 31(1):40-48.[6] Morley JE, Anker SD, von Haehling S. Prevalence, incidence, and clinical impact of sarcopenia: facts, numbers, and epidemiology- update 2014.J Cachexia Sarcopenia Muscle. 2014; 5(4):253-259.[7] Naseeb MA, Volpe SL. Protein and exercise in the prevention of sarcopenia and aging. Nutr Res.2017;40:1-20.[8] Makanae Y, Fujita S. Role of exercise and nutrition in the prevention of sarcopenia. J Nutr Sci Vitaminol (Tokyo).2015;61 Suppl:S125-127.[9] Binkley N, Krueger D, Buehring B. What's in a name revisited: should osteoporosis and sarcopenia be considered components of "dysmobility syndrome?". Osteoporos Int.2013;24(12):2955-2959.[10] DiGirolamo DJ, Clemens TL, Kousteni S. The skeleton as an endocrine organ. Nat Rev Rheumatol. 2012;8(11):674-683.[11] Frost HM. Muscle, bone, and the Utah paradigm: a 1999 overview. Med Sci Sports Exerc.2000; 32(5):911-917.[12] Bogl LH, Latvala A, Kaprio J, et al. An investigation into the relationship between soft tissue body composition and bone mineral density in a young adult twin sample.J Bone Miner Res.2011;26(1): 79-87.[13] Frost HM. The Utah paradigm of skeletal physiology: an overview of its insights for bone, cartilage and collagenous tissue organs.J Bone Miner Metab.2000;18(6): 305-316.[14] Tagliaferri C, Wittrant Y, Davicco MJ, et al. Muscle and bone, two interconnected tissues. Ageing Res Rev.2015;21: 55-70.[15] Laurent MR, Dubois V, Claessens F, et al. Muscle-bone interactions: From experimental models to the clinic? A critical update. Mol Cell Endocrinol.2016;432: 14-36.[16] Qin W, Sun L, Cao J, et al. The central nervous system (CNS)- independent anti-bone-resorptive activity of muscle contraction and the underlying molecular and cellular signatures. J Biol Chem. 2013; 288(19):13511-13521.[17] Edwards WB, Schnitzer TJ, Troy KL. Bone mineral loss at the proximal femur in acute spinal cord injury. Osteoporos Int.2013;24(9): 2461-2469.[18] Gomez C, David V, Peet NM, et al. Absence of mechanical loading in utero influences bone mass and architecture but not innervation in Myod-Myf5-deficient mice. J Anat. 2007;210(3):259-271.[19] Kawao N, Kaji H. Interactions between muscle tissues and bone metabolism. J Cell Biochem.2015;116(5):687-695.[20] Colaianni G, Cuscito C, Mongelli T, et al. The myokine irisin increases cortical bone mass. Proc Natl Acad Sci U S A. 2015;112(39): 12157-12162.[21] Karsenty G, Olson EN. Bone and Muscle Endocrine Functions: Unexpected Paradigms of Inter-organ Communication. Cell.2016; 164(6):1248-1256.[22] Schiaffino S, Mammucari C. Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: insights from genetic models. Skelet Muscle. 2011; 1(1): 4.[23] Govoni KE, Wergedal JE, Florin L, et al. Conditional deletion of insulin-like growth factor-I in collagen type 1alpha2-expressing cells results in postnatal lethality and a dramatic reduction in bone accretion. Endocrinology. 2007;148(12):5706-5715.[24] Bren-Mattison Y, Hausburg M, Olwin BB. Growth of limb muscle is dependent on skeletal-derived Indian hedgehog. Dev Biol. 2011; 356(2):486-495.[25] Quarles LD. Role of FGF23 in vitamin D and phosphate metabolism: implications in chronic kidney disease. Exp Cell Res.2012;318(9):1040-1048.[26] Borsheim E, Herndon DN, Hawkins HK, et al. Pamidronate attenuates muscle loss after pediatric burn injury. J Bone Miner Res.2014;29(6): 1369-1372.[27] Stewart JW, Alekel DL, Ritland LM, et al. Serum 25-hydroxyvitamin D is related to indicators of overall physical fitness in healthy postmenopausal women. Menopause. 2009;16(6):1093-1101.[28] Mastaglia SR, Seijo M, Muzio D, et al. Effect of vitamin D nutritional status on muscle function and strength in healthy women aged over sixty-five years. J Nutr Health Aging. 2011;15(5):349-354.[29] Kuchuk NO, Schoor NMV, Pluijm SM, et al. Vitamin D status, parathyroid function, bone turnover, and BMD in postmenopausal women with osteoporosis: global perspective. J Bone Miner Res.2009; 24(4):693-701.[30] Meier C, Nguyen TV, Handelsman DJ, et al. Endogenous sex hormones and incident fracture risk in older men: the Dubbo Osteoporosis Epidemiology Study. Arch Intern Med. 2008;168(1):47-54.[31] Ryan CW, Huo D, Bylow K, et al. Suppression of bone density loss and bone turnover in patients with hormone-sensitive prostate cancer and receiving zoledronic acid.BJU Int. 2007;100(1):70-75.[32] Storer TW, Miciek R, Travison TG. Muscle function, physical performance and body composition changes in men with prostate cancer undergoing androgen deprivation therapy. Asian J Androl. 2012;14(2):204-221.[33] LeBlanc ES, Wang PY, Lee CG, et al. Higher testosterone levels are associated with less loss of lean body mass in older men. J Clin Endocrinol Metab.2011;96(12): 3855-3863.[34] Hallangerås M, Angerås U, Zamir O, et al. Effect of the glucocorticoid receptor antagonist RU 38486 on muscle protein breakdown in sepsis. Surgery.1991;109(4): 468.[35] Hall-Angerås M, Angerås U, Zamir O, et al. Interaction between corticosterone and tumor necrosis factor stimulated protein breakdown in rat skeletal muscle, similar to sepsis. Surgery. Surgery. 1990;108(2): 460.[36] Zhou H, Mak W, Zheng Y, et al. Osteoblasts directly control lineage commitment of mesenchymal progenitor cells through Wnt signaling.J Biol Chem.2008;283(4): 1936-1945.[37] Murphy DR, Smolen LJ, Klein TM, et al. The cost effectiveness of teriparatide as a first-line treatment for glucocorticoid-induced and postmenopausal osteoporosis patients in Sweden. BMC Musculoskelet Disord.2012;13(1):213.[38] Kauh E, Mixson L, Malice MP, et al. Prednisone affects inflammation, glucose tolerance, and bone turnover within hours of treatment in healthy individuals. Eur J Endocrinol.2012;166(3):459-467.[39] Zhou H, Mak W, Kalak R, et al. Glucocorticoid-dependent Wnt signaling by mature osteoblasts is a key regulator of cranial skeletal development in mice. Development. 2009;136(3):427-436.[40] Bellido T. Antagonistic interplay between mechanical forces and glucocorticoids in bone: A tale of kinases. J Cell Biochem.2010;111(1):1-6.[41] Henneicke H, Herrmann M, Kalak R, et al. Corticosterone selectively targets endo-cortical surfaces by an osteoblast-dependent mechanism. Bone. 2011;49(4): 733.[42] Miyakoshi N, Hongo M, Mizutani Y, et al. Prevalence of sarcopenia in Japanese women with osteopenia and osteoporosis. J Bone Miner Metab.2013;31(5):556-561.[43] Monaco MD, Vallero F, Monaco RD, et al. Prevalence of sarcopenia and its association with osteoporosis in 313 older women following a hip fracture. Arch Gerontol Geriatr.2011;52(1):71-74.[44] Go SW, Cha YH, Lee JA, et al. Association between Sarcopenia, Bone Density, and Health-Related Quality of Life in Korean Men. Korean J Fam Med. 2013; 34(4): 281-288.[45] Pereira FB, Leite AF, de Paula AP. Relationship between pre-sarcopenia, sarcopenia and bone mineral density in elderly men. Arch Endocrinol Metab. 2015;59(1):59-65.[46] Verschueren S, Gielen E, O'Neill TW, et al. Sarcopenia and its relationship with bone mineral density in middle-aged and elderly European men. Osteoporos Int.2013; 24(1): 87-98.[47] 帅波,沈霖,杨艳萍,等.武汉地区中老年男性肌肉减少症与骨密度的相关性[J].中华骨质疏松和骨矿盐疾病杂志, 2016,9(3):257-263.[48] Bischoff-Ferrari HA, Orav JE, Kanis JA, et al. Comparative performance of current definitions of sarcopenia against the prospective incidence of falls among community-dwelling seniors age 65 and older. Osteoporos Int.2015;26(12): 2793-2802.[49] Szulc P, Feyt C, Chapurlat R. High risk of fall, poor physical function, and low grip strength in men with fracture—the STRAMBO study. J Cachexia Sarcopenia Muscle. 2015;7(3): 299-311[50] Tarantino U, Piccirilli E, Fantini M, et al. Sarcopenia and fragility fractures: molecular and clinical evidence of the bone-muscle interaction. J Bone Joint Surg Am.2015; 97(5):429-437. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Lü Zhen, Bai Jinzhu. A prospective study on the application of staged lumbar motion chain rehabilitation based on McKenzie’s technique after lumbar percutaneous transforaminal endoscopic discectomy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1398-1403. |
| [3] | Fan Jiabing, Zhang Junmei. Morphological measurement and analysis of the mandible in adult females with different vertical skeletal types [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1177-1183. |
| [4] | Song Liming, Su Hailong. Muscle force response characteristics of the slipping leg after an unexpected slip [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1184-1189. |
| [5] | Tang Hui, Yao Zhihao, Luo Daowen, Peng Shuanglin, Yang Shuanglin, Wang Lang, Xiao Jingang. High fat and high sugar diet combined with streptozotocin to establish a rat model of type 2 diabetic osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1207-1211. |
| [6] | Li Zhongfeng, Chen Minghai, Fan Yinuo, Wei Qiushi, He Wei, Chen Zhenqiu. Mechanism of Yougui Yin for steroid-induced femoral head necrosis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1256-1263. |
| [7] | Wang Mengting, Gu Yanping, Ren Wenbo, Qin Qian, Bai Bingyi, Liao Yuanpeng. Research hotspots of blood flow restriction training for dyskinesia based on visualization analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1264-1269. |
| [8] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [9] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [10] | Hou Guangyuan, Zhang Jixue, Zhang Zhijun, Meng Xianghui, Duan Wen, Gao Weilu. Bone cement pedicle screw fixation and fusion in the treatment of degenerative spinal disease with osteoporosis: one-year follow-up [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 878-883. |
| [11] | Yang Weiqiang, Ding Tong, Yang Weike, Jiang Zhengang. Combined variable stress plate internal fixation affects changes of bone histiocyte function and bone mineral density at the fractured end of goat femur [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 890-894. |
| [12] | Li Shibin, Lai Yu, Zhou Yi, Liao Jianzhao, Zhang Xiaoyun, Zhang Xuan. Pathogenesis of hormonal osteonecrosis of the femoral head and the target effect of related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 935-941. |
| [13] | Xiao Fangjun, Chen Shudong, Luan Jiyao, Hou Yu, He Kun, Lin Dingkun. An insight into the mechanism of Salvia miltiorrhiza intervention on osteoporosis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 772-778. |
| [14] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [15] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||