Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (16): 2496-2501.doi: 10.3969/j.issn.2095-4344.0245
Previous Articles Next Articles
Effect of treadmill training on the fitness and activation of cerebral cortex in older adults and youth
Li Jin-tian
- College of Physical Education, Shaoguan University, Shaoguan 512026, Guangdong Province, China
-
Received:2018-04-04Online:2018-06-08Published:2018-06-08 -
About author:Li Jin-tian, Associate professor, College of Physical Education, Shaoguan University, Shaoguan 512026, Guangdong Province, China -
Supported by:the Social Science Foundation of Guangdong Province, No. GD17CTY02
CLC Number:
Cite this article
Li Jin-tian. Effect of treadmill training on the fitness and activation of cerebral cortex in older adults and youth[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(16): 2496-2501.
share this article
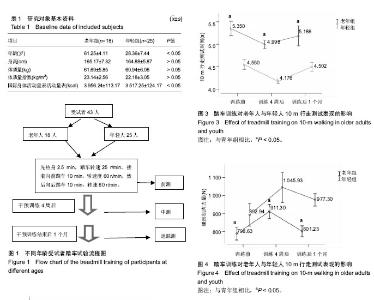
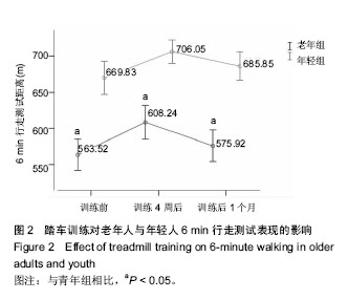
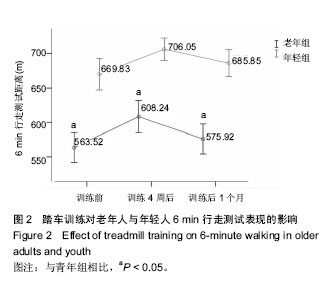
2.1 受试者基线资料分析 表1显示,2组受试者的人口学资料,除了年龄与国际身体活动量表活动量差异有显著性意义外(P < 0.05),其余皆无显著性意义,但老年人组体质量指数稍高于年轻人,但仍在正常范围。试验流程见图1。 2.2 老年人与年轻人6 min行走测试结果 如图2所示,老年组训练前6 min行走测试成绩比年轻人差(P < 0.05)。经过4周踏车训练后,老年组6 min行走测试成绩显著提升至608.24 m;年轻组6 min行走测试成绩显著提升至 706.05 m,2组提升率无显著性差异(P > 0.05)。停止训练1个月后,老年组6 min行走测试成绩显著下降至575.92 m,该成绩与训练前情况接近(P > 0.05);年轻组6 min行走测试成绩同样呈显著下降到685.85 m,但依旧显著高于训练前(P < 0.05)。 2.3 老年人与年轻人10 m行走测试结果 如图3所示,老年组训练前10 m行走结果比年轻组慢(P < 0.05)。而经过4周踏车训练后,老年人组行走结果降至4.998 s,成绩提升率约6%;年轻组行走结果降至4.176 s,成绩提升幅度约8%;年轻组成绩提升率显著高于老年组(P < 0.05)。训练"
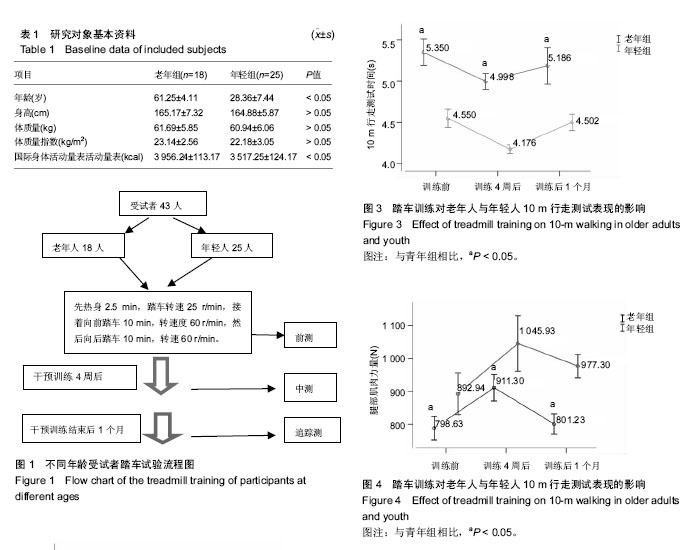
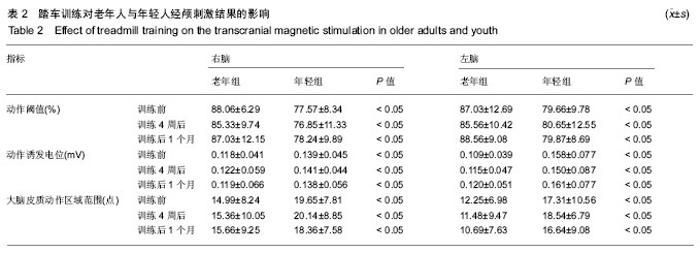
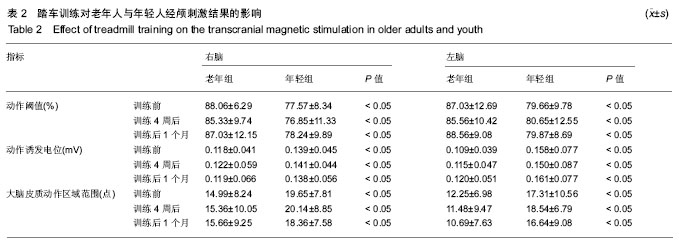
后1个月时,老年组10 m行走结果显著下降至5.186 s,但仍显著低于训练前(P < 0.05);而年轻组10m行走结果下降到4.502 s,与训练前接近(P > 0.05)。 2.4 老年人与年轻人腿肌力测试结果 如图4所示,老年组训练前腿部肌肉力量显著低于年轻组(P < 0.05)。经4周训练后,老年组腿部肌肉力量显著提升到911.30 N,提升率约15.0%,年轻显著提升到1 045.93 N,提升率约17.0%,且2组腿部肌肉力量提升率接近(P > 0.05)。停止训练1个月后,老年组腿部肌肉力量下降至81.76 kg,与训练前接近(P > 0.05);年轻组腿部肌肉力量下降至99.24 kg,仍显著高于训练前(P < 0.05)。 2.5 经颅磁刺激检测结果 表2显示,在训练前、训练4周及训练后1个月时,不论是左脑或右脑,老年组动作阈值显著高于年轻组(P < 0.05),动作诱发电位显著小于年轻组(P < 0.05),大脑皮质动作区域范围小于年轻组(P < 0.05)。在训练前、训练4周及训练后1月时,老年组左右脑动作阈值、动作诱发电位及大脑皮质区域范围接近(P > 0.05)。年轻组所表现的规律与老年组完全相同。"
| [1] Brown BM, Rainey-Smith SR, Castalanelli N, et al. Study protocol of the Intense Physical Activity and Cognition study: The effect of high-intensity exercise training on cognitive function in older adults. Alzheimers Dement (N Y). 2017;3(4):562-570. [2] Deal JA, Albert MS, Arnold M, et al. A randomized feasibility pilot trial of hearing treatment for reducing cognitive decline: Results from the Aging and Cognitive Health Evaluation in Elders Pilot Study. Alzheimers Dement (N Y). 2017;3(3):410-415. [3] Mariño RJ, Marwaha P, Barrow SY. Web-based oral health promotion program for older adults: Development and preliminary evaluation. Int J Med Inform. 2016;91:e9-e15. [4] Hong J, Kim J, Kim SW, et al. Effects of home-based tele-exercise on sarcopenia among community-dwelling elderly adults: Body composition and functional fitness. Exp Gerontol. 2017;87(Pt A):33-39. [5] Pijnappels M, Reeves ND, Maganaris CN, et al. Tripping without falling; lower limb strength, a limitation for balance recovery and a target for training in the elderly. J Electromyogr Kinesiol. 2008;18(2):188-196. [6] Lemmer JT, Martel GF, Hurlbut DE, et al. Age and sex differentially affect regional changes in one repetition maximum strength. J Strength Cond Res. 2007;21(3):731-737.[7] Ciolac EG, Garcez-Leme LE, Greve JM. Resistance exercise intensity progression in older men. Int J Sports Med. 2010;31(6):433-438. [8] Clark BC, Taylor JL. Age-related changes in motor cortical properties and voluntary activation of skeletal muscle. Curr Aging Sci. 2011;4(3): 192-199.[9] Lin PY, Lin SI, Chen JJ. Functional near infrared spectroscopy study of age-related difference in cortical activation patterns during cycling with speed feedback. IEEE Trans Neural Syst Rehabil Eng. 2012;20(1):78-84. [10] Muellbacher W, Ziemann U, Boroojerdi B, et al. Role of the human motor cortex in rapid motor learning. Exp Brain Res. 2001;136(4):431-438.[11] Ngomo S, Leonard G, Mercier C. Influence of the amount of use on hand motor cortex representation: effects of immobilization and motor training. Neuroscience. 2012;220:208-214. [12] Yang YR, Chen IH, Liao KK, et al. Cortical reorganization induced by body weight-supported treadmill training in patients with hemiparesis of different stroke durations. Arch Phys Med Rehabil. 2010;91(4):513-518. [13] Lovell DI, Cuneo R, Gass GC. Can aerobic training improve muscle strength and power in older men? J Aging Phys Act. 2010;18(1):14-26.[14] Janssen TW, Beltman JM, Elich P, et al. Effects of electric stimulation- assisted cycling training in people with chronic stroke. Arch Phys Med Rehabil. 2008;89(3):463-469. [15] Yamamoto S, Hotta K, Ota E, et al. Effects of resistance training on muscle strength, exercise capacity, and mobility in middle-aged and elderly patients with coronary artery disease: A meta-analysis. J Cardiol. 2016;68(2):125-134.[16] Yang HC, Lee CL, Lin R, et al. Effect of biofeedback cycling training on functional recovery and walking ability of lower extremity in patients with stroke. Kaohsiung J Med Sci. 2014;30(1):35-42.[17] Bruce RA. Exercise, functional aerobic capacity, and aging--another viewpoint. Med Sci Sports Exerc. 1984;16(1):8-13.[18] Santana MG, de Lira CA, Passos GS, et al. Is the six-minute walk test appropriate for detecting changes in cardiorespiratory fitness in healthy elderly men? J Sci Med Sport. 2012;15(3):259-265.[19] Holviala JH, Sallinen JM, Kraemer WJ, et al. Effects of strength training on muscle strength characteristics, functional capabilities, and balance in middle-aged and older women. J Strength Cond Res. 2006; 20(2):336-344.[20] Heo S, Prakash RS, Voss MW, et al. Resting hippocampal blood flow, spatial memory and aging. Brain Res. 2010;1315:119-127. [21] Ogoh S. Relationship between cognitive function and regulation of cerebral blood flow. J Physiol Sci. 2017;67(3):345-351. [22] Kaup AR, Mirzakhanian H, Jeste DV, et al. A review of the brain structure correlates of successful cognitive aging. J Neuropsychiatry Clin Neurosci. 2011;23(1):6-15. [23] Bertsch K, Hagemann D, Hermes M, et al. Resting cerebral blood flow, attention, and aging. Brain Res. 2009;1267:77-88.[24] Mitschelen M, Garteiser P, Carnes BA, et al. Basal and hypercapnia- altered cerebrovascular perfusion predict mild cognitive impairment in aging rodents. Neuroscience. 2009;164(3):918-928. [25] Steffener J, Brickman AM, Habeck CG, et al. Cerebral blood flow and gray matter volume covariance patterns of cognition in aging. Hum Brain Mapp. 2013;34(12):3267-3279. [26] Lucas SJ, Ainslie PN, Murrell CJ, et al. Effect of age on exercise-induced alterations in cognitive executive function: relationship to cerebral perfusion. Exp Gerontol. 2012;47(8):541-551. [27] Griffin ÉW, Mullally S, Foley C, et al. Aerobic exercise improves hippocampal function and increases BDNF in the serum of young adult males. Physiol Behav. 2011;104(5):934-941. [28] Whiteman AS, Young DE, He X, et al. Interaction between serum BDNF and aerobic fitness predicts recognition memory in healthy young adults. Behav Brain Res. 2014;259:302-312. [29] Griffin EW, Bechara RG, Birch AM, et al. Exercise enhances hippocampal-dependent learning in the rat: evidence for a BDNF-related mechanism. Hippocampus. 2009;19(10):973-980. [30] Lee TM, Wong ML, Lau BW, et al. Aerobic exercise interacts with neurotrophic factors to predict cognitive functioning in adolescents. Psychoneuroendocrinology. 2014;39:214-224. [31] Tsai CL, Pan CY, Chen FC, et al. Effects of acute aerobic exercise on a task-switching protocol and brain-derived neurotrophic factor concentrations in young adults with different levels of cardiorespiratory fitness. Exp Physiol. 2016;101(7):836-850. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Zhao Xiang, Wei Cuilan, Zhang Yeting. Neurogenesis and neuroinflammation under exercise: alteration and regulation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 813-820. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Fan Jin, Zeng Luyao, Zhong Dongling, Li Yuxi, Tian Yanping, Huang Yijie, Jin Rongjiang. Development of functional near-infrared spectroscopy in recent 10 years: a visual analysis using CiteSpace [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3711-3717. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||