Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (28): 4587-4592.doi: 10.3969/j.issn.2095-4344.0241
Expression and role of wnt signaling pathway in neurogenesis and neurodegeneration
Wang Xian-bin1, Tang Hong-yan2, Li Xing-tong1, Ma Wei1, Yang Jin-wei1, 3, Dai Yun-fei1, Zhang Tong3, Guo Jian-hui3, Li Li-yan1
- 1Institute for Neuroscience, Kunming Medical University, Kunming 650500, Yunnan Province, China; 2Department of Neurosurgery, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China; 3Second Department of General Surgery, the First People’s Hospital of Yunnan Province, Kunming 650032, Yunnan Province, China
-
Received:2018-02-03 -
Contact:Guo Jian-hui, Professor, Second Department of General Surgery, the First People’s Hospital of Yunnan Province, China Corresponding author: Li Li-yan, M.D., Professor, Institute for Neuroscience, Kunming Medical University, Kunming 650500, Yunnan Province, China -
About author:Wang Xian-bin, Institute for Neuroscience, Kunming Medical University, Kunming 650500, Yunnan Province, China Tang Hong-yan, Nurse-in-charge, Department of Neurosurgery, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China Wang Xian-bin and Tang Hong-yan contributed equally to this work. -
Supported by:the National Natural Science Foundation of China, No. 31560295; the Health Science Technology Program of Yunnan Province, No. 2014NS202; the Combined Project of Kunming Medical University, No. 2015FB098
CLC Number:
Cite this article
Wang Xian-bin1, Tang Hong-yan2, Li Xing-tong1, Ma Wei1, Yang Jin-wei1, 3, Dai Yun-fei1, Zhang Tong3, . Expression and role of wnt signaling pathway in neurogenesis and neurodegeneration[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(28): 4587-4592.
share this article
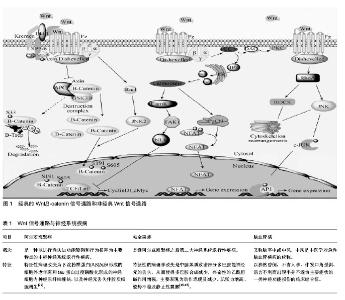
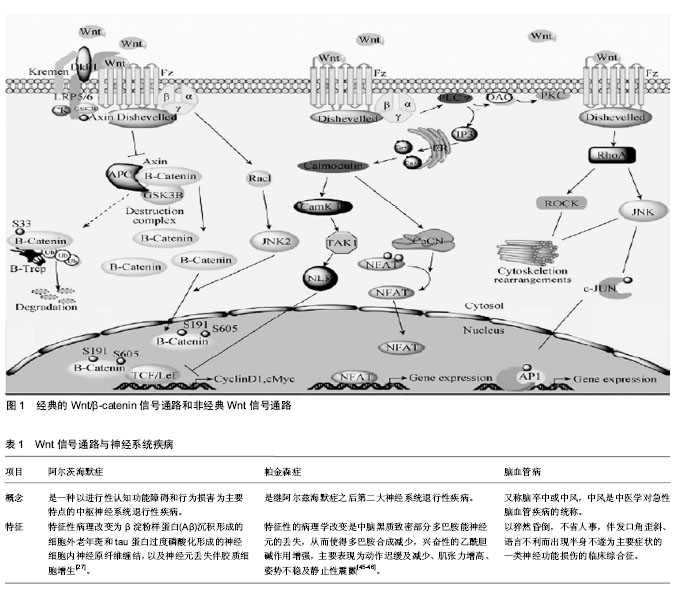
2.1 Wnt信号通路概述 Wnt信号通路是广泛存在于多细胞真核生物中,并且在进化上高度保守的信号通路。1982年Nusse在研究中发现了Wnt基因[3],该基因被命名为Int1,其编码的蛋白在细胞间传递生长和发育信号。随后在果蝇中的研究发现,该基因可以影响果蝇的发育,导致果蝇表现为无翅,因此将wingless与Int1合称为Wnt1[4]。Wnt蛋白由350-400个氨基酸组成,其羧基末端含有23或24个保守的半胱氨酸残基。Wnt蛋白作为Wnt信号通路中的配体蛋白,可与受体结合激活通路,调控下游靶基因cyclinD1、ngn-1、ngn-2、c-myc的转录和翻译[5-6],进而调控细胞的增殖和分化。 目前认为Wnt信号通路主要由以下几种蛋白构成:细胞外因子Wnt家族分泌蛋白、特异性跨膜受体卷曲蛋白、散乱蛋白、β-连环蛋白(β-catenin)、结肠腺瘤性息肉病基因蛋白、糖原合成酶激酶3β、Axin或Conductin(轴蛋白或传导蛋白)及TCF/LEF(T细胞因子/淋巴增强因子)等[7]。Wnt信号通路的构成及调控见图1。 2.1.1 经典Wnt信号通路 经典Wnt信号通路即Wnt/β-catenin信号通路,参与了细胞增殖、凋亡及细胞命运调控,是Wnt信号通路中研究的比较透彻的一条通路。在没有Wnt蛋白存在时,GSK-3β、β-catenin、APC蛋白、Axin及CK1组成的复合物在GSK-3β的作用下降解β-catenin,而不引起下游基因的表达。当存在分泌的Wnt蛋白时,Wnt蛋白通过与特异性跨膜受体卷曲蛋白受体Fzd、LRP5和LRP6结合,抑制β-catenin复合物的活性而使β-catenin免于被降解,胞浆内β-catenin累积并稳定后会转移至细胞核与Lef/Tcf家族的转录因子结合[8],并与细胞内的其他因子共同作用下激活T细胞因子/淋巴增强因子,特异性的启动、激活下游靶基因的转录[9-11]。 2.1.2 非经典Wnt信号通路 经典Wnt信号通路转导的关键是β-catenin在胞浆中积累的量,但研究发现Wnt蛋白家族的一些成员可不通过β-catenin来行使生物学功能,主要包括Wnt-Ca2+信号通路、平面极细胞通路及调节纺锤体的方向和非对称细胞分裂的胞内通路。 Wnt/Ca2+通路是通过Wnt5a和Wntl1激活一个激酶的级联反应。Wnt通过Fzd家族,在G蛋白的介导下促进细胞内Ca2+的释放,从而激活磷脂酶C(PLC)和蛋白激酶C(PKC),引起细胞内Ca2+浓度增加和Ca2+敏感信号成分的激活,以调控细胞的迁徙和细胞的黏附性,由于Ca2+在整个过程中起了关键作用,所以叫Wnt-Ca2+[12]。该通路能够拮抗经典的Wnt通路,在细胞生长与胚胎发育中具有极其重要的作用,它可以影响细胞极性、引导细胞移形、诱导细胞凋亡以及参与胚胎发育等过程[10,13]。 平面极细胞通路又称为Wnt-JNK通路,Wnt/polarity通路主要是通过Wnt蛋白激活Dsh蛋白的DEP结构域、Rac、小GTP酶、Rho和Cde42等,再由GTPase RhoA介导,激活JNK,JNK一旦被激活,胞质中的JNK进入到细胞核,活化的JNK与转录因子ATF2及c-jun的氨基末端区域结合,使转录因子ATF2及c-jun的活性区域磷酸化,从而调控基因表达[13]。该通路主要与细胞极性的产生和细胞骨架重排,以及细胞骨架的不对称分布的调控和上皮细胞的协同极化密切相关[14-15]。 2.1.3 Wnt信号通路与神经发生 神经发生(neurogenesis)是包括从神经干细胞增殖并经历均衡和不均衡性分裂成为定向祖细胞,并逐渐向功能区域迁移、不断发生可塑性变化并与其他神经元建立突触联系从而产生神经功能的完整的过程。在神经系统中,Wnt信号通路能够调控神经细胞的分化、损伤以及凋亡[16]。 在干细胞、神经前体细胞的分化及神经嵴、神经板的形成中,Wnt信号通路起着关键的作用[17-18]。在胚胎干细胞的研究中,APC基因的突变可通过胞内β-catenin浓度的升高来抑制胚胎干细胞向三胚层组织分化而维持其增殖[19]。Wnt-3a蛋白可通过上调β-catenin的表达来促进骨髓干细胞的增殖,阻断Wnt信号通路则导致干细胞增殖受阻[20]。在中枢神经系统中,β-catenin能调 控细胞增殖、分化的平衡,同时调控神经前体细胞的生长[9]。有研究表明,在β-catenin基因被敲除后,将出现小鼠脑和脊髓神经发育缺陷,神经前体细胞增殖的数量减少;而过表达β-catenin,神经前体细胞增殖的数量则明显增加[21]。在神经管的发生过程中,主要表达于神经管背侧中心线的Wnt1和Wnt3a浓度从背侧至腹侧逐渐降低,因此导致越靠近背侧的细胞增殖越快,靠近神经管腹侧的细胞则趋于分化,这与神经管发育过程中细胞增殖分化的趋势一致[22]。研究发现,在正常情况下Wnt1、Wnt3a表达于神经管背侧区域,当它们过表达则使神经嵴细胞增多,抑制其表达将导致小鼠神经嵴衍生物不足及背外侧神经管中的神经前体细胞明显减少;GSK-3β的过表达则导致神经嵴标志物Krox-20,AP-2和slug的表达显著下调,由此引起的EMT会导致细胞恶性变[23]。同样,干扰诱导神经嵴发生的Fzd3、LRP6及β-catenin的表达将影响神经嵴的正常发育及分化,神经嵴干细胞中不表达β-catenin则会导致色素细胞和背根神经节的缺失[22-23]。在神经板和神经管头尾模式的建立中,Wnt信号通路同样起着关键作用。小脑颗粒细胞分泌的Wnt-7a突变导致小鼠突触素(synapsin)I等在神经纤维末梢特定区域的聚集及小脑小球发生成熟的延迟,表明Wnt-7a能促进突触的发生[24-26]。 2.2 Wnt信号通路与神经系统疾病 2.2.1 阿尔茨海默症(Alzheimer’s Disease,AD) 研究表明,β淀粉样蛋白大量沉积时,阻断了细胞信号向胞内转导,从而导致阿尔茨海默病的发生[27-28]。对于阿尔茨海默症,Wnt信号通路中某些因子的表达水平发生变化,将直接影响神经元形成,从而加重阿尔茨海默病程度[29]。 在Wnt/β-catenin信号通路中,β淀粉样蛋白能够激活GSK-3β,GSK-3β的表达水平增高,将促进Tau蛋白和β-catenin过度磷酸化,从而神经细胞增殖受到抑制,同时变性凋亡坏死[30]。此外,Fzd也参与包括神经管闭合、轴突生长和神经元存活在内的神经系统发育[31],β淀粉样蛋白通过结合Fzd位于胞外的N端半胱氨酸富集区抑制Wnt信号通路参与阿尔茨海默病的发生[32]。Fzd1通过Wnt-3a, Fzd5通过Wnt-5a介导阿尔茨海默病的发生,在体外培养细胞中,β淀粉样蛋白上调Wnt-3a、Wnt-5a、Fzd1和Fzd5的表达[33-34]。LRP6作为Wnt信号通路的受体之一,其突变与迟发性阿尔茨海默病有一定的关系[35]。Dsh为胞浆内支架蛋白,其与Tau蛋白的共表达可显著降低Tau蛋白的磷酸化水平和由Tau引起的轴突传递缺陷及运动功能障碍[36],这表明Dsh在阿尔茨海默病的发生中发挥着重要作用。GSK-3β的磷酸化和去磷酸化对调控Wnt信号通路至关重要,其活性的异常导致Tau蛋白磷酸化及记忆功能损害[37],同时促进APP蛋白的剪切,使β淀粉样蛋白在胞内积累[38];抑制GSK-3β则显著降低β淀粉样蛋白的水平[39],其磷酸化早老素-1(Presenilin-1,PS1)将导致家族性阿尔茨海默 病[40]。β-catenin作为在Wnt信号通路中起着核心作用的分子,上调该分子的表达可以抑制β淀粉样蛋白诱导的细胞凋亡,反之敲除该基因则拮抗Tau蛋白的抗凋亡作用[41]。Dkk1作为Wnt信号通路的抑制分子,在阿尔茨海默病患者及动物模型脑内显著上调[41-42]。 以上证据表明阿尔茨海默病的发生同Wnt信号通路中多环节,多位点存在密切的关系。β淀粉样蛋白可以通过抑制Wnt信号通路的功能来影响靶基因的表达,从而产生神经毒性最终导致大量神经元的缺失。那么研究某种药物来激活Wnt信号通路从而抑制β淀粉样蛋白的神经毒性可成为治疗阿尔茨海默病的一种思路。同时Wnt信号通路的负调控因子GSK-3β可以通过对tau蛋白的过度磷酸化破坏神经元产生认知功能障碍,GSK-3β的抑制剂的研究可作为治疗阿尔茨海默病的再一突破口。突触损伤所导致的神经元的坏死凋亡,也将引起阿尔茨海默病发生[43],促进突触的发生可起到保护神经元的作用,从而减轻阿尔茨海默病的病情程度。此外,Wnt信号在神经干细"
| [1] Salinas PC. Wnt signaling in the vertebrate central nervous system: from axon guidance to synaptic function. Cold Spring Harb Perspect Biol.2012;4(2). pii: a008003.[2] Harrisonuy SJ, Pleasure SJ. Wnt signaling and forebrain development. Cold Spring Harb Perspect Biol.2012;4(7):a008094.[3] Clevers H,Nusse R.Wnt/β-Catenin Signaling and Disease. Cell.2012; 149(6):1192-1205.[4] Mohammed MK,Shao C, Wang J,et al. Wnt/β-catenin signaling playsan ever-expanding role in stem cell self-renewal, tumorigenesis and cancer chemoresistance. Genes Dis.2016;3(1):11-40.[5] Rennoll S, Yochum G.Regulation of MYC gene expression by aberrant Wnt/beta-cateninsignaling in colorectal cancer.World J Biol Chem. 2015;6(4):290-300.[6] Bertrand V. β-catenin-driven binary cell fate decisions in animal development. Wiley Interdiscip Rev Dev Biol. 2016;5(3):377-88.[7] 曹旻.神经发生中的Wnt信号通路[J].中国组织工程研究, 2013,17(2): 358-362.[8] Song X, Xin N, Wang W, et al. Wnt/beta-catenin, an oncogenic pathway targeted by H. pylori ingastric carcinogenesis. Oncotarget. 2015;6(34):35579-35588.[9] 文静,王建,罗世兰,等.Wnt/β-catenin 信号通路参与脑缺血后神经血管单元调控及相关药物的研究进展[J].中国药理学通报,2016,32( 3) : 310-314.[10] Prakash S,Swaminathan U.beta catenin in health: A review.J Oral Maxillofac Pathol.2015; 19(2):230-238.[11] FuentesRG,Arai MA,Ishibashi M. Natural compounds withWntsignal modulating activity.Nat Prod Rep. 2015;32(12): 1622-1628.[12] Hohendanner F,Mcculloch AD,Blatter LA,et al.Calcium and IP3 dynamics in cardiacmyocytes: experimental and computational perspectives and approaches. Front Pharmacol.2014 ;5:35.[13] Endo M,Nishita M,Fujii M,et al.Insight into the role of Wnt5a-induced signaling in normal and cancer cells. Int Rev Cell Mol Biol.2015;314: 117-148.[14] Devenport D. Tissue morphodynamics: Translating planar polarity cues into polarized cell behaviors. Semin Cell Dev Biol. 2016;55: 99-110.[15] Carvajal-Gonzalez JM, Mlodzik M. Mechanisms of planar cell polarity establishment in Drosophila. F1000Prime Rep.2014;6:98.[16] Bielen H,Houart C.TheWntcries many:Wntregulation ofneurogenesis through tissue patterning, proliferation, and asymmetriccell division. Dev Neurobiol. 2014;74(8):772-780.[17] Nouri N,Patel MJ,Joksimovic M,et al.Excessive Wnt/beta-catenin signaling promotes midbrain floor plate neurogenesis, but results in vacillating dopamine progenitors. Mol Cell Neurosci.2015;68:131-142.[18] Undi RB,Gutti U,Sahu I,et al.WntSignaling: Role in Regulation of Haematopoiesis. Indian J Hematol Blood Transfus. 2016;32(2): 123-134.[19] Kielman MF,Rindap M,Gaspar C,et al.Apc modulates embryonic stem-cell differentiation by controlling the dosage of beta-catenin signaling. Nature Genetics.2002;33(1):594-605.[20] Du Y, Zhang S, Yu T, et al. Wnt3a is critical forendothelial progenitor cell-mediated neural stem cell proliferation and differentiation. Mol Med Rep.2016;14 (3): 2473-2482.[21] Liu X, Yun F, Shi L, et al. Roles of Signaling Pathways in the Epithelial-Mesenchymal Transition in Cancer.Asian Pac J Cancer Prev. 2015;16(15):6201-6206.[22] Wu J,Saintjeannet JP,Klein PS.Wnt-frizzled signaling in neural crest formation.Trends in Neurosciences.2003;26(26): 40-45.[23] Mayor R,Theveneau E.The role of the non-canonical Wnt-planar cell polarity pathway in neural crest migration. Biochem J.2014;457(1): 19-26.[24] Hall AC,Lucas FR,Salinas PC.Axonal remodeling and synaptic differentiation in the cerebellum is regulated by Wnt-7a signaling. Cell. 2000;100(5):525-535.[25] 王建材,冯达云,李洋,等.DKK1 对慢性吗啡和纳洛酮处理的培养海马神经元树突棘的影响[J].神经解剖学杂志,2016,32(1):1-6.[26] Bian WJ,Miao WY,He SJ,et al.A novel Wnt5a-Frizzled4 signaling pathway mediates activity-independent dendrite morphogenesis via the distal PDZ motif of Frizzled 4.Dev Neurobiol.2015;75(8):805-822.[27] Martin L,Latypova X,Terro F.Post-translational modifications of tau protein: Implications for Alzheimer's disease. Neurochem Int. 2011; 58(4):458-471.[28] 霍江涛,张小乔,严洁,等.wnt信号传导通路在阿尔茨海默病中的作用[J].中国临床研究,2015,28(6):709-716.[29] Inestrosa NC,Varela-Nallar L.Wntsignaling in the nervous system and in Alzheimer's disease.J Mol Cell Biol.2014;6(1):64-74. 215-227. [30] 姚宏波,廉洁,张善强,等.基于wnt信号通路研究骨髓基质干细胞对阿尔茨海默病的治疗作用[J].世界最新医学信息文摘, 2016,16(48):62.[31] Chacn MA,Varelanallar L,Inestrosa NC.Frizzled-1 is involved in the neuroprotective effect of Wnt3a against Abeta oligomers. J Cell Physiol. 2008;217(1):215-227.[32] Magdesian MH,Carvalho MM,Mendes FA,et al.Amyloid-beta binds to the extracellular cysteine-rich domain of Frizzled and inhibits Wnt/beta-catenin signaling. J Biol Chem.2008;283(14):9359-9368. [33] Inestrosa NC,Montecinosoliva C,Fuenzalida M. Wnt Signaling:Role in Alzheimer Disease and Schizophrenia. J Neuroimmune Pharmacol. 2012;7(4):788-807.[34] Li B,Zhong L,Yang X,et al.Wnt5A Signaling Contributes to Abeta-Induced Neuroinflammation and Neurotoxicity.Plos One. 2011; 6(8):e22920.[35] De Ferrari GV,Papassotiropoulos A,Biechele T,et al.Common genetic variation within the low-density lipoprotein receptor-related protein 6 and late-onset Alzheimer's disease.Proc Natl Acad Sci U S A. 2007; 104(22):9434-9439.[36] Folwell J,Cowan CM,Ubhi KK,et al.A beta exacerbates the neuronal dysfunction caused by human tau expression in a Drosophila model of Alzheimer's disease. Exp Neurol. 2010;223(2):401-409.[37] Xu J,Zhang R,Zuo P,et al. Aggravation Effect of Isoflurane on Abeta- Induced Apoptosis and Tau Hyperphosphorylation in PC12 Cells. Cell Mol Neurobiol.2012;32(8):1343-1351.[38] Hooper C,Killick R,Lovestone S.The GSK3 hypothesis of Alzheimer's disease. J Neurochem. 2008;104(6):1433-1439.[39] Kramer T,Schmidt B,Lo MF.Small-Molecule Inhibitors of GSK-3: Structural Insights and Their Application to Alzheimer's Disease Models. Int J Alzheimers Dis. 2012;2012:381029.[40] Maesako M,Uemura K,Kuzuya A,et al.Gain of function by phosphorylation in Presenilin 1-mediated regulation of insulin signaling. J Neurochem. 2012;121(6):964-973.[41] Caricasole A,Copani A,Caraci F,et al.Induction of Dickkopf-1, a negative modulator of theWntpathway, is associated with neuronal degeneration in Alzheimer's brain. J Neurosci. 2004;24(26):6021-6027.[42] Rosi MC, Luccarini I, Grossi C, et al.Increased Dickkopf-1 expression in transgenic mouse models of neurodegenerative disease. J Neurochem. 2010 ;112(6):1539-1551.[43] Zhou Y,Lou Y,Dai J.Axonal and dendritic changes are associcated with diabetic encephalopathy in rats: an improtanr risk factor for Alzheimer’s disease.J Alzheimers Dis.2013;34(4): 937-947.[44] 王薇,张海廷,王淑辉,等.阿尔茨海默病与Wnt信号通路及神经干细胞的关系[J].中国组织工程研究,2013,17(19):3566-3572. [45] Kojovic M, Higgins A, Jahanshahi M. In Parkinson's disease Stn stimulation enhances responsiveness of movement initiation speed to high reward value.Neuropsychologia.2016;89: 273-280.[46] Okada Y,Murata M,Toda T.Effects of levodopa on vowel articulation in patients with Parkinson's disease.Kobe J Med Sci.2016;61 (5): E144-154.[47] Sancho RM,Law BK.Mutations in the LRRK2 Roc-COR tandem domain link Parkinson's disease toWntsignalling pathways. 2009; 18(20): 3955-3968.[48] Wang W, Yang Y, Ying C, et al.Inhibition of glycogen synthase kinase-3beta protects dopaminergic neurons from MPTP toxicity. Neuropharmacology.2007;52(8): 1678-1684.[49] Petitpaitel A,Brau F,Cazareth J,et al.Involvment of cytosolic and mitochondrial GSK-3beta in mitochondrial dysfunction and neuronal cell death of MPTP/MPP-treated neurons. Plos One.2009;4(5): e5491.[50] 张凯辉,徐宝山,杨强.骨髓间充质干细胞在多种疾病治疗中的应用进展[J].中国组织工程研究,2017,21(21):3400-3406.[51] Kernie SG, Parent JM. Forebrain neurogenesis after focal Ischemic and traumatic brain injury. Neurobiology of Disease. 2010; 37(37): 267-274.[52] Kreuzberg M,Kanov E,Timofeev O,et al. Increased subventricular zone-derived cortical neurogenesis after ischemic lesion. Experimental Neurology. 2010;226(1): 90-99.[53] 李慧,黄景阳,陈海丽,等. 脑缺血再灌注后大鼠海马Wnt7b的表达[J].山东大学学报:医学版,2013,51(2):7-11.[54] Mastroiacovo F,Busceti CL,Biagioni F,et al.Induction of the Wntantagonist, Dickkopf-1, contributes to the development of neuronal death in models of brain focal ischemia. J Cereb Blood Flow Metab. 2009;29(2):264-276.[55] Shruster A, Ben-Zur T, Melamed E, et al.Wnt Signaling Enhances Neurogenesis and Improves Neurological Function after Focal Ischemic Injury. Plos One.2012;7(7): e40843.[56] Calió ML,Marinho DS,Ko GM,et al.Transplantation of bone marrow mesenchymal stem cells decreases oxidative stress, apoptosis, and hippocampal damage in brain of a spontaneous stroke model. Free Radic Biol Med. 2014;70:141-154.[57] 张慧玲,彭会珍,陈素艳,等. Wnt信号通路在缺血性脑卒中后神经发生中的作用研究进展[J]. 实用医学杂志, 2015,31(17):2922-2923.[58] Aimone JB, Li Y, Lee SW, et al. Regulation and function of adult neurogenesis: from genes to cognition.Physiol Rev.2014;94(4):991-1026.[59] De La Rosa-Prieto C, De Moya-Pinilla M, Saiz-Sanchez D, et al. Olfactory and cortical projections to bulbar and hippocampal adult-born neurons.Front Neuroanat.2015;9: 4.[60] Kriska J,Honsa P,Dzamba D,et al.ManipulatingWntsignaling at dif-ferent subcellular levels affects the fate of neonatal neural stem/progeni-tor cells.Brain Res.2016;1651:73-87.[61] Inestrosa NC, Varela-Nallar L.Wntsignalling in neuronal differentia-tion and development.Cell Tissue Res.2015;359(1): 215-223.[62] 张东圆,李明,王正则,等.缺氧诱导因子-1α在缺血性脑卒中模型中介导神经干-祖细胞作用机制的研究进展[J]. 中国康复理论与实践, 2017, 23(3): 319-322. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||