Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (8): 1287-1293.doi: 10.3969/j.issn.2095-4344.0150
Previous Articles Next Articles
Physical activities promote recovery from spinal cord injury in patients
Wang Ji-chao, Zhu Jun-peng, Wu Xue-ping
- School of Physical Education and Sport Training, Shanghai University of Sport, Shanghai 200438, China
-
Received:2018-01-10Online:2018-03-18Published:2018-03-18 -
Contact:Wu Xue-ping, Ph.D., Professor, Doctoral supervisor, School of Physical Education and Sport Training, Shanghai University of Sport, Shanghai 200438, China -
About author:Wang Ji-chao, Master candidate, School of Physical Education and Sport Training, Shanghai University of Sport, Shanghai 200438, China -
Supported by:the Postgraduate Education Innovation Plan of Shanghai University of Sport, No. yjscx2016036; the Science and Technology Program of Shanghai Science and Technology Commission, No. 14490503600; the Shanghai Key Laboratory of Development and Guarantee of Human Sports Capacity (Shanghai University of Sport), No. 11DZ2261100
CLC Number:
Cite this article
Wang Ji-chao, Zhu Jun-peng, Wu Xue-ping. Physical activities promote recovery from spinal cord injury in patients[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1287-1293.
share this article
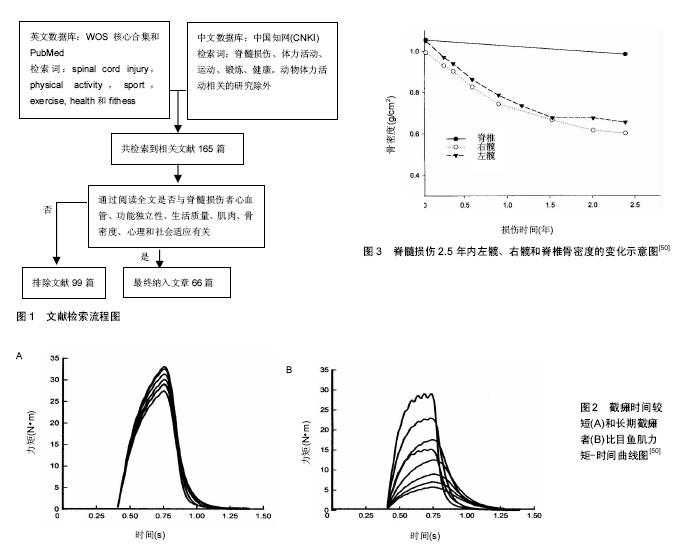
2.1 脊髓损伤对体力活动的影响 脊髓损伤者受伤较为突然且肢体功能受损严重,现有医疗水平难以治愈,因此,脊髓损伤常常给受伤者造成行动不便、生活难以自理的结果。利晓莲等[10]研究认为脊髓损伤者受伤7 d内日常生活活动能力最低,3个月后,脊髓损伤者日常生活活动能力有所提升,此时,医院护理人员或康复师应该鼓励他们进行康复锻炼,争取最大程度的提高日常生活活动能力。施红梅[11]利用世界卫生组织《残疾评定方案2.0》对脊髓损伤者活动和参与功能进行评估结果显示,脊髓损伤者认知功能存在轻度障碍,认知功能障碍产生的原因和焦虑和抑郁情绪有关;活动功能存在重度障碍,主要功能障碍表现在移动、站立和行走等方面;自我照护功能存在重度障碍,日常生活独立性差;与他人相处功能表现出中等障碍,且该功能障碍与抑郁呈正相关关系;与生活相关的各项活动功能主要体现在家务、工作和学习方面的障碍,脊髓损伤者出现重度障碍;社会参与功能存在重度障碍,脊髓损伤者心理状态与应对方式对社会参与功能有着重要的作用。 2.2 体力活动对脊髓损伤者心血管健康的影响 心脏和血管构成了机体血液循环系统,心脏收缩是血液在血液循环系统周而复始流动的保障。心血管疾病又称循环系统疾病,是一系列涉及循环系统的疾病。如今心血管疾病是威胁人类,特别是50岁以上中老年人健康的主要疾病。调查显示,近年来心脑血管疾病患病率、死亡率持续增长,心脑血管疾病已经成为国内致死率最高的因 素[12]。脊髓损伤者由于损伤导致神经系统控制的局部血液流动、心输出量和最大心率受限。研究显示,脊髓损伤者最大心率可降低至110-130次/min[13],最大心率下降导致运动时心输出量减少,从而影响循环系统的效率。此外,损伤平面以下结构(小动脉和毛细血管数量减少,阻力血管的直径减少)和功能(交感神经血管调节)改变、腿部血管阻力增加和下肢血流减少也会影响脊髓损伤者血管功能[14]。以上由于病理原因引起的心血管功能变化,会因缺乏运动而恶化,从而使心血管发病的概率增加。研究表明,参与一定强度的体力活动有利于控制血脂水平、减少冠状动脉粥样硬化性心脏病和动脉硬化的发生率,从而提高心血管健康水平[13]。 高血脂是世界公认的导致冠状动脉粥样硬化性心脏病和动脉硬化的主要危险因素,脂代谢异常是动脉硬化等心血管疾病发病率的重要原因,参与体力活动对控制血脂有着良好的效果[15]。Hooker等[16]研究证明,每次20 min,每周3次,共8周的中等强度的体力活动可以改善脊髓损伤者的血脂水平。体力活动改善脊髓损伤者血脂水平的干预研究中,更多的研究支持至少每周3次、每次30min的中等强度体力活动才对血脂有良好的效果,而且研究证明了不同运动模式下这种剂量反应的有效性[17-19]。 参加体力活动可以增加高密度脂蛋白的含量,降低冠状动脉粥样硬化性心脏病的发病率[20]。脊髓损伤者久坐的生活方式会引起一系列的心血管并发症,如冠状动脉粥样硬化性心脏病[13]。冠状动脉粥样硬化性心脏病和高密度脂蛋白胆固醇含量有密切的关系,高密度脂蛋白胆固醇含量减少,冠状动脉粥样硬化性心脏病的发病率增加[21]。Brenes等[22]研究认为,体力活动水平高的脊髓损伤轮椅运动员比久坐不动的脊髓损伤者高密度脂蛋白含量高。Dallmeijer等[23]对参与体力活动的四肢瘫痪男性和久坐不动的四肢瘫痪男性进行对比观察,发现前者高密度脂蛋白胆固醇和载脂蛋白A1/载脂蛋白B含量均比后者高。一些横向研究也得到了相同的结果[24-25]。高密度脂蛋白胆固醇含量和冠状动脉粥样硬化性心脏病有密切的联系,体力活动是高密度脂蛋白胆固醇含量的主要影响因素,因此,体力活动在减少冠状动脉粥样硬化性心脏病的风险方面有着重要的作用。脊髓损伤者参与体力活动增加高密度脂蛋白胆固醇含量的机制尚不确定,但似乎和增强胆固醇运输脂蛋白脂肪酶和酰基转移酶有关[26]。 脊髓损伤者常年久坐且缺乏锻炼,动脉硬化水平较高,体力活动不足是导致动脉硬化发病率的主要原因,而经常参与体力活动可以减少脊髓损伤者的动脉硬化发病率。动脉硬化会引起动脉管壁增厚、变硬、失去弹性,导致管腔变窄,它是随着年龄而发生的一种心血管疾病,也是造成老年人死亡的主要原因之一。Wecht 等[27]研究显示,久坐不动的脊髓损伤者比他们年龄相同的健全人动脉硬化水平高,但是,体力活动量多的轮椅运动员比他们年龄相同的健全人动脉硬化水平低[28],这说明脊髓损伤者动脉硬化水平比健全人高的原因与缺乏体力活动有一定联系。研究显示,参与体力活动可以提高脊髓损伤者20%的最大摄氧量(maximal oxygen consumption,VO2 max)[29],动脉硬化与VO2 max呈负相关,而参与体力活动可以一定程度的提高VO2 max[30],因此体力活动可以降低动脉硬化的发病率。血小板活化后释放的一些炎症递质直接参与动脉硬化的形成和发展,因此血小板在形成动脉硬化方面起着重要的作用。血小板凝集是一个复杂的过程,受到很多内在和外在因素影响,如儿茶酚胺、肾上腺素水平等。一些研究尝试探索训练对血小板凝集的影响,但结果存在争议[31-33]。 综上所述,保持一定强度的体力活动可以控制血脂水平、预防冠状动脉粥样硬化性心脏病和动脉硬化等慢性疾病,从而改善脊髓损伤者心血管健康。但是脊髓损伤者受损的感觉功能、心血管功能、自主神经功能和稳定调节功能在激烈的运动中有一定的危险,在未来的研究中针对不同损伤水平脊髓损伤者体力活动的研究必将成为焦点。 2.3 体力活动对脊髓损伤者功能独立性的影响 脊髓损伤常导致高度失能,如失去行走能力、生活不能自理等,导致功能独立性下降,从而给他们今后的生活带来巨大挑战。保持功能独立性能够给脊髓损伤者带来精神上的安慰,同时增加他们面对未来的勇气,研究显示,体力活动可以改善脊髓损伤者功能独立性,是促进身体重塑的有效方式。Melo[34]认为,脊髓损伤者参与体力活动有利于增加肌肉力量,同时改善功能独立性。Barbin等[35]将84名男性截瘫者分成体力活动组和非体力活动组进行研究,结果显示,体力活动组功能能力(功能独立量表)得到优化,自理能力提高。该作者在损伤时间的对比上发现,体力活动对于损伤时间10年以内的脊髓损伤者效果更好。 体力活动对于脊髓损伤者功能独立性的影响与损伤程度、损伤时间和活动量存在一定的联系。Manns 等[36]认为,在一定范围内,损伤程度越重或损伤时间越短,体力活动对功能独立性的效益越好。但是也有学者提出不同的意见,他们认为损伤时间较短的脊髓损伤者影响体力活动对功能独立性的分析,原因是损伤时间较短的脊髓损伤者需要康复过程和一定的时间去适应新的变化,这种康复和适应过程可能比体力活动对脊髓损伤者的影响更大[37]。因此,为了使研究结果更具可信度,在以后的研究中建议选择损伤时间1年以上的脊髓损伤者。此外,体力活动对功能独立性的影响和活动量也有一定的关系。已有研究显示,每周2次,每次30 min的体力活动对功能独立性没有影响[38];每周2次,每次 45 min的体力活动对功能独立性有一定程度的影响[39];每周累计4 h的体力活动对功能独立性有积极的影 响[40]。以上结果表明,每周累计150 min的中等强度活动量也适合脊髓损伤者。 2.4 体力活动对脊髓损伤者生活质量的影响 脊髓损伤者损伤平面以下感觉和运动功能出现障碍,同时还伴随着一些病理变化如反射异常和尿便失禁等,导致自理能力严重下降。另外脊髓损伤者夫妻关系、社交生活和经济条件较差,这些状况对脊髓损伤者生活造成很大的影响,从而导致其生活质量下降。李妍等[41]学者认为,40.5%的脊髓损伤者存在严重的功能障碍,无法独自一人完成日常生活。Glickman等[42]对115名脊髓损伤者进行的研究显示,脊髓损伤者自理能力差,49%的脊髓损伤者需要30 min以上时间来完成如厕,这些状况给他们的日常生活带来很大不便,会造成生活质量下降。有学者对脊髓损伤者生活质量进行调查,其结果表明,脊髓损伤者生活质量总分为(66.4±14.7)分,生活质量总分较低,且4个维度得分均较低[43]。 研究表明,适当的参加一定强度的体力活动可以提高脊髓损伤者生活质量。Mulroy等[44]对80例脊髓损伤者进行为期12周的体力活动干预,每周3次,每次60 min,其生活质量得分提高了10%。Ginis等[45]对34例脊髓损伤者进行3个月的抗阻和有氧训练,每周2次,每次65- 95 min,结果显示,与对照组相比,训练组在疼痛、压力、抑郁和总的生活质量得分方面都有所改善。该学者认为,对生活质量和抑郁方面的改善主要是由于疼痛和压力方面的改善。但是也有学者对体力活动改善生活质量这种结论提出不同意见。Yildirim等[46]利用循环抗阻训练法对26例截瘫者进行干预,每次60 min,每周5次,共6周的训练,结果显示与对照组相比,训练组功能独立性有显著性提高,生活质量却没有显著性变化。脊髓损伤者个人状况通常会出现较大的差异,如损伤程度、受教育程度、有无配偶、生活收入等,这种差异可能会对实验结果产生较大的影响,从而导致研究者得出不同的结论。 2.5 体力活动对脊髓损伤者肌肉和骨密度的影响 肌肉力量是体力活动的动力源泉,是完成一切目标任务的前提,脊髓损伤者由于病理和缺少运动,导致肌肉在形态学、骨骼肌收缩和新陈代谢上发生变化。脊髓损伤者下肢肌纤维类型改变,Ⅱb肌纤维(快缩/糖酵解型,易疲劳)类型数量增加,Ⅰ型肌纤维数量(慢氧化型、不易疲劳)减少[47]。损伤平面以下肌肉完整性受到破坏,如交感神经功能受损、神经输入受到影响等,使参与肌肉收缩的运动神经元和肌纤维数量减少,肌肉力量下降[48]。此外,损伤平面以下肌肉内线粒体酶活性和毛细血管密度下降,导致有氧能力降低[49]。Shields等[50]对截瘫时间较短和长期截瘫者的比目鱼肌力量进行研究,结果显示,长期截瘫者比截瘫时间较短者比目鱼肌更易疲劳(图2)。 肌纤维数量增多、增粗是肌肉力量增长的主要因素,体力活动干预时肌肉活动增强,为了完成运动负荷,肌肉收缩时募集的运动纤维类型和数量增加。而且,蛋白质是肌纤维的重要构成元素,体力活动可以刺激肌肉,使代谢旺盛,加快蛋白质合成,因此,体力活动对增加肌肉力量有积极的作用。Mohr等[47]对10例脊髓损伤者进行为期1年的训练,每周3次(平均为2.3次),每次30 min,结果显示,干预组肌纤维类型有良好的分布,训练前后相比,肌肉(臀肌、股四头肌)中同类型Ⅱb肌纤维肌球蛋白重链由63%减少至32%、同类型Ⅱa肌纤维肌球蛋白重链(快缩,不易疲劳)由33%增加至61%和同类型Ⅰ型肌纤维的肌球蛋白重链(慢缩)由5%增加至7%,而且柠檬酸合酶和线粒体氧化能力是原来的2倍。孙启涛等[51]应用等速肌力训练结合综合康复训练对60例不完全性脊髓损伤者下肢进行干预研究,将受试者随机分为治疗组和对照组各30例,对照组只进行综合康复训练,治疗组接受综合康复训练结合等速肌力训练。每次干预时间为30 min,每周训练5次,共4周,结果显示,治疗组膝关节屈伸肌肌力和对照组相比有显著性提高。Jacops等[52]对神经损伤水平在T5-L1的10例脊髓损伤受试者上肢进行循环抗阻训练,每周3次,共12周,结果表明,受试者手臂峰值摄氧量、疲劳时间和最大输出功率增加,差异有显著性意义。 骨质疏松症是一种全身代谢疾病,骨骼缺乏外力刺激被认为是影响骨健康的重要因素。有研究显示,大多数轮椅使用者由于下肢失去对骨的刺激,其骨质流失是单纯代谢病因引起骨质流失的5-20倍[53]。而且,脊髓损伤者骨小梁含量越丰富的地方骨质流失越严重,如胫骨近端和远端等。Shields等[50]研究了脊髓损伤后两年半时间内骨量的变化(图3),图3显示1年后,股骨近端骨密度丢失30%,但是腰椎骨密度变化不大。该作者认为,脊髓损伤者坐位姿势时,腰椎经常承受来自身体重量的负荷,故骨量变化不大,而股骨失去了外部负荷对它的刺激,导致骨量丢失[50]。以上腰椎和股骨骨密度不同的变化表明,脊髓损伤者骨量的丢失是骨骼失去了外界负荷刺激的结果。"
| [1] 李妍,李小妹.脊髓损伤患者生活质量及影响因素研究进展[J].齐鲁护理杂志,2014(21):50-52.[2] Wyndaele M, Wyndaele JJ. Incidence, prevalence and epidemiology of spinal cord injury: what learns a worldwide literature survey? Spinal Cord. 2006;44(9):523-529.[3] 唐强,张安仁.临床康复学[M].北京:人民卫生出版社,2012:63.[4] 卫波.2005年北京市脊髓损伤流行病学初步调查研宄[D].北京:首都医科大学,2007.[5] Anneken V, Hanssen-Doose A, Hirschfeld S, et al. Influence of physical exercise on quality of life in individuals with spinal cord injury. Spinal Cord. 2010;48(5):393-399. [6] 全明辉,陈佩杰,王茹,等.体力活动对认知能力影响及其机制研究进展[J].体育科学,2014,9(34):56-65.[7] Garshick E, Kelley A, Cohen SA, et al. A prospective assessment of mortality in chronic spinal cord injury. Spinal Cord. 2005;43(7):408-416. [8] Dost G, Dulgeroglu D, Yildirim A, et al. The effects of upper extremity progressive resistance and endurance exercises in patients with spinal cord injury. J Back Musculoskelet Rehabil. 2014;27(4):419-426. [9] Kloosterman MG, Snoek GJ, Jannink MJ. Systematic review of the effects of exercise therapy on the upper extremity of patients with spinal-cord injury. Spinal Cord. 2009;47(3):196-203. [10] 李晓莲,肖智真,张虹,等. 50例脊髓损伤患者心理状况?日常生活活动能力及社会支持情况调查[J].护理学报,2014,21(5):40-42.[11] 施红梅.脊髓损伤患者情绪状态与活动和参与功能研究[J].中国康复理论与实践,2017,23(8):946-949.[12] 赵丽丽,李唐棣,马洪颖,等.唐山地震37年后脊髓损伤患者死亡原因调查[J].中国康复理论与实践,2014,(10):975-978.[13] Devillard X, Rimaud D, Roche F, et al. Effects of training programs for spinal cord injury. Ann Readapt Med Phys. 2007;50(6):490-498, 480-489. [14] Hopman MT, Groothuis JT, Flendrie M, et al. Increased vascular resistance in paralyzed legs after spinal cord injury is reversible by training. J Appl Physiol (1985). 2002;93(6):1966-1972.[15] 周景,周倩,王东平,等. 静坐行为和体力活动与血脂异常[J].北京大学学报:医学版,2017,49(3):418-423.[16] Hooker SP, Wells CL. Effects of low- and moderate-intensity training in spinal cord-injured persons. Med Sci Sports Exerc. 1989;21(1):18-22.[17] Cowan RE, Nash MS. Cardiovascular disease, SCI and exercise: unique risks and focused countermeasures. Disabil Rehabil. 2010; 32(26):2228-2236. [18] 晁敏,梁丰,王尊,等.不同强度有氧运动对2型糖尿病患者生理指标的影响[J].中国康复医学杂志,2015,30(9):883-887.[19] 张荷,周越,张一民,等.低氧运动对肥胖大鼠胰岛素抵抗及血脂代谢的影响[J].北京体育大学学报,2016,39(9):44-56.[20] 苏中军,张肃,姜军.高密度脂蛋白生物学功能及运动调节的研究与进展[J].中国组织工程研究,2015,19(37):6048-6054.[21] 张诗岚,杜晓,刘玲.高密度脂蛋白胆固醇水平与高密度脂蛋白功能: 孰是孰非?[J].中国动脉硬化杂志,2017,25(1):90-94. [22] Brenes G, Dearwater S, Shapera R, et al. High density lipoprotein cholesterol concentrations in physically active and sedentary spinal cord injured patients. Arch Phys Med Rehabil. 1986;67(7):445-450. [23] Dallmeijer AJ, Hopman MT, van der Woude LH. Lipid, lipoprotein, and apolipoprotein profiles in active and sedentary men with tetraplegia. Arch Phys Med Rehabil. 1997;78(11):1173-1176. [24] Dearwater SR, LaPorte RE, Robertson RJ, et al. Activity in the spinal cord-injured patient: an epidemiologic analysis of metabolic parameters. Med Sci Sports Exerc.1986;18(5):541-544.[25] Maki KC, Briones ER, Langbein WE, et al. Associations between serum lipids and indicators of adiposity in men with spinal cord injury. Paraplegia. 1995;33(2):102-109. [26] 李俊,冯丽洁.抗阻训练对心血管疾病风险因素的影响[J].体育科学研究, 2015,19(6):41-46.[27] Wecht JM, Weir JP, DeMeersman RE, et al. Arterial stiffness in persons with paraplegia. J Spinal Cord Med. 2004;27(3):255-259. [28] Fernhall B, Heffernan K, Jae SY, et al. Health implications of physical activity in individuals with spinal cord injury: a literature review. J Health Hum Serv Adm. 2008;30(4):468-502. [29] Rimaud D, Calmels P, Devillard X. Training programs in spinal cord injury. Ann Readapt Med Phys. 2005;48(5):259-269. [30] 易学,刘光辉,高志红.老年人与有氧运动能力的相关研究[J].河北体育学院学报,2005,19(1):6-8.[31] El-Sayed MS, Younesian A, Rahman K, et al. The effects of arm cranking exercise and training on platelet aggregation in male spinal cord individuals. Thromb Res. 2004;113(2):129-136. [32] 罗军,魏燕璇,王毅,等.有氧运动对老年稳定性心绞痛患者血浆炎症指标及血小板功能的影响[J].中国老年学杂志,2015,35(3):577-579.[33] 李日行,罗军,梁菊艳,等.有氧运动对冠心病患者血小板受体密度及血黏度的影响[J].广州医学,2015,36(14):2200-2202.[34] Melo AC. Description of initial fitness for swimming in spinal cord injured. Revista Brasileira De Medicina Do Esporte. 2009;15(6): 441-445.[35] Barbin JM, Bilard J, Gaviria M, et al. La mesured’indépendancefonctionnelle chez le paraplégiquetraumatique: étudedifférentielle d’un groupesportif et non sportif. Ann Readapt Med Phys. 1999;42(6):297-305.[36] Manns PJ, Chad KE. Determining the relation between quality of life, handicap, fitness, and physical activity for persons with spinal cord injury. Arch Phys Med Rehabil. 1999;80(12):1566-1571. [37] Durán FS, Lugo L, Ramírez L, et al. Effects of an exercise program on the rehabilitation of patients with spinal cord injury. Arch Phys Med Rehabil. 2001;82(10):1349-1354. [38] Durán FS, Lugo L, Ramírez L, et al. Effects of an exercise program on the rehabilitation of patients with spinal cord injury. Arch Phys Med Rehabil. 2001;82(10):1349-1354. [39] Silva MCR, Oliveira RJ, Conceição MIG. Effects of swimming on the functional independence of patients with spinal cord injury. Revista Brasileira De Medicina Do Esporte. 2005;11(4):251-256.[40] Miki Y, Kanayama C, Nakashima S, et al. Health-Related Quality of Life in Active Persons with Spinal Cord Injury. Jpn J Phys Fit Sports Med. 2012;61(2):177-182.[41] 李妍,鱼星峰,杨磊,等.脊髓损伤病人康复期心理健康?自理能力及生活质量的相关性研究[J]. 护理研究, 2014,28(5):1691-1693.[42] Glickman S, Kamm MA. Bowel dysfunction in spinal-cord-injury patients. Lancet. 1996;347(9016):1651-1653.[43] 陈银海,靳安民,姚红华.脊髓损伤患者的抑郁情绪及其对功能恢复的影响[J].南方医科大学学报,2007,27(6):903-904.[44] Mulroy SJ, Thompson L, Kemp B, et al. Strengthening and optimal movements for painful shoulders (STOMPS) in chronic spinal cord injury: a randomized controlled trial. Phys Ther. 2011;91(3):305-324. [45] Martin Ginis KA, Jetha A, Mack DE, et al. Physical activity and subjective well-being among people with spinal cord injury: a meta-analysis. Spinal Cord. 2010;48(1):65-72. [46] Yildirim A, Sürücü GD, Karamercan A, et al. Short-term effects of upper extremity circuit resistance training on muscle strength and functional independence in patients with paraplegia. J Back Musculoskelet Rehabil. 2016;29(4):817-823. [47] Mohr T, Andersen JL, Biering-Sørensen F, et al. Long-term adaptation to electrically induced cycle training in severe spinal cord injured individuals. Spinal Cord. 1997;35(1):1-16. [48] Hoffman MD. Cardiorespiratory fitness and training in quadriplegics and paraplegics. Sports Med. 1986;3(5):312-330. [49] Hartkopp A, Harridge SD, Mizuno M, et al. Effect of training on contractile and metabolic properties of wrist extensors in spinal cord-injured individuals. Muscle Nerve. 2003;27(1):72-80. [50] Shields RK. Muscular, skeletal, and neural adaptations following spinal cord injury. J Orthop Sports Phys Ther. 2002;32(2):65-74. [51] 孙启涛,陈延荣.不完全性脊髓损伤等速肌力训练的疗效[J].菏泽医学专科学校学报,2014,26(1):27-28.[52] Jacobs PL, Nash MS, Rusinowski JW. Circuit training provides cardiorespiratory and strength benefits in persons with paraplegia. Med Sci Sports Exerc. 2001;33(5):711-717. [53] Noce F, Simim MAM, Mello MT. The perception of quality of life of people with physical disabilities can be influenced by physical activity? Revista Brasileira De Medicina Do Esporte. 2009;15(3):174-178.[54] Bloomfield SA, Mysiw WJ, Jackson RD. Bone mass and endocrine adaptations to training in spinal cord injured individuals. Bone. 1996; 19(1):61-68. [55] Ben M, Harvey L, Denis S, et al. Does 12 weeks of regular standing prevent loss of ankle mobility and bone mineral density in people with recent spinal cord injuries? Aust J Physiother. 2005;51(4):251-256. [56] de Bruin ED, Frey-Rindova P, Herzog RE, et al. Changes of tibia bone properties after spinal cord injury: effects of early intervention. Arch Phys Med Rehabil. 1999;80(2):214-220. [57] Clark JM, Jelbart M, Rischbieth H, et al. Physiological effects of lower extremity functional electrical stimulation in early spinal cord injury: lack of efficacy to prevent bone loss. Spinal Cord. 2007;45(1):78-85. [58] 宓忠祥,刘松怀,祁长凤.脊髓损伤患者的心理问题及康复策略[J].中国康复理论与实践,2003,9(2):97-99.[59] Ditor DS, Latimer AE, Ginis KA, et al. Maintenance of exercise participation in individuals with spinal cord injury: effects on quality of life, stress and pain. Spinal Cord. 2003;41(8):446-450.[60] Cao Y, Massaro JF, Krause JS, et al. Suicide mortality after spinal cord injury in the United States: injury cohorts analysis. Arch Phys Med Rehabil. 2014;95(2):230-235. [61] Curtis K, Hitzig SL, Bechsgaard G, et al. Evaluation of a specialized yoga program for persons with a spinal cord injury: a pilot randomized controlled trial. J Pain Res. 2017;10:999-1017. [62] Ginis KAM, Latimer AE, Mckechnie K, et al. Using exercise to enhance subjective well-being among people with spinal cord injury: The mediating influences of stress and pain. Rehabil Psychol. 2003;48(3): 157-164.[63] Kim YS. Physical Activity and Mental Health. West J Med. 2014;34(2): 60.[64] Renwick R, Yoshida K, Self H, et al. Final report getting on with life: meeting consumers’ social adaptation needs. Toronto: The Ontario Neurotrauma Foundation. 2007;2(6):23-27.[65] Yoshida KK, Self HM, Renwick RM, et al. A value-based practice model of rehabilitation: consumers' recommendations in action. Disabil Rehabil. 2015;37(20):1825-1833. [66] Nooijen CF, Stam HJ, Sluis T, et al. A behavioral intervention promoting physical activity in people with subacute spinal cord injury: secondary effects on health, social participation and quality of life. Clin Rehabil. 2017;31(6):772-780. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||