Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (8): 1196-1204.doi: 10.3969/j.issn.2095-4344.0137
Previous Articles Next Articles
Influence of autophagy-mediated high-intensity interval training on skeletal muscle mass and aerobic capacity of middle-aged rats
Cui Xin-wen, Zhang Yi-min, Wang Zan, Kong Zhen-xing
- Key Laboratory of Exercise and Physical Fitness of Education Department, Beijing Sport University, Beijing 100084, China
-
Received:2017-10-19Online:2018-03-18Published:2018-03-18 -
Contact:Zhang Yi-min, Ph.D., Doctoral supervisor, Key Laboratory of Exercise and Physical Fitness of Education Department, Beijing Sport University, Beijing 100084, China -
About author:Cui Xin-wen, Doctoral candidate, Key Laboratory of Exercise and Physical Fitness of Education Department, Beijing Sport University, Beijing 100084, China -
Supported by:the National Scientific and Technological Supporting Program during the Twelfth Five-Year Plan Period, No. 2012BAK21B00; the Fundamental Research Funds for the Central Universities of China, No. 2015ZD007 and 2016SYS009
CLC Number:
Cite this article
Cui Xin-wen, Zhang Yi-min, Wang Zan, Kong Zhen-xing. Influence of autophagy-mediated high-intensity interval training on skeletal muscle mass and aerobic capacity of middle-aged rats[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1196-1204.
share this article
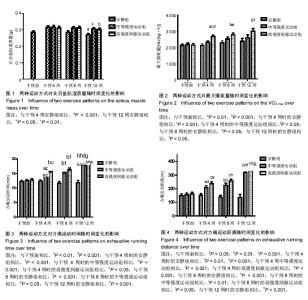
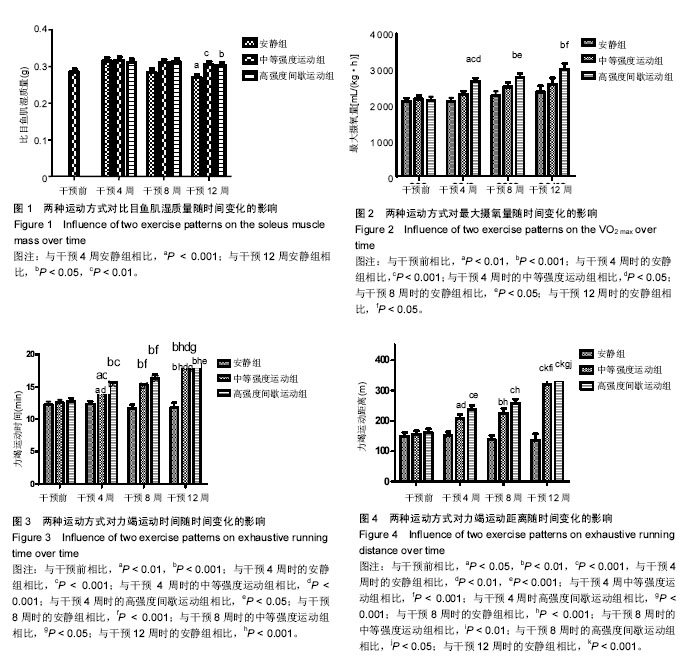
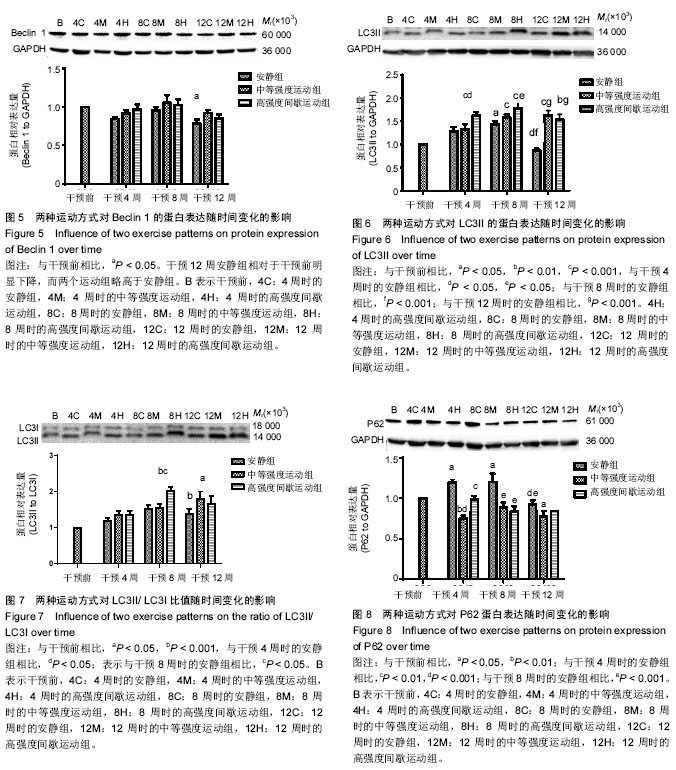
2.1 实验动物数量分析 除中等强度运动组和高强度间歇运动组个别大鼠在训练期间尾巴被跑台夹伤外,无其他大鼠受伤、感染,生病等意外情况,最终80只大鼠进入结果分析。 2.2 两种运动方式对中年大鼠骨骼肌质量随时间变化的影响 采用双因素方差分析,以体质量为协变量,在体质量=600.17 g时估计两种运动方式对比目鱼肌湿质量随时间变化的影响。发现不同运动方式和时间都对比目鱼肌湿质量产生了影响。安静组大鼠比目鱼肌湿质量在干预4周时达到峰值,从8周开始略有下降,到干预12周(大鼠12月龄)时,相对于干预4周时下降了15%(P=0.003,95%CI:4%-25%)而此时中等强度运动组和高强度间歇运动组同样继续保持较高水平,分别高于同期安静组14%和12% (P=0.012,95%CI:2%-24%;P=0.014,95%CI:2%-22%),中等强度运动组与高强度间歇运动组无差异,见图1。高强度间歇运动和中等强度运动均有效抑制了12月龄大鼠比目鱼肌湿质量的下降,但两种运动方式对比目鱼肌湿质量的影响无明显差异。 2.3 两种运动方式对中年大鼠有氧运动能力随时间变化的影响 双因素方差分析两种运动方式对最大摄氧量和力竭运动时间和力竭运动距离随时间变化的影响,发现不同运动方式和时间都对最大摄氧量和力竭运动时间和力竭运动距离产生了影响。 在对最大摄氧量的影响中,干预4周时高强度间歇运动组相对于干预前提高了25%(P=0.002,95%CI:7%-42%),相对于安静组提高了26%(P=0.000,95%CI:10%-43%),相对于中等强度运动组提高了16%(P=0.031,95%CI:1%-31%);干预8周时高强度间歇运动组相对于干预前提高了30%(P=0.001,95%CI:10%-50%;P=0.000,95%CI:13%-43%),相对于安静组提高了22%(P=0.013,95%CI:3.6%-40.6%);12周时高强度间歇运动组相对于干预前提高了40%(P=0.000,95%CI:15%-65%),相对于安静组提高了26%(P=0.034,95%CI:1.4%-51.5%),见图2。安静组在整个干预过程中无明显波动,中等强度运动组从干预第4周开始有上升趋势,但差异无显著性意义,而高强度间歇运动组从干预第4周时开始,最大摄氧量显著提高,12周时达到最高水平,可知,高强度间歇运动较中等强度运动明显提高了最大摄氧量。 在对力竭运动时间的影响中,4周时中等强度运动组和高强度间歇运动组相对于干预前分别提高了17%和23%(P=0.006,95%CI:4%-30%;P=0.000,95%CI:10%-30%),相对于安静组分别提高了18%和27% (P=0.001,95%CI:6%-31%;P=0.000,95%CI:15%- 27%),但二者无显著性差异;8周时中等强度运动组和高强度间歇运动组相对于干预前分别提高了22%和28% (P=0.001,95% CI:7%-37%),P=0.000,95%CI:13%-43%),相对于安静组分别提高了31%和40% (P=0.000,95%CI:15%-47%;P=0.000,95%CI:24%- 56%),但二者无显著性差异;干预12周中等强度运动组和高强度间歇运动组相对于干预前分别提高了45%和46% (P=0.001,95%CI:26%-64%,P=0.000,95%CI:27%- 64%),相对于安静组分别提高了55%和57% (P=0.000,95%CI:32%-77%;P=0.000,95%CI:35%-80%),但二者无显著性差异,见图3。可见,安静组在整个干预过程中没有明显波动,而中等强度运动组和高强度间歇运动组从干预4周时开始,力竭运动时间逐步提高,直到12周时达到最高水平,分别超出对照组55%和57%,但两种运动方式在提高力竭运动时间上无明显的差异。 在对力竭运动距离的影响中,干预4周中等强度运动组和高强度间歇运动组相对于干预前分别提高了35%和48% (P=0.014,95%CI:5%-64%;P=0.000,95%CI:20%-76%),相对于安静组分别提高了38%和58% (P=0.001,95%CI:10%-66%;P=0.000,95%CI:30%-85%),高强度间歇运动组有高于中等强度运动组的趋势(P=0.242);8周时 中等强度运动组和高强度间歇运动组相对于干预前分别提高了45%和60%(P=0.002,95%CI:12%-78%),P=0.000,95%CI:27%-91%),相对于安静组分别提高了64%和87%(P=0.000,95%CI:27%-101%;P=0.000,95%CI:50%-124%),但二者无显著性差异;12周时 中等强度运动组和高强度间歇运动组相对于干预前分别提高了106%和105%(P=0.001,95%CI:64%- 148%,P=0.000,95%CI:65%-146%),相对于安静组分别提高了135%和144%(P=0.000,95%CI:82%-188%;P=0.000,95%CI:90%-197%),但二者无显著性差异,见图4。可见,安静组在整个干预过程中没有明显波动,而中等强度运动组和高强度间歇运动组从干预4周时开始,力竭运动距离逐步提高,直到12周时达到最高水平,分别超出对照组135%和144%,且高强度间歇运动在提高力竭运动距离上有优于中等强度运动的趋势。 2.4 两种运动方式对中年大鼠骨骼肌Beclin 1、LC3和P62蛋白表达随时间变化的影响 采用双因素方差分析两种运动方式对比目鱼肌Beclin 1、LC3II的蛋白表达,LC3II/LC3I比值和P62蛋白表达随时间变化的影响。在对比目鱼肌Beclin 1的蛋白表达的影响中,可见12周时安静组相对于干预前有明显的下降趋势(P=0.025),而两个运动组略高于安静组,见图5。 在对比目鱼肌对LC3II蛋白表达的影响中,干预4周时,高强度间歇运动组LC3II蛋白表达显著高于干预前和同期的安静组,分别提升了47%和28%(P=0.003,95%CI:12%-82%;P=0.048,95%CI:0%-55%);8周时,高强度间歇运动组LC3II蛋白表达继续提升,显著高于干预前和同期的安静组,分别提升了60%和25%(P=0.000,95%CI:24%-94%;P=0.05,95%CI:0%-50%),此时中等强度运动组也显著高于干预前40%(P=0.015,95%CI:5%-75%),安静组相对于干预前和4周有提升的趋势,但是无显著变化;12周时,安静组LC3II蛋白表相对于4周和8周时期显著下降33%和40%(P=0.025,95%CI:3%-64%;P=0.001,95%CI:12%-67%),而同期的中等强度运动组和高强度间歇运动组则在8周的基础上继续保持较高水平,分别高于安静组88%和78%(P=0.000,95%CI:48%- 130%;P=0.000,95%CI:38%-120%),见图6。可见,随时间的变化,安静组LC3II蛋白表达在干预12周(大鼠12月龄)出现显著降低。高强度间歇运动组则从干预4周时起显著提升,直到实验结束,而中等强度运动组的提升相对滞后,说明两种运动方式有效促进了LC3II蛋白表达,缓解了安静组的下降趋势,且高强度间歇运动较中等强度运动更具有时效性。"
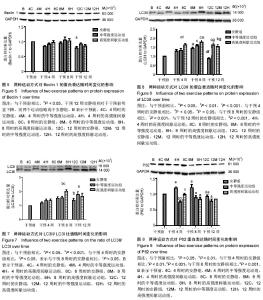
两种运动方式和时间对LC3II/LC3I比值产生了影响(P=0.05,P=0.000),在干预8周时,高强度间歇运动组的LC3II/LC3I比值明显高于干预前(100%,P=0.000,95%CI:44%-157%)和同期安静组(34%,P=0.048, 95%CI:0%-64%),到了干预第12周,两个运动组均维持在较高水平,分别高于干预前80%和66%(P=0.002,95%CI:23%- 136%;P=0.014,95%CI:9%-122%),且有高于同期安静组的趋势,见图7。 双因素方差分析发现,不同运动方式和时间对比目鱼肌P62蛋白相对含量产生了影响,干预4周时,安静组相对于干预前的P62蛋白含量显著上升(P < 0.05),而中等强度运动组和高强度间歇运动组显著降低了此时安静组的P62蛋白含量(36%,P=0.000,95%CI:24%-50%;18%,P=0.000,95%CI:5%-32%);干预8周时,相对于干预前,安静组P62蛋白水平仍保持较高的水平(P < 0.05),而中等强度运动组和高强度间歇运动组同样显著降低了此时安静组的P62蛋白含量(26%,P=0.000,95%CI:12%-40%;30%,P=0.000,95%CI:16%-44%);到了干预12周,安静组P62蛋白含量显著低于了干预4,8周时(P < 0.001),此时中等强度运动组和高强度间歇运动组有低于安静组的趋势,但差异无显著性意义,见图8。 安静组在干预4,8周时P62蛋白含量显著上升,这可能与大鼠比目鱼肌自噬体的产生增多有关,而两种运动方式均有效降低了P62蛋白含量。在干预12周时,由于安静组比目鱼肌自噬体产生减少,P62蛋白含量也相应下降了,但仍有较高自噬水平的两个运动组保持了较低P62蛋白含量,说明两种运动方式促进了该时期自噬活性的通畅性。"
| [1] Sandri M. Autophagy in health and disease. 3. Involvement of autophagy in muscle atrophy. Am J Physiol Cell Physiol. 2010;298(6): C1291-C1297.[2] Masiero E, Agatea L, Mammucari C, et al. Autophagy is required to maintain muscle mass. Cell Metab. 2009;10(6):507-515.[3] Papackova Z, Cahova M. Important role of autophagy in regulation of metabolic processes in health, disease and aging. Physiol Res. 2014; 63(4):409-420.[4] Tachtsis B, Smiles WJ, Lane SC, et al. Acute Endurance Exercise Induces Nuclear p53 Abundance in Human Skeletal Muscle. Front Physiol. 2016;7:144.[5] Pigna E, Berardi E, Aulino P, et al. Aerobic Exercise and Pharmacological Treatments Counteract Cachexia by Modulating Autophagy in Colon Cancer. Sci Rep. 2016;6:26991.[6] Kim JS, Lee YH, Yi HK. Gradual downhill running improves age-related skeletal muscle and bone weakness: implication of autophagy and bone morphogenetic proteins. Exp Physiol. 2016;101(12):1528-1540.[7] Vainshtein A, Hood DA. The regulation of autophagy during exercise in skeletal muscle. J Appl Physiol (1985). 2016;120(6):664-673.[8] White Z, Terrill J, White RB, et al. Voluntary resistance wheel exercise from mid-life prevents sarcopenia and increases markers of mitochondrial function and autophagy in muscles of old male and female C57BL/6J mice. Skelet Muscle. 2016;6(1):45.[9] 黎涌明.高强度间歇训练对不同训练人群的应用效果[J].体育科学, 2015, 35(8):59-75.[10] Billat L V. Interval training for performance: a scientific and empirical practice. Special recommendations for middle- and long-distance running. Part I: aerobic interval training. Sports Med. 2001;31(1):13-31.[11] Nybo L, Sundstrup E, Jakobsen MD, et al. High-intensity training versus traditional exercise interventions for promoting health. Med Sci Sports Exerc. 2010;42(10):1951-1958.[12] Gibala MJ, Mcgee SL. Metabolic adaptations to short-term high-intensity interval training: a little pain for a lot of gain? Exerc Sport Sci Rev. 2008;36(2):58-63.[13] 王京京,韩涵,张海峰.高强度间歇训练对青年肥胖女性腹部脂肪含量的影响[J].中国运动医学杂志,2015,34(1):15-20.[14] Heinrich KM, Patel PM, O'neal JL, et al. High-intensity compared to moderate-intensity training for exercise initiation, enjoyment, adherence, and intentions: an intervention study. BMC Public Health. 2014;14:789.[15] Bartlett JD, Close GL, Maclaren DP, et al. High-intensity interval running is perceived to be more enjoyable than moderate-intensity continuous exercise: implications for exercise adherence. J Sports Sci. 2011;29(6):547-553.[16] Tjonna AE, Lee SJ, Rognmo O, et al. Aerobic interval training versus continuous moderate exercise as a treatment for the metabolic syndrome: a pilot study. Circulation. 2008;118(4):346-354.[17] Burgomaster KA, Howarth KR, Phillips SM, et al. Similar metabolic adaptations during exercise after low volume sprint interval and traditional endurance training in humans. J Physiol. 2008;586(1): 151-160.[18] Holloway TM, Bloemberg D, Da Silva ML, et al. High intensity interval and endurance training have opposing effects on markers of heart failure and cardiac remodeling in hypertensive rats. PLoS One. 2015; 10(3):e0121138.[19] Bedford TG, Tipton CM, Wilson NC, et al. Maximum oxygen consumption of rats and its changes with various experimental procedures. J Appl Physiol Respir Environ Exerc Physiol. 1979;47(6): 1278-1283.[20] Angadi SS, Mookadam F, Lee CD, et al. High-intensity interval training vs. moderate-intensity continuous exercise training in heart failure with preserved ejection fraction: a pilot study. J Appl Physiol (1985). 2015; 119(6):753-758.[21] Smart NA, Dieberg G, Giallauria F. Intermittent versus continuous exercise training in chronic heart failure: a meta-analysis. Int J Cardiol. 2013;166(2):352-358.[22] Arena R, Myers J, Forman DE, et al. Should high-intensity-aerobic interval training become the clinical standard in heart failure?Heart Fail Rev. 2013;18(1):95-105.[23] Leandro CG, Levada AC, Hirabara SM, et al. A program of moderate physical training for Wistar rats based on maximal oxygen consumption. J Strength Cond Res. 2007;21(3):751-756.[24] He C, Bassik M C, Moresi V, et al. Exercise-induced BCL2-regulated autophagy is required for muscle glucose homeostasis. Nature. 2012;481(7382):511-515.[25] Roubenoff R. Physical activity, inflammation, and muscle loss. Nutr Rev. 2007;65(12 Pt 2):S208-S212.[26] Betik AC, Baker DJ, Krause DJ, et al. Exercise training in late middle-aged male Fischer 344 x Brown Norway F1-hybrid rats improves skeletal muscle aerobic function. Exp Physiol. 2008; 93(7):863-871.[27] Kim YA, Kim YS, Oh SL, et al. Autophagic response to exercise training in skeletal muscle with age. J Physiol Biochem. 2013; 69(4):697-705.[28] Stoggl TL, Bjorklund G. High Intensity Interval Training Leads to Greater Improvements in Acute Heart Rate Recovery and Anaerobic Power as High Volume Low Intensity Training. Front Physiol. 2017;8: 562.[29] Moholdt T, Aamot I L, Granoien I, et al. Aerobic interval training increases peak oxygen uptake more than usual care exercise training in myocardial infarction patients: a randomized controlled study. Clin Rehabil, 2012;26(1):33-44.[30] Freyssin C, Verkindt C, Prieur F, et al. Cardiac rehabilitation in chronic heart failure: effect of an 8-week, high-intensity interval training versus continuous training. Arch Phys Med Rehabil. 2012;93(8):1359-1364.[31] Scharf M, Schmid A, Kemmler W, et al. Myocardial adaptation to high-intensity (interval) training in previously untrained men with a longitudinal cardiovascular magnetic resonance imaging study (Running Study and Heart Trial). Circ Cardiovasc Imaging. 2015; 8(4):e002566.[32] Metcalfe RS, Babraj JA, Fawkner SG, et al. Towards the minimal amount of exercise for improving metabolic health: beneficial effects of reduced-exertion high-intensity interval training. Eur J Appl Physiol. 2012;112(7):2767-2775.[33] Cocks M, Shaw CS, Shepherd SO, et al. Sprint interval and endurance training are equally effective in increasing muscle microvascular density and eNOS content in sedentary males. J Physiol. 2013;591(3): 641-656.[34] Little JP, Safdar A, Bishop D, et al. An acute bout of high-intensity interval training increases the nuclear abundance of PGC-1alpha and activates mitochondrial biogenesis in human skeletal muscle. Am J Physiol Regul Integr Comp Physiol. 2011;300(6):R1303-R1310.[35] Bartlett JD, Hwa Joo C, Jeong TS, et al. Matched work high-intensity interval and continuous running induce similar increases in PGC-1alpha mRNA, AMPK, p38, and p53 phosphorylation in human skeletal muscle. J Appl Physiol (1985). 2012;112(7):1135-1143.[36] Taylor CW, Ingham SA, Ferguson RA. Acute and chronic effect of sprint interval training combined with postexercise blood-flow restriction in trained individuals. Exp Physiol. 2016;101(1):143-154.[37] Robinson MM, Dasari S, Konopka AR, et al. Enhanced Protein Translation Underlies Improved Metabolic and Physical Adaptations to Different Exercise Training Modes in Young and Old Humans. Cell Metab. 2017;25(3):581-592.[38] Scribbans TD, Edgett BA, Vorobej K, et al. Fibre-specific responses to endurance and low volume high intensity interval training: striking similarities in acute and chronic adaptation. PLoS One. 2014;9(6): e98119.[39] Mueller SM, Aguayo D, Zuercher M, et al. High-intensity interval training with vibration as rest intervals attenuates fiber atrophy and prevents decreases in anaerobic performance. PLoS One. 2015;10(2): e0116764.[40] Burgomaster KA, Cermak NM, Phillips SM, et al. Divergent response of metabolite transport proteins in human skeletal muscle after sprint interval training and detraining. Am J Physiol Regul Integr Comp Physiol. 2007;292(5):R1970-R1976.[41] Burgomaster KA, Heigenhauser GJ, Gibala MJ. Effect of short-term sprint interval training on human skeletal muscle carbohydrate metabolism during exercise and time-trial performance. J Appl Physiol (1985). 2006;100(6):2041-2047.[42] Kim YA, Kim YS, Song W. Autophagic response to a single bout of moderate exercise in murine skeletal muscle. J Physiol Biochem. 2012;68(2):229-235.[43] Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132(1):27-42.[44] 刘文锋,魏霞,张冰侠,等.规律运动对增龄大鼠比目鱼肌细胞凋亡与自噬及PGC-1α信号调控的影响[J].中国运动医学杂志,2017,36(2):128-135.[45] 马晓雯,常芸,王世强,等.不同强度不同时间耐力训练对于大鼠心肌细胞自噬发生程度的影响[J].中国运动医学杂志,2016,35(1):27-31.[46] Klionsky DJ. Autophagy: from phenomenology to molecular understanding in less than a decade. Nat Rev Mol Cell Biol. 2007;8(11): 931-937.[47] Wirawan E, Lippens S, Vanden Berghe T, et al. Beclin1: a role in membrane dynamics and beyond. Autophagy. 2012;8(1):6-17.[48] Lira VA, Okutsu M, Zhang M, et al. Autophagy is required for exercise training-induced skeletal muscle adaptation and improvement of physical performance. FASEB J. 2013;27(10):4184-4193.[49] Kim J, Kundu M, Viollet B, et al. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol. 2011;13(2): 132-141.[50] Zhao J, Brault JJ, Schild A, et al. Coordinate activation of autophagy and the proteasome pathway by FoxO transcription factor. Autophagy. 2008;4(3):378-380.[51] Zhao J, Brault JJ, Schild A, et al. FoxO3 coordinately activates protein degradation by the autophagic/lysosomal and proteasomal pathways in atrophying muscle cells. Cell Metab. 2007;6(6):472-483.[52] Mizushima N, Yoshimori T. How to interpret LC3 immunoblotting. Autophagy. 2007;3(6):542-545.[53] Kabeya Y, Mizushima N, Ueno T, et al. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 2000;19(21):5720-5728.[54] Kabeya Y, Mizushima N, Yamamoto A, et al. LC3, GABARAP and GATE16 localize to autophagosomal membrane depending on form-II formation. J Cell Sci. 2004;117(Pt 13):2805-2812. |
| [1] | Mao Yu-rong, Chen Na, Chen Pei-ming, Chen Song-bin, Li Li-fang, Huang Dong-feng. Three-dimensional motion analysis of dominant and nondominant hands under weight-bearing conditions in health elderly people [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(42): 6776-6781. |
| [2] | Bi Lian-yong. Neuroprotective effects of salvianolic acid B against Schwann cell injury induced by lipopolysaccharide [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(42): 6813-6818. |
| [3] | Wang Ying-wei, Qin Zi-xi, Wu Zheng. Fabrication and functional characteristics of cardiac patches [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(38): 6211-6216. |
| [4] | Sun Ming-yu, Yan Cheng-hui, Tian Xiao-xiang, Li Yang, Han Ya-ling. Interactions between the recombinant human CREG protein and cathepsins and M6P/IGFIIR [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(37): 5961-5965. |
| [5] | Liu Xing-zhen, Jin Wen-jie, Shen Kang-ping, Fu Zhi-yi, Wu Yu-jie. Expression of hypoxia-inducible factor 1 alpha and its relationship to apoptosis in human lumbar nucleus pulposus [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(33): 5279-5283. |
| [6] | Luo Qing-qing. Effect of Dll4 at different concentrations on the biological functions of extravillous trophoblasts [J]. Chinese Journal of Tissue Engineering Research, 2015, 19(24): 3860-3864. |
| [7] | Tan Yu-lin1, Bao Tong-zhu, Liu Qin, Han Yu, Wang Yan-lin, Liu Yang, Zhou Na-xin, Yan Fei,Zhao Long-tao. Influence of geniposide on collagen II synthesis of cultured chondrocytes in vitro [J]. Chinese Journal of Tissue Engineering Research, 2014, 18(20): 3149-3152. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||