Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (4): 619-624.doi: 10.3969/j.issn.2095-4344.0100
Previous Articles Next Articles
Animal models of intervertebral disc degeneration: economy, feasibility, reliability and controllability
Yin Xun-lu1, Feng Min-shan1, 2, Zhu Li-guo1, 2, Li Xue-peng1, Chen Lin3, Li Ling-hui1, Zhan Jia-wen1, Wei Xu1
- 1Wangjing Hospital of China Academy of Chinese Medical Sciences, Beijing 100102, China; 2Beijing Key Laboratory of Palasy Technology, Beijing 100102, China; 3Beijing University of Chinese Medicine, Beijing 100102, China
-
Received:2017-10-20Online:2018-02-08Published:2018-02-08 -
Contact:Feng Min-shan, M.D., Associate chief physician, Wangjing Hospital of China Academy of Chinese Medical Sciences, Beijing 100102, China; Beijing Key Laboratory of Palasy Technology, Beijing 100102, China -
About author:Yin Xun-lu, Studying for doctorate, Physician, Wangjing Hospital of China Academy of Chinese Medical Sciences, Beijing 100102, China -
Supported by:the Project of State Administration of Traditional Chinese Medicine of China, No. JDZX2015274; the National Natural Science Foundation of China, No. 81674005 and 81774330; the Natural Science Foundation of Beijing, No. 7164313; the Postdoctoral Science Foundation of China, No. 2016M591364
CLC Number:
Cite this article
Yin Xun-lu1, Feng Min-shan1, 2, Zhu Li-guo1, 2, Li Xue-peng1, Chen Lin3, Li Ling-hui1, Zhan Jia-wen1, Wei Xu1. Animal models of intervertebral disc degeneration: economy, feasibility, reliability and controllability[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(4): 619-624.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 模式动物的选择 用于研究椎间盘退行性变动物模型的模式动物种类很多,但主要分为两大类,即小型动物和大型动物。选择合适的模式动物对后续的实验研究至关重要,如在体外器官模型中,考虑到营养物质的弥散距离,小型动物椎间盘的体表比大,有利于营养及代谢物质的渗透,适合做长期体外培养;而大型动物椎间盘体积大,适合生物力学的研究。 2.1.1 小型动物 (1)鼠类:当前,常用于椎间盘退行性变研究的鼠类主要是大鼠和小鼠[4-6],此外还有沙鼠、仓鼠等。鼠类作为动物模型,具有经济适用、种系纯、基因相似度高、抗感染、生命力强等优势;然而,受其体型小、椎间盘体积小等客观因素的影响,造成影像学指标测量困难、生物学检测标本获取量少等不足。Moskowitz等[7]研究发现,沙鼠受长期高盐低水习惯的影响,椎间盘普遍较早退变(椎间盘退变率在3月龄时约为10%,18月龄时约为50%),且该性状具有较强的遗传性。Silberberg等[8]对糖尿病发生率较高的仓鼠进行脊柱学相关研究发现:糖尿病仓鼠相比于非糖尿病仓鼠,脊柱炎的发生率分别为60%和39%,而椎间盘突出的发生率分别为9%和30%。 (2)兔类:作为常用的实验动物之一,具有取材便利、经济适用等特点;相比于鼠类,其椎间盘体积较大,操作方便、便于影像学指标测量,且结构与人类基本相同。Lipson等[9]研究发现:造模前兔椎间盘含水量及蛋白多糖单体大小与人类相似,但其透明质酸浓度及蛋白多糖聚合体占蛋白多糖总量的百分比与人类存有差异;造模12个月后,各项观察指标(除蛋白多糖单体大小外)变化规律与人类相似。 2.1.2 大型动物 (1)犬类:实验犬的种类比较多,主要包括德克萨斯犬、哈巴狗、猎犬、长毛耳垂犬等。相比于小型动物,犬椎间盘及椎体体积更接近于人类,操作性及可行性更好;然而其经济适用性相对较低。Gillett等[10]通过对不同年龄阶段的猎犬进行研究后,认为犬的椎间盘退行性变的进程与人类相似;Goggin等[11]则对不同的种犬进行比较研究,发现德克萨斯犬患椎间盘突出的相对危险度最大。 (2)羊类:羊的种系较多,取材较为方便,其脊柱解剖结构、椎间盘及椎体大小与人类较为相似;而且羊的胸腰段脊柱在某些指标上与人类具有相似之处,因此可以作为理想的实验动物;然而,相比于小型动物,羊的经济成本相对较高,从而限制了羊作为体内模型动物的大量使用。Paul等[12]研究发现,山羊椎间盘中只有软骨细胞和成纤维细胞,而没有脊索细胞,这与人类在生物学方面有一定的相似性;Gantenbein等[13]认为,羊尾椎间盘在营养和代谢产物渗透方面与人类存在相似性,故羊尾椎间盘适合作人类椎间盘退变模型;任东风等[14]通过对羊腰椎破裂模型进行研究后,认为经山羊腰椎间盘前方人为损伤后部纤维环内层,可以可靠、一致地制备椎间盘内核破裂模型。 (3)猪类:猪由于在解剖学、生理学方面与人类有极大的相似性,且体型相对较大、易于操作,且部分种类的猪经济适用,因此在国外也是被广泛应用的模型动物之一。当前由于缺少关于猪和人类腰脊柱剖学之间的对比资料,国内将其作为实验动物的研究还是比较少[15],但国外许多学者如Kappa等[16-17]已将猪作为动物模型进行研究相关脊柱疾病,并积累了一定的经验。 (4)灵长类:用作模型的灵长类动物主要包括恒河猴、狒狒等。该类动物与人类亲缘关系最近,在生理特性、解剖结构和生活习性等方面也都极为相似,可以作为研究对象直接进行观察[15]。然而,由于其种类、数量的有限性,尤其是价格的高昂性,限制了灵长类作为模型动物的广泛应用。Simon等[18-19]学者应用恒河猴进行了脊柱生物力学方面的研究,郭常安等[20]用恒河猴作椎间盘退变动物模型研究;Lauerman等[21]通过对不同年龄段的狒狒进行影像学研究,发现狒狒椎间盘具有退变早、发生率高的特点,且接近人类椎间盘退行性变的规律。 2.2 动物模型的建立 在椎间盘退行性变动物模型中,虽然建立模型方法很多,但归纳起来只有体内模型和体外模型之别;体内模型根据损伤机制的不同分为:生物力学改变模型、生理结构破坏模型、自发退变模型及全身疾病模型[22];体外模型根据培养组织水平的不同分为体外细胞模型、体外器官模型[23]。 2.2.1 体内模型 (1)生物力学改变模型:该模型主要是通过改变椎间盘与椎体等结构间的生物力学状态,造成椎间盘的退变。其类型包括:去前肢动物模型、应力改变模型、脊柱失稳模型。 去前肢动物模型:当前,该模型动物主要为新生大鼠。将其双前肢结扎后,使其双后肢站立活动;造模时间一般为14-18个月。Bailey等[24]研究后认为,该动物模型存在时间成本高、椎间盘退变率不稳定等不足,且临床相似性低;因此,该模型目前未被广泛应用。 应力改变模型:其模型动物主要为小型动物,一般为大鼠;也有应用大型动物,如羊。该模型通过异常应力破坏椎间盘的结构,导致椎间盘退变[25]。早期应力改变模型是将鼠尾弯成“U”型[26],造模时间一般为2周至14个月;后期改良模型为轴向加压动物模型[27]:将轴性加压装置置于鼠尾,通过轴向某一频率的动态或静态加压,从而破坏椎间盘静水压,进而改变椎间盘基质成分,最终导致椎间盘退行性变。早期“U”型应力退变模型由于会出现尾部凹侧结缔组织破坏、细胞数目减少等不良反应,故逐渐被轴向加压动物模型替代。虽然改良模型也存在一些需要问题,如:造模过程中有克氏针劈裂椎体使脊髓损伤、针道感染等可能,轴向加压装置尚不能模拟人体椎间盘所受的剪切力和扭转力等[28];但其具有良好的可控性和可重复性、造模周期短,可以模拟重力对椎间盘的影响等优势,其应用前景较为广阔。 脊柱失稳模型:该类造模方法对大、小型动物均适合。通过咬除椎体后柱部分结构(如关节突、棘突)或破坏一侧椎旁肌(切断或反复电刺激等)[29],破坏脊柱的动态、静态平衡,导致椎间盘退变[30];造模时间一般为3-8个月。脊柱失稳模型虽然具有较好的操作性,但是也存在造模时间长、不易控制、可重复性差等问题[1]。 (2)生理结构破坏模型:即通过物理或化学方法破坏椎间盘的生理结构,最终导致椎间盘退变;其模型种类分为:机械损伤模型、化学损伤模型。 机械损伤模型:根据其损伤椎间盘位置的不同又可分为纤维环损伤模型、终板损伤模型及髓核损伤模型。 纤维环损伤模型:纤维环损伤方法分为切开法及穿刺法。虽然自Key等[31]首次将兔椎间盘纤维环直接切开造成退变模型后,后人不断对其进行改进,但该法在可控性、标准化等方面仍存在诸多问题。近年来,纤维环穿刺法发展不断成熟且应用相对广泛。该法以一定的次数和深度(兔:5 mm;鼠:1-5 mm;牛:15 mm)将直径固定的穿刺针刺入椎间盘,间接诱发椎间盘退行性变[28,32]。虽然该法所造模型退变的程度和速度受操作者操作手法(深度和次数)的影响,但其具有较高的可重复性、可操作性及适用性,造模周期短(鼠类:2-4周;兔:8-12周),因而被广泛应用。 终板损伤模型:通常是经过椎体和软骨终板斜钻一个到达椎间盘的的孔洞[33],也有学者将骨水泥直接打入椎间盘的上下终板区域[34],从而通过破坏终板与椎间盘之间营养及代谢物质通道达到椎间盘发生退变的目的。然而,由于该种造模方法钻孔时角度、深度以及对终板的损伤程度难以控制,因而影响了该种模型的可靠性、可控性、可重复性等,因而并不常被采用。 髓核损伤模型:是用注射器将髓核组织部分抽吸,通过破坏其静水压达到造模目的;造模时间一般为4-8周。该种方法所造椎间盘退变模型效果显著,尤其适合大型动物,其病理及影像学表现与人椎间盘退变十分相似[35]。 化学损伤模型:即通过向椎间盘内注射化学物质,从而使髓核细胞死亡、基质急剧减少,最终达到使椎间盘退变的目的。常用的化学物质主要是酶类,包括:Cp、CABC、肿瘤坏死因子α等[36-38];此外还有非酶类化学物质,如:博来霉素等。该法虽然可以在较短时间内造成显著的退变模型,但是这与人类椎间盘退变的发生机制差异较大,并且对后续的生化检测指标、生物学疗效等均造成一定程度的影响[28]。因此,虽然该法具有较好的可重复性、可操作性,但上述弊端的存在严重降低了该模型的临床相似性及可靠性,使其应用范围逐渐减小。 (3)自发退变模型:自发退变模型主要包括3类:特定基因敲除模型、椎间盘自发退变的特种动物模型及缺血模型(表1)。"

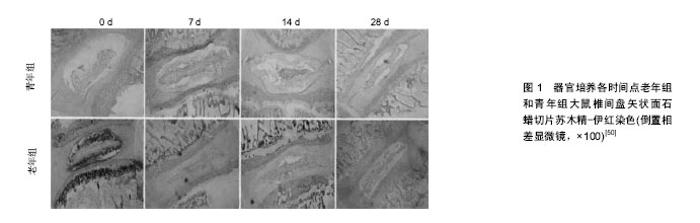
(4)全身疾病模型:包括吸烟模型[40]、卵巢切除模型等[41]。虽然这些模型能较好的拟合导致人类椎间盘退变中的某一因素,但其可重复性差、造模成功率低。 2.2.2 体外模型 (1)体外细胞模型:即采用细胞生物学技术对椎间盘细胞进行体外培养,并观察其生长、增殖和分化的规律及特点。该模型通过采用外部干预处理,探索单一因素对椎间盘退变的影响。当前,体外细胞造模技术已较成熟,且相关检测技术也日臻完善,尤其是近年来基因组学、高通量测序等技术的高速发展,为椎间盘退变的深入研究提供了基础。然而,在细胞培养过程中也面临诸多问题:单层细胞培养多次传代后,易出现细胞形态改变、活力下降[42];离体环境下细胞基质的缺失,导致细胞增殖率低、表型缺失[43],甚至发生分化或丧失功能[44]。 (2)体外器官模型:根据是否带有椎体分为两类:椎间盘器官离体模型和脊柱运动节段离体模型。 椎间盘器官离体模型根据有无终板分为:带终板椎间盘器官离体模型和去终板椎间盘器官离体模型。椎间盘由髓核、纤维环、终板3部分组成,且终板是椎间盘进行营养和代谢物质的通道,具有维持椎间盘完整性及限制组织膨胀压的作用。当前对于该模型是否应带有终板,不同学者间仍存在争议。Korecki,Lee等[45-46]认为,去除终板后可以使营养物质直接扩散到髓核,但不适合长期培养研究;Gawri等[47]则认为,通过预处理终板可以在保持椎间盘器官完整性的同时,限制组织肿胀并保证营养物质供给。 脊柱运动节段离体模型,在体外状态下最大限度的保留了组织器官的完整性,更好的拟合了椎间盘在人体内的生理环境、力学负荷等因素;同时,椎体的保留为脊柱生物力学加载装置提供了着力点,有利于对复杂力学与椎间盘退变之间关系的研究[48]。 椎间盘退变体外器官模型作为体内模型和体外细胞模型之间的桥梁,能够在外界干预下的原生细胞外基质中维持细胞的活性[49];而且,该模型为研究椎间盘对外界刺激的反应和观察正常与退变椎间盘的组织学变化创造了良好的实验平台(图1)[50-51]。"
| [1] 王海莹,张旭,丁文元,等.椎间盘退变动物模型的研究进展[J].中国脊柱脊髓杂志,2015,25(3):279-281.[2] 高乾,程英武.腰椎间盘突出动物模型的研究进展[J].中华中医药杂志,2012,27(12):2165-68.[3] Lob. Die zusammenhänge zwischen der Bandscheiben und der Spondylosis deformans im Tieversuch. Deutsche Zeitschrift for Chirurgie.1933:240-421.[4] Risbud MV, Izzo MW, Adams CS,et al. An organ culture system for the study of the nucleus pulposus: description of the system and evaluation of the cells. Spine (Phila Pa 1976).2003;28(24): 2652-2658; discussion 2658-2659. [5] Lim TH, Ramakrishnan PS, Kurriger GL, et al. Rat spinal motion segment in organ culture: a cell viability study. Spine (Phila Pa 1976).2006;31(12):1291-1297; discussion 1298.[6] Ariga K, Yonenobu K, Nakase T,et al.Mechanical stress-induced apoptosis of endplate chondrocytes in organ-cultured mouse intervertebral discs: an ex vivo study. Spine (Phila Pa 1976). 2003;28(14):1528-33.[7] Moskowitz RW, Ziv I, Denko CW, et al. Spondylosis in sand rats: a model of intervertebral disc degeneration and hyperostosis.J Orthop Res.1990;8(3):401-411.[8] Silberberg R, Gerritsen G. Aging changes in intervertebral discs and spondylosis in Chinese hamsters. Diabetes. 1976; 25(6):477-83.[9] Lipson SJ, Muir H.1980 Volvo award in basic science. Proteoglycans in experimental disc degeneration.Spine (Phila Pa 1976).1981;6(3):194-210.[10] Gillett NA, Gerlach R, Cassidy JJ, et al. Age-related changes in the beagle spine. Acta Orthop Scand.1988;59(5):503-507.[11] Goggin JE, Li AS, Franti CE. Canine intervertebral disk disease: characterization by age, sex, breed and anatomic site of involvement.Am J Vet Res.1970;31(9):1687-1692.[12] Paul CP, Zuiderbaan HA, Zandieh Doulabi B, et al. Simulated-physiological loading conditions preserve biological and mechanical properties of caprine lumbar intervertebral discs in ex vivo culture. PLoS One.2012; 7(3):e33147.[13] Gantenbein B, Grünhagen T, Lee CR, et al. An in vitro organ culturing system for intervertebral disc explants with vertebral endplates: a feasibility study with ovine caudal discs. Spine (Phila Pa 1976).2006;31(23):2665-2673.[14] 任东风,侯树勋,彭宝淦,等.腰椎间盘内破裂模型的建立[J].中国疼痛医学杂志,2006,12(6):353-356.[15] 陈亮,吕朝辉.腰椎间盘退变动物模型的研究概况[J].中国中医骨伤科杂志,2008,16(12):59-60.[16] Kääpä E, Holm S, Han X, et al. Collagens in the injuries porcine intervertebral disc. J Orthop Res.1994;12(1):93-102.[17] Pfeiffer M, Griss P, Franke P, et al. Degeneration model of the porcine lunbar motion segment: effects of various intradiscal procedures. Eur Spine J.1994;3(1): 8-16.[18] Simon BR, Wu JS. Poroelastic dynamic structural models of rhesus spinal motion segments. Spine (Phila Pa 1976).1985; 10(6):494-507.[19] 阮狄克,费正奇,陆瓞骥,等.冷冻保存异体椎间盘移植的实验研究[J].中华骨科杂志,1999,19(1):54-58.[20] 郭常安,胡有谷,吴新彦,等.腰椎间盘退变动物模型的建立[J].中华外科杂志,2000,38(7):548-551.[21] Lauerman WC, Platenberg RC, Cain JE, et al. Age-related disk degeneration: Preliminaryrepot of a naturally occurring baboon model.J Spinal Disord.1992;5(2): 170-174.[22] 刘彬彬,张柳,田发明,等.椎间盘退变动物模型及降钙素治疗的研究进展[J].中国修复重建外科杂志,2014,28(8):1051-1053.[23] 杨松波,高春华,庞晓东,等.椎间盘退变模型的研究进展[J].脊柱外科杂志,2012, 10(5):315-317[24] Bailey AS, Adler F, Min Lai S, et al. A comparison between bipedal and quadrupedal rats: do bipedal rats actually assume an upright posture. Spine (Phila Pa 1976).2001;26(14):E308-13.[25] Kelsey JL, Githens PB, O’Conner T, et al. Acute prolapsed lumbar intervertebral disc: an epidemiologic study with special reference to driving automobiles and cigarette smoking. Spine (Phila Pa 1976).1984;9(6):608-613.[26] Lindblom K. Intervertebral-disc degeneration considered as a pressure atrophy. J Bone Joint Surg Am. 1957;39-A(4): 933-945.[27] Lai A, Chow DH, Siu SW, et al. Effects of static compression with different loading magnitudes and durations on the intervertebral disc: an in vivo rat-tail study. Spine (Phila Pa 1976). 2008;33 (25):2721-2727. [28] 顾韬,张超,何勍,等.不同类型椎间盘退变动物模型的评价与比较[J].脊柱外科杂志,2015,13(2):115-120.[29] Sullivan JD,Farfan HF,Kahn DS. Pathologicchanges with intervertebral join rotational instability in the rabbit. Can J Surg. 1971;14:71-79.[30] Wang YJ, Shi Q, Lu WW, et al. Cervical intervertebral disc degeneration induced by unbalanced dynamic and static forces: a novel in vivo rat model. Spine (Phila Pa 1976).2006; 31(14):1532-1538.[31] Key JA, Ford LT. Experimental interverbral disc lesion. J Bone Joint Surg Am.1948;30(3):621-629.[32] Aoki Y, Akeda K, An H, et al. Nerve fiber ingrowth into scar tissue formed following nucleus pulposus extrusion in the rabbit anular-puncture disc degeneration model: effects of depth of puncture.Spine (Paila Pa 1976).2006;31(21):E774-80. [33] 吕浩然,杨进顺,黄文铎,等.终板损伤建立兔腰椎间盘退变模型[J].广东医学,2008,29(1):44-45.[34] Hutton WC, Murakami H, Li J, et al. The effect of blocking a nutritional pathway to the intervertebral disc in the dog model. J Spinal Disord Tech.2004;17(1):53-63. [35] 周松,李锋,陈安民,等.抽洗法诱导椎间盘退变模型的病理及影像表现[J].中国组织工程研究与临床康复,2009,13(46):9125-9128.[36] Lotz JC.Animal models of intervertebral disc degeneration: lessons learned. Spine (Phila Pa 1976).2004;29(23):2742-2750.[37] Hoogendoom RJ, Wuisman PI, Smit TH, et al. Experimental intervertebral disc degeneration induced by chondroitinase ABC in the goat. Spine (Phila Pa 1976).2007;32(17):1816-1825.[38] Kang R, Li H, Rickers K, et al. Intervertebral disc degenerative changes after intradiscal injection of TNF-α in a porcine model. Eur Spine J.2015;24(9):2010-2016.[39] Boyd LM, Richardson WJ, Allen KD, et al. Early-onset degeneration of the intervertebral disc and vertebral end plate in mice deficient in type IX collagen. Arthritis Rheum. 2008; 58(1):164-171.[40] Oda H, Matsuzaki H, Tokuhashi Y, et al. Degeneration of intervertebral discs due to smoking: experimental assessment in a rat-smoking model.J Orthop Sci.2004;9(2):135-141.[41] Wang T, Zhang L, Huang C, et al. Relationship between osteopenia and lumbar intervertebral disc degeneration in ovariectomized rats. Calcif Tissue Int.2004;75(3):205-213.[42] Preradovic A, Kleinpeter G, Feichtinger H, et al. Quantitation of collagen I, collagen II and aggrecan mRNA and expression of the corresponding proteins in human nucleus pulposus cells in monolayer cultures. Cell Tissue Res.2005;321(3):459-464.[43] Gruber HE, Hanley EN, Jr. Human dics cells in monolayer vs 3D culture: cell shape, division and matrix formation. BMC Musculoskelet Disord.2000,1:1.[44] Zhang Y, Phillips FM, Thonar EJ, et al. Cell therapy using articular chondrocytes overexpressing BMP-7 or BMP-10 in a rabbit disc organ culture model. Spine (Phila Pa 1976).2008; 33(8):831-838.[45] Korecki CL, MacLean JJ, Iatridis JC. Characterization of an in vitro intervertebral disc organ culture system. Eur Spine J.2007; 16(7):1029-37.[46] Lee CR, Iatridis JC, Poveda L, et al. In vitro organ culture of the bovine intervertebral disc: effects of vertebral endplate and potential for mechanobiology studies. Spine (Phila Pa 1976).2006;31(5):515-522.[47] Gawri R, Mwale F,Ouellet J, et al. Development of an organ culture system for long-term survival of the intact human intervertebral disc. Spine (Phila Pa 1976).2011;36(22):1835-1842. [48] Seol D, Choe H, Ramakrishnan PS, et al. Organ culture stability of the intervertebral disc: rat versus rabbit. J Orthop Res.2013;31(6):838-46.[49] Risbud MV, Izzo MW, Adams CS, et al. An organ culture system for the study of the nucleus pulposus: description of the system and evaluation of the cells. Spine (Phila Pa 1976).2003; 28(24): 2652-2658; discussion 2658-2659.[50] 徐宏光,章平治,宋俊兴,等.退变大鼠椎间盘器官培养模型的建立及其意义[J].中国骨与关节外科,2012,5(3):233-237+197.[51] 冯敏山,展嘉文,朱立国,等.体外培养条件下脊柱运动节段髓核组织的变化[J].中国组织工程研究,2015,19(51):8241-46.[52] Sun YQ, Zheng S, Yu J, et al. Effect of total disc replacement on atypical symptoms associated with cervical spondylosis. Eur Spine J.2013;22(7):1553-1557.[53] Oguntona SA. Cervical spondylosis in South West Nigerian farmers and female traders. Ann Afr Med.2014;13(2):61-64.[54] Yoshimura S, Shimosato A, Imanami M, et al. Awake intubation using Pentax-airway scope combined with surface airway anesthesia in the cases of two obese patients with cervical spondylosis and the author himself. Masui.2012;61(7): 738-741.[55] 宁斌,整修军,胡有谷.慢性压迫性脊髓损伤动物模型的制作方法[J].中国脊柱脊髓杂志,2005,15(5):316-318.[56] 陈智,魏显招.脊髓型颈椎病动物模型的研究进展[J].中国矫形外科杂志,2010,18(21):1784-1786. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||