Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (26): 6779-6798.doi: 10.12307/2026.803
Previous Articles Next Articles
Curcumin-loaded chitosan/beta-glycerophosphate sodium thermoresponsive hydrogel promotes tendon healing in rats
Zhang Yibo1, Li Jian2, Wang Peng1, Jiang Qing1
- 1Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, Nanjing 210000, Jiangsu Province, China; 2School of Medicine, Jiangsu Medical College, Yancheng 224005, Jiangsu Province, China
-
Accepted:2025-12-18Online:2026-09-18Published:2026-03-11 -
Contact:Zhang Yibo, Doctoral candidate, Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, Nanjing 210000, Jiangsu Province, China -
About author:Jiang Qing, PhD, Professor, Nanjing Drum Tower Hospital, Affiliated Hospital of Medical School, Nanjing University, Nanjing 210000, Jiangsu Province, China -
Supported by:Yancheng Basic Research Program, No. YCBK2024048 (to LJ); National Natural Science Foundation of China, No. 32271413 (to WP)
CLC Number:
Cite this article
Zhang Yibo, Li Jian, Wang Peng, Jiang Qing. Curcumin-loaded chitosan/beta-glycerophosphate sodium thermoresponsive hydrogel promotes tendon healing in rats[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6779-6798.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
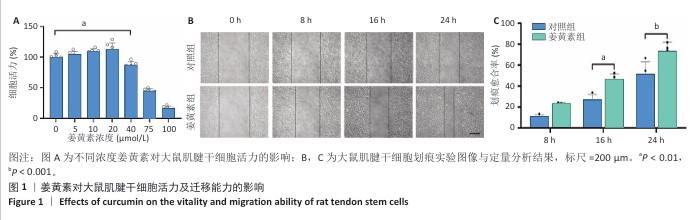
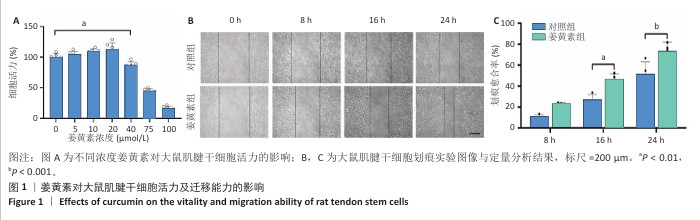
2.1 姜黄素对大鼠肌腱干细胞活力及迁移能力的影响 为探究姜黄素对肌腱干细胞增殖特性及细胞毒性的剂量效应,研究检测了0-100 μmol/L浓度梯度姜黄素对细胞活力的作用,结果显示:当姜黄素浓度≤20 μmol/L时,细胞吸光度值呈浓度依赖性增加,提示药物具有一定的促增殖效应;当姜黄素浓度达到40 μmol/L时,细胞活力下降至对照组的87.15%,并且随姜黄素浓度升高表现出显著的细胞毒性作用(图1A)。基于此结果,后续实验选择20 μmol/L作为姜黄素最佳处理浓度。 在组织修复过程中,定向迁移能力是干细胞参与损伤修复的关键生物学特性。为评估姜黄素对大鼠肌腱干细胞迁移功能的影响,研究通过划痕实验动态观察细胞迁移行为,结果显示姜黄素组16,24 h后的划痕愈合率显著高于对照组,表明20 μmol/L姜黄素能有效增强大鼠肌腱干细胞的迁移能力(图1B,C)。"
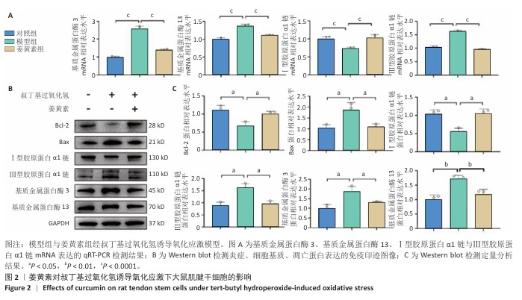
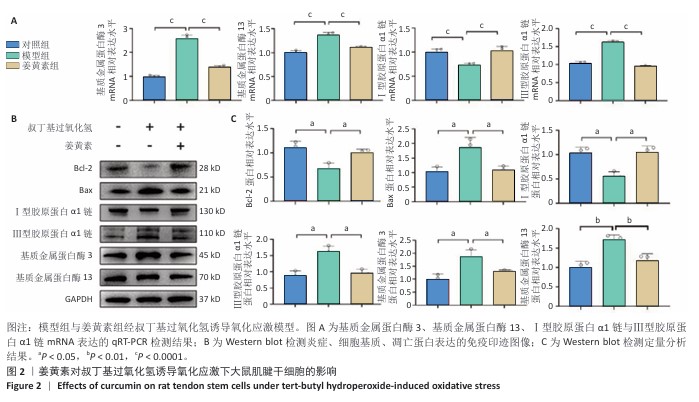
2.2 姜黄素提高大鼠肌腱干细胞的抗炎、抗氧化应激能力 肌腱损伤修复机制涉及炎症反应、细胞增殖及组织重塑3个阶段的级联生物学反应,值得注意的是,损伤微环境常存在血供障碍及炎性递质异常聚集现象。此次研究采用125 μmol/L叔丁基过氧化氢构建体外氧化应激模型,该浓度已在前人研究中证实可有效模拟肌腱干细胞的病理微环境[32-33]。 qRT-PCR检测显示,与对照组比较,模型组基质金属蛋白酶3、基质金属蛋白酶13、Ⅲ型胶原蛋白α1链mRNA表达升高,Ⅰ型胶原蛋白α1链mRNA表达降低,差异均有显著性意义;与模型组比较,姜黄素基质金属蛋白酶3、基质金属蛋白酶13、Ⅲ型胶原蛋白α1链mRNA表达降低,Ⅰ型胶原蛋白α1链mRNA表达升高,差异均有显著性意义,见图2A。 Western blot检测显示,与对照组比较,模型组基质金属蛋白酶3、基质金属蛋白酶13、Ⅲ型胶原蛋白α1链、Bax蛋白表达升高,Ⅰ型胶原蛋白α1链、Bcl-2蛋白表达降低,差异均有显著性意义;与模型组比较,姜黄素组基质金属蛋白酶3、基质金属蛋白酶13、Ⅲ型胶原蛋白α1链、Bax蛋白表达降低,Ⅰ型胶原蛋白α1链、Bcl-2蛋白表达升高,差异均有显著性意义,见图2B,C。 加入叔丁基过氧化氢造模后,一方面,炎症因子通过抑制Ⅰ型胶原转录影响正常胶原合成;另一方面,激活的蛋白酶加速细胞外基质降解,同时氧化应激可诱发肌腱干细胞凋亡级联反应;值得注意的是, Ⅲ型胶原虽在损伤早期发挥临时支架作用,但它的过量积累将导致组织力学性能下降。引入姜黄素干预后表现出显著调控作用:①有效抑制叔丁基过氧化氢诱导的炎性相关基质金属蛋白酶家族表达;②双向调节胶原代谢,恢复Ⅰ型胶原表达同时降低Ⅲ型胶原表达;③逆转凋亡相关蛋白表达失衡,提高抗凋亡蛋白Bcl-2的表达同时抑制促凋亡蛋白Bax表达,提示姜黄素可能通过多靶点调控维护肌腱干细胞的功能稳态。 "

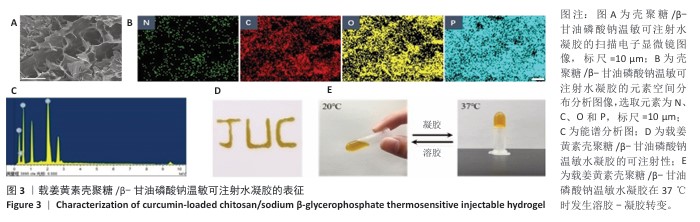
2.3 水凝胶的构建和表征结果 扫描电镜下可见壳聚糖/β-甘油磷酸钠水凝胶呈现典型的三维多孔网络形貌,孔径分布均匀且孔隙相互贯通,如图3A所示。元素空间分布扫描分析证实碳(C)、氧(O)、磷(P)、氮(N)4种组分在水凝胶基质中呈高度均匀分散状态(图3B);能谱定量分析结果显示(图3C),各元素质量分数依次为35.04%,5.48%,50.06%和9.41%,对应原子百分比分别为43.28%,5.80%,46.41%和4.51%,与材料理论组成具有良好一致性。基于前期优化的制备工艺,通过动态共混法将姜黄素负载于壳聚糖/β-甘油磷酸钠温敏体系中,流变学测试表明,载姜黄素壳聚糖/β-甘油磷酸钠温敏水凝胶体系在25 ℃条件下表现出优异的剪切稀化特性,可顺利通过26 G注射针头实现精准递送(图3D);当温度升至生理温度条件(37 ℃)时,体系在120 s内完成溶胶-凝胶相转变(图3E),形成具有空间稳定性的三维网络结构,这种智能温敏特性为局部药物缓释提供了理想载体。 "
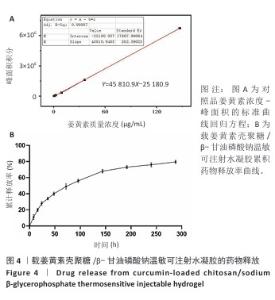
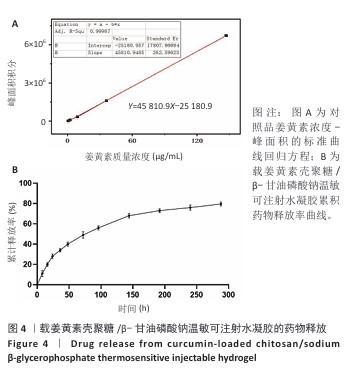
2.4 载姜黄素壳聚糖/β-甘油磷酸钠温敏可注射水凝胶的药物释放研究 通过超高效液相色谱法建立姜黄素定量检测体系,以系列梯度浓度(0.575 6-147.351 4 μg/mL)的姜黄素对照品溶液为横坐标(X),对应色谱峰面积为纵坐标(Y)进行线性回归,获得标准曲线方程Y=45 810.9X-25 180.9(R2=0.999 87),证实检测系统在宽浓度范围内具有优异线性响应(图4A)。方法学验证显示:连续3次进样的峰面积相对标准偏差均低于2%,三组平行样品溶液检测结果的相对标准偏差亦小于2%,证实该方法具有良好精密度与重复性。 通过透析法持续监测载姜黄素壳聚糖/β-甘油磷酸钠水凝胶体系288 h内姜黄素的释放行为,如图4B所示。载姜黄素壳聚糖/β-甘油磷酸钠水凝胶体系呈现典型缓释特性,初始24 h为快速释放阶段,随后释放速率显著下降,96 h姜黄素累计释放量为56.01%,至144,240 h分别达68.3%,76.04%;在288 h时释放趋于平衡(79.51%)。整个释放过程未观察到药物突然释放现象,符合一级动力学释放模型特征。基于体外药物释放动力学数据,结合药物半衰期及组织修复周期特点,确定在大鼠跟腱断裂模型中采用4 d间隔的局部皮下重复注射策略,以确保治疗窗口期内的有效药物浓度。 "
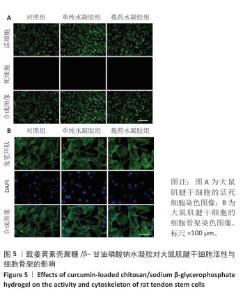
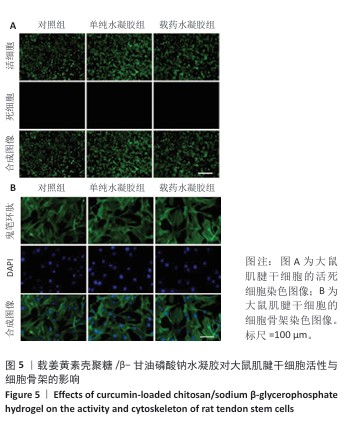
2.5 载姜黄素壳聚糖/β-甘油磷酸钠温敏可注射水凝胶的体外生物安全性评估 为评估载姜黄素壳聚糖/β-甘油磷酸钠温敏可注射水凝胶的细胞相容性,此次研究通过细胞形态学观察结合荧光标记技术对大鼠肌腱干细胞行为进行系统分析。当细胞生长融合度达90%以上时,各组细胞均呈现贴壁生长特性,未发现明显悬浮细胞。活/死染色显示(图5A),水凝胶处理组因37 ℃成胶过程形成的三维微拓扑结构导致荧光信号呈现梯度分布特征,但各组间细胞存活情况无明显差异,表明温敏水凝胶体系未产生细胞毒性效应,并且能构建适宜细胞生长的仿生微环境。 为进一步解析材料对细胞骨架的影响,采用鬼笔环肽染色试剂盒对细胞骨架进行染色(鬼笔环肽染色:绿色;DAPI:蓝色),如图5B所示。对照组肌腱干细胞呈现典型肌腱干细胞形态学特征,F-肌动蛋白纤维沿细胞长轴有序排列,形成致密应力纤维束。单纯水凝胶组、载药水凝胶组细胞虽受三维基质表面拓扑结构影响出现荧光强度区域差异,但细胞整体形态及骨架排布模式与对照组具有高度一致性。结果证实,载姜黄素壳聚糖/β-甘油磷酸钠温敏可注射水凝胶不仅能维持大鼠肌腱干细胞正常增殖,还可为细胞提供仿生支架功能,具备作为组织工程载体的应用潜力。 "
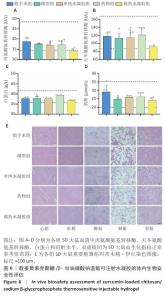
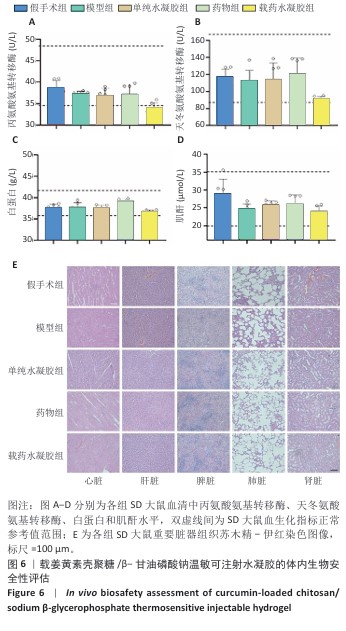
2.6 载姜黄素壳聚糖/β-甘油磷酸钠温敏可注射水凝胶的体内生物安全性评估结果 为验证载姜黄素壳聚糖/β-甘油磷酸钠温敏水凝胶的体内安全性,此次研究在术后8周取材进行血液生化检测及重要脏器病理学分析。血液生化检测结果显示(图6A-D),各组大鼠血清中丙氨酸氨基转移酶、天冬氨酸氨基转移酶、白蛋白和肌酐等关键指标均维持在正常参考区间内[34-36]。重要脏器苏木精-伊红染色显示(图6E),各组大鼠心、肝、脾、肺、肾等实质器官均保持完整组织结构,未见炎性浸润、细胞空泡化或纤维化等病理性改变。结果说明载姜黄素壳聚糖/β-甘油磷酸钠温敏水凝胶在实验剂量范围内的生物相容性良好,血液毒性及器官特异性毒性均符合生物材料临床应用标准,为后续转化研究提供了重要安全性依据。 "
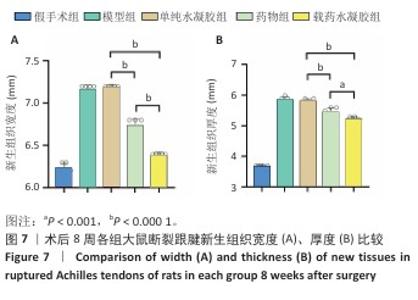
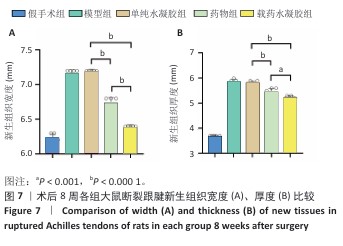
2.7 各组跟腱吻合端形态及腱周组织粘连度评定结果 此次研究构建SD大鼠跟腱离断模型,术后8周参照肌腱粘连分级标准对侧愈合组织进行形态学评估[30-31]。60只大鼠全部进入结果分析。术后8周取材时所有大鼠均未表现运动功能障碍,切口愈合良好无感染征象,未发生缝线反应及再断裂。大体观察显示,各造模组肌腱连续性恢复,可吸收缝线完全降解,模型组与单纯水凝胶组新生腱组织质地坚硬伴炎性增生,与周围组织致密粘连,分离需锐性操作;药物组及其载药水凝胶组织边界较清晰,钝性分离比例显著增加(表2)。各组粘连等级分布具有显著异质性(H=38.633,P < 0.001),值得注意的是,载药水凝胶组粘连等级分布与假手术组比较差异无显著性意义(P=0.283),显著优于其他造模组(P < 0.05)。 跟腱横断面测量显示,模型组、单纯水凝胶组右侧跟腱新生组织宽度、厚度高于假手术组,载药水凝胶组右侧跟腱新生组织宽度、厚度低于其他4组,差异均有显著性意义(图7)。结果表明,载姜黄素壳聚糖/β-甘油磷酸钠温敏水凝胶可显著减轻肌腱愈合过程中的瘢痕增生。 "
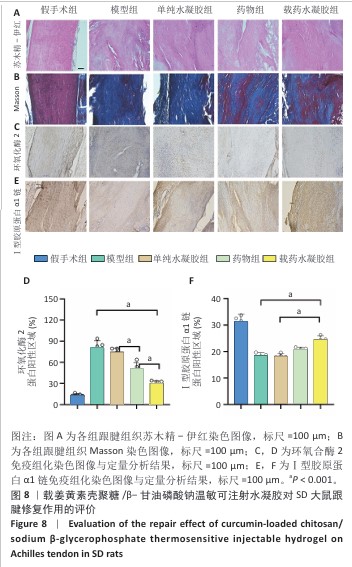
2.8 各组跟腱组织切片分析结果 苏木精-伊红染色显示,各造模组跟腱炎性增生体积均比假手术组大,其中药物组和载药水凝胶组跟腱炎性增生体积明显更薄、范围更小,并且跟腱内胶原纤维排列较模型组和单纯水凝胶组更加有序、紧密,见图8A所示。Masson染色显示,模型组和单纯水凝胶组胶原纤维增生、水肿(苯胺蓝染色区域纹理增粗)最为明显,结构无序、紊乱,载药水凝胶组在造模组中蓝染区域占比最小,纹理相对清晰、有序,见图8B所示。 免疫组化染色显示,假手术组仅在少量细胞中检测到环氧合酶2弱阳性信号,模型组和单纯水凝胶组呈现环氧合酶2强阳性表达,药物组与载药水凝胶组环氧合酶2表达受到抑制,尤以载药水凝胶组抑制效果最为显著,见图8C,D。Ⅰ型胶原作为肌腱力学承载的核心组分,假手术组Ⅰ型胶原呈现典型致密束状分布;横断损伤后,模型组和单纯水凝胶组Ⅰ型胶原沉积显著减少,载药水凝胶组Ⅰ型胶原表达较模型组和单纯水凝胶组显著提升,并且纤维排列方向性明显改善,见图8E,F。"
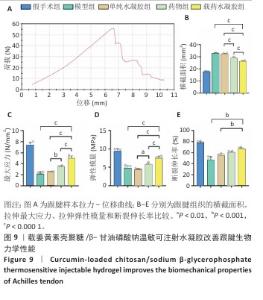
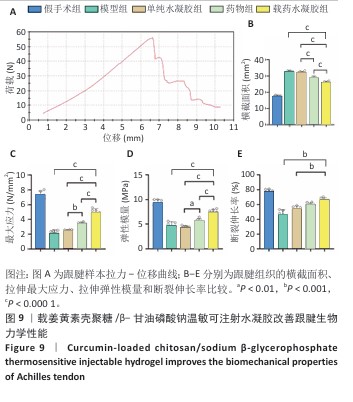
2.9 各组跟腱生物力学分析结果 跟腱组织拉力-位移曲线,如图9A所示。方差分析显示,各组间跟腱组织横截面参数存在显著差异(F=827.2,P < 0.001),载药水凝胶组跟腱组织横截面积低于模型组、单纯水凝胶组、药物组,差异有显著性意义,见图9B所示。载药水凝胶组跟腱组织拉伸最大应力与弹性模量均高于模型组、单纯水凝胶组、药物组,差异有显著性意义,见图9C,D所示。载药水凝胶组跟腱组织拉伸断裂伸长率大于模型组、单纯水凝胶组,差异有显著性意义,见图9E所示。值得注意的是,单纯水凝胶组与模型组跟腱组织弹性模量比较差异无显著性意义(P=0.90),提示单纯材料植入对组织刚度的改善有限,药物与水凝胶联合治疗组的优异力学表现证实姜黄素在提升组织抗拉强度、弹性恢复及延展性方面具有关键作用,进一步结合温敏水凝胶的缓释特性使修复组织的结构完整性显著提升,有效降低修复组织再断裂风险。"
| [1] LUO S, HUANG S, LI S. Review of Preclinical and Clinical Studies Supporting the Role of Polydeoxyribonucleotide in the Treatment of Tendon Disorders. Med Sci Monit. 2025;31:e945743. [2] THOMOPOULOS S, PARKS WC, RIFKIN DB, et al. Mechanisms of tendon injury and repair. J Orthop Res. 2015;33(6):832-839. [3] ZHANG Q, YANG Y, YILDIRIMER L, et al. Advanced technology-driven therapeutic interventions for prevention of tendon adhesion: Design, intrinsic and extrinsic factor considerations. Acta Biomater. 2021;124:15-32. [4] LIU J, DAI G, XIAO L, et al. Tendon-Derived Stem Cell Sheet Promotes Early-Stage Tendon-Bone Healing. J Musculoskelet Neuronal Interact. 2025;25(1):133-141. [5] LIANG Y, XU K, ZHANG P, et al. Quercetin reduces tendon adhesion in rat through suppression of oxidative stress. BMC Musculoskelet Disord. 2020;21(1):608. [6] LEE YW, FU SC, MOK TY, et al. Local administration of Trolox, a vitamin E analog, reduced tendon adhesion in a chicken model of flexor digitorum profundus tendon injury. J Orthop Translat. 2017;10:102-107. [7] ZHANG W, LI X, COMES FRANCHINI M, et al. Controlled release of curcumin from curcumin-loaded nanomicelles to prevent peritendinous adhesion during Achilles tendon healing in rats. Int J Nanomedicine. 2016;11:2873-2881. [8] HE Y, YUE Y, ZHENG X, et al. Curcumin, inflammation, and chronic diseases: how are they linked? Molecules. 2015;20(5):9183-9213. [9] GÜLEÇ A, TÜRK Y, AYDIN BK, et al. Effect of curcumin on tendon healing: an experimental study in a rat model of Achilles tendon injury. Int Orthop. 2018; 42(8):1905-1910. [10] GOEL A, KUNNUMAKKARA AB, AGGARWAL BB. Curcumin as “Curecumin”: from kitchen to clinic. Biochem Pharmacol. 2008;75(4):787-809. [11] ARAÚJO CC, LEON LL. Biological activities of Curcuma longa L. Mem Inst Oswaldo Cruz. 2001;96(5):723-728. [12] AMALRAJ A, PIUS A, GOPI S, et al. Biological activities of curcuminoids, other biomolecules from turmeric and their derivatives - A review. J Tradit Complement Med. 2016;7(2):205-233. [13] GOPI S, JACOB J, VARMA K, et al. Comparative Oral Absorption of Curcumin in a Natural Turmeric Matrix with Two Other Curcumin Formulations: An Open-label Parallel-arm Study. Phytother Res. 2017;31(12):1883-1891. [14] KUNNUMAKKARA AB, BORDOLOI D, PADMAVATHI G, et al. Curcumin, the golden nutraceutical: multitargeting for multiple chronic diseases. Br J Pharmacol. 2016; 174(11):1325-1348. [15] HUSSAIN Z, THU HE, NG SF, et al. Nanoencapsulation, an efficient and promising approach to maximize wound healing efficacy of curcumin: A review of new trends and state-of-the-art. Colloids Surf B Biointerfaces. 2017;150:223-241. [16] KULAC M, AKTAS C, TULUBAS F, et al. The effects of topical treatment with curcumin on burn wound healing in rats. J Mol Histol. 2013;44(1):83-90. [17] MEHRABANI D, FARJAM M, GERAMIZADEH B, et al. The healing effect of curcumin on burn wounds in rat. World J Plast Surg. 2015;4(1):29-35. [18] DUAN Y, CHEN H L, LING M, et al. The Curcumin Analog EF24 Inhibits Proliferation and Invasion of Triple-Negative Breast Cancer Cells by Targeting the Long Noncoding RNA HCG11/Sp1 Axis. Mol Cell Biol. 2022;42(1):e0016321. [19] RAHMANIAN-DEVIN P, BARADARAN RAHIMI V, ASKARI VR. Thermosensitive chitosan-β-glycerophosphate hydrogels as targeted drug delivery systems: An overview on preparation and their applications. Adv Pharmacol Pharm Sci. 2021; 2021:6640893. [20] BAJPAI AK, SHUKLA SK, BHANU S, et al. Responsive polymers in controlled drug delivery. Prog Polym Sci. 2008;33(11):1088-1118. [21] RUEL-GARIEPY E, LEROUX JC. In situ-forming hydrogels—review of temperature-sensitive systems. Eur J Pharm Biopharm. 2004;58(2):409-426. [22] CHO E, QIAO Y, CHEN C, et al. Injectable FHE+BP composites hydrogel with enhanced regenerative capacity of tendon-bone interface for anterior cruciate ligament reconstruction. Front Bioeng Biotechnol. 2023;11:1117090. [23] SATHESH D, SATHESH KUMAR K, DEVADASAN V, et al. Formulation of a thermo-sensitive hydro-gel for ulcerative colitis treatment. Bioinformation. 2022;18(10):925-937. [24] FELT O, BURI P, GURNY R. Chitosan: a unique polysaccharide for drug delivery. Drug Dev Ind Pharm. 1998;24(11):979-993. [25] 王雲蛟.阿司匹林对肌腱干细胞和肌腱病损伤修复的作用和机制研究[D].重庆:中国人民解放军陆军军医大学,2019. [26] 蒋涛.肌腱干细胞来源外泌体在肌腱损伤修复中的作用及其机制研究[D].南京:南京大学,2019. [27] GUO D, LI H, LIU Y, et al. Fibroblast growth factor-2 promotes the function of tendon-derived stem cells in Achilles tendon restoration in an Achilles tendon injury rat model. Biochem Biophys Res Commun. 2020;521(1):91-97. [28] XU K, AL-ANI MK, SUN Y, et al. Platelet-rich plasma activates tendon-derived stem cells to promote regeneration of Achilles tendon rupture in rats. J Tissue Eng Regen Med. 2017;11(4):1173-1184. [29] AL-ANI M, XU K, SUN Y, et al. Study of Bone Marrow Mesenchymal and Tendon-Derived Stem Cells Transplantation on the Regenerating Effect of Achilles Tendon Ruptures in Rats. Stem Cells Int. 2015;2015:984146. [30] 汤锦波.各种伤情下屈肌腱的愈合及粘连形成:肌腱愈合[J].手外科杂志, 1992,8(1):31-35. [31] MULHALL KJ, MCLAUGHLIN R, KAY E, et al. Thermal preconditioning prevents peritendinous adhesions and inflammation. Clin Orthop Relat Res. 2002; 405(405):258-266. [32] INGOLD I, BERNDT C, SCHMITT S, et al. Selenium utilization by GPX4 is required to prevent hydroperoxide-induced ferroptosis. Cell. 2018;172(3):409-422.e21. [33] LU S, SONG Y, LUO R, et al. Ferroportin-Dependent Iron Homeostasis Protects against Oxidative Stress-Induced Nucleus Pulposus Cell Ferroptosis and Ameliorates Intervertebral Disc Degeneration In Vivo. Oxid Med Cell Longev. 2021;2021:6670497. [34] 靳苏香,程洁,钱雯,等.试验相关因素对SD大鼠临床生化学指标的影响[J].毒理学杂志,2019,33(5):366-369,80. [35] WIEDMEYER CE, RUBEN D, FRANKLIN C. Complete blood count, clinical chemistry, and serology profile by using a single tube of whole blood from mice. J Am Assoc Lab Anim Sci. 2007;46(2):59-64. [36] BOEHM O, ZUR B, KOCH A, et al. Clinical chemistry reference database for Wistar rats and C57/BL6 mice. Biol Chem. 2007;388(5):547-554. [37] NOURISSAT G, BERENBAUM F, DUPREZ D. Tendon injury: from biology to tendon repair. Nat Rev Rheumatol. 2015;11(4):223-233. [38] MAGNUSSON SP, LANGBERG H, KJAER M. The pathogenesis of tendinopathy: balancing the response to loading. Nat Rev Rheumatol. 2010;6(5):262-268. [39] CHEN L, LIU JP, TANG KL, et al. Tendon derived stem cells promote platelet-rich plasma healing in collagenase-induced rat achilles tendinopathy. Cell Physiol Biochem. 2014;34(6):2153-2168. [40] DE JONGE S, VAN DEN BERG C, DE VOS RJ, et al. Incidence of midportion Achilles tendinopathy in the general population. Br J Sports Med. 2011;45(13):1026-108. [41] RILEY G. Tendinopathy--from basic science to treatment. Nat Clin Pract Rheumatol. 2008;4(2):82-89. [42] MILLAR NL, SILBERNAGEL KG, THORBORG K, et al. Tendinopathy. Nat Rev Dis Primers. 2021;7(1):1. [43] LIU A, WANG Q, ZHAO Z, et al. Nitric Oxide Nanomotor Driving Exosomes-Loaded Microneedles for Achilles Tendinopathy Healing. ACS Nano. 2021;15(8): 13339-13350. [44] DE MOS M, VAN EL B, DEGROOT J, et al. Achilles tendinosis: changes in biochemical composition and collagen turnover rate. Am J Sports Med. 2007; 35(9):1549-1556. [45] VINCENTI MP. The matrix metalloproteinase (MMP) and tissue inhibitor of metalloproteinase (TIMP) genes. Transcriptional and posttranscriptional regulation, signal transduction and cell-type-specific expression. Methods Mol Biol. 2001;151:121-148. [46] MARCOS RL, ARNOLD G, MAGNENET V, et al. Biomechanical and biochemical protective effect of low-level laser therapy for Achilles tendinitis. J Mech Behav Biomed Mater. 2014;29:272-285. [47] DUAN Y, LI K, WANG H, et al. Preparation and evaluation of curcumin grafted hyaluronic acid modified pullulan polymers as a functional wound dressing material. Carbohydr Polym. 2020;238:116195. [48] ZHOU Y, MING J, DENG M, et al. Chemically modified curcumin (CMC2.24) alleviates osteoarthritis progression by restoring cartilage homeostasis and inhibiting chondrocyte apoptosis via the NF-kappaB/HIF-2alpha axis. J Mol Med (Berl). 2020;98(10):1479-1491. [49] BLAINE TA, KIM YS, VOLOSHIN I, et al. The molecular pathophysiology of subacromial bursitis in rotator cuff disease. J Shoulder Elbow Surg. 2005; 14(1 Suppl S):84S-89S. [50] SHOHRATI M, ABEDI F, BAGHERI M, et al. Effects of curcumin on vascular smooth muscle cells: implications for health and disease. Pharmacol Rep. 2025. doi: 10.1007/s43440-025-00744-3. [51] HAYDEN MS, GHOSH S. NF-κB, the first quarter-century: remarkable progress and outstanding questions. Genes Dev. 2012;26(3):203-234. [52] PASPARAKIS M. Regulation of tissue homeostasis by NF-kappaB signalling: implications for inflammatory diseases. Nat Rev Immunol. 2009;9(11):778-788. [53] LAWRENCE T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb Perspect Biol. 2009;1(6):a001651. [54] YANYAN Y, SEUNG CHEOL K, TAO Y, et al. Functional roles of p38 mitogen-activated protein kinase in macrophage-mediated inflammatory responses. Mediators Inflamm. 2014;2014:352371. [55] RAMAN M, CHEN W, COBB MH. Differential regulation and properties of MAPKs. Oncogene. 2007;26(22):3100-3112. [56] LI J, TIANZHU L, KEXIN L, et al. Inflammation-related signaling pathways in tendinopathy. Open Life Sci. 2023;18(1):20220729. [57] MULLER SA, DURSELEN L, HEISTERBACH P, et al. Effect of a Simple Collagen Type I Sponge for Achilles Tendon Repair in a Rat Model. Am J Sports Med. 2016;44(8):1998-2004. [58] ZHANG K, ZHANG S, LI Q, et al. Effects of celecoxib on proliferation and tenocytic differentiation of tendon-derived stem cells. Biochem Biophys Res Commun. 2014;450(1):762-766. [59] ABDOLLAHIYAN P, OROOJALIAN F, MOKHTARZADEH A. The triad of nanotechnology, cell signalling, and scaffold implantation for the successful repair of damaged organs: An overview on soft-tissue engineering. J Control Release. 2021;332:460-492. [60] VERONESI F, BORSARI V, CONTARTESE D, et al. The clinical strategies for tendon repair with biomaterials: A review on rotator cuff and Achilles tendons. J Biomed Mater Res B Appl Biomater. 2020;108(5):1826-1843. [61] ABDUL ALIM M, DOMEIJ-ARVERUD E, NILSSON G, et al. Achilles tendon rupture healing is enhanced by intermittent pneumatic compression upregulating collagen type I synthesis. Knee Surg Sports Traumatol Arthrosc. 2018;26: 2021-2029. [62] YAO L, BESTWICK CS, BESTWICK L, et al. Non-immortalized human tenocyte cultures as a vehicle for understanding cellular aspects to tendinopathy. Transl Med UniSa. 2011;1:173. [63] MAO Z, FAN B, WANG X, et al. A Systematic Review of Tissue Engineering Scaffold in Tendon Bone Healing in vivo. Front Bioeng Biotechnol. 2021;9:621483. [64] YANG L, TANG C, CHEN Y, et al. Pharmacological Inhibition of Rac1 Activity Prevents Pathological Calcification and Enhances Tendon Regeneration. ACS Biomater Sci Eng. 2019;5(7):3511-3522. |
| [1] | Yang Xuetao, Zhu Menghan, Zhang Chenxi, Sun Yimin, Ye Ling. Applications and limitations of antioxidant nanomaterials in oral cavity [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2044-2053. |
| [2] | Liu Yang, Liu Donghui , Xu Lei, Zhan Xu, Sun Haobo, Kang Kai. Role and trend of stimuli-responsive injectable hydrogels in precise myocardial infarction therapy [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2072-2080. |
| [3] |
Dong Chunyang, Zhou Tianen, Mo Mengxue, Lyu Wenquan, Gao Ming, Zhu Ruikai, Gao Zhiwei.
Action mechanism of metformin combined with Eomecon chionantha Hance dressing in treatment of deep second-degree burn wounds#br#
#br#
[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 2001-2013.
|
| [4] | Zhou Xiaohui, Wang Siyi, Zhou Qiyun, He Zhao, Jia Yujuan, Wang Yuanbin, Ma Jianwu, Chen Gang, Zheng Feng, Chu Genglei. Nanohydroxyapatite-polyether carbonate urethane electrospinning membrane promotes bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5134-5142. |
| [5] | He Zhenzhen, Huang Hanji, Wang Jiawei, Xie Qingtiao, Jiang Xianfang. Role of bioscaffolds in the repair of inflammation-driven bone and cartilage destruction and structural damage in temporomandibular joint [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5312-5320. |
| [6] | Wang Jieyan, Yao Jiayi, Xin Yingtong, Zhang Xinwen, Li Riwang, Liu Dahai. Chitosan hydrogel drug delivery system is a safer and more effective solution for treating oral ulcers [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5331-5340. |
| [7] | Liu Shuting, Qiu Muen, Li Wei. Application and development trend of hydrogels in ophthalmic diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5349-5360. |
| [8] | Bai Xiangyu, Huo Feng, Hao Yan, Wang Zecheng, Guo Xiaoyu. Platelet-derived growth factor BB-loaded chitosan/reduced graphene oxide scaffold for repairing alveolar bone defects [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 329-337. |
| [9] | Dong Chao, Zhao Mohan, Liu Yunan, Yang Zeli, Chen Leqin, Wang Lanfang. Effects of magnetic nano-drug carriers on exercise-induced muscle injury and inflammatory response in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 345-353. |
| [10] | Wang Yuhang, Zhang Han, Zhang Chaojing, Kou Xurong, Jing Tongtong, Lin Rimei, Liu Xinyu, Lou Shilei, Yan Hui, Sun Cong. Curcumin extraction and preparation and optimization of curcumin nanoparticles [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 362-374. |
| [11] | Liu Xiaohong, Zhao Tian, Mu Yunping, Feng Wenjin, Lyu Cunsheng, Zhang Zhiyong, Zhao Zijian, Li Fanghong. Acellular dermal matrix hydrogel promotes skin wound healing in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 395-403. |
| [12] | Yuan Qian, Zhang Hao, Pang Jie. Characterization and biological properties of naringin-loaded chitosan/beta-tricalcium phosphate scaffold [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 424-432. |
| [13] | Jiang Kan, Alimujiang·Abudourousuli, Shalayiding·Aierxiding, Aikebaierjiang·Aisaiti, Kutiluke·Shoukeer, Aikeremujiang·Muheremu. Biomaterials and bone regeneration: research hotspots and analysis of 500 influential papers [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(2): 528-536. |
| [14] | Shi Xiaonan, Wu Xuan, Zhang Daxu, Hu Jingjing, Zheng Yazhe, Liu Yutong, Zhao Shuo, Li Weilong, Ye Shujun, Wang Jingyi, Yan Li. Preparation and characterization of 3D printed microstructured silk fibroin scaffold for liver injury repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3618-3625. |
| [15] | Li Liang, Yang Han, Suo Hairui, Guan Lu, Wang Zhenlin. 3D printed methacrylated gelatin/chitosan scaffolds: evaluation of antibacterial, mechanical properties and cytocompatibility [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(14): 3636-3642. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||