Chinese Journal of Tissue Engineering Research ›› 2026, Vol. 30 ›› Issue (19): 5033-5039.doi: 10.12307/2026.244
Previous Articles Next Articles
Application and prospects of precision-medicine-driven breast cancer organoids in therapeutic drug discovery
Mou Jiancheng1, Luo Jie1, Liu Haotian1, Yang Zhuotao1, Mu Yuxiao1, Qian Da2, Meng Xuli1
- 1Department of Breast Surgery, Affiliated Hospital of Hangzhou Medical College/Zhejiang Provincial People's Hospital, Hangzhou 310000, Zhejiang Province, China; 2Department of Plastic Surgery, Changshu Hospital Affiliated to Soochow University/Changshu First People's Hospital, Changshu 215500, Jiangsu Province, China
-
Received:2025-10-15Accepted:2025-11-18Online:2026-07-08Published:2026-02-24 -
Contact:Qian Da, MD, Attending physician, Department of Plastic Surgery, Changshu Hospital Affiliated to Soochow University/Changshu First People's Hospital, Changshu 215500, Jiangsu Province, China; Corresponding author: Meng Xuli, MS, Professor, Department of Breast Surgery, Affiliated Hospital of Hangzhou Medical College/Zhejiang Provincial People's Hospital, Hangzhou 310000, Zhejiang Province, China -
About author:Mou Jiancheng, MS candidate, Department of Breast Surgery, Affiliated Hospital of Hangzhou Medical College/Zhejiang Provincial People's Hospital, Hangzhou 310000, Zhejiang Province, China -
Supported by:National Natural Science Foundation of China, No. 82404685 (to QD); Zhejiang Provincial Department of Science and Technology 2023 "Pioneer" and "Leader" Projects, No. 2023C03044 (to MXL)
CLC Number:
Cite this article
Mou Jiancheng, Luo Jie, Liu Haotian, Yang Zhuotao, Mu Yuxiao, Qian Da, Meng Xuli. Application and prospects of precision-medicine-driven breast cancer organoids in therapeutic drug discovery[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(19): 5033-5039.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
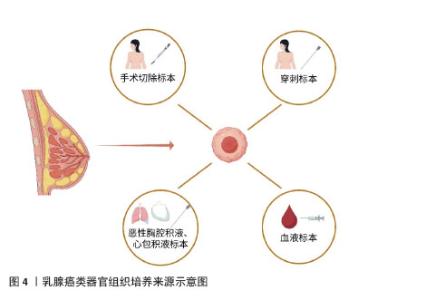
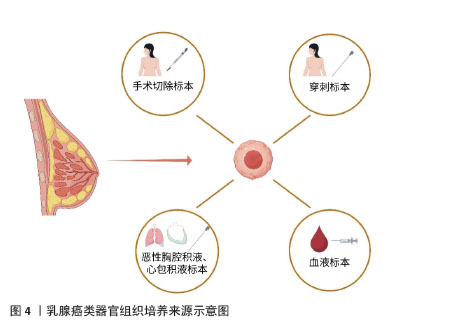
2.1 乳腺癌研究模型的创新 以往乳腺癌的研究通常依赖已构建的细胞系来探究相关药物的作用以及细胞内相关信号通路的传递与表达。虽然已有的细胞系也涵盖了目前大部分乳腺癌的各个分子分型,包括三阴性的MDA-MB-231、A549、激素受体阳性的MCF-7细胞系等,它们在一定程度上体现了肿瘤的异质性,但是围绕细胞系的探索始终不能体现肿瘤微环境、不同细胞之间的通讯等因素对肿瘤细胞带来的影响。此外,还有一种实验模型是将患者来源的肿瘤组织移植到免疫缺陷的小鼠上建立的,虽然可以在一定程度上维持肿瘤细胞的遗传特性,但其个体差异的存在以及构建的高成本仍使得相关的研究成果难以应用于临床实践。 乳腺癌类器官是一种模拟了与乳腺癌组织有类似空间结构、具有相似功能的三维立体结构,是由相应干细胞或对应的癌变组织在特定的培养条件下发展而来。它不仅可以模拟肿瘤微环境、展现细胞间的交流互动,并且构建成功后生长速度快,保持肿瘤较高还原度的临床特征。CHEN等[13]从耐药的乳腺癌患者中获得肿瘤组织并培养成类器官模型,他们发现该类器官模型可以很好地还原患者对于某种药物的临床反应,并且准确地体现了肿瘤组织的组织病理学和遗传学特征。此外,乳腺癌类器官相对的高经济性使得它成为筛选药物敏感性的实用模型之一。 现有的乳腺癌类器官培养,通常是先将患者的肿瘤组织用胰酶消化成单个或成团的肿瘤细胞,然后种植于基底膜提取物中,加入含有多种生长因子的特定培养基,在特殊的培养条件下构建而成。乳腺癌类器官的组织来源种类众多,包括手术或穿刺标本、恶性胸腔积液等,共培养的免疫细胞也可能来源于血液标本等,见图4[12,14]。传统的类器官培养往往会受到患者来源肿瘤组织的批次间差异以及细胞外基质稳定性的影响,面临可重复性低、失败风险高等困难。探索新型的标准化内容以及稳定的类器官培养体系将是未来类器官培养领域的热点。AREF等[15]运用3D微流控系统,通过不同大小的过滤器获得分别为S1(> 100 μm)、S2(40–100 μm)和 S3(< 40 μm)3个肿瘤成分,而后收集富含S2组分的肿瘤组织与胶原蛋白混合,加入到微流控系统中。这种培养方法可以保留天然的淋巴细胞和骨髓细胞,模拟复杂的肿瘤微环境[15]。此外,HERNANDEZ-GORDILLO带领团队[16]将聚乙二醇水凝胶与基质金属蛋白酶可降解肽交联,并结合α2β1整合素结合肽,它们的组合对类器官培养与生长的支持完全可以与基质胶媲美甚至超越。创新的共培养方式有助于更好地模拟体内肿瘤生长微环境,研究细胞间的相互作用及其对肿瘤生长、侵袭和治疗反应的影响。 综上,乳腺癌类器官作为乳腺癌研究方面的新模型,其原始肿瘤细胞来源的多样性,培养体系的不断创新,使得它越来越成为乳腺癌研究领域最具潜力的工具之一,同时其对药物敏感性筛选的有效性也让它改善临床上乳腺癌患者治疗耐药成为可能。 "
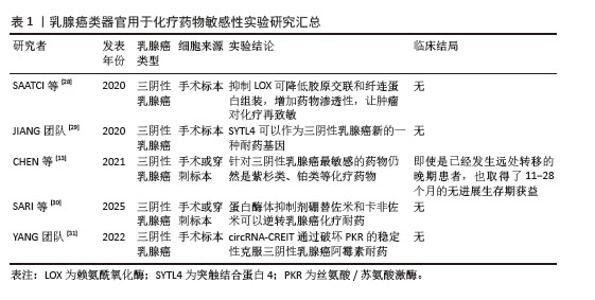
2.2 乳腺癌类器官基于精准医疗背景下的药物筛选 目前乳腺癌的精准治疗主要基于其肿瘤的分子分型、生物标志物以及个体化差异,为患者制定个性化的治疗方案。一般将乳腺癌分为HER2阳性型、激素受体阳性型以及三阴性乳腺癌(triple negative breast cancer,TNBC)[17]。针对HER2阳性型乳腺癌,通常使用曲妥珠单抗/帕妥珠单抗靶向HER2有效治疗这类患者。针对雌激素受体阳性型(HR+)患者,内分泌治疗常用于治疗该类患者,包括芳香化酶抑制剂、雌激素受体调节剂等。目前针对高复发风险的HR+型乳腺癌患者,CDK4/6抑制剂(如瑞博西利、阿贝西利和达拉西利)联合内分泌治疗在改善此类患者的无病生存期取得了令人惊喜的效果[18-19]。三阴性乳腺癌目前尚无针对性的治疗策略,化疗往往是临床上采取的相对有效的治疗方法,但是三阴性乳腺癌对化疗的敏感性也因人而异,这也导致患者的预后相对较差[20]。学者们为提高三阴性乳腺癌的疗效做出了巨大的努力,上海复旦大学附属肿瘤医院邵志敏教授团队基于三阴性乳腺癌基因组与转录组景观,提出了三阴性乳腺癌的“复旦分型”,针对不同的亚型,也提出了不同的针对性治疗策略[21]。免疫治疗,特别是针对程序性细胞死亡蛋白1/程序性细胞死亡配体1的治疗,目前在三阴性乳腺癌中取得了一定的疗效。在KEYNOTE-522试验中,帕博利珠单抗联合新辅助化疗显著提高了病理完全缓解率(64.8% vs. 51.2%),并延长了无事件生存期[22]。但无论是化疗,亦或者是免疫治疗,许多患者往往因无法耐受药物副反应而中止治疗,导致三阴性乳腺癌类型乳腺癌患者5年生存率仍然偏低。 如何选择针对肿瘤异质性如此显著的乳腺癌患者治疗药物,以及判断相关药物的治疗毒性,仍旧是目前乳腺癌治疗领域等待突破的关键问题之一。类器官作为一种高效的药物敏感性筛选模型能在很大程度上改变这种局面,其生物学信息的高保真性、与其亲本肿瘤高度一致的药物反应性,进一步指导临床药物使用,更好地推动对乳腺癌的精准治疗。CHEN团队[13]从99个样本中建立了132个乳腺癌类器官模型,总体成功率为75%,随后使用这些模型进行药物敏感性测试,最后的结果显示从类器官模型上获得的药物反应在很大程度上反映原发性肿瘤的反应。WU等[23]建立的70余例乳腺癌类器官模型检测药物疗效也取得了近似的结果,为后续乳腺癌的精准治疗提供了依据。此外,可以从药物筛选单独角度出发,利用类器官技术观察类器官培养上清液中循环肿瘤DNA(ctDNA)变异等位基因频率的动态变化并且联合药敏性实验多维度指导治疗策略[24]。 2.2.1 化疗药物 化疗是目前乳腺癌治疗的常用手段,尤其是针对三阴性乳腺癌,化疗是其治疗策略的核心[25]。乳腺癌化疗通常有蒽环类、紫杉类以及铂类药物,各类化疗药物往往引起各不相同的副反应,比如说蒽环类药物不可逆的心脏毒性,以及紫杉类药物的神经毒性等等,都在很大程度上威胁患者的身心健康,影响患者的生活质量,导致患者依从性差。如何精准筛选药敏性强的化疗药物,在保证杀伤效果的同时,尽可能减低化疗的毒副反应,是目前制定化疗策略的关键。基于类器官检测药物敏感性的结果,修正具体的临床用药,选择最适合患者的化疗方案,获得临床最大收益。同时,使用类器官这个工具也可以评估化疗联合其他药物作用于不同类型乳腺癌的获益。 针对三阴性乳腺癌患者,虽然化疗是目前相对有效的治疗手段,但达到病理完全缓解的三阴性乳腺癌患者仍然只有30%-40%,这与三阴性乳腺癌的肿瘤特性所导致的化疗耐药密切相关[26]。学者们利用类器官寻找针对三阴性乳腺癌高药敏性的化疗药物,亦或是基于类器官寻找化疗联合其他药物的三阴性乳腺癌治疗策略仍然是目前研究的热点[27]。SAATCI等[28]发现在阿霉素耐药的三阴性乳腺癌类器官中,LOX抑制剂与阿霉素的组合显著减小了类器官大小,抑制LOX可降低胶原交联和纤连蛋白组装,增加药物渗透性,并下调ITGA5/FN1表达,导致抑制FAK/Src信号传导,诱导细胞凋亡和对化疗的再致敏。JIANG团队[29]用患者来源三阴性乳腺癌类器官模型鉴定了SYTL4可以作为三阴性乳腺癌新的一种耐药基因。在CHEN等[13]的研究中,他们建立了13例类器官样本,其中包括8例三阴性乳腺癌,应用类器官进行药敏实验发现针对三阴性乳腺癌最敏感的药物仍然是紫杉类、铂类等化疗药物,将药敏实验结果应用于临床实践,同样也获得了与预期一样不错的疗效,即使是已经发生远处转移的晚期患者,也取得了11-28个月的无进展生存期的获益。利用乳腺癌类器官检测化疗药物敏感性的相关文献结论总结如表1所示。 "


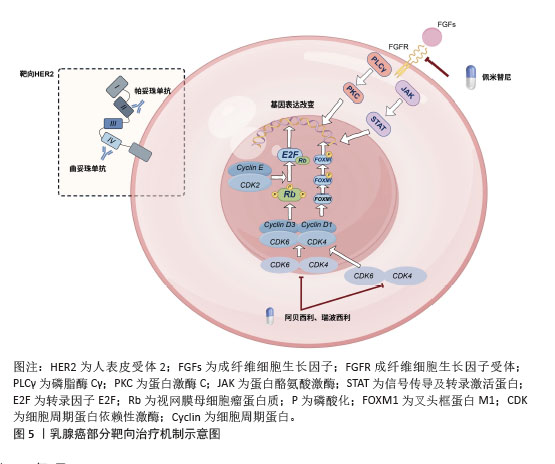
传统的乳腺癌靶向治疗主要在HER2阳性型乳腺癌患者中开展,而HER2阳性在总的乳腺癌患者中约占20%,相当大的一部分患者可以在针对HER2的靶向治疗中获益[32]。针对HER2的靶向药物种类也在不停地更新换代,从刚起步时曲妥珠、帕妥珠单抗的应用,到现在针对转移性的HER2阳性乳腺癌患者仍有T-DM1应用于靶向HER2治疗,为此类乳腺癌患者带来了福音。除了针对HER2的靶向治疗,核糖聚合酶(PARP)、细胞周期蛋白依赖性激酶(CDK4/6)、成纤维细胞生长因子受体等也是公认的治疗靶点,一直是乳腺癌靶向治疗药物开发的主要重点[11,33]。 类器官模型是目前相对有效、使用较多且准确率较高的验证靶向药物疗效的工具。NAVARRO-YEPES等[34]使用类器官模型研究两种CDK4/6抑制剂在激素受体阳性乳腺癌中的有效性,得出一个有趣的结论,对帕博西利耐药的激素受体阳性乳腺癌仍可对阿贝西利敏感;细胞周期蛋白E、白细胞介素6/JAK/STAT3和自噬途径的上调都与帕博西利耐药有关[35-36]。然而由于阿贝西利的多激酶抑制活性使得对帕博西利耐药的激素受体阳性乳腺癌类器官及PDX模型仍对阿贝西利有反应,且其疗效在临床患者的用药实践中得到了验证[36]。同时有研究利用类器官模型验证得出结论,表皮生长因子受体和HER2通路的表达上调可能与ER+的乳腺癌对CDK4/6抑制剂的耐药相关[37]。此外,还有一篇类器官的研究中报道了他汀类药物与AKT靶向药物联用的协同作用可以诱导PDO模型中体外三阴性乳腺癌的细胞毒性,为三阴性乳腺癌靶向治疗提供了新的治疗策略[38]。在SHAO教授团队[39]的研究中,类器官技术被充分应用于乳腺癌药敏筛选领域,他通过对HER2阳性型的乳腺癌患者进行多组学分析,根据其分子特征,将HER2阳性型乳腺癌分为4类,经典HER2(HER2-CLA)亚型、免疫调节(HER2-IM)亚型、管腔样(HER2-LUM)亚型和基底/间充质样(HER2-BM)亚型。SHAO教授团队获取并建立多个乳腺癌患者组织来源的肿瘤类器官,验证了肿瘤异质性的存在以及其对不同药物敏感性的特点,比如说管腔样(HER2-LUM)亚型的HER2阳性乳腺癌与激素受体阳性型乳腺癌的分子分型类似,在类器官验证的基础上,他们得出结论,内分泌治疗对该类型HER2阳性乳腺癌患者有效。总的来说,类器官在靶向药物在乳腺癌中的疗效验证中发挥了重要作用[40],对不同类型的乳腺癌患者个性化药物选择提供了新的策略,同时也为类器官技术在乳腺癌治疗领域带来了更广泛、更实用的应用。利用乳腺癌类器官检测靶向药物敏感性的相关文献结论总结如表2所示。 "

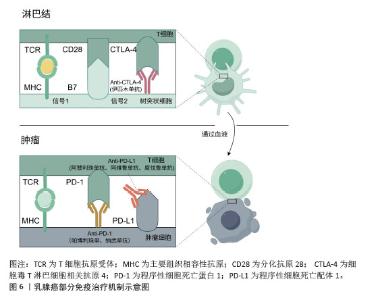
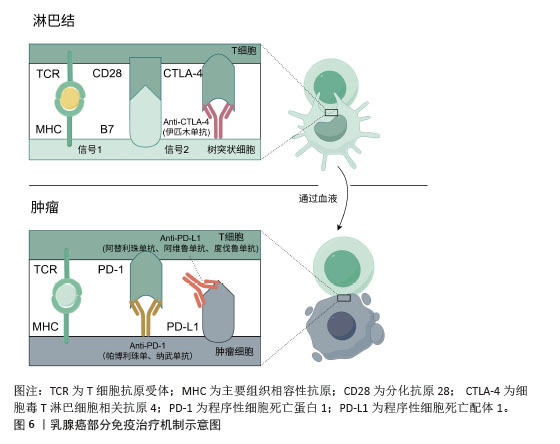
2.2.3 免疫治疗药物 免疫治疗是在癌症治疗领域新兴的治疗手段,有望在癌症治疗上取得突破性进展。与其他癌症免疫治疗类似,程序性细胞死亡蛋白1与程序性细胞死亡配体1的单克隆抗体也是乳腺癌免疫治疗的主要治疗药物之一[44],见图6。然而免疫治疗在目前的乳腺癌治疗临床实践中获得的收益有限,这可能与诱导有效的免疫细胞难度较大有关,后续寻找更有效的免疫治疗标志物可能是预测免疫治疗疗效的手段之一。传统的细胞实验往往难以模拟肿瘤免疫微环境,而动物实验模型往往与人类的免疫系统相比,存在较大差异,两者均难以真正评估免疫治疗在乳腺癌中的疗效,类器官技术较高程度地还原了肿瘤微环境,包括免疫微环境,在乳腺癌免疫治疗过程中发挥的作用。类器官技术为探究免疫治疗药物在乳腺癌治疗中的敏感性以及后续相关药物的开发与应用于临床提供了重要保障[45]。 "

截至目前,三阴性乳腺癌仍缺乏针对性的治疗方案,其中位生存期仍然较短,免疫治疗的兴起为三阴性乳腺癌的精准治疗提供了新的策略。DIVOUX等[46]使用患者肿瘤来源类器官评估三阴性乳腺癌对免疫治疗的疗效。 JIANG教授团队[47]发现鸟苷二磷酸-甘露糖是一种重要的高同源重组修复缺陷相关代谢物,并提出了一种有前途的治疗策略,使用鸟苷二磷酸-甘露糖、PARP抑制剂和抗程序性细胞死亡蛋白1免疫疗法的组合来治疗低同源重组修复缺陷评分三阴性乳腺癌,并且该治疗方案在患者类器官模型中取得了不错的疗效。此外,XU等[44]使用类器官模型验证葫芦素通过激活cGAS-STING 先天免疫途径,与程序性细胞死亡蛋白1阻断剂的联合给药显现出显著的协同功效。 使用类器官模型探索免疫治疗在乳腺癌治疗中的疗效,无论是其相关的机制研究,亦或是其它药物与免疫检查点单克隆抗体联用的疗效,均取得了一定的成效。发现更多的免疫标志物,亦或是多种药物联用,或许可以是未来乳腺癌精准治疗的发展与研究方向之一。利用乳腺癌类器官检测免疫治疗药物敏感性的相关文献结论总结如表3所示。 "

| [1] GIAQUINTO AN, SUNG H, MILLER KD, et al. Breast Cancer Statistics,2022. CA Cancer J Clin. 2022;72(6):524-541. [2] BODAI BI, TUSO P. Breast cancer survivorship: a comprehensive review of long-term medical issues and lifestyle recommendations. Perm J. 2015;19(2):48-79. [3] BAE SY, KIM S, LEE JH, et al. Poor prognosis of single hormone receptor- positive breast cancer: similar outcome as triple-negative breast cancer.BMC Cancer. 2015;15:138. [4] 刘继伟,刘伟慈,毛文君.肿瘤类器官在精准肺癌药物筛选中的研发与进展[J].中国组织工程研究,2024,28(31):5078-5084. [5] DESANTIS CE, MA J, GAUDET MM, et al.Breast cancer statistics,2019. CA Cancer J Clin. 2019; 69(6):438-451. [6] ROPER J, TAMMELA T, CETINBAS NM, et al.In vivo genome editing and organoid transplantation models of colorectal cancer and metastasis. Nat Biotechnol. 2017;35(6):569-576. [7] BYRNE AT, ALFÉREZ DG, AMANT F, et al.Interrogating open issues in cancer precision medicine with patient-derived xenografts. Nat Rev Cancer. 2017;17(4):254-268. [8] SACHS N, DE LIGT J, KOPPER O, et al. A Living Biobank of Breast Cancer Organoids Captures Disease Heterogeneity. Cell. 2018;172(1-2): 373-386.e10. [9] LEE SH, HU W, MATULAY JT, et al. Tumor Evolution and Drug Response in Patient-Derived Organoid Models of Bladder Cancer. Cell. 2018;173(2): 515-528.e17. [10] HIDALGO M, AMANT F, BIANKIN AV, et al. Patient-derived xenograft models: an emerging platform for translational cancer research. Cancer Discov. 2014;4(9):998-1013. [11] YE F, DEWANJEE S, LI Y, et al. Advancements in clinical aspects of targeted therapy and immunotherapy in breast cancer. Mol Cancer. 2023;22(1):105. [12] GOLDHAMMER N, KIM J, TIMMERMANS-WIELENGA V, et al. Characterization of organoid cultured human breast cancer. Breast Cancer Res. 2019;21(1):141. [13] CHEN P, ZHANG X, DING R, et al. Patient-Derived Organoids Can Guide Personalized-Therapies for Patients with Advanced Breast Cancer. Adv Sci (Weinh). 2021;8(22):e2101176. [14] PRANAV P, PALANIYANDI T, BASKAR G, et al. Gene expressions and their significance in organoid cultures obtained from breast cancer patient-derived biopsies. Acta Histochem. 2022; 124(5):151910. [15] AREF AR, CAMPISI M, IVANOVA E, et al. 3D microfluidic ex vivo culture of organotypic tumor spheroids to model immune checkpoint blockade.Lab Chip. 2018;18(20):3129-3143. [16] HERNANDEZ-GORDILLO V, KASSIS T, LAMPEJO A, et al. Fully synthetic matrices for in vitro culture of primary human intestinal enteroids and endometrial organoids. Biomaterials. 2020; 254:120125. [17] PEROU CM, SØRLIE T, EISEN MB, et al. Molecular portraits of human breast tumours. Nature. 2000; 406(6797):747-752. [18] SLAMON D, LIPATOV O, NOWECKI Z, et al. Ribociclib plus Endocrine Therapy in Early Breast Cancer. N Engl J Med. 2024;390(12):1080-1091. [19] OLIVEIRA M, POMINCHUK D, NOWECKI Z, et al. Camizestrant, a next-generation oral SERD, versus fulvestrant in post-menopausal women with oestrogen receptor-positive, HER2-negative advanced breast cancer (SERENA-2): a multi-dose, open-label, randomised, phase 2 trial. Lancet Oncol. 2024;25(11):1424-1439. [20] BIANCHINI G, DE ANGELIS C, LICATA L, et al.Treatment landscape of triple-negative breast cancer - expanded options, evolving needs. Nat Rev Clin Oncol. 2022;19(2):91-113. [21] JIANG YZ, MA D, SUO C, et al. Genomic and Transcriptomic Landscape of Triple-Negative Breast Cancers: Subtypes and Treatment Strategies. Cancer Cell. 2019;35(3):428-440.e5. [22] SCHMID P, CORTES J, PUSZTAI L, et al. Pembrolizumab for Early Triple-Negative Breast Cancer. N Engl J Med. 2020;382(9):810-821. [23] WU H, WANG W, ZHANG Y, et al. Establishment of patient-derived organoids for guiding personalized therapies in breast cancer patients. Int J Cancer. 2024;155(2):324-338. [24] LIU J, FANG F, YUAN W, et al.Integrating Circulating Tumor DNA Dynamics and Sequential Organoid Drug Screening Enhances Breast Cancer Treatment Efficacy. Cancer Res. 2025;24.doi:10.1158/0008-5472.CAN-25-0608 [25] MAYER EL, BURSTEIN HJ. Chemotherapy for Triple-Negative Breast Cancer: Is More Better?. J Clin Oncol. 2016;34(28):3369-3371. [26] LIEDTKE C, MAZOUNI C,HESS KR,et al.Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26(8):1275-1281. [27] PSILOPATIS I, MANTZARI A, VRETTOU K, et al. The Role of Patient-Derived Organoids in Triple-Negative Breast Cancer Drug Screening. Biomedicines. 2023;11(3):773. [28] SAATCI O, KAYMAK A, RAZA U, et al. Targeting lysyl oxidase (LOX) overcomes chemotherapy resistance in triple negative breast cancer. Nat Commun. 2020;11(1):2416. [29] LIU XY, JIANG W, MA D, et al. SYTL4 downregulates microtubule stability and confers paclitaxel resistance in triple-negative breast cancer.Theranostics. 2020;10(24):10940-10956. [30] SARI AIP, ROYTRAKUL S, CHITTAVANICH P, et al. Drug repurposing identifies proteasome inhibitors as antiproliferative agents counteracting inflammation-driven chemoresistance in triple-negative breast cancer organoids. Biomed Pharmacother. 2025;190:118359. [31] WANG X, CHEN T, LI C, et al. CircRNA-CREIT inhibits stress granule assembly and overcomes doxorubicin resistance in TNBC by destabilizing PKR. J Hematol Oncol. 2022;15(1):122. [32] CRONIN KA, HARLAN LC, DODD KW, et al. Population-based estimate of the prevalence of HER-2 positive breast cancer tumors for early stage patients in the US. Cancer Invest. 2010; 28(9):963-968. [33] 黄慧琦,伍秋苑,张昆,等.川楝素联合奥拉帕尼在三阴性乳腺癌中的抗肿瘤机制研究[J].天津医药,2025,53(9):897-903. [34] NAVARRO-YEPES J, KETTNER NM, RAO X, et al. Abemaciclib Is Effective in Palbociclib-Resistant Hormone Receptor-Positive Metastatic Breast Cancers. Cancer Res. 2023;83(19):3264-3283. [35] TURNER NC, LIU Y, ZHU Z, et al. Cyclin E1 Expression and Palbociclib Efficacy in Previously Treated Hormone Receptor-Positive Metastatic Breast Cancer. J Clin Oncol. 2019;37(14):1169-1178. [36] KETTNER NM, VIJAYARAGHAVAN S, DURAK MG, et al. Combined Inhibition of STAT3 and DNA Repair in Palbociclib-Resistant ER-Positive Breast Cancer. Clin Cancer Res. 2019;25(13):3996-4013. [37] BELLI S, ESPOSITO D, ASCIONE CM, et al. EGFR and HER2 hyper-activation mediates resistance to endocrine therapy and CDK4/6 inhibitors in ER+ breast cancer. Cancer Lett. 2024;593:216968. [38] HILLIS AL, MARTIN TD, MANCHESTER HE, et al. Targeting Cholesterol Biosynthesis with Statins Synergizes with AKT Inhibitors in Triple-Negative Breast Cancer. Cancer Res. 2024;84(19): 3250-3266. [39] LI YW, DAI LJ, WU XR, et al. Molecular Characterization and Classification of HER2-Positive Breast Cancer Inform Tailored Therapeutic Strategies. Cancer Res. 2024;84(21):3669-3683. [40] CUI Y, RAN R, DA Y, et al. The combination of breast cancer PDO and mini-PDX platform for drug screening and individualized treatment. J Cell Mol Med. 2024;28(9):e18374. [41] TAGHIZADEH-TABARSI R, AKBARI-BIRGANI S, AMJADI M, et al. Aptamer-guided graphene oxide quantum dots for targeted suicide gene therapy in an organoid model of luminal breast cancer. Sci Rep. 2024;14(1):24104. [42] MADORSKY ROWDO FP, XIAO G, KHRAMTSOVA GF, et al. Patient-derived tumor organoids with p53 mutations, and not wild-type p53, are sensitive to synergistic combination PARP inhibitor treatment. Cancer Lett. 2024;584:216608. [43] LIU Z, SHI Y, LV L, et al. Small Molecular Inhibitors Reverse Cancer Metastasis by Blockading Oncogenic PITPNM3. Adv Sci (Weinh). 2022;9(35): e2204649. [44] XU Q, JIANG Z, PAN Y, et al.Cucurbitacin B stimulates PD-1 immunotherapy response in malignant breast cancer by covalent targeting MTCH2. Phytomedicine. 2025;145:157017. [45] YUKI K, CHENG N, NAKANO M, et al. Organoid Models of Tumor Immunology. Trends Immunol. 2020;41(8):652-664. [46] DIVOUX J, FLORENT R, JACOBS M, et al. The TRIPLEX study: use of patient-derived tumor organoids as an innovative tool for precision medicine in triple-negative breast cancer. BMC Cancer. 2023;23(1):883. [47] DING JH, XIAO Y, YANG F, et al. Guanosine diphosphate-mannose suppresses homologous recombination repair and potentiates antitumor immunity in triple-negative breast cancer. Sci Transl Med. 2024;16(728):eadg7740. [48] HUANG L, RONG Y, TANG X, et al. Engineered exosomes as an in situ DC-primed vaccine to boost antitumor immunity in breast cancer. Mol Cancer. 2022;21(1):45. [49] LIN YZ, LIU CH, WU WR, et al. Memory-promoting function of miR-379-5p attenuates CD8+ T cell exhaustion by targeting immune checkpoints. J Immunother Cancer. 2025;13(4):e010363. [50] DING JH, XIAO Y, YANG F, et al. Guanosine diphosphate-mannose suppresses homologous recombination repair and potentiates antitumor immunity in triple-negative breast cancer. Sci Transl Med. 2024;16(728):eadg7740. [51] LI X, PAN B, SONG X, et al. Breast cancer organoids from a patient with giant papillary carcinoma as a high-fidelity model. Cancer Cell Int. 2020;20:86. [52] SEGATTO I, MATTEVI MC, RAMPIONI VINCIGUERRA GL, et al.A comprehensive luminal breast cancer patient-derived xenografts (PDX) library to capture tumor heterogeneity and explore the mechanisms of resistance to CDK4/6 inhibitors. J Pathol. 2024;264(4):434-447. [53] 谭景云,兰坚强,白跃宗,等.2024年度肿瘤类器官研究进展[J].肿瘤综合治疗电子杂志, 2025,11(2):77-84. [54] 中国抗癌协会肿瘤多学科诊疗专业委员会,中国抗癌协会肿瘤内分泌专业委员会,赵冰,等.肿瘤类器官诊治平台的质量控制标准中国专家共识(2022年版)[J].中国癌症杂志,2022, 32(7):657-668. [55] MAZZUCCHELLI S, PICCOTTI F, ALLEVI R, et al. Establishment and Morphological Characterization of Patient-Derived Organoids from Breast Cancer. Biol Proced Online. 2019;21:12. [56] MOU J, LI C, ZHENG Q, et al. Research progress in tumor angiogenesis and drug resistance in breast cancer. Cancer Biol Med. 2024;21(7):571-585. [57] DEKKERS JF, VAN VLIET EJ, SACHS N, et al. Long-term culture, genetic manipulation and xenotransplantation of human normal and breast cancer organoids. Nat Protoc. 2021;16(4):1936-1965. [58] MAULANA TI, TEUFEL C, CIPRIANO M, et al. Breast cancer-on-chip for patient-specific efficacy and safety testing of CAR-T cells. Cell Stem Cell. 2024;31(7):989-1002.e9. |
| [1] | Min Changqin, Huang Ying. Construction of pH/near-infrared laser stimuli-responsive drug delivery system and its application in treatment of oral squamous cell carcinoma [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(8): 1940-1951. |
| [2] | Zhang Qian, Huang Dongfeng. Weighted gene co-expression network analysis combined with machine learning to screen and validate biomarkers for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(5): 1096-1105. |
| [3] | Li Huaying, Li Hao, Peng Wuxun, Dong Wentao. Mechanism of cuproptosis in the diagnosis and treatment of orthopedic-related diseases [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6275-6281. |
| [4] | Kan Weiyi, Wang Linrong, Cheng Leping. The interaction and balance between cellular senescence and tissue repair [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6297-6305. |
| [5] | Li Shu, Zhao Zhengyi, Zeng Qin, Zhu Xiangdong. Nanohydroxyapatite induces immunogenic cell death in tumors [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(20): 5143-5151. |
| [6] | Jiang Linglong, Zhang Yuan, Shen Yuwei, Pan Jiawei, Sun Yangyang, Zhu Jundong, Fan Min, Shi Jian. Natural killer cells in treatment of prostate cancer: a bibliometric analysis from basic research to clinical applications [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(13): 3474-3484. |
| [7] | Xu Fanping, Li Qinchun, Tang Dongfang. Targeting diverse chimeric antigen receptor T cell-related targets in treatment of B-cell hematological malignancies: a review of long-term follow-up data [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 248-259. |
| [8] | Yu Shuai, Liu Jiawei, Zhu Bin, Pan Tan, Li Xinglong, Sun Guangfeng, Yu Haiyang, Ding Ya, Wang Hongliang. Hot issues and application prospects of small molecule drugs in treatment of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(9): 1913-1922. |
| [9] | Chen Yaodong, Ren Jiayi, Cao Jingwei, Fan Wenwen, Chen Wu. Near-infrared photoresponsive h-PCuNF nanoparticles mediate multimodal therapeutics against malignant tumors [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(4): 780-788. |
| [10] | Han Mengjun, Xu Fang. Hematopoietic stem cell mobilization: advantages and disadvantages of different plans and improvements in predictive models and technologies [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(36): 7863-7871. |
| [11] | Guo Zhao, Zhuang Haoyan, Shi Xuewen. Role of exosomes derived from mesenchymal stem cells in treatment of colorectal cancer [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(36): 7872-7879. |
| [12] | Wu Qingyun, Su Qiang. Antioxidant nanomedicine-mediated targeted therapy for myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(34): 7431-7438. |
| [13] | Wang Jianxu, Dong Zihao, Huang Zishuai, Li Siying, Yang Guang. Interaction between immune microenvironment and bone aging and treatment strategies [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(30): 6509-6519. |
| [14] | Fan Hongkai, Guan Yingying, Wang Lumin, Zeng Fanwei, Yin Yirui. Feasibility of gastric cancer organoid models for personalized drug screening [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(25): 5345-5350. |
| [15] | Guo Zhao, Zhuang Haoyan, Shi Xuewen. Crossroads of colorectal cancer progression and suppression: efficacy and challenges of mesenchymal stem cell therapy interventions [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(23): 5022-5030. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||