Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (17): 2667-2674.doi: 10.12307/2024.480
Previous Articles Next Articles
Polycaprolactone-polydopamine-AOPDM1 scaffold promotes bone formation in a high-glucose environment
Liu Ziyang, Lao An, Xu Chenci, AiRi Shin, Wu Jiaqing, Liu Jiaqiang
- Department of Oral & Cranio-Maxillofacial Surgery, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine; College of Stomatology, Shanghai Jiao Tong University; National Center for Stomatology; National Clinical Research Center for Oral Diseases; Shanghai Key Laboratory of Stomatology, Shanghai Research Institute of Stomatology, Shanghai 200011, China
-
Received:2023-08-02Accepted:2023-09-18Online:2024-06-18Published:2023-12-14 -
Contact:Liu Jiaqiang, Chief physician, Professor, Department of Oral & Cranio-Maxillofacial Surgery, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine; College of Stomatology, Shanghai Jiao Tong University; National Center for Stomatology; National Clinical Research Center for Oral Diseases; Shanghai Key Laboratory of Stomatology, Shanghai Research Institute of Stomatology, Shanghai 200011, China -
About author:Liu Ziyang, Master candidate, Department of Oral & Cranio-Maxillofacial Surgery, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine; College of Stomatology, Shanghai Jiao Tong University; National Center for Stomatology; National Clinical Research Center for Oral Diseases; Shanghai Key Laboratory of Stomatology, Shanghai Research Institute of Stomatology, Shanghai 200011, China Lao An, Doctoral candidate, Department of Oral & Cranio-Maxillofacial Surgery, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine; College of Stomatology, Shanghai Jiao Tong University; National Center for Stomatology; National Clinical Research Center for Oral Diseases; Shanghai Key Laboratory of Stomatology, Shanghai Research Institute of Stomatology, Shanghai 200011, China -
Supported by:National Natural Science Foundation of China (General Program), No. 82071081 (to LJQ); Shanghai Municipal Health Commission Health Industry Clinical Research Project (Excellence Project), No. 20224Z0010 (to LJQ)
CLC Number:
Cite this article
Liu Ziyang, Lao An, Xu Chenci, AiRi Shin, Wu Jiaqing, Liu Jiaqiang. Polycaprolactone-polydopamine-AOPDM1 scaffold promotes bone formation in a high-glucose environment[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2667-2674.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
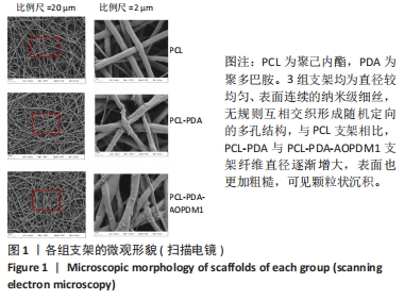
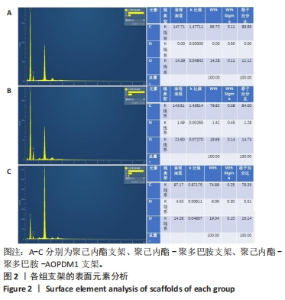
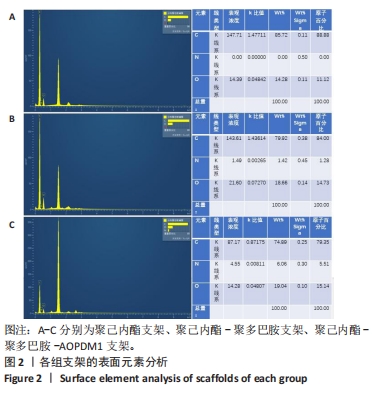
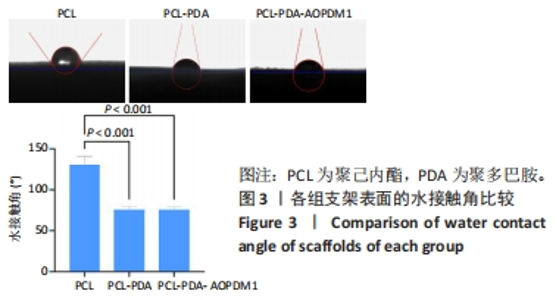
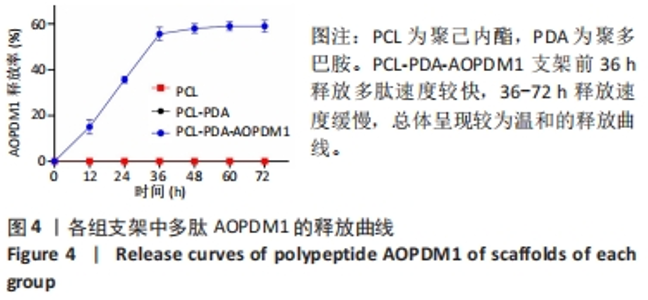
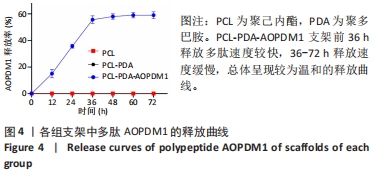
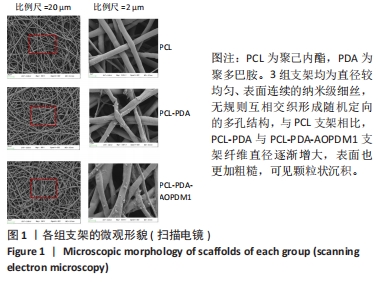
2.1 支架表征结果 2.1.1 支架形貌 扫描电镜显示3组支架均为直径较均匀、表面连续的纳米级细丝,无规则互相交织形成随机定向的多孔结构,见图1,该多孔结构有利于细胞的浸润、营养物质及气体的输送,并且在结构上仿生了细胞外基质[33-34]。与PCL支架相比,PCL-PDA与PCL-PDA-AOPDM1支架纤维直径逐渐增大,表面也更加粗糙,可见颗粒状沉积,沉积的颗粒分别为PDA和PDA-AOPDM1。图2表面元素分析显示,PCL支架表面的元素主要为C和O;PCL-PDA支架表面的主要元素仍为C和O,出现少量N元素;PCL-PDA-AOPDM1支架表面的元素中出现较多的N,提示多肽成功负载于电纺膜上。"
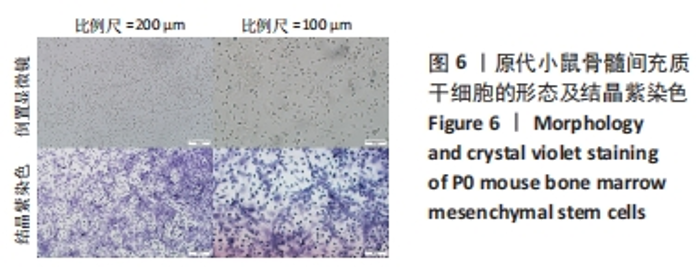
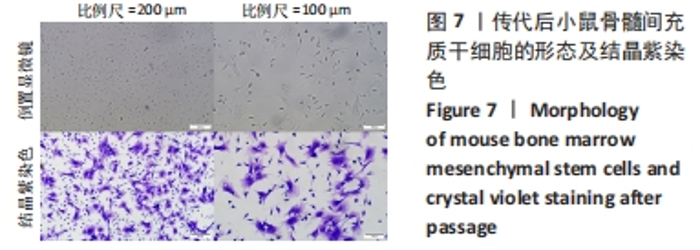
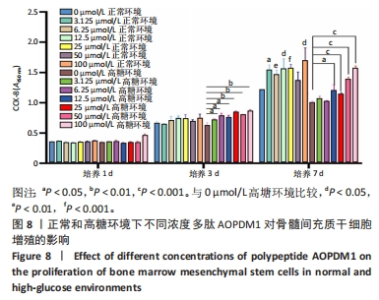
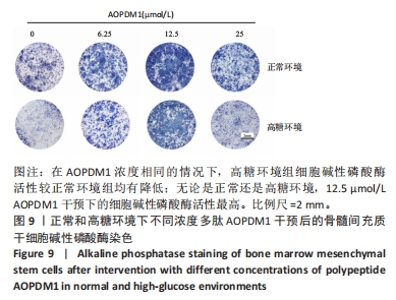
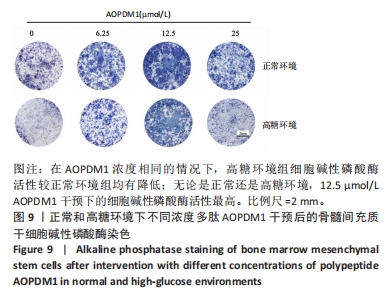
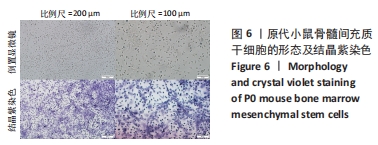
2.2 AOPDM1对骨髓间充质干细胞增殖与成骨分化的影响 2.2.1 小鼠骨髓间充质干细胞形态观察 图6,7显示小鼠骨髓间充质干细胞的形态,在原代培养初期有一些杂细胞,贴壁细胞于1周左右长满瓶底,随着换液与细胞传代,杂细胞数目逐渐减少,细胞形态逐渐均一化,细胞多为梭形、纺锤形。 2.2.2 AOPDM1对骨髓间充质干细胞增殖的影响 图8显示正常环境与高糖环境下不同浓度AOPDM1对骨髓间充质干细胞增殖的影响。培养第1天,各组细胞增殖无明显差异(P > 0.05)。培养第3天,高糖环境下的骨髓间充质干细胞增殖与正常对照组比较差异无显著性意义(P > 0.05);高糖环境下,6.25,12.5,25,50,100 μmol/L AOPDM1均可促进骨髓间充质干细胞的增殖(P < 0.05,P < 0.01)。培养第7天,高糖环境下0 μmol/L AOPDM1组骨髓间充质干细胞的增殖低于正常对照组(P < 0.001);高糖环境下,12.5,25,50,100 μmol/L的AOPDM1可促进骨髓间充质干细胞的增殖(P < 0.05,P < 0.001)。 2.2.3 AOPDM1对骨髓间充质干细胞碱性磷酸酶活性的影响 预实验发现12.5 μmol/L AOPDM1为对骨髓间充质干细胞碱性磷酸酶活性影响发生转折的浓度,因此选择附近的4个浓度说明问题。"
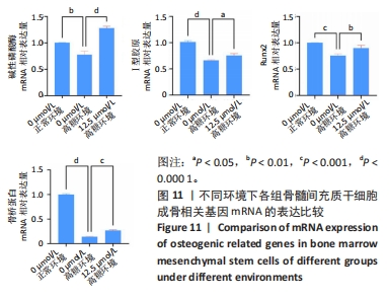
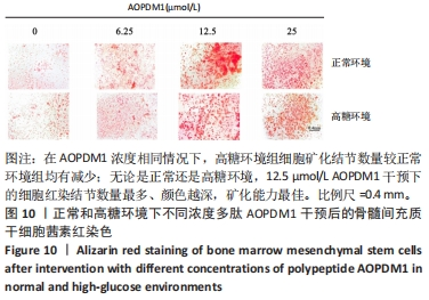
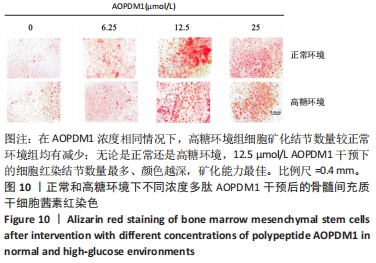
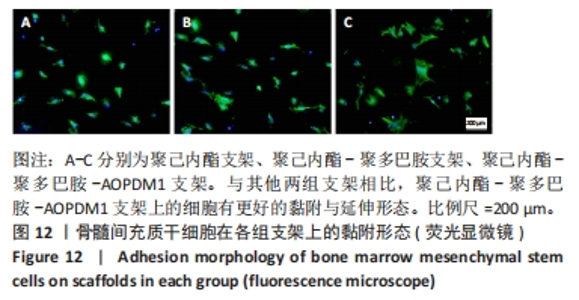
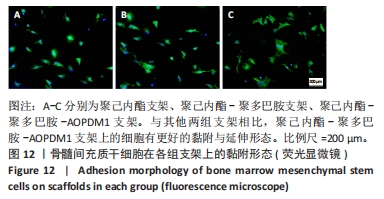
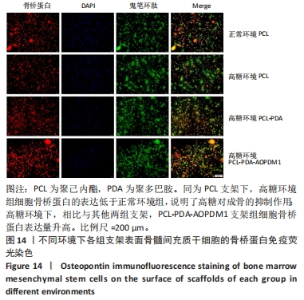
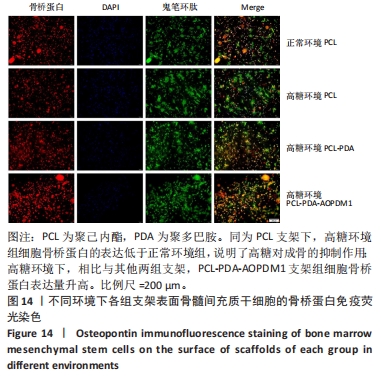
2.2.5 AOPDM1对骨髓间充质干细胞成骨分化基因表达的影响 碱性磷酸酶染色及茜素红染色结果表明,12.5 μmol/L是AOPDM1促进成骨的最佳浓度,因此采用12.5 μmol/L浓度进行后续实验。图11显示高糖环境下12.5 μmol/L AOPDM1对骨髓间充质干细胞成骨基因Ⅰ型胶原(成骨诱导7 d)、骨桥蛋白(成骨诱导14 d)、Runx2(成骨诱导7 d)及碱性磷酸酶(成骨诱导7 d)mRNA表达的影响。在不加入AOPDM1的情况下,高糖环境组细胞内Ⅰ型胶原、骨桥蛋白、Runx2及碱性磷酸酶 mRNA的表达均低于正常环境组(P < 0.01,P < 0.001,P < 0.000 1);高糖环境下,加入12.5 μmol/L AOPDM1可提高细胞内Ⅰ型胶原、骨桥蛋白、Runx2及碱性磷酸酶 mRNA的表达(P < 0.05,P < 0.01,P < 0.001,P < 0.000 1)。"
| [1] 刘建国,周宏博.医用β钛合金的性能研究现状[J].口腔医学研究, 2020,36(6):501-508. [2] 王振铭,叶玲.生物活性组织工程材料在口腔颌面部骨修复中的应用研究进展[J].口腔生物医学,2021,12(2):71-76. [3] ZHAO QM, LI XK, GUO S, et al. Osteogenic activity of a titanium surface modified with silicon-doped titanium dioxide. Mater Sci Eng C Mater Biol Appl. 2020;110:110682. [4] CHEN Y, ZHOU Y, LIN J, et al. Challenges to Improve Bone Healing Under Diabetic Conditions. Front Endocrinol (Lausanne). 2022;13:861878. [5] PAPACHRISTOFOROU E, LAMBADIARI V, MARATOU E, et al. Association of Glycemic Indices (Hyperglycemia, Glucose Variability, and Hypoglycemia) with Oxidative Stress and Diabetic Complications. J Diabetes Res. 2020;2020:7489795. [6] PILLON NJ, LOOS RJF, MARSHALL SM, et al. Metabolic consequences of obesity and type 2 diabetes: Balancing genes and environment for personalized care. Cell. 2021;184(6):1530-1544. [7] KAUR J, KHOSLA S, FARR JN. Effects of diabetes on osteocytes. Curr Opin Endocrinol Diabetes Obes. 2022;29(4):310-317. [8] RENDRA E, RIABOV V, MOSSEL DM, et al. Reactive oxygen species (ROS) in macrophage activation and function in diabetes. Immunobiology. 2019;224(2):242-253. [9] LEI C, SONG JH, LI S, et al. Advances in materials-based therapeutic strategies against osteoporosis. Biomaterials. 2023;296:122066. [10] LIU X, CHEN M, LUO J, et al. Immunopolarization-regulated 3D printed-electrospun fibrous scaffolds for bone regeneration. Biomaterials. 2021;276:121037. [11] MA L, YU Y, LIU H, et al. Berberine-releasing electrospun scaffold induces osteogenic differentiation of DPSCs and accelerates bone repair. Sci Rep. 2021;11(1):1027. [12] DING H, CHENG Y, NIU X, et al. Application of electrospun nanofibers in bone, cartilage and osteochondral tissue engineering. J Biomater Sci Polym Ed. 2021;32(4):536-561. [13] HE C, LV Q, LIU Z, et al. Random and aligned electrostatically spun PLLA nanofibrous membranes enhance bone repair in mouse femur midshaft defects. J Biomater Appl. 2023;37(9):1582-1592. [14] MA K, LIAO C, HUANG L, et al. Electrospun PCL/MoS(2) Nanofiber Membranes Combined with NIR-Triggered Photothermal Therapy to Accelerate Bone Regeneration. Small. 2021;17(51):e2104747. [15] JANG HY, SHIN JY, OH SH, et al. PCL/HA Hybrid Microspheres for Effective Osteogenic Differentiation and Bone Regeneration. ACS Biomater Sci Eng. 2020;6(9):5172-5180. [16] YUAN B, WANG Z, ZHAO Y, et al. In Vitro and In Vivo Study of a Novel Nanoscale Demineralized Bone Matrix Coated PCL/β-TCP Scaffold for Bone Regeneration. Macromol Biosci. 2021;21(3):e2000336. [17] REN X, GAO X, CHENG Y, et al. Maintenance of multipotency of bone marrow mesenchymal stem cells on poly(ε-caprolactone) nanoneedle arrays through the enhancement of cell-cell interaction. Front Bioeng Biotechnol. 2022;10:1076345. [18] PARK J, LEE SJ, JUNG TG, et al. Surface modification of a three-dimensional polycaprolactone scaffold by polydopamine, biomineralization, and BMP-2 immobilization for potential bone tissue applications. Colloids Surf B Biointerfaces. 2021;199:111528. [19] LI Y, YANG L, HOU Y, et al. Polydopamine-mediated graphene oxide and nanohydroxyapatite-incorporated conductive scaffold with an immunomodulatory ability accelerates periodontal bone regeneration in diabetes. Bioact Mater. 2022;18:213-227. [20] XU Z, WANG N, LIU P, et al. Poly(Dopamine) Coating on 3D-Printed Poly-Lactic-Co-Glycolic Acid/β-Tricalcium Phosphate Scaffolds for Bone Tissue Engineering. Molecules. 2019;24(23):4397. [21] LU Y, WAN Y, GAN D, et al. Enwrapping Polydopamine on Doxorubicin-Loaded Lamellar Hydroxyapatite/Poly(lactic-co-glycolic acid) Composite Fibers for Inhibiting Bone Tumor Recurrence and Enhancing Bone Regeneration. ACS Appl Bio Mater. 2021;4(8):6036-6045. [22] TANG Y, TAN Y, LIN K, et al. Research Progress on Polydopamine Nanoparticles for Tissue Engineering. Front Chem. 2021;9:727123. [23] KANG Z, LI D, SHU C, et al. Polydopamine Coating-Mediated Immobilization of BMP-2 on Polyethylene Terephthalate-Based Artificial Ligaments for Enhanced Bioactivity. Front Bioeng Biotechnol. 2021;9:749221. [24] NUZZI G, TRAMBUSTI I, DI CICCO ME, et al. Breast milk: more than just nutrition! Minerva Pediatr (Torino). 2021;73(2):111-114. [25] THAI JD, GREGORY KE. Bioactive Factors in Human Breast Milk Attenuate Intestinal Inflammation during Early Life. Nutrients. 2020; 12(2):581. [26] YI DY, KIM SY. Human Breast Milk Composition and Function in Human Health: From Nutritional Components to Microbiome and MicroRNAs. Nutrients. 2021;13(9):3094. [27] 张玉梅,石羽杰,张健,等.母乳α-乳清蛋白、β-酪蛋白与婴幼儿健康的研究进展[J].营养学报,2020,42(1):78-82. [28] LI Y, CUI X, WANG X, et al. Human milk derived peptide AOPDM1 attenuates obesity by restricting adipogenic differentiation through MAPK signalling. Biochim Biophys Acta Gen Subj. 2021;1865(3): 129836. [29] LIU F, YUAN Y, BAI L, et al. LRRc17 controls BMSC senescence via mitophagy and inhibits the therapeutic effect of BMSCs on ovariectomy-induced bone loss. Redox Biol. 2021;43:101963. [30] ZHANG X, CAO D, XU L, et al. Harnessing matrix stiffness to engineer a bone marrow niche for hematopoietic stem cell rejuvenation. Cell Stem Cell. 2023;30(4):378-395.e378. [31] WANG ZX, LUO ZW, LI FX, et al. Aged bone matrix-derived extracellular vesicles as a messenger for calcification paradox. Nat Commun. 2022; 13(1):1453. [32] WU J, CAO L, LIU Y, et al. Functionalization of Silk Fibroin Electrospun Scaffolds via BMSC Affinity Peptide Grafting through Oxidative Self-Polymerization of Dopamine for Bone Regeneration. ACS Appl Mater Interfaces. 2019;11(9):8878-8895. [33] BHARADWAZ A, JAYASURIYA AC. Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110698. [34] FADAIE M, MIRZAEI E, GERAMIZADEH B, et al. Incorporation of nanofibrillated chitosan into electrospun PCL nanofibers makes scaffolds with enhanced mechanical and biological properties. Carbohydr Polym. 2018;199:628-640. [35] Correction to Lancet Diabetes Endocrinol 2022; 10: 58-74. Lancet Diabetes Endocrinol. 2022;10(1):e1. [36] SUN Y, ZHU Y, LIU X, et al. Morroniside attenuates high glucose-induced BMSC dysfunction by regulating the Glo1/AGE/RAGE axis. Cell Prolif. 2020;53(8):e12866. [37] LI Y, WANG X. Chrysin Attenuates High Glucose-Induced BMSC Dysfunction via the Activation of the PI3K/AKT/Nrf2 Signaling Pathway. Drug Des Devel Ther. 2022;16:165-182. [38] CHEN M, JING D, YE R, et al. PPARβ/δ accelerates bone regeneration in diabetic mellitus by enhancing AMPK/mTOR pathway-mediated autophagy. Stem Cell Res Ther. 2021;12(1):566. [39] DIEMAR SS, MØLLEHAVE LT, QUARDON N, et al. Effects of age and sex on osteocalcin and bone-specific alkaline phosphatase-reference intervals and confounders for two bone formation markers. Arch Osteoporos. 2020;15(1):26. [40] ZHANG J, TONG D, SONG H, et al. Osteoimmunity-Regulating Biomimetically Hierarchical Scaffold for Augmented Bone Regeneration. Adv Mater. 2022;34(36):e2202044. |
| [1] | Wang Wen, Zheng Pengpeng, Meng Haohao, Liu Hao, Yuan Changyong. Overexpression of Sema3A promotes osteogenic differentiation of dental pulp stem cells and MC3T3-E1 [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 993-999. |
| [2] | Mei Jingyi, Liu Jiang, Xiao Cong, Liu Peng, Zhou Haohao, Lin Zhanyi. Proliferation and metabolic patterns of smooth muscle cells during construction of tissue-engineered blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1043-1049. |
| [3] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [4] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [5] | Lin Feng, Cheng Ling, Gao Yong, Zhou Jianye, Shang Qingqing. Hyaluronic acid hydrogel-encapsulated bone marrow mesenchymal stem cells promote cardiac function in myocardial infarction rats (III) [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 355-359. |
| [6] | Bi Yujie, Ma Dujun, Peng Liping, Zhou Ziqiong, Zhao Jing, Zhu Houjun, Zhong Qiuhui, Yang Yuxin. Strategy and significance of Chinese medicine combined with medical hydrogel for disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 419-425. |
| [7] | Chen Xiangshan, Liu Hua, Sun Weikang, Li Huanan. Mechanism of m6A methylation regulating bone metabolism for prevention and treatment of osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(28): 4572-4577. |
| [8] | Yang Qipei, Chen Feng, Cui Wei, Zhang Chi, Wu Ruiqi, Song Zhenheng, Meng Xin. Signaling pathways related to kaempferol active monomers in the treatment of osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4242-4249. |
| [9] | Yang Shanshan, Ouyang Renjun, Tian Jia, Linghu Min, Wang Zhen, Yang Xiaohong. Detection of immune-related cytokines of bone marrow mesenchymal stem cells in postmenopausal osteoporosis mice by antibody chip and analysis of key differential genes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 3947-3954. |
| [10] | Zhang Jie, Xiao Tianjiao, Li Li, Kang Jiabing, Zhan Jifan, Wei Yan, Tian Ai. Interleukin-4 regulates macrophage polarization and osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 3960-3966. |
| [11] | Han Dunzheng, Qin Xiaozhou, Pan Xiudi, Lu Waner, Dai Ying, Chen Yanxun, Cheng Xianfei, Tang Muhan. Effect of apoptosis-inducing factor gene knockdown on bone marrow mesenchymal stem cell transplantation for myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 3967-3973. |
| [12] | Zhang Min, Lou Guo, Fu Changxi. Aerobic exercise preconditioning improves therapeutic effect of bone marrow mesenchymal stem cells on acute myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 3988-3993. |
| [13] | Han Yue, Wang Yufei, Liu Wanqing, Dong Ming, Niu Weidong. Effects of icariin on proliferation and differentiation of MC3T3-E1 cells in an inflammatory environment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3709-3714. |
| [14] | Wang Qian, Lu Ziang, Li Lihe, Lyu Chaoliang, Wang Meng, Zhang Cunxin. Sinomenine effectively inhibits interleukin-1beta-induced apoptosis in nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 224-230. |
| [15] | Chen Na, Wang Yanlin, Sun Huifang, Fan Feiyan, Li Donghong, Zhang Yunke. Shexiang Huangqi compound dripping pills-containing serum promotes proliferation and differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 2960-2966. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||