Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (26): 4113-4120.doi: 10.12307/2024.393
Previous Articles Next Articles
Effects of aerobic exercise on the expression of Notch1 and Caspase-3 in the hippocampus of Alzheimer’s disease mice
Yang Liyuan1, Zhang Yeting2, Li Chuikun1, Wei Cuilan3
- 1College of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China; 2Civil Aviation Flight University of China, Guanghan 618307, Sichuan Province, China; 3Sports Institute of Chengdu University of Technology, Chengdu 610059, Sichuan Province, China
-
Received:2023-06-06Accepted:2023-07-11Online:2024-09-18Published:2023-09-28 -
Contact:Zhang Yeting, PhD, Lecturer, Civil Aviation Flight University of China, Guanghan 618307, Sichuan Province, China -
About author:Yang Liyuan, Master, Experimentalist, College of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China -
Supported by:Sichuan Provincial Science and Technology Program, No. 2020YFH0184 (to ZYT); the Fundamental Research Fund for the Central Universities, No. PHD2023-003 (to ZYT)
CLC Number:
Cite this article
Yang Liyuan, Zhang Yeting, Li Chuikun, Wei Cuilan. Effects of aerobic exercise on the expression of Notch1 and Caspase-3 in the hippocampus of Alzheimer’s disease mice[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4113-4120.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
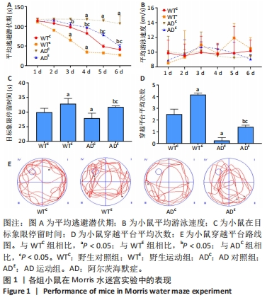
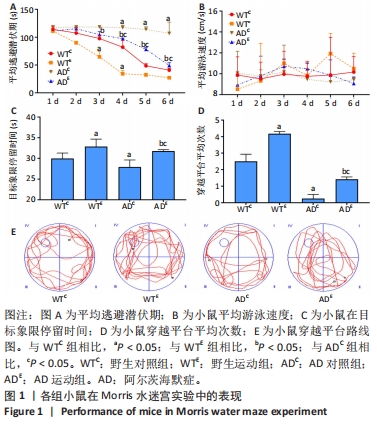
2.1 实验动物数量分析 实验选用小鼠80只,分为4组,实验过程中,AD运动组有1只小鼠死亡,最终进入结果分析72只(每组最终各选取18只小鼠进行取材)。 2.2 小鼠空间学习记忆能力检测结果 在Morris水迷宫定位航行训练实验过程中,伴随训练实验天数的增加,野生对照组、野生运动组与AD运动组小鼠的平均逃避潜伏期出现逐渐减小的趋势,而AD对照组小鼠的平均逃避潜伏期并未出现显著减小的趋势。通过重复方差分析逃避潜伏期结果表明:干预方式、基因型与时间的交互作用显著。以基因型和时间为主效应量,分析干预方式对小鼠潜伏期的影响:在野生型小鼠中,野生运动组从第2天开始,每天的逃避潜伏期均显著低于野生对照组(P < 0.05);在AD小鼠中,AD运动组从第1天开始,每天的逃避潜伏期均显著低于AD对照组(P < 0.05)。以干预方式和时间为主效应量,分析基因型对小鼠潜伏期的影响:在对照组中,AD对照组小鼠逃避潜伏期从第3天开始显著高于野生对照组(P < 0.05);在运动组中,AD运动组小鼠逃避潜伏期从第3天开始显著高于野生运动组(P < 0.05)。以干预方式和基因型为主效应量,分析时间对小鼠潜伏期的影响:只有AD对照组小鼠各时间点的逃避潜伏期均无显著差异(P > 0.05),见图1A。以上的数据能够说明AD对照组小鼠具有较低的空间参考学习能力,而其余各组小鼠则具有一定的空间参考学习能力,并且随着训练实验次数和天数的增加,能够通过参照水迷宫内壁的标记物建立寻找水下隐藏平台位置的记忆。 通过重复方差分析泳速结果表明:干预方式与基因型的交互作用显著。以基因型为主效应量,分析干预方式对小鼠泳速的影响:无论是野生型小鼠,还是AD小鼠,运动都没有显著影响小鼠的泳速(P > 0.05);以干预方式为主效应量,分析基因型对小鼠泳速的影响:无论是对照组小鼠,还是运动组小鼠,基因型都没有显著影响小鼠的泳速(P > 0.05),见图1B。这表明各组小鼠并无游泳运动障碍,且在Morris水迷宫定位航行训练实验中的逃避潜伏期差异并非由运动障碍所造成。 在60 s的空间探索实验中,通过两因素被试间方差分析小鼠在目标象限中所用时间:野生运动组在目标象限中停留的时间显著高于野生对照组(P=0.037),AD运动组在目标象限中停留的时间显著高于AD对照组(P=0.010);AD对照组在目标象限中停留的时间显著低于野生对照组(P=0.025),AD运动组在目标象限中停留的时间显著低于野生运动组(P=0.043),见图1C。通过两因素被试间方差分析小鼠穿越平台次数:野生运动组穿越原有平台区域的次数显著高于野生对照组(P < 0.005),AD运动组穿越原有平台区域的次数显著高于AD对照组(P=0.001);AD对照组穿越原有平台区域的次数显著低于野生对照组(P < 0.005),AD运动组穿越原有平台区域的次数显著低于野生运动组(P < 0.005),见图1D。小鼠穿越平台路线图,见图1E。"
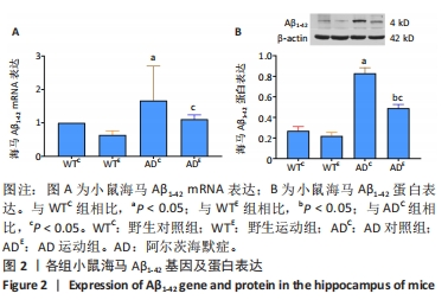
2.3 小鼠海马Aβ的表达 图2A为小鼠海马Aβ1-42 mRNA表达。通过两因素被试间方差分析小鼠海马Aβ1-42 mRNA表达结果:野生运动组Aβ1-42 mRNA表达与野生对照组相比没有显著差异,AD运动组Aβ1-42 mRNA表达显著低于AD对照组(P=0.032);AD对照组Aβ1-42 mRNA表达显著高于野生对照组,AD运动组Aβ1-42 mRNA表达与野生运动组相比没有显著差异(P=0.129)。图2B为小鼠海马Aβ1-42 蛋白表达。通过两因素被试间方差分析小鼠海马Aβ1-42蛋白表达结果:野生运动组Aβ1-42蛋白表达与野生对照组相比没有显著差异,AD运动组Aβ1-42蛋白表达显著低于AD对照组(P < 0.000 5);AD对照组Aβ1-42 蛋白表达显著高于野生对照组,AD运动组Aβ1-42蛋白表达显著高于野生运动组(P < 0.000 5)。"


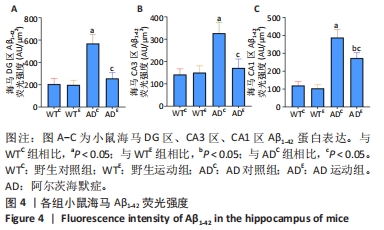
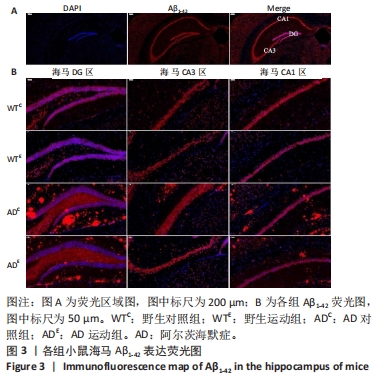
利用荧光显微镜对免疫荧光切片进行扫描成像,并利用CaseViewer软件选取所需区域图片。图3中蓝色代表DAPI标记的细胞核,红色代表Aβ1-42免疫阳性产物表达。采用Image-Pro Plus 6.0软件测定荧光强度。通过两因素被试间方差分析小鼠海马DG区的Aβ1-42免疫阳性产物荧光强度:野生运动组Aβ1-42免疫阳性产物荧光强度与野生对照组相比没有显著差异,AD运动组Aβ1-42免疫阳性产物荧光强度显著低于AD对照组(P < 0.000 5);AD对照组Aβ1-42免疫阳性产物荧光强度显著高于野生对照组,AD运动组Aβ1-42免疫阳性产物荧光强度与野生运动组相比无显著差异(P=0.104),见图4A。通过两因素被试间方差分析小鼠海马CA3区的Aβ1-42免疫阳性产物荧光强度:野生运动组Aβ1-42免疫阳性产物荧光强度与野生对照组相比没有显著差异,AD运动组Aβ1-42免疫阳性产物荧光强度显著低于AD对照组(P < 0.000 5);AD对照组Aβ1-42免疫阳性产物荧光强度显著高于野生对照组,AD运动组Aβ1-42免疫阳性产物荧光强度与野生运动组相比无显著差异(P=0.353),见图4B。通过两因素被试间方差分析小鼠海马CA1区的Aβ1-42免疫阳性产物荧光强度:野生运动组Aβ1-42免疫阳性产物荧光强度与野生对照组相比没有显著差异,AD运动组Aβ1-42免疫阳性产物荧光强度显著低于AD对照组(P < 0.000 5);AD对照组Aβ1-42免疫阳性产物荧光强度显著高于野生对照组(P < 0.000 5),AD运动组Aβ1-42免疫阳性产物荧光强度显著高于野生运动组(P < 0.000 5),见图4C。这表明AD转基因小鼠海马组织中出现了大量的Aβ1-42沉积,而长期有氧运动似乎可以减少其海马组织中Aβ1-42的沉积。"

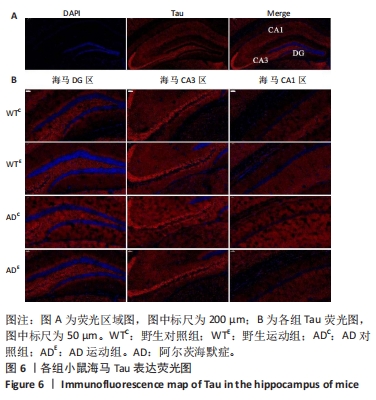
2.4 小鼠海马Tau的表达 图5A为小鼠海马Tau mRNA表达。通过两因素被试间方差分析小鼠海马Tau mRNA表达结果:野生运动组Tau mRNA表达显著低于野生对照组,AD运动组Tau mRNA表达显著低于AD对照组(P < 0.000 5);AD对照组Tau mRNA表达显著高于野生对照组,AD运动组Tau mRNA表达显著高于野生运动组(P=0.002)。图5B为小鼠海马Tau蛋白表达。通过两因素被试间方差分析小鼠海马Tau蛋白表达结果:野生运动组Tau蛋白表达与野生对照组相比没有显著差异,AD运动组Tau蛋白表达显著低于AD对照组(P < 0.000 5);AD对照组Tau蛋白表达显著高于野生对照组,AD运动组Tau蛋白表达与野生运动组相比没有显著差异(P=0.125)。"
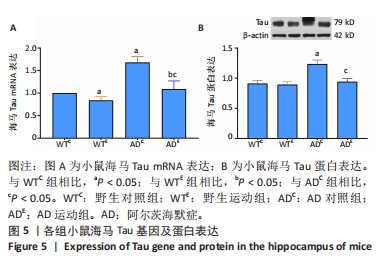

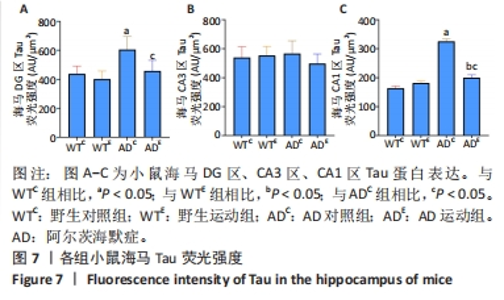
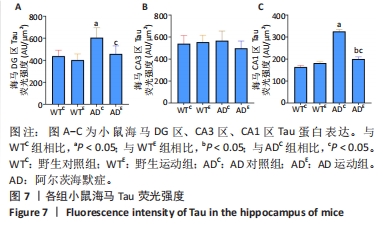
图6中蓝色代表DAPI标记的细胞核,红色代表Tau免疫阳性产物表达。通过两因素被试间方差分析小鼠海马DG区的Tau免疫阳性产物荧光强度:野生运动组Tau免疫阳性产物荧光强度与野生对照组相比没有显著差异,AD运动组Tau免疫阳性产物荧光强度显著低于AD对照组(P=0.002);AD对照组Tau免疫阳性产物荧光强度显著高于野生对照组,AD运动组Tau免疫阳性产物荧光强度与野生运动组相比无显著差异(P=0.188),见图7A。通过两因素被试间方差分析小鼠海马CA3区的Tau免疫阳性产物荧光强度:各组之间免疫阳性产物荧光强度相比无显著差异,见图7B。通过两因素被试间方差分析小鼠海马CA1区的Tau免疫阳性产物荧光强度:野生运动组Tau免疫阳性产物荧光强度与野生对照组相比无显著差异,AD运动组Tau免疫阳性产物荧光强度显著低于AD对照组(P=0.002);AD对照组Tau免疫阳性产物荧光强度显著高于野生对照组,AD运动组Tau免疫阳性产物荧光强度显著高于野生运动组(P=0.001),见图7C。这表明AD转基因小鼠海马,尤其是DG区、CA1区组织中出现了大量的Tau蛋白,而长期有氧运动似乎可以减少Tau蛋白的表达。"
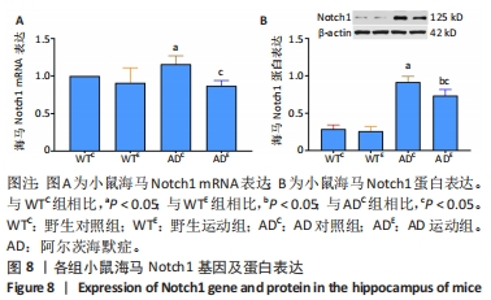
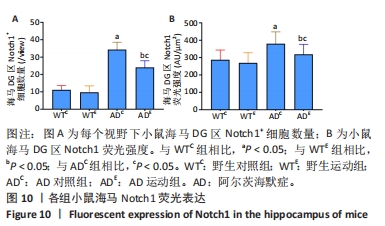
2.5 小鼠海马Notch1的表达 图8A为小鼠海马Notch1 mRNA表达。野生运动组海马Notch1 mRNA表达与野生对照组相比没有显著差异(P > 0.05);AD对照组海马Notch1 mRNA表达显著高于野生对照组 (P < 0.05);而AD运动组海马Notch1 mRNA表达显著低于AD对照组(P < 0.05),但AD运动组海马Notch1 mRNA表达与野生运动组相比没有显著差异(P > 0.05)。图8B为小鼠海马Notch1蛋白表达。野生运动组Notch1蛋白表达与野生对照组相比没有显著差异(P > 0.05);AD对照组Notch1蛋白表达显著高于野生对照组 (P < 0.05);而AD运动组Notch1蛋白表达显著低于AD对照组(P < 0.05),且AD运动组Notch1蛋白表达显著高于野生运动组 (P < 0.05)。"
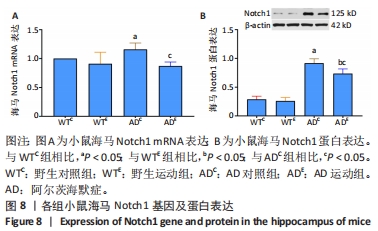
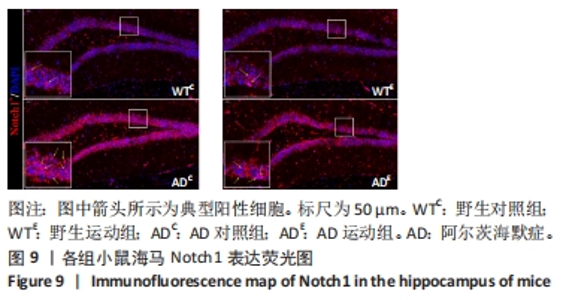
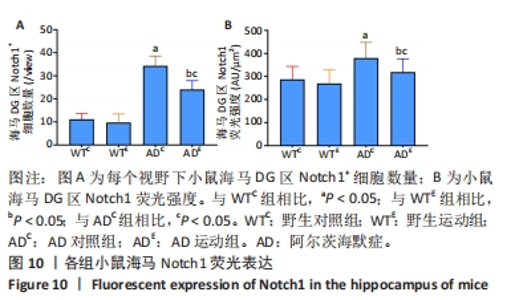
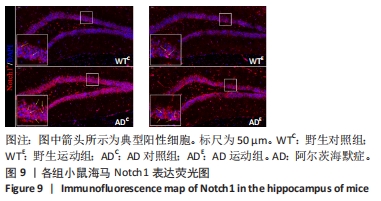
图9中蓝色代表DAPI标记的细胞核,红色代表Notch1免疫阳性产物表达。图10A为每个视野下海马DG区Notch1阳性细胞数量,野生运动组海马DG区Notch1阳性细胞数量与野生对照组相比没有显著差异(P > 0.05);AD对照组海马DG区的Notch1阳性细胞数量显著高于野生对照组(P < 0.05);AD运动组海马DG区Notch1阳性细胞数量显著低于AD对照组(P < 0.05),但AD运动组海马DG区Notch1阳性细胞数量显著高于野生运动组(P < 0.05)。图10B为小鼠海马DG区Notch1荧光强度:野生运动组海马DG区Notch1免疫阳性产物荧光强度与野生对照组相比没有显著差异(P > 0.05);AD对照组海马DG区Notch1免疫阳性产物荧光强度显著高于野生对照组(P < 0.05);AD运动组海马DG区Notch1免疫阳性产物荧光强度显著低于AD对照组 (P < 0.05),但AD运动组海马DG区的Notch1免疫阳性产物荧光强度显著高于野生运动组(P < 0.05)。这表明野生型小鼠海马DG区Notch1的表达并未受到运动的影响,而AD转基因小鼠海马Notch1的表达相较于野生型小鼠显著升高,且长期有氧运动能够显著减少其海马Notch1的表达,这可能预示着运动会通过影响AD转基因小鼠Notch信号通路,从而调节其成年海马神经发生。"
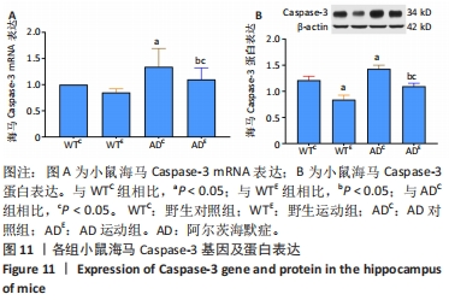
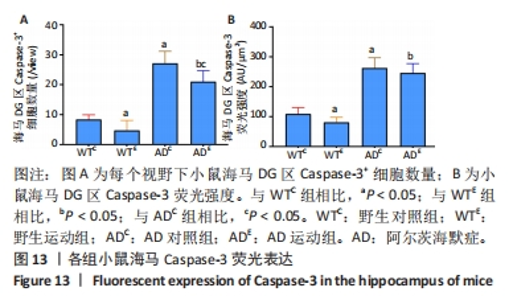
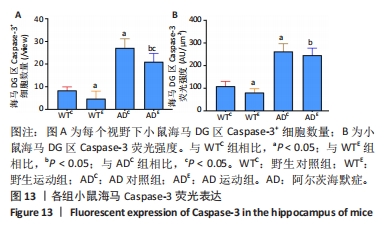
2.6 小鼠海马Caspase-3的表达 图11A为小鼠海马Caspase-3 mRNA表达。野生运动组海马Caspase-3 mRNA表达与野生对照组相比没有显著差异(P > 0.05);AD对照组海马Caspase-3 mRNA表达显著高于野生对照组(P < 0.05);而AD运动组海马Caspase-3 mRNA表达显著低于AD对照组(P < 0.05),且AD运动组海马Caspase-3 mRNA表达显著高于野生运动组(P < 0.05)。图11B为小鼠海马Caspase-3蛋白表达。野生运动组海马Caspase-3蛋白表达显著低于野生对照组(P < 0.05);AD对照组海马Caspase-3蛋白表达显著高于野生对照组(P < 0.05);而AD运动组海马Caspase-3蛋白表达显著低于AD对照组(P < 0.05),且AD运动组海马Caspase-3蛋白表达显著高于野生运动组 (P < 0.05)。"
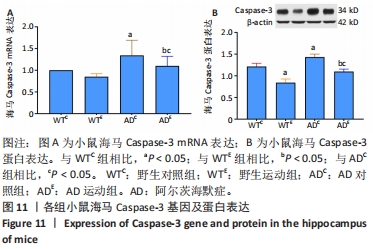
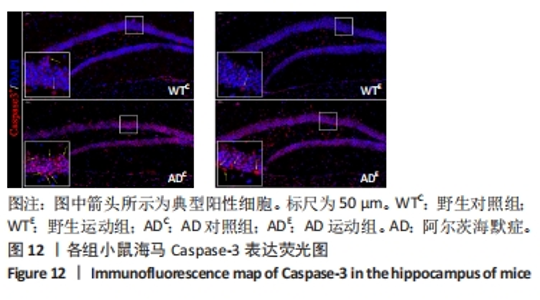
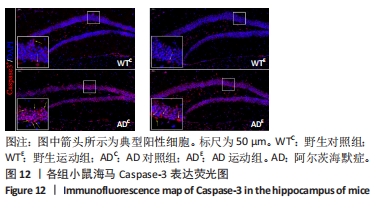
图12中蓝色代表DAPI标记的细胞核,红色代表Caspase-3免疫阳性产物表达。图13A为每个视野下小鼠海马DG区Caspase-3阳性细胞数量,野生运动组海马DG区Caspase-3阳性细胞数量显著低于野生对照组 (P < 0.05);AD对照组海马DG区的Caspase-3阳性细胞数量显著高于野生对照组(P < 0.05);AD运动组海马DG区Caspase-3阳性细胞数量显著低于AD对照组(P < 0.05),但AD运动组海马DG区Caspase-3阳性细胞数量显著高于野生运动组(P < 0.05)。图13B为小鼠海马DG区Caspase-3荧光强度:野生运动组海马DG区Caspase-3免疫阳性产物荧光强度显著低于野生对照组(P < 0.05);AD对照组海马DG区Caspase-3免疫阳性产物荧光强度显著高于野生对照组(P < 0.05);AD运动组海马DG区Caspase-3免疫阳性产物荧光强度与AD对照组相比没有显著差异(P > 0.05),但AD运动组海马DG区的Caspase-3免疫阳性产物荧光强度显著高于野生运动组(P < 0.05)。这表明长期有氧运动可以显著降低野生型小鼠海马DG区Caspase-3的表达,AD转基因小鼠海马Caspase-3的表达相较于野生型小鼠显著升高,而长期有氧运动能够显著减少其海马Caspase-3阳性细胞的数量。"
| [1] SCHELTENS P, BLENNOW K, BRETELER MM, et al. Alzheimer’s disease. Lancet. 2016;388(10043):505-517. [2] BOOTH FW, ROBERTS CK, LAYE MJ. Lack of exercise is a major cause of chronic diseases. Compr Physiol. 2012;2(2):1143-1211. [3] VALENZUELA PL, CASTILLO-GARCÍA A, MORALES JS, et al. Exercise benefits on Alzheimer’s disease: State-of-the-science. Ageing Res Rev. 2020;62:101108. [4] DEPP C, SUN T, SASMITA AO, et al. Myelin dysfunction drives amyloid-β deposition in models of Alzheimer’s disease. Nature. 2023;618(7964):349-357. [5] LEAKE I. Oligomeric tau might spread trans-synaptically in Alzheimer disease. Nat Rev Neurosci. 2023;24(7):393. [6] 2022 Alzheimer’s disease facts and figures. Alzheimers Dement. 2022;18(4):700-789. [7] AQUILA G, MARRACINO L, MARTINO V, et al. The Use of Nutraceuticals to Counteract Atherosclerosis: The Role of the Notch Pathway. Oxid Med Cell Longev. 2019;2019:5470470. [8] PENG X, WANG S, CHEN H, et al. Role of the Notch1 signaling pathway in ischemic heart disease (Review). Int J Mol Med. 2023;51(3):27. [9] ROHN TT, HEAD E. Caspase activation in Alzheimer’s disease: early to rise and late to bed. Rev Neurosci. 2008;19(6):383-393. [10] COTMAN CW, POON WW, RISSMAN RA, et al. The role of caspase cleavage of tau in Alzheimer disease neuropathology. J Neuropathol Exp Neurol. 2005;64(2):104-112. [11] 张象,张业廷.运动改善阿尔茨海默症模型小鼠病程的剂量效应关系[J].中国组织工程研究,2021,25(17):2761-2766. [12] REARDON S. Alzheimer’s drug donanemab: what promising trial means for treatments. Nature. 2023;617(7960):232-233. [13] The Lancet Neurology. Treatment for Alzheimer’s disease: time to get ready. Lancet Neurol. 2023;22(6):455. [14] LOURENCO MV, FROZZA RL, DE FREITAS GB, et al. Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models. Nat Med. 2019;25(1):165-175. [15] CHEN X, GAN L. An exercise-induced messenger boosts memory in Alzheimer’s disease. Nat Med. 2019;25(1):20-21. [16] MARX J. Alzheimer’s disease. Play and exercise protect mouse brain from amyloid buildup. Science. 2005;307(5715):1547. [17] DEVANAND DP, MASURKAR AV, WISNIEWSKI T. Vigorous, regular physical exercise may slow disease progression in Alzheimer’s disease. Alzheimers Dement. 2023; 19(4):1592-1597. [18] SOBOL NA, HOFFMANN K, FREDERIKSEN KS, et al. Effect of aerobic exercise on physical performance in patients with Alzheimer’s disease. Alzheimers Dement. 2016;12(12):1207-1215. [19] BLUMENTHAL JA, BABYAK MA, SHERWOOD A. Diet, exercise habits, and risk of Alzheimer disease. JAMA. 2009;302(22):2431; author reply 2431-2432. [20] HIRSCH C. ACP Journal Club. A 12-month, in-home exercise program delayed functional deterioration in Alzheimer disease. Ann Intern Med. 2013;159(4):JC10. [21] HUUHA AM, NOREVIK CS, MOREIRA JBN, et al. Can exercise training teach us how to treat Alzheimer’s disease? Ageing Res Rev. 2022;75:101559. [22] Exercise slows decline in adults with Alzheimer’s disease. BMJ. 2013;346:f2422. [23] TERI L, GIBBONS LE, MCCURRY SM, et al. Exercise plus behavioral management in patients with Alzheimer disease: a randomized controlled trial. JAMA. 2003; 290(15):2015-2022. [24] LÓPEZ-ORTIZ S, VALENZUELA PL, SEISDEDOS MM, et al. Exercise interventions in Alzheimer’s disease: A systematic review and meta-analysis of randomized controlled trials. Ageing Res Rev. 2021;72:101479. [25] FIRTH J, STUBBS B, VANCAMPFORT D, et al. Effect of aerobic exercise on hippocampal volume in humans: A systematic review and meta-analysis. Neuroimage. 2018;166:230-238. [26] 李垂坤,张业廷,魏翠兰,等.不同强度有氧运动对APP/PS1双转基因小鼠海马双皮质素及β-淀粉样蛋白的影响[J].西安体育学院学报,2022,39(2):214-223. [27] CRUZ HERNÁNDEZ JC, BRACKO O, KERSBERGEN CJ, et al. Neutrophil adhesion in brain capillaries reduces cortical blood flow and impairs memory function in Alzheimer’s disease mouse models. Nat Neurosci. 2019;22(3):413-420. [28] SUN X, HE G, QING H, et al. Hypoxia facilitates Alzheimer’s disease pathogenesis by up-regulating BACE1 gene expression. Proc Natl Acad Sci U S A. 2006;103(49):18727-18732. [29] BRAAK H, BRAAK E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991;82(4):239-259. [30] CAMPOS HC, RIBEIRO DE, HASHIGUCHI D, et al. Neuroprotective effects of resistance physical exercise on the APP/PS1 mouse model of Alzheimer’s disease. Front Neurosci. 2023;17:1132825. [31] BRANDT MD, MAASS A, KEMPERMANN G, et al. Physical exercise increases Notch activity, proliferation and cell cycle exit of type-3 progenitor cells in adult hippocampal neurogenesis. Eur J Neurosci. 2010;32(8):1256-1264. |
| [1] | Wang Ji, Zhang Min, Li Wenbo, Yang Zhongya, Zhang Long. Effect of aerobic exercise on glycolipid metabolism, skeletal muscle inflammation and autophagy in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1200-1205. |
| [2] | Li Longyang, Zhang Songjiang, Zhao Xianmin, Zhou Chunguang, Gao Jianfeng. Electroacupuncture intervention on the proliferation and differentiation of hippocampal neurons and oligodendrocytes in Alzheimer’s disease model mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1029-1035. |
| [3] | Chai Shifan, Li Xinru, Ye Yucai, Sun Junli, Cai Hongyan, Wang Zhaojun. Silent information regulator 1: A potential target of semaglutide in the treatment of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3235-3239. |
| [4] | Sun Yuan, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Zhang Yan. Effects of early and late aerobic exercise on right heart failure induced by monocrotaline in rats with pulmonary hypertension [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 177-185. |
| [5] | Wang Jingfeng, Wen Dengtai, Wang Shijie, Gao Yinghui. Atg-mediated autophagy, exercise and skeletal muscle aging [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 295-301. |
| [6] | Xiao Yi, Lu Shuo, Ge Lite, Lu Ming. Interaction between autophagy and mesenchymal stem cells in treatment of neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 3111-3116. |
| [7] | Zhang Shasha, Wang Na, Li Yongjun. Grape seed extract inhibits apoptosis of rat growth plate chondrocytes and promotes tibial bone growth [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2494-2499. |
| [8] | Tang Liang, Wang Hexia, Wang Qingbo, Pi Yihua, Zhang Yan. Aerobic exercise modulates mitochondrial quality control system to reverse cardiac pathological remodeling in aging rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2534-2541. |
| [9] | Han Weina, Xu Xiaoqing, Shi Jinning, Li Xinru, Cai Hongyan. Prediction and validation of potential targets for the glucagon-like peptide-1 receptor agonist in the treatment of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2568-2573. |
| [10] | Wu Yuzhen, Sun Qing, Liu Xia, Zhou Yu, Jin Qiguan. Variation in renal function of type 2 diabetic rats undergoing aerobic exercise [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2145-2151. |
| [11] | Deng Longfei, Zhang Yeting, Fu Yan. Aerobic exercise inhibits neuroinflammation and alleviates cognitive impairment in Alzheimer’s disease model mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2209-2214. |
| [12] | Zhang Yeting, Li Chuikun, Wei Cuilan, Fu Yan, Zhang Feifei. Effects of aerobic exercise on adult hippocampal neurogenesis in Alzheimer’s disease mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(13): 2068-2075. |
| [13] | Long Qingxi, Zhang Pingshu, Liu Qing, Ou Ya, Zhang Lili, Yuan Xiaodong. Single-cell RNA sequencing reveals the heterogeneity of astrocytes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 139-146. |
| [14] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [15] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||