Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (13): 2068-2075.doi: 10.12307/2024.131
Previous Articles Next Articles
Effects of aerobic exercise on adult hippocampal neurogenesis in Alzheimer’s disease mice
Zhang Yeting1, Li Chuikun2, Wei Cuilan3, Fu Yan4, Zhang Feifei4
- 1Civil Aviation Flight University of China, Guanghan 618307, Sichuan Province, China; 2College of Physical Education, Chengdu University, Chengdu 610106, Sichuan Province, China; 3Sports Institute of Chengdu University of Technology, Chengdu 610059, Sichuan Province, China; 4College of Physical Education, Southwest Minzu University, Chengdu 610041, Sichuan Province, China
-
Received:2023-02-14Accepted:2023-04-12Online:2024-05-08Published:2023-08-28 -
Contact:Fu Yan, PhD, Professor, College of Physical Education, Southwest Minzu University, Chengdu 610041, Sichuan Province, China -
About author:Zhang Yeting, PhD, Lecturer, Civil Aviation Flight University of China, Guanghan 618307, Sichuan Province, China -
Supported by:Sichuan Provincial Science and Technology Plan Project, No. 2020YFH0184 (to ZYT); Special Funding for Basic Scientific Research Business Expenses in Central Universities, No. PHD2023-003 (to ZYT)
CLC Number:
Cite this article
Zhang Yeting, Li Chuikun, Wei Cuilan, Fu Yan, Zhang Feifei. Effects of aerobic exercise on adult hippocampal neurogenesis in Alzheimer’s disease mice[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(13): 2068-2075.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
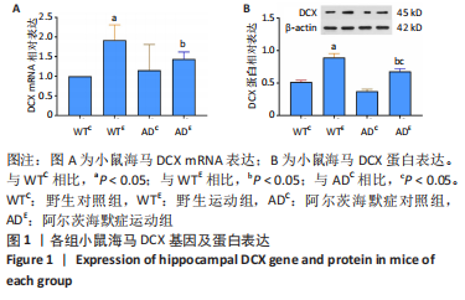
2.1 实验动物数量分析 参加实验小鼠数量共计80只,随机分为4组,每组20只,每组随机取12只小鼠进入结果分析,中途无脱落。 2.2 小鼠海马DCX的表达 图1A为小鼠海马DCX mRNA表达。野生运动组DCX mRNA表达显著高于野生对照组(P < 0.05),AD运动组DCX mRNA表达与AD对照组相比无显著差异(P=0.35);AD对照组DCX mRNA表达与野生对照组相比无显著差异,AD运动组DCX mRNA表达显著低于野生运动组(P=0.044)。图1B为小鼠海马DCX蛋白表达。野生运动组DCX蛋白表达显著高于野生对照组(P < 0.05),AD运动组DCX蛋白表达显著高于AD对照组(P < 0.05);AD对照组DCX蛋白表达与野生对照组相比无显著差异, AD运动组DCX蛋白表达显著低于野生运动组(P < 0.05)。"
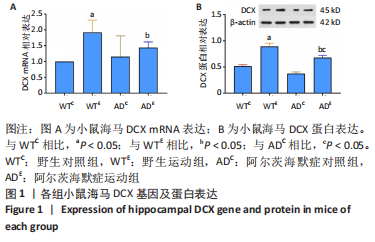
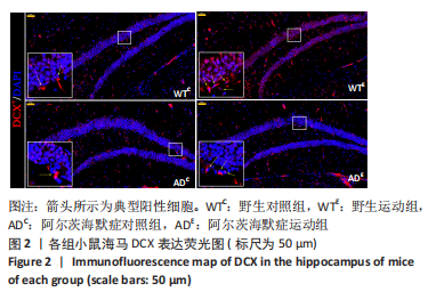
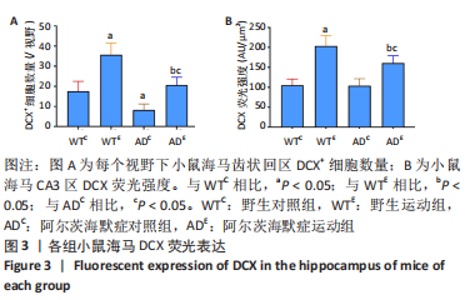
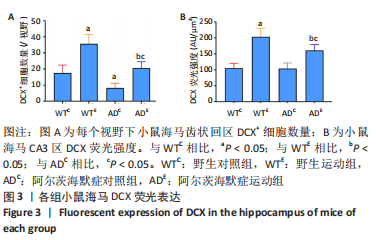
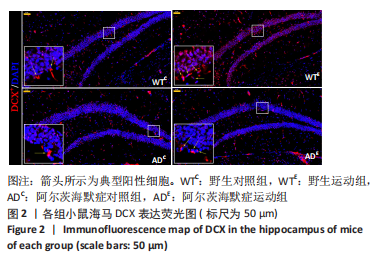
图2为小鼠海马DCX免疫荧光染色,图中蓝色代表DAPI标记的细胞核,红色代表小鼠海马齿状回区DCX免疫阳性产物表达。图3A为小鼠海马齿状回区DCX+细胞数量。野生运动组DCX+细胞数量显著高于野生对照组(P < 0.005),AD运动组DCX+细胞数量显著高于AD对照组(P < 0.005);AD对照组DCX+细胞数量显著低于野生对照组(P=0.002),AD运动组DCX+细胞数量显著低于野生运动组(P < 0.005)。图3B为小鼠海马齿状回区DCX荧光强度。野生运动组DCX荧光强度显著高于野生对照组(P < 0.005),AD运动组DCX荧光强度显著高于AD对照组(P < 0.005);AD对照组DCX荧光强度与野生对照组相比无显著差异(P=0.903),AD运动组DCX荧光强度显著低于野生运动组(P=0.001)。这表明长期有氧运动可以提高野生型小鼠及AD小鼠海马齿状回区DCX+细胞数量及DCX表达。 "
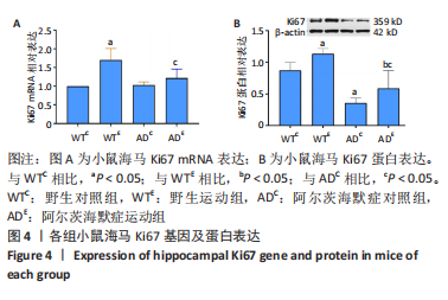
2.3 小鼠海马Ki67的表达 图4A为小鼠海马Ki67 mRNA表达。野生运动组Ki67 mRNA表达显著高于野生对照组(P < 0.000 5),AD运动组Ki67 mRNA显著高于AD对照组(P=0.039);AD对照组Ki67 mRNA表达与野生对照组相比差异不显著(P=0.829),AD运动组Ki67 mRNA表达与野生运动组相比差异不显著(P=0.051)。图4B为小鼠海马Ki67蛋白表达。野生运动组Ki67 蛋白表达显著高于野生对照组(P=0.012),AD运动组Ki67蛋白显著高于AD对照组(P=0.024);AD对照组Ki67蛋白表达显著低于野生对照组(P < 0.000 5),AD运动组Ki67蛋白表达显著低于野生运动组(P < 0.000 5)。"
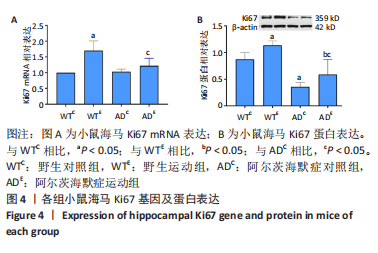
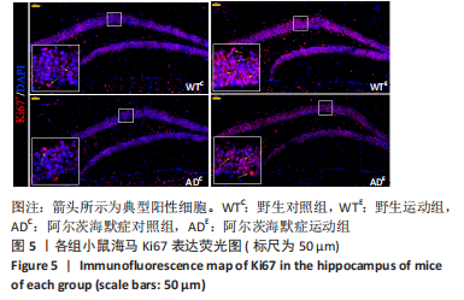
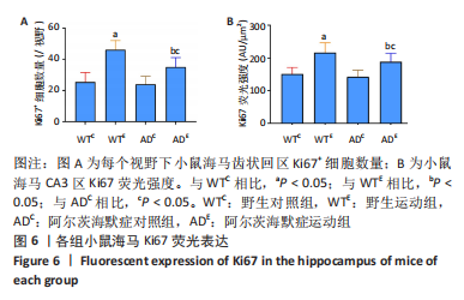
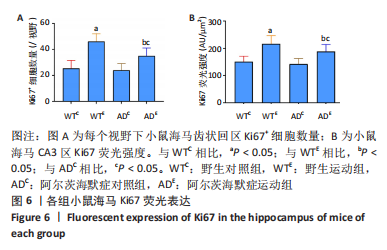
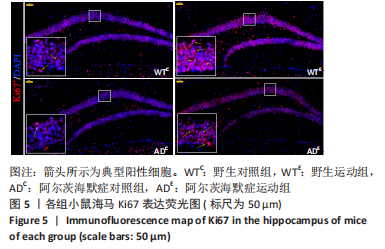
图5为小鼠海马Ki67免疫荧光染色,图中蓝色代表DAPI标记的细胞核,红色代表Ki67免疫阳性产物表达。图6A为每个视野下小鼠海马齿状回区Ki67+细胞数量。野生运动组Ki67+细胞数量显著高于野生对照组(P < 0.005),AD运动组Ki67+细胞数量显著高于AD对照组(P=0.004);AD对照组Ki67+细胞数量与野生对照组相比无显著差异(P=0.68),AD运动组Ki67+细胞数量显著低于野生运动组(P=0.003)。图6B为小鼠海马齿状回区Ki67荧光强度。野生运动组Ki67荧光强度显著高于野生对照组(P < 0.005),AD运动组Ki67荧光强度显著高于AD对照组(P=0.008);AD对照组Ki67荧光强度与野生对照组相比无显著差异(P=0.597),AD运动组Ki67荧光强度显著低于野生运动组(P=0.048)。这表明长期有氧运动可以提高野生型小鼠海马Ki67的表达,AD转基因小鼠海马Ki67的表达相较于野生型小鼠没有显著差异,而长期有氧运动能够显著提高AD小鼠海马Ki67的表达。 "
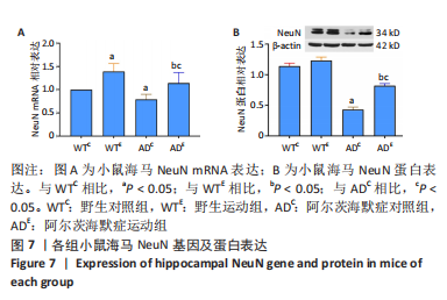
2.4 小鼠海马NeuN的表达 图7A为小鼠海马NeuN mRNA表达。野生运动组NeuN mRNA表达显著高于野生对照组(P < 0.000 5),AD运动组NeuN mRNA表达显著高于AD对照组(P=0.001);AD对照组NeuN mRNA表达显著低于野生对照组(P=0.025),AD运动组NeuN mRNA表达显著低于野生运动组(P=0.008)。图7B为小鼠海马NeuN蛋白表达。野生运动组NeuN 蛋白表达与野生对照组相比无显著差异(P=0.072),AD运动组NeuN蛋白表达显著高于AD对照组(P < 0.000 5);AD对照组NeuN蛋白表达显著低于野生对照组(P < 0.000 5),AD运动组NeuN蛋白表达显著低于野生运动组(P < 0.000 5)。"
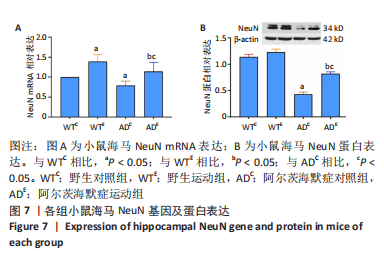
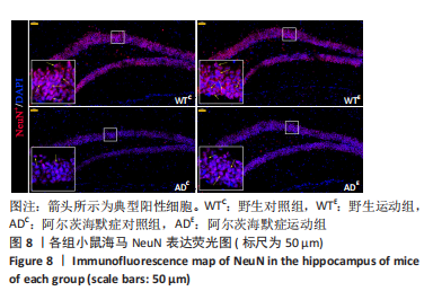
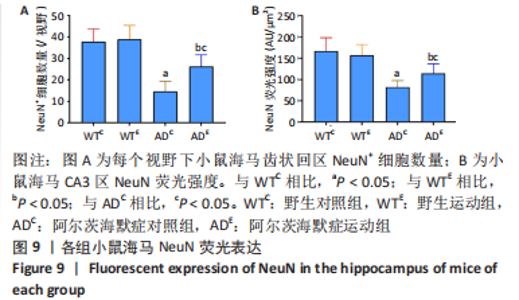
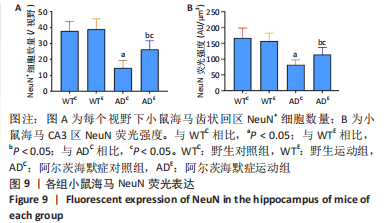
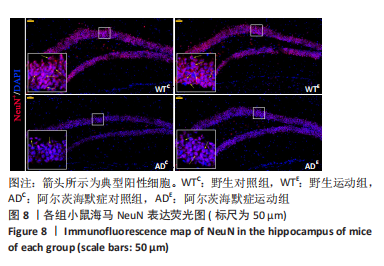
图8为小鼠海马NeuN免疫荧光染色,图中蓝色代表DAPI标记的细胞核,红色代表NeuN免疫阳性产物表达。图9A为每个视野下小鼠海马齿状回区NeuN+细胞数量。野生运动组NeuN+细胞数量与野生对照组相比无显著差异(P=0.748),AD运动组NeuN+细胞数量显著高于AD对照组(P=0.002);AD对照组NeuN+细胞数量显著低于野生对照组(P < 0.000 5),AD运动组NeuN+细胞数量显著低于野生运动组(P=0.001)。图9B为小鼠海马齿状回区NeuN荧光强度。野生运动组NeuN荧光强度与野生对照组相比无显著差异(P=0.504),AD运动组NeuN荧光强度显著高于AD对照组(P=0.032);AD对照组NeuN荧光强度显著低于野生对照组(P < 0.000 5),AD运动组NeuN荧光强度显著低于野生运动组(P=0.007)。这表明长期有氧运动并未影响到野生型小鼠海马NeuN的表达,AD转基因小鼠海马NeuN的表达相较于野生型小鼠显著降低,而长期有氧运动能够显著提高其海马齿状回区NeuN的表达。 "
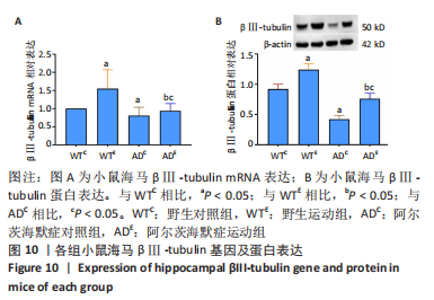
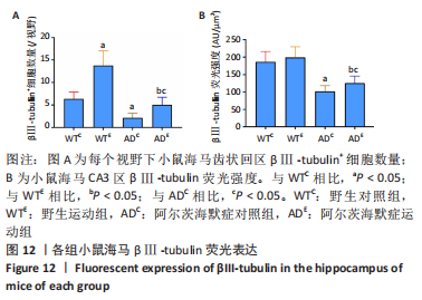
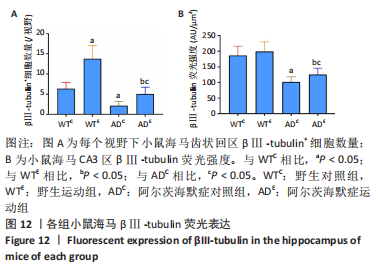
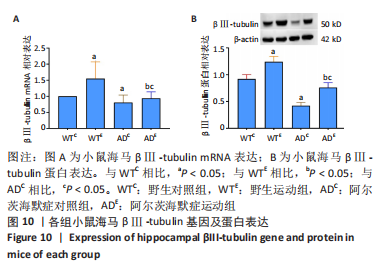
2.5 小鼠海马βⅢ-tubulin的表达 图10A为小鼠海马βⅢ-tubulin mRNA表达。野生运动组βⅢ-tubulin mRNA表达显著高于野生对照组(P=0.006),AD运动组βⅢ-tubulin mRNA表达显著高于AD对照组(P=0.02);AD对照组βⅢ-tubulin mRNA表达显著低于野生对照组(P=0.025),AD运动组βⅢ-tubulin mRNA表达显著低于野生运动组(P=0.008)。图10B为小鼠海马βⅢ-tubulin蛋白表达。野生运动组βⅢ-tubulin蛋白表达显著高于野生对照组(P < 0.000 5),AD运动组βⅢ-tubulin蛋白表达显著高于AD对照组(P < 0.000 5);AD对照组βⅢ-tubulin蛋白表达显著低于野生对照组(P < 0.000 5),AD运动组βⅢ-tubulin蛋白表达显著低于野生运动组(P < 0.000 5)。 "
| [1] GAGE FH. Mammalian neural stem cells. Science. 2000;287(5457): 1433-1438. [2] BOLDRINI M, FULMORE CA, TARTT AN, et al. Human Hippocampal Neurogenesis Persists throughout Aging. Cell Stem Cell. 2018;22(4): 589-599.e5. [3] AUGUSTO-OLIVEIRA M, ARRIFANO GPF, MALVA JO, et al. Adult Hippocampal Neurogenesis in Different Taxonomic Groups: Possible Functional Similarities and Striking Controversies. Cells. 2019;8(2):125. [4] JESSBERGER S, CLARK RE, BROADBENT NJ, et al. Dentate gyrus-specific knockdown of adult neurogenesis impairs spatial and object recognition memory in adult rats. Learn Mem. 2009;16(2):147-154. [5] DAHLIN E, ANDERSSON M, THORÉN A, et al. Effects of physical exercise and stress on hippocampal CA1 and dentate gyrus synaptic transmission and long-term potentiation in adolescent and adult Wistar rats. Neuroscience. 2019;408:22-30. [6] ZHANG C, MCNEIL E, DRESSLER L, et al. Long-lasting impairment in hippocampal neurogenesis associated with amyloid deposition in a knock-in mouse model of familial Alzheimer’s disease. Exp Neurol. 2007;204(1):77-87. [7] VOSS MW, HEO S, PRAKASH RS, et al. The influence of aerobic fitness on cerebral white matter integrity and cognitive function in older adults: results of a one-year exercise intervention. Hum Brain Mapp. 2013;34(11):2972-2985. [8] LAUTENSCHLAGER NT, COX KL, ELLIS KA. Physical activity for cognitive health: what advice can we give to older adults with subjective cognitive decline and mild cognitive impairment? Dialogues Clin Neurosci. 2019;21(1):61-68. [9] 张象,张业廷.运动改善阿尔茨海默症模型小鼠病程的剂量效应关系[J].中国组织工程研究,2021,25(17):2761-2766. [10] VERMUNT L, SIKKES SAM, VAN DEN HOUT A, et al. Duration of preclinical, prodromal, and dementia stages of Alzheimer’s disease in relation to age, sex, and APOE genotype. Alzheimers Dement. 2019;15(7):888-898. [11] MONTGOMERY W, GOREN A, KAHLE-WROBLESKI K, et al. Detection, diagnosis, and treatment of Alzheimer’s disease dementia stratified by severity as reported by caregivers in Japan. Neuropsychiatr Dis Treat. 2018;14:1843-1854. [12] HOFFMANN K, SOBOL NA, FREDERIKSEN KS, et al. Moderate-to-High Intensity Physical Exercise in Patients with Alzheimer’s Disease: A Randomized Controlled Trial. J Alzheimers Dis. 2016;50(2):443-453. [13] TOLPPANEN AM, SOLOMON A, KULMALA J, et al. Leisure-time physical activity from mid- to late life, body mass index, and risk of dementia. Alzheimers Dement. 2015;11(4):434-443.e6. [14] SOFI F, VALECCHI D, BACCI D, et al. Physical activity and risk of cognitive decline: a meta-analysis of prospective studies. J Intern Med. 2011;269(1):107-117. [15] PANZA GA, TAYLOR BA, MACDONALD HV, et al. Can Exercise Improve Cognitive Symptoms of Alzheimer’s Disease? J Am Geriatr Soc. 2018; 66(3):487-495. [16] 赵祥,魏翠兰,张业廷.神经发生和炎性环境在运动条件下的改变与调节[J].中国组织工程研究,2021,25(5):813-820. [17] JU Y, TAM KY. Pathological mechanisms and therapeutic strategies for Alzheimer’s disease. Neural Regen Res. 2022;17(3):543-549. [18] YANG X, YAO C, TIAN T, et al. A novel mechanism of memory loss in Alzheimer’s disease mice via the degeneration of entorhinal-CA1 synapses. Mol Psychiatry. 2018;23(2):199-210. [19] MORENO-JIMÉNEZ EP, FLOR-GARCÍA M, TERREROS-RONCAL J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019;25(4):554-560. [20] TOBIN MK, MUSARACA K, DISOUKY A, et al. Human Hippocampal Neurogenesis Persists in Aged Adults and Alzheimer’s Disease Patients. Cell Stem Cell. 2019;24(6):974-982.e3. [21] LEI X, WU Y, XU M, et al. Physical exercise: bulking up neurogenesis in human adults. Cell Biosci. 2019;9:74. [22] VAN PRAAG H, KEMPERMANN G, GAGE FH. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat Neurosci. 1999;2(3):266-270. [23] VAN PRAAG H, CHRISTIE BR, SEJNOWSKI TJ, et al. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc Natl Acad Sci U S A. 1999;96(23):13427-13431. [24] CASSILHAS RC, TUFIK S, DE MELLO MT. Physical exercise, neuroplasticity, spatial learning and memory. Cell Mol Life Sci. 2016;73(5):975-983. [25] 孙开宏,王坤.运动对应激的保护效应与成年神经发生的关系[J].中国康复医学杂志,2018,33(11):1374-1377. [26] LIANG YY, ZHANG LD, LUO X, et al. All roads lead to Rome - a review of the potential mechanisms by which exerkines exhibit neuroprotective effects in Alzheimer’s disease. Neural Regen Res. 2022;17(6):1210-1227. [27] CLARK PJ, BRZEZINSKA WJ, THOMAS MW, et al. Intact neurogenesis is required for benefits of exercise on spatial memory but not motor performance or contextual fear conditioning in C57BL/6J mice. Neuroscience. 2008;155(4):1048-1058. [28] CHOI SH, BYLYKBASHI E, CHATILA ZK, et al. Combined adult neurogenesis and BDNF mimic exercise effects on cognition in an Alzheimer’s mouse model. Science. 2018;361(6406):eaan8821. [29] LAFENÊTRE P, LESKE O, MA-HÖGEMEIE Z, et al. Exercise can rescue recognition memory impairment in a model with reduced adult hippocampal neurogenesis. Front Behav Neurosci. 2010;3:34. [30] PEREIRA AC, HUDDLESTON DE, BRICKMAN AM, et al. An in vivo correlate of exercise-induced neurogenesis in the adult dentate gyrus. Proc Natl Acad Sci U S A. 2007;104(13):5638-5643. [31] GRIFFIN ÉW, MULLALLY S, FOLEY C, et al. Aerobic exercise improves hippocampal function and increases BDNF in the serum of young adult males. Physiol Behav. 2011;104(5):934-941. [32] SZYMURA J, KUBICA J, WIECEK M, et al. The Immunomodulary Effects of Systematic Exercise in Older Adults and People with Parkinson’s Disease. J Clin Med. 2020;9(1):184. [33] ERICKSON KI, VOSS MW, PRAKASH RS, et al. Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci U S A. 2011;108(7):3017-3022. [34] CHAPMAN SB, ASLAN S, SPENCE JS, et al. Shorter term aerobic exercise improves brain, cognition, and cardiovascular fitness in aging. Front Aging Neurosci. 2013;5:75. [35] YANG C, MOORE A, MPOFU E, et al. Effectiveness of Combined Cognitive and Physical Interventions to Enhance Functioning in Older Adults With Mild Cognitive Impairment: A Systematic Review of Randomized Controlled Trials. Gerontologist. 2020;60(8):633-642. |
| [1] | Wang Ji, Zhang Min, Li Wenbo, Yang Zhongya, Zhang Long. Effect of aerobic exercise on glycolipid metabolism, skeletal muscle inflammation and autophagy in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1200-1205. |
| [2] | Li Longyang, Zhang Songjiang, Zhao Xianmin, Zhou Chunguang, Gao Jianfeng. Electroacupuncture intervention on the proliferation and differentiation of hippocampal neurons and oligodendrocytes in Alzheimer’s disease model mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1029-1035. |
| [3] | Sun Yuan, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Zhang Yan. Effects of early and late aerobic exercise on right heart failure induced by monocrotaline in rats with pulmonary hypertension [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 177-185. |
| [4] | Wang Jingfeng, Wen Dengtai, Wang Shijie, Gao Yinghui. Atg-mediated autophagy, exercise and skeletal muscle aging [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 295-301. |
| [5] | Tang Liang, Wang Hexia, Wang Qingbo, Pi Yihua, Zhang Yan. Aerobic exercise modulates mitochondrial quality control system to reverse cardiac pathological remodeling in aging rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2534-2541. |
| [6] | Han Weina, Xu Xiaoqing, Shi Jinning, Li Xinru, Cai Hongyan. Prediction and validation of potential targets for the glucagon-like peptide-1 receptor agonist in the treatment of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2568-2573. |
| [7] | Wu Yuzhen, Sun Qing, Liu Xia, Zhou Yu, Jin Qiguan. Variation in renal function of type 2 diabetic rats undergoing aerobic exercise [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2145-2151. |
| [8] | Deng Longfei, Zhang Yeting, Fu Yan. Aerobic exercise inhibits neuroinflammation and alleviates cognitive impairment in Alzheimer’s disease model mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2209-2214. |
| [9] | Long Qingxi, Zhang Pingshu, Liu Qing, Ou Ya, Zhang Lili, Yuan Xiaodong. Single-cell RNA sequencing reveals the heterogeneity of astrocytes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 139-146. |
| [10] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [11] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [12] | Li Xinru, Chai Shifan, Li Weiran, Cai Hongyan, Ye Yucai, Li Shuo, Hou Meng, Wang Zhaojun. Bioinformatics analysis and experimental validation of genes related to the pathogenesis of Alzheimer's disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5653-5658. |
| [13] | Tang Lu, Lin Haiqi. The power-law model-based analysis of aerobic exercise against Alzheimer’s disease in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5695-5700. |
| [14] | Du Jia, Fu Yan, Fan Jia, Zhou Miaorong, Zhang Yeting. Aerobic exercise intervenes with beta-amyloid 1-42 induced changes in hippocampal synaptic structure and proteins in a rat model of Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5256-5262. |
| [15] | Li Mengdi, Li Na, Guo Minfang, Meng Tao, Yu Jingwen, Li Yanbing, Ma Cungen, Yu Jiezhong. Eriodictyol improves cognitive function of 5×FAD mice by regulating Nogo-A/NgR/ROCK2 signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(32): 5097-5102. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||