Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (2): 307-313.doi: 10.12307/2023.052
Previous Articles Next Articles
Mechanism and implication of angiogenesis in osteoarthritis
Liu Yinghong1, Yi Yating2
- 1Jinjiang Clinic, West China Hospital of Stomatology, Sichuan University, Chengdu 610011, Sichuan Province, China; 2Department of Orthodontics, West China Hospital of Stomatology, Sichuan University, National Key Laboratory of Oral Disease Research, National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China
-
Received:2022-01-07Accepted:2022-03-07Online:2023-01-18Published:2022-06-20 -
Contact:Yin Yating, MD, Department of Orthodontics, West China Hospital of Stomatology, Sichuan University, National Key Laboratory of Oral Disease Research, National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China -
About author:Liu Yinghong, MD, Attending physician, Jinjiang Clinic, West China Hospital of Stomatology, Sichuan University, Chengdu 610011, Sichuan Province, China -
Supported by:the National Natural Science Foundation of China (Youth Foundation), No. 82101059 (to YYT); Research Funding from West China School/Hospital of Stomatology, Sichuan University, No. RCDWJS2022-11 (to YYT)
CLC Number:
Cite this article
Liu Yinghong, Yi Yating. Mechanism and implication of angiogenesis in osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 307-313.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
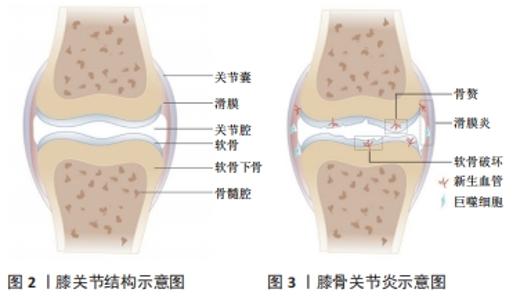
2.1 正常滑膜关节的解剖及血管分布 膝关节为典型的滑膜关节(见图2),也是骨关节炎的好发部位之一。膝关节外部为关节囊,内部为关节软骨,二者之间通过滑膜相连;上下两侧关节软骨之间为半月板,关节软骨的下方为软骨下骨,其下有髓腔来容纳血管,为周围组织提供营养。正常情况下,关节软骨和半月板的内2/3处均无血管结构,其营养供给和氧气来源依赖于邻近富含毛细血管网的组织结构(如骨膜和滑膜),而关节腔内的负压及关节的运动恰好方便了营养物质的传输[7]。关节软骨和半月板处之所以无血管结构,可能与应力承载有关,血管侵入会降低其应力承载能力。同时由于血管结构的缺乏,一旦关节软骨遭到破坏需要重新修复便十分困难[8]。 2.2 血管生成的病理性改变参与骨关节炎的发生发展过程 血管生成几乎参与了骨关节炎各个过程(见图3),从滑膜出现新生血管引起的滑膜炎,到骨软骨联合处出现新生血管引起的软骨破坏,甚至骨赘形成,都与血管生成密切相关[9],研究时间脉络图见图4。"
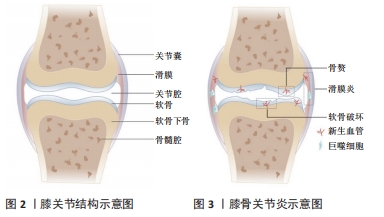
2.2.1 滑膜炎 滑膜炎是骨关节炎的主要临床症状之一,可见于约1/3的有症状的骨关节炎患者。滑膜炎的组织学表现为滑膜增生、巨噬细胞浸润和血管生成增加[16],其中,血管生成的量和巨噬细胞浸润程度均与滑膜炎的严重程度呈正相关,而炎症反过来也能够进一步促进血管生成[17-18]。其主要机制为:滑膜中新生的血管一方面为炎症细胞提供了扩散渠道,另一方面向炎症组织持续供应营养物质和氧气[19];巨噬细胞能够分泌血管内皮生长因子(vascular endothelial growth factor,VEGF),并刺激内皮细胞、成纤维细胞等分泌促进血管生成的细胞因子[20],形成“炎症-血管生成”的正反馈,促进滑膜炎的发生发展。 2.2.2 软骨破坏及退化 骨关节炎患者的软骨破坏及退化一方面是由于关节软骨本身蛋白多糖缺失和降解造成[21],另一方面与软骨下骨中新生的血管入侵密切相关,新生血管会导致软骨细胞肥大和基质中矿物质降解[22]。炎症细胞可分泌大量VEGF,减少软骨细胞内胶原蛋白和蛋白聚糖的表达,削弱基质形成[23];同时,VEGF能够刺激软骨细胞和血管内皮细胞分泌一氧化氮(NO),引起软骨细胞的凋亡,进而导致软骨退化[24]。更重要地,VEGF能直接促进软骨下骨及软骨内血管新生。有证据表明,从软骨下骨自下而上新生的血管相较于从滑膜组织自上而下新生的血管在软骨破坏中发挥更重要的作用[25]。在骨软骨联合处,破骨细胞首先穿过潮线入侵到无血管未钙化的软骨中,形成一个通向软骨的通道,新生的血管和感觉神经可沿该通道进入到软骨中,促进软骨的钙化[26]。 2.2.3 骨赘形成 随着血管在软骨内的侵入,Ⅰ型、X型胶原蛋白、成骨细胞和破骨样细胞在血管周围聚集[27-28]。破骨样细胞降解软骨,为新生血管侵入提供通道,成骨细胞促进软骨中钙化的骨组织生成。同时,新生血管可促进成骨前体细胞向关节表面移动,在有炎症的骨关节边缘形成类似于骺板的骨赘结构。另一方面,骨赘中存在过度肥大的软骨细胞,能够进一步产生VEGF,促进血管生成并导致软骨细胞凋亡[29]。 2.2.4 关节疼痛 在生理和病理条件下,神经常伴随着血管生长,血管周围的细胞可以刺激神经生长并且帮助新的轴突达到指定位置[30]。因此,感觉神经伴随血管入侵软骨与关节疼痛有一定相关性。与此同时,血管以及其周围的神经也可以通过纤维软骨联合入侵本无血管的半月板,以削弱半月板的功能,并引起关节区的疼痛和功能障碍[31]。骨关节炎患者中高表达的VEGF和转化生长因子β在促进血管生成的同时也促进了神经生长[25]。 2.3 骨关节炎中血管生成的调控因素 "

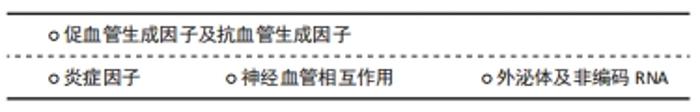
2.3.1 促血管生成因子及抗血管生成因子 滑膜关节的稳态维持依赖于促血管生成因子和抗血管生成因子的平衡。促血管生成因子表达增加、该平衡被打破时,将直接导致病理性血管生成增加,启动或促进骨关节炎的发生。促血管生成因子可由软骨细胞、滑膜、软骨下骨、骨髓腔和骨软骨联合产生,主要包括前列腺素、一氧化氮、调节肽、多种细胞因子、趋化因子和生长因子,其中,VEGF在骨关节炎患者病理性血管生成中发挥核心作用[32]。VEGF可与内皮细胞上的受体VEGFR-2结合,刺激内皮细胞的增殖、迁徙、成熟和存活,促进血管生成。值得注意的是,内皮细胞上亦存在VEGF的另一受体——VEGFR-1,该受体是一种诱骗受体,可减少VEGF和VEGFR-2的结合率,减少血管的生成[33]。但研究表明,外源性刺激促进VEGFR-1激活的同时,VEGFR-2也相应激活[34];同时,VEGFR-1可募集和激活巨噬细胞,继而促进VEGF的产生。 血小板衍生生长因子BB由骨髓内的单核破骨前体细胞分泌,在血管生成-骨形成偶联过程中发挥重要作用。新近研究发现其亦可作为骨关节炎进展中的一种促血管生成因子。在小鼠骨关节炎模型早期即可见血小板衍生生长因子BB分泌大量增加,促进软骨下骨血管形成及神经长入;敲除血小板衍生生长因子BB可明显减弱关节损伤症状,而过表达血小板衍生生长因子BB后,可见软骨下骨血管大量增生、侵入关节软骨,继而软骨下骨成骨活性增强,关节软骨钙化[35],提示血小板衍生生长因子BB在骨关节炎血管生成及骨软骨改变中的地位十分关键。 抗血管生成因子主要由软骨细胞产生,包括肌钙蛋白Ⅰ、血小板反应蛋白1(thrombospondin 1,TSP-1)、金属蛋白酶组织抑制剂、纤溶酶原激活物抑制剂1等。骨关节炎发生时,这些抑制剂在关节软骨的表达亦上调,但其作用局限于浅表软骨细胞局部,无法真正发挥抑制血管生成的作用[30]。 2.3.2 炎症因子 如前所述,炎症与血管生成可形成正反馈效应,炎症因子大多通过促进VEGF的产生从而达到促进血管生成的情况。研究显示,炎症递质结缔组织生长因子可以激活磷脂酰肌醇3-激酶(phosphatidylinositol 3 kinae,PI3K),蛋白激酶B(protein kinase B,PKB或AKT),细胞外信号调节激酶(extracellular signal regulated kinase,ERK)和核转录因子κB (nuclear factor kappaB,NF-κB)/ELK1通路,上调miR-210,从而抑制GPD1L的表达和脯氨酰羟化酶-2的活性,最终促进VEGF表达引起骨关节炎中血管生成增加[36]。肝细胞生长因子(HGF)也可以通过c-Met受体通路促进人滑膜成纤维细胞中VEGF的表达,进而促进血管生成[37]。炎症因子白细胞介素6和白细胞介素1β同样可以诱导VEGF在细胞核内转录来增加VEGF表达水平[38]。肿瘤坏死因子α可促进新发现的病理性血管促进因子——富亮氨酸-2-糖蛋白1的表达增加软骨下骨血管生成和改变间充质干细胞的迁徙[14]。与此同时,这些炎症因子通过新生血管被运输至被破坏的骨软骨联合处,维持炎症状态,进一步促进血管生成[39]。 2.3.3 神经血管相互作用 神经生长因子是一种能够调节神经生长、分化、修复和存活的介质[40]。在骨关节炎患者的血液、滑膜液和软骨细胞中均能检测到神经生长因子及其高亲和力受体原肌球蛋白受体激酶A(tropomyosin-receptor kinase A,TrkA)表达量升高[41]。越来越多研究表明,神经生长因子与血管生成有一定相关性,随着血管中神经生长因子的表达增加,骨软骨联合处血管密度也随之增加[42]。 神经生长因子促进血管生成的原因有几方面,一方面是神经生长因子与内皮细胞表面的TrkA直接结合刺激内皮细胞的增殖、迁移和毛细血管形成;其次神经生长因子可与感觉神经末梢的TrkA结合促进血管生成神经肽(SP和CGRP-1)的释放,它们与内皮细胞表面的NK1受体和CGRPR结合刺激血管生成;与此同时,神经生长因子与巨噬细胞上的TrkA受体结合后会促进VEGF的释放,从而促进血管生成[30]。另一方面,体外实验表明神经生长因子可通过人体软骨细胞中PI3K/Akt和ERK/丝裂原活化蛋白激酶(mitogen-activated protein kinases,MAPKs)通路促进成纤维细胞生长因子2的表达,而成纤维细胞生长因子2可以刺激内皮细胞的迁移和侵袭从而在体内促进血管的生成[43]。虽然神经生长因子对于血管生成的促进作用较明显,但是神经生长因子可同时与内皮细胞表面的p75结合,诱导内皮细胞凋亡并促进血管的退化[44]。因此,神经生长因子对血管生成具有双向作用。 2.3.4 外泌体及非编码RNA 外泌体是一种由大多数真核细胞(例如骨髓间充质干细胞、成骨细胞、破骨细胞等)形成并分泌到细胞外的纳米级小囊泡,它内部含有丰富的生物活性分子,可以通过配体受体的相互作用、内吞作用、直接膜融合和介导信号通路来完成细胞间的信息传递[45]。值得注意的是,外泌体中存在非编码RNA。新近研究显示,外泌体非编码RNA与骨代谢相关疾病息息相关[46]。 非编码RNA是一群能从基因组上转录而来但不翻译成蛋白质的RNA[47],根据其长度可以划分为3类:小于50 nt[包括微小RNA(miRNA)、小干扰RNA(siRNA)、新型非编码小RNA(piRNA)],长度在50-500 nt之间[包括核糖体RNA(rRNA),转运RNA(tRNA)],长度大于500 nt[包括长链非编码RNA(long noncoding RNA,lncRNA)、环状RNA(circRNA)][48]。 微小RNA是一群高度保守的内源性非编码RNA,它可以结合到3’-UTR位点,降解目标mRNA,或阻断翻译过程,从而影响细胞的生物学功能。微小RNA可以调控细胞外基质的降解或软骨细胞的凋亡,引起骨关节炎的发生。近年来,亦有研究显示微小RNA可以通过调节VEGF的表达介导关节区血管生成,进而影响骨关节炎进程[15]。软骨细胞内miR-214-3p可靶向结合-酪氨酸激酶受体B(TrkB)的mRNA。对骨关节炎患者软骨细胞的RT-qPCR结果显示,其miR-214-3p表达明显减少,其关节区TrkB表达水平明显升高,从而刺激VEGF的旁分泌,通过激活内皮细胞PI3K/Akt通路蛋白促进内皮细胞的迁移和血管生成[15]。miR-210是一类重要的在缺氧环境下产生的微小RNA,在肿瘤中,其表达受低氧诱导因子2的调控,能够通过限制血管生成抑制因子EFNA3和PTP1B的表达促进血管形成。XIE等[49]研究发现,骨关节炎患者关节滑液中miR-210的水平与VEGF的表达水平成正相关,提示miR-210可能通过促进VEGF的表达促进血管生成及早期骨关节炎的进展。 lncRNA可以在mRNA转录前、转录中、转录后通过折叠成特殊构象或与DNA、RNA、蛋白质互作对基因表达进行调控。lncRNA-MEG3是一种印迹基因,WEI等[50]研究显示,骨关节炎患者关节软骨中MEG3显著下调,而VEGF表达明显提高。同时,MEG3可促进p53的转录,从而通过结合VEGF启动子上的转录位点Sp1抑制VEGF的表达[51-52]。因此,在骨关节炎患者的软骨中,MEG3的下调可能通过该途径间接促进VEGF的产生及软骨内血管新生。lncRNA H19是一种能够调节软骨细胞和成骨细胞的生长以及细胞合成代谢过程的基因[53]。有研究表明,lncRNA H19在骨关节炎患者的外周血液中高表达,有望成为诊断骨关节炎的新指标[54]。而骨关节炎患者体内H19高表达的机制在体外实验中显示,外泌体中的lncRNA H19能吸引大量miR-106来促进血管生成素1(Angpt1)的表达,从而激活内皮细胞中的lnc-H19/Tie2-NO信号通路,产生NO促进内皮血管生成[55]。 2.4 血管生成抑制剂在骨关节炎的治疗研究进展 "


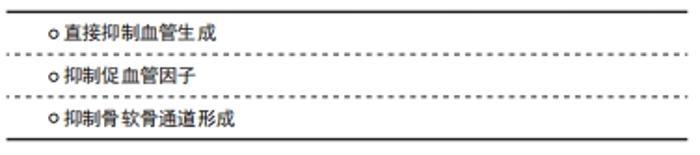
血管生成抑制剂可以大致分为3类:第一类是直接作用于血管内皮细胞抑制血管生成的,第二类是消除刺激血管生成的因素来间接抑制血管生成的,第三类是抑制基质退化以及骨软骨通道的形成(这是血管新生的必要条件)来间接抑制血管生成。如前所述,血管生成与骨关节炎患者的组织改变和疼痛程度具有一定相关性,因此血管生成抑制剂成为目前治疗骨关节炎的研究热点之一。 2.4.1 直接抑制血管生成 常见的直接抑制血管生成的物质有血小板反应蛋白TSP-1和PPI-2458,TSP-1是一种存在于关节中的糖蛋白,可由软骨细胞分泌。TSP-1可直接与VEGF结合,或与VEGF竞争性结合内皮细胞表面硫酸肝素蛋白聚糖结合位点,抑制内皮细胞的增殖和迁移同时导致内皮细胞的凋亡[56]。在骨关节炎动物模型中,关节腔内注射包裹TSP-1的腺病毒能够减轻滑膜炎症、软骨退化和骨赘形成等症状[57]。血管生成抑制剂 PPI-2458是夫马菌素类似物,能够抑制内皮细胞的增殖和细胞周期进展[58]。骨关节炎动物模型中使用PPI-2458可以减轻滑膜炎及结构损伤,缓解疼痛,疼痛的减少可能与其抑制骨软骨通道里的血管神经生成的功能有关[16]。近期有研究发现,中药黄芩素被注射入关节腔中可以抑制内皮细胞减少血管新生和抑制滑膜细胞的增殖[59]。XIONG等[60]研究发现白藜芦醇可通过下调VEGFA和Ang-1的表达来抑制血管的异常增殖。 2.4.2 抑制促血管因子 VEGF是常见的促血管生成因子,因此抑制VEGF的表达可间接抑制血管生成。贝伐单抗是一种已被应用于肿瘤治疗的VEGF活性抑制药物,也是目前唯一能够促进软骨修复的抗血管生成药物。动物实验表明,贝伐单抗可以加速软骨修复,促进透明软骨的形成[61]。NAGAI等[62]在兔骨关节炎模型上低剂量注射贝伐单抗后发现,软骨退化、骨赘形成和滑膜炎的症状均有减轻,以关节腔内注射效果相对最佳,且低剂量的疗效优于高剂量。LI等[63]利用兔骨关节炎模型证实,贝伐单抗主要通过抑制VEGF和基质金属蛋白酶1的表达、减少血管生成等减轻关节局部炎症。REMON等[64]病例报告显示,使用克唑替尼(治疗非小细胞肺癌药物)抑制肝细胞生长因子(HGF)通路16个月内能够通过减少VEGF的表达减轻骨关节炎的临床症状。除直接抑制VEGF活性外,抑制VEGF受体亦能实现VEGF的阻断。sFlt-1是一种内生性可溶受体,可以模拟VEGFR-1与VEGF结合,发挥VEGFR-1抑制血管的作用,从而减轻骨关节炎的症状[65]。 2.4.3 抑制骨软骨通道形成 抑制骨软骨通道的形成也是阻止软骨内血管生成的一个有力干预靶点。双膦酸盐可以抑制破骨细胞或破软骨细胞的活动,抑制软骨下骨代谢,减少软骨下骨丢失、软骨病和骨软骨通道的形成[66]。然而,双膦酸盐治疗骨关节炎患者的初步临床研究结果并不十分乐观,其在减少骨关节炎的症状或体征方面的临床疗效不佳,原因可能是现有临床试验所用药物剂量不足,没有充分抑制骨改建,或纳入的参与者没有软骨下骨的病理活动[67]。MOBASHERI等[68]研究表明,姜黄素和白藜芦醇可以阻断白细胞介素1β介导的蛋白多糖降解和软骨细胞的凋亡,从而为阻断血管生成的通道形成提供可能性。青蒿酯能够通过调节OPG-RANKL轴抑制破骨细胞介导的骨吸收,并减少软骨下骨CD31(hi)Emcn(hi)血管的生成[69]。WANG等[70]在体外实验中发现,甜菜碱具有抑制破骨细胞形成、减少软骨下骨吸收的效果,该作用主要是通过对活性氧和MAPK通路的下调实现的;体内实验同样证实,甜菜碱对破骨细胞形成具有抑制作用,有利于维护关节软骨的完整性。此外,甜菜碱能够通过抑制血管形成相关酶(例如基质金属蛋白酶2)的表达直接抑制软骨下骨血管形成[71-72]。亦有研究发现,甜菜碱可抑制白细胞介素1β的产生、抑制炎症小体的激活[72-73],从而产生一定的抗炎作用。因此,甜菜碱可能成为骨关节炎治疗的潜在有效药物之一。 干细胞治疗是骨相关疾病治疗的新近研究热点。骨髓间充质干细胞在软骨形成过程中具有重要作用,加之其易分离获取,因此骨髓间充质干细胞局部注射有可能成为骨关节炎患者软骨损伤修复的有效手段。PRASADAM等[74]将骨髓间充质干细胞与关节软骨细胞以1∶1进行混合培养后植入免疫缺陷小鼠皮下的软骨修复体,3周后可见软骨损伤修复良好,表面光滑,无血管侵入;而基质胶组或单纯骨髓间充质干细胞组均可见血管入侵;相关分子定量检测显示,骨髓间充质干细胞与软骨细胞混合培养组软骨形成相关基因表达量更高,血管生成和软骨肥大相关基因表达量更低,且显示出更好的软骨保护及软骨再生能力。该结果提示,骨髓间充质干细胞与软骨细胞混合培养产生的作用可能是多维度的,骨髓间充质干细胞在体内除直接参与软骨形成,亦可能通过调节局部微环境(如抑制血管入侵)发挥软骨保护作用。 值得注意的是,尽管血管生成在骨关节炎中有许多不良影响,但它在机体生理过程(如孕期和儿童的生长发育、女性生殖周期和伤口愈合等)中也有极其重要的作用。FDA批准使用的抗血管生成药物目前主要局限于晚期癌症的治疗,因为其潜在的好处可能大于风险。而临床上抗血管生成用于治疗骨关节炎的研究主要集中于体外实验和动物实验,临床应用较少且效果尚不明确。因此,对于骨关节炎这一慢性非致命性疾病,如何减少抗血管生成药物的全身应用、提高血管生成抑制剂在局部的疗效、抑制病理性而非生理性血管生成将是未来研究的重要方向。 血管生成抑制剂在骨关节炎中的部分研究汇总见表1。 "

| [1] MARTEL-PELLETIER J, BARR AJ, CICUTTINI FM, et al. Osteoarthritis. Nat Rev Dis Primers. 2016;2:16072. [2] STAGNIC, ROCCHI M, MAZZOTTA A, et al. Randomised, double-blind comparison of a fixed co-formulation of intr a-articular polyn ucleotides and hyaluronic acid versus hyaluronic acid alone in the treatment of knee osteoarthritis: two-year follow-up. BMC Musculoskelet Disord. 2021;22:773. [3] CADET C, MAHEU E, The French AGRHUM Group (Association Geriatric and RHeUMatology). Non-steroidal anti-inflammatory drugs in the pharmacological management of osteoarthritis in the very old: prescribe or proscribe. Ther Adv Musculoskelet Dis. 2021;13: 1759720X211022149. [4] 陈朝蔚,陈永强.骨关节炎血管生成与炎症的关系[J].国际骨科学杂志,2007,28(1):33-35. [5] ASHRAF S, WIBBERLEY H, MAPP PI, et al. Increased vascular penetration and nerve growth in the meniscus: a potential source of pain in osteoarthritis. Ann Rheum Dis. 2011;70:523-529. [6] FRANSÈS RE, MCWILLIAMS DF, MAPP PI, et al. Osteochondral angiogenesis and increased protease inhibitor expression in OA. Osteoarthritis Cartilage. 2010;18:563-571. [7] STEVENSC CR, BLAKE DR, MERRY P, et al. A comparative study by morphometry of the microvasculature in normal and rheumatoid synovium. Arthritis Rheum. 1991;34(12):1508-1513. [8] TOH WS, LAI RC, HUI JHP, et al. MSC Exosome as a Cell-free MSC Therapy for Cartilage Regeneration: Implications for Osteoarthritis Treatment. Semin Cel Dev Biol. 2017;67:56-64. [9] ASHRAF S, MAPP PI, WALSH DA. Contributions of angiogenesis to inflammation, joint damage, and pain in a rat model of osteoarthritis. Arthritis Rheum. 2011;63(9):2700-2710. [10] BROWN R, WEISS J, TOMLINSON I, et al. Angiogenic factor from synovial fluid resembling that from tumours. Lancet. 1980;315(8170):682-685. [11] HAYWOOD L, MCWILLIAMS DF, PEARSON CI, et al. Inflammation and angiogenesis in osteoarthritis. Arthritis Rheum. 2003;48(8):2173-2177. [12] WALSH DA, BONNET CS, TURNER EL, et al. Angiogenesis in the synovium and at the osteochondral junction in osteoarthritis. Osteoarthritis Cartilage. 2007;15(7):743-751. [13] ASHRAF S, WIBBERLEY H, MAPP PI, et al. Increased vascular penetration and nerve growth in the meniscus: a potential source of pain in osteoarthritis. Ann Rheum Dis. 2011;70:523-529. [14] WANG Y, XU J, ZHANG X, et al. TNF-α-induced LRG1 promotes angiogenesis and mesenchymal stem cell migration in the subchondral bone during osteoarthritis. Cell Death Disease. 2017;8(3):e2715. [15] XIAO P, ZHU X, SUN J, et al. Cartilage tissue miR-214-3p regulates the TrkB/ShcB pathway paracrine VEGF to promote endothelial cell migration and angiogenesis. Bone. 2021;151:116034. [16] BONDESON J, BLOM AB, WAINWRIGHT, et al. The role of synovial macrophages and macrophage‐produced mediators in driving inflammatory and destructive responses in osteoarthritis. Arthritis Rheum. 2010;62:647-657. [17] PIROSA A, TANKUS EB, MAINARDI A, et al. Modeling In Vitro Osteoarthritis Phenotypes in a Vascularized Bone Model Based on a Bone-Marrow Derived Mesenchymal Cell Line and Endothelial Cells. Int J Mol Sci. 2021;22:9581-9597. [18] KO JY, LEE MS, LIAN WS, et al. MicroRNA-29a Counteracts Synovitis in Knee Osteoarthritis Pathogenesis by Targeting VEGF. Scientific Reports. 2017;7(1):3584. [19] KONISTI S, KIRIAKIDIS S, PALEOLOG EM. Hypoxia--a key regulator of angiogenesis and inflammation in rheumatoid arthritis. Nat Rev Rheumatol. 2012;8(3):153-162. [20] ASHRAF S, WALSH DA. Angiogenesis in osteoarthritis. Curr Opin Rheumatol. 2008;20(5):573-580. [21] GOLDRING MB. Articular Cartilage Degradation in Osteoarthritis. Hss J. 2012;8(1):7-9. [22] LI B, GUAN G, MEI L, et al. Pathological mechanism of chondrocytes and the surrounding environment during osteoarthritis of temporomandibular joint. J Cell Mol Med. 2021;25:4902-4911. [23] HAMILTON JL, NAGAO M, LEVINE BR, et al. Targeting VEGF and Its Receptors for the Treatment of Osteoarthritis and Associated Pain. J Bone Miner Res. 2016;31(5):911-924. [24] AKARAPHUTIPORN E, SUNAGA T, BWALYA EC, et al. In Insight into the Role of Apoptosis and Autophagy in Nitric Oxide-Induced Articular Chondrocyte Cell Death. Cartilage. 2021;13(2_suppl):826S-838S. [25] HU Y, CHEN X, WANG S, et al. Subchondral bone microenvironment in osteoarthritis and pain. Bone Res. 2021;9(1):20. [26] SURI S, GILL SE, MASSENAD CS, et al. Neurovascular invasion at the osteochondral junction and in osteophytes in osteoarthritis. Ann Rheum Dis. 2007;66(11):1423-1428. [27] HE Y, SIEBUHR AS, BRANDT-HANSEN NU, et al. Type X collagen levels are elevated in serum from human osteoarthritis patients and associated with biomarkers of cartilage degradation and inflammation. BMC Musculoskelet Disord. 2014;15:309. [28] INTEMANN J, DE GORTER DJJ, NAYLOR AJ, et al. Importance of osteocyte-mediated regulation of bone remodelling in inflammatory bone disease. Swiss Med Wkly. 2020;150:w20187. [29] WANG S, ZHOU C, ZHENG H, et al. Chondrogenic progenitor cells promote vascular endothelial growth factor expression through stromal-derived factor-1. Osteoarthritis Cartilage. 2016;25(5):742-749. [30] MAPP PI, WALSH DA. Mechanisms and targets of angiogenesis and nerve growth in osteoarthritis. Nat Rev Rheumatol. 2012;8:390-398. [31] ANDREWS S, RATTNER JB, ABUSARA Z, et al. Tie‐fibre structure and organization in the knee menisci. J Anatomy. 2014;224(5):531-537. [32] JANSEN H, MEFFERT RH, BIRKENFELD F, et al. Detection of vascular endothelial growth factor (VEGF) in moderate osteoarthritis in a rabbit model. Ann Anat. 2012;194(5):452-456. [33] SUGIMOTO M, KNODO M, KAMIMOTO Y, et al. Changes in VEGF-related factors are associated with presence of inflammatory factors in carbohydrate metabolism disorders during pregnancy. PloS one. 2019;14(8):e0220650. [34] CYDZIK M, ABDUL-WAHID A, PARK S, et al. Slow binding kinetics of secreted protein, acidic, rich in cysteine-VEGF interaction limit VEGF activation of VEGF receptor 2 and attenuate angiogenesis. FASEB J. 2015;29(8):3493-3505. [35] SU W, LIU G, LIU X, et al. Angiogenesis stimulated by elevated PDGF-BB in subchondral bone contributes to osteoarthritis development. JCI Insight. 2020;5(8):e135466. [36] LIU SC, CHUANG SM, HSU CJ, et al. Ctgf increases vascular endothelial growth factor-dependent angiogenesis in human synovial fibroblasts by increasing mir-210 expression. Cell Death Dis. 2014;5: e1485. [37] LIN YM, HUANG YL, FONG YC, et al. Hepatocyte Growth Factor Increases Vascular Endothelial Growth Factor-A Production in Human Synovial Fibroblasts through c-Met Receptor Pathway. PLOS ONE. 2012; 7(11):es50924. [38] ZHAO H, LIU S, MA C, et al. Estrogen-related receptor γ induces angio- genesis and extracellular matrix degradation of temporomandibular joint osteoarthritis in rats. Front Pharmacol. 2019;10:1290. [39] PESESSE L, SANCHEZ C, HENROTIN Y. Osteochondral plate angio- genesis: a new treatment target in osteoarthritis. Joint bone spine. 2011;78(2):144-149. [40] HUANG EJ, REICHARDT LF. Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci. 2001;24:677e736. [41] MONTAGNOLI C, TIRIBUZI R, CRISPOLTONI L, et al. beta-NGF and beta-NGF receptor upregulation in blood and synovial fluid in osteoarthritis. Biol Chem. 2017;398:1045e54. [42] WALSH DA, MCWILLIAMS DF, TURLEY MJ, et al. Angiogenesis and nerve growth factor at the osteochondral junction in rheumatoid arthritis and osteoarthritis. Rheumatology (Oxford). 2010;49:1852e61. [43] XINNING, YIYING, TENGFEI, et al. NGF Increases FGF2 Expression and Promotes Endothelial Cell Migration and Tube Formation Through PI3K/Akt and ERK/MAPK Pathways in Human Chondrocytes. Osteoarthritis and cartilage,2018. [44] KUO MH, LEE HF, TU YF, et al. Astaxanthin Ameliorates Ischemic-Hypoxic-Induced Neurotrophin Receptor p75 Upregulation in the Endothelial Cells of Neonatal Mouse Brains. Int J Mol Scie. 2019;20(24):6168. [45] KALAMVOKI M, DU T, ROIZMAN B. Cells Infected with Herpes Simplex Virus 1 export to Uninfected Cells Exosomes Containing STING, Viral mRNAs, and microRNAs. Proc Nat Acad Sci USA. 2014; 111(46):E4991-E4996. [46] KOWAL J, TKACH M, THÉRY C. Biogenesis and Secretion of Exosomes. Curr Opin Cel Biol. 2014;29:116-125. [47] GUPTA SK, THUM T. Non-coding RNAs as Orchestrators of Autophagic Processes. J Mol Cell Cardiol. 2016;95:26-30. [48] KNOWLING S, MORRIS KV. Non-coding RNA and Antisense RNA. Nature’s Trash or Treasure? Biochimie. 2011;93(11):1922-1927. [49] XIE W, SU W, XIA H, et al. Synovial Fluid MicroRNA-210 as a Potential Biomarker for Early Prediction of Osteoarthritis. Biomed Res Int. 2019; 2019:1-4. [50] WEI S, XIE W, SHANG Q, et al. The Long Noncoding RNA MEG3 Is Downregulated and Inversely Associated with VEGF Levels in Osteoarthritis. Biomed Res Int. 2015;2015:356893. [51] KIM HA, SEO KH, KANG YR, et al. Mechanisms of platelet-activating factor-induced enhancement of VEGF expression. Cell Physiol Biochem. 2011;27(1):55-62. [52] ZHANG Y, WU J, JING H, et al. Long noncoding RNA MEG3 inhibits breast cancer growth via upregulating endoplasmic reticulum stress and activating NF‐κB and p53. J Cell Biochem. 2019;120(4):6789-6797. [53] HE P, ZHANG Z, HUANG G, et al. miR-141 modulates osteoblastic cell proliferation by regulating the tar-get gene of lncRNA H19 and lncRNA H19-derived miR-675. Am J Transl Res. 2016;8:1780. [54] ZHOU L, WAN Y, CHENG Q, et al. The Expression and Diagnostic Value of LncRNA H19 in the Blood of Patients with Osteoarthritis. Iran J Public Health. 2020;49(8):1494-1501. [55] BEHERA J, KUMAR A, VOOR MJ, et al. Exosomal lncRNA-H19 Promotes Osteogenesis and Angiogenesis through Mediating Angpt1/Tie2-NO Signaling in CBS-Heterozygous Mice. Theranostics. 2021;11(16): 7715-7734. [56] BORNSTEIN P. Thrombospondins function as regulators of angiogenesis. J Cell Commun Signal. 2009;3(3-4):189-200. [57] GELSE K, KLINGER P, KOCH M, et al. Thrombospondin-1 prevents excessive ossification in cartilage repair tissue induced by osteogenic protein-1. Tissue Eng Part A. 2011;17(15-16):2101-2112. [58] ARICO-MUENDEL CC, BELANGER B, BENJAMIN D, et al. Metabolites of PPI-2458, a Selective, Irreversible Inhibitor of Methionine Aminopeptidase-2: Structure Determination and In Vivo Activity. Drug Metab Dispos. 2013;41(4):814-826. [59] LI B, CHEN KC, QIAN ND, et al. Baicalein alleviates osteoarthritis by protecting subchondral bone, inhibiting angiogenesis and synovial proliferation. J Cell Mol Med. 2021;25(11):5283-5294. [60] XIONG G, YANG Y, GUO M. Effect of resveratrol on abnormal bone remodeling and angiogenesis of subchondral bone in osteoarthritis. Int J Clin Exp Pathol. 2021;14(4):417-425. [61] VADALA G, AMBROSIO L, CATTANI C, et al. Bevacizumab Arrests Osteoarthritis Progression in a Rabbit Model: A Dose-Escalation Study. J Clin Med. 2021;10(13):2825. [62] NAGAI T, SATO M, KOBAYASHI M, et al. Bevacizumab, an anti-vascular endothelial growth factor antibody, inhibits osteoarthritis. Arthritis Res Ther. 2014;16(5):427. [63] LI W, LIN J, WANG Z, et al. Bevacizumab tested for treatment of knee osteoarthritis via inhibition of synovial vascular hyperplasia in rabbits. J Orthop Translat. 2019;19:38-46. [64] REMON J, GAZZAH A, BESSE B, et al. Crizotinib improves osteoarthritis symptoms in a ROS1-fusion advanced non-small cell lung cancer patient. J Thorac Oncol. 2015;10(8):e72-e73. [65] MIFUNE Y, MATSUMOTO T, TAKAYAMA K, et al. The effect of platelet-rich plasma on the regenerative therapy of muscle derived stem cells for articular cartilage repair. Osteoarthritis Cartilage. 2013;21(1):175-185. [66] LI MY, NIE Y, ZENG Y, et al. Does Bisphosphonate Increase the Sclerosis of Tibial Subchondral Bone in the Progression of Knee Osteoarthritis-A Propensity Score Matching Cohort Study Based on Osteoarthritis Initiative. Front Med (Lausanne). 2021;8:781219. [67] FERNÁNDEZ-MARTÍN S, LÓPEZ-PEÑA M, MUÑOZ F, et al. Bisphosphonates as disease-modifying drugs in osteoarthritis preclinical studies: a systematic review from 2000 to 2020. Arthritis Res Ther. 2021;23:60. [68] MOBASHERI A, HENROTIN Y, BIESALSKI HK, et al. Scientific Evidence and Rationale for the Development of Curcumin and Resveratrol as Nutraceutricals for Joint Health. Int J Mol Sci. 2012;13(4):4202-4232. [69] LI Y, MU W, XU B, et al. Artesunate, an Anti-Malaria Agent, Attenuates Experimental Osteoarthritis by Inhibiting Bone Resorption and CD31hiEmcnhi Vessel Formation in Subchondral Bone. Front Pharmacol. 2019;10:685. [70] WANG YJ, CUI J, GU ZG, et al. Betaine Attenuates Osteoarthritis by Inhibiting Osteoclastogenesis and Angiogenesis in Subchondral Bone. Front Pharmacol. 2021;12. [71] BINGÜL İ, BAŞARAN-KÜÇÜKGERGIN C, AYDIN AF, et al. Betaine Treatment Decreased Oxidative Stress, Inflammation, and Stellate Cell Activation in Rats with Alcoholic Liver Fibrosis. Environ. Toxicol. Pharmacol. 2016;45:170-178. [72] GE CX, YU R, XU MX, et al. Betaine Prevented Fructose-Induced NAFLD by Regulating LXRα/PPARα Pathway and Alleviating ER Stress in Rats. Eur J Pharmacol. 2016;770:154-164. [73] XIA Y, CHEN S, ZHU G, et al. Betaine Inhibits Interleukin-1β Production and Release: Potential Mechanisms. Front Immunol. 2018;9:2670. [74] PRASADAM I, AKUIEN A, FRIIS TE, et al. Mixed cell therapy of bone marrow-derived mesenchymal stem cells and articular cartilage chondrocytes ameliorates osteoarthritis development. Lab Invest. 2018;98:106-116. |
| [1] | Zhou Jie, Pei Xibo, Wan Qianbing. Advances and biological application of asymmetric dressings [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 434-440. |
| [2] | Chen Jingqiao, Li Ying, Meng Maohua, Xu Xingxing, Wang Qinying, Wang Huan, Lu Jing, Shu Jiayu, Dong Qiang. Research progress in platelet-rich fibrin in stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 441-446. |
| [3] | Wang Kaiyu, Hu Peng, Wei Zairong, Huang Guangtao, Zhou Jian, He Guijia, Nie Kaiyu. Use of expanders and implants in breast reconstruction complicated with infection [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 461-469. |
| [4] | Han Tao, Hao Jianqiang, Li Wenbo, Shi Jie, Gao Qiuming. Advantages and problems of antibiotic-loaded bone cements for bone and joint infections [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 470-477. |
| [5] | Shui Xiaoping, Li Chunying, Li Mingjuan, Li Shunchang, Sun Junzhi, Su Quansheng. Effects of aerobic and resistance exercise on antioxidant stress index and brain-derived neurotrophic factor expression in the hippocampus of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 264-269. |
| [6] | Liu Gang, Deng Bowen, Jiang Shengyuan, Xu Lin, Fan Xiao, Tao Jingwei, Zhang Houjun, He Feng, Zhao Yi, Mu Xiaohong. Tetramethylpyrazine improves hemorheological indexes in rats with complete spinal cord transection: a dynamic observation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 282-286. |
| [7] | Zhang Qian, Bian Minjie, He Qin, Huang Dongfeng. Predicting vascular mild cognitive impairment based on vascular risk factors: construction and application of a support vector machine model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 287-292. |
| [8] | Chen Feng, Ren Guowu, Zhang Xiaoyun, Chen Yueping, Shi Rusheng. Receptor activator of nuclear factor-kappa B ligand signal transduction mechanism and osteoclast activation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 293-299. |
| [9] | Li Mingxiu, Wang Xuan, Yang Jie, Li Yi. An osteoarthritis model in vitro: characteristics and new design idea [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 300-306. |
| [10] | Zhang Jie, Tian Ai. Advances in the signaling pathway of M2 macrophages involved in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 314-321. |
| [11] | Zhu Miaomiao, Kong Fanming, Zhao Qian. Exercise regulates lactic acid metabolism [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 322-328. |
| [12] | Xie Pingjin, Luo Zhen, Lu Qigui, Guo Yanxing, Chen Qunqun, Li Feilong. Effect of ligustrazine and overexpression of miR-20b-5p on synovial, cartilage and subchondral bone angiogenesis in rats with early-stage knee osteoarthritis: a histological observation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 237-245. |
| [13] | Wang Huanhuan, Wang Qing, Tang Peng, Zhang Rui, Min Hongwei. Effects of extracorporeal shock wave on the proliferation and autophagy of chondrocytes from osteoarthritis rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 252-257. |
| [14] | Zhang Luyao, Yang Kang. Effects of different exercises on renal interstitial fibrosis in type 2 diabetic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 200-207. |
| [15] | Niu Zihan, Yu Yang, Ai Jiang, Bu Panpan, Li Wenbo, Suriye·Reheman, Ma Shaolin. Total flavonoids of Hippophae rhamnoides L. interfere with the regression of hypertrophic scar tissue blocks in a rabbit ear model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 258-263. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

