Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (30): 4773-4779.doi: 10.12307/2022.756
Previous Articles Next Articles
Effects of miR-126-3p from adipose-derived mesenchymal stem cell released exosomes on human umbilical vein endothelial cell glucolipotoxicity
Lin Bo, Chen Xinyu, Jin Qiu, Zhu Zhiman, Zhao Wenhui
- Basic Medical Department, Jiangsu Vocational College of Nursing, Huaian 223005, Jiangsu Province, China
-
Received:2021-09-17Accepted:2021-11-03Online:2022-10-28Published:2022-03-29 -
Contact:Zhao Wenhui, PhD, Associate professor, Basic Medical Department, Jiangsu Vocational College of Nursing, Huaian 223005, Jiangsu Province, China -
About author:Lin Bo, Master, Lecturer, Basic Medical Department, Jiangsu Vocational College of Nursing, Huaian 223005, Jiangsu Province, China -
Supported by:Science and Technology Project of Jiangxi Provincial Department of Education, No. GJJ201243 (to LB)
CLC Number:
Cite this article
Lin Bo, Chen Xinyu, Jin Qiu, Zhu Zhiman, Zhao Wenhui. Effects of miR-126-3p from adipose-derived mesenchymal stem cell released exosomes on human umbilical vein endothelial cell glucolipotoxicity[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4773-4779.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
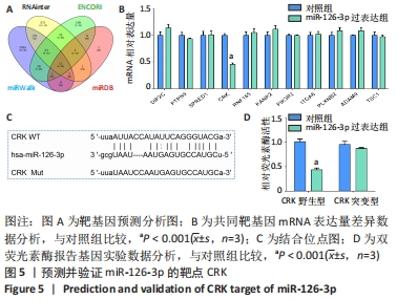
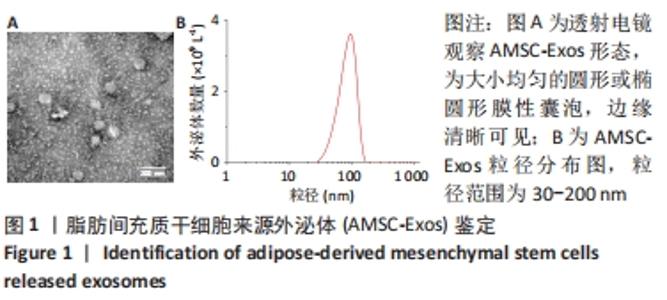
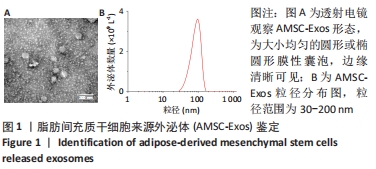
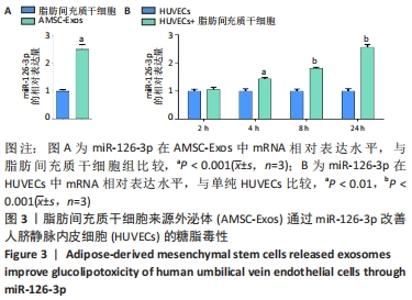
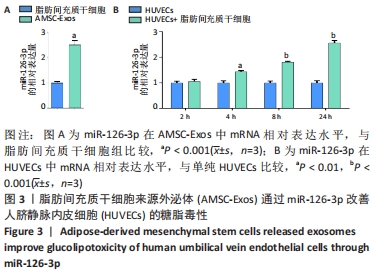
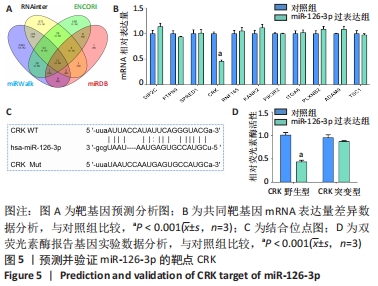
2.5 MiR-126-3p的靶基因分析 利用生物信息学软件预测miR-126-3p靶基因。通过4种软件分析预测结果的交集作为共同靶基因,共有11种基因(DIP2C、PTPN9、SPRED1、CRK、RNF165、KANK2、PIK3R2、ITGA6、PLXNB2、ADAM9和TSC1)符合,占总量0.2%,见图5A。采用RT-qPCR分析11种共同靶基因在miR-126-3p过表达细胞模型中有差异表达的靶基因,结果表明,只有CRK mRNA相对表达水平降低,差异有显著性意义(P < 0.001),见图5B。MiR-126-3p能够与野生型CRK的3’非编码区结合,见图5C。双荧光素酶基因报告实验结果表明,与对照组相比,miR-126-3p能够负向调控野生型CRK基因的荧光素酶活性(P < 0.001),而对突变型没有影响,见图5D,并对靶基因CRK相关信号通路进行预测,最终选取AKT信号通路进行后续研究。"
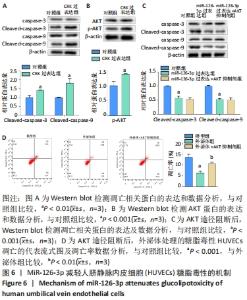
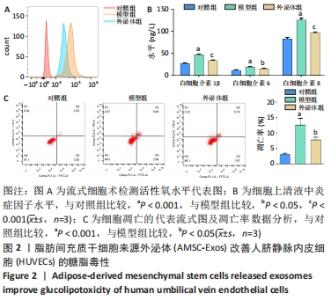
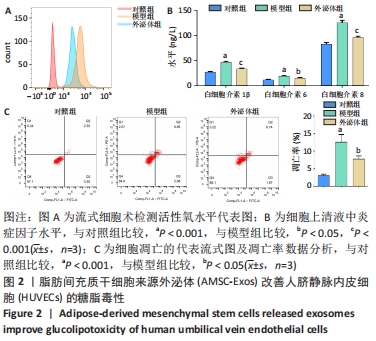
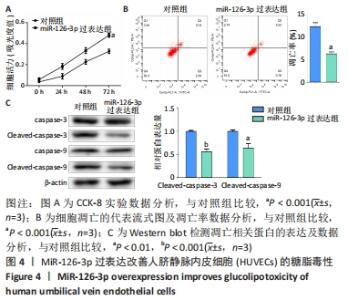
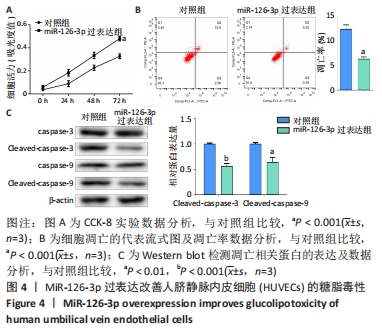
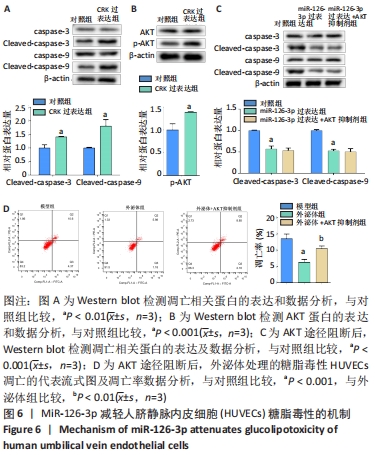
2.6 预测并验证 miR-126-3p作用机制 为验证miR-126-3p改善HUVECs糖脂毒性的可能作用机制,构建miR-126-3p靶基因CRK过表达转染HUVECs糖脂毒性细胞模型,Western blot检测凋亡相关蛋白,结果表明,CRK过表达能够显著提高细胞凋亡相关蛋白Cleaved caspase 3,9的表达量,表明CRK基因大量异常表达可促进细胞凋亡,见图6A。p-AKT蛋白磷酸化水平显著提高(P < 0.01),证实AKT途径能够被CRK激活,见图6B。使用AKT途径抑制剂RG7440与miR-126-3p共处理糖脂毒性HUVECs,结果表明,与对照组相比,miR-126-3p可显著降低Cleaved caspase 3,9蛋白的表达量(P < 0.001),但与miR-126-3p过表达组相比,RG7440与miR-126-3p共处理对Cleaved caspase 3,9的表达水平没有明显作用,见图6C。与外泌体组相比,RG7440与外泌体共处理能够显著提高细胞凋亡的能力(P < 0.01),见图6D。"
| [1] REUSCH JE, MANSON JE. Management of Type 2 Diabetes in 2017: Getting to Goal. JAMA. 2017;317(10):1015-1016. [2] MUREA M, FREEDMAN BI, PARKS JS, et al. Lipotoxicity in diabetic nephropathy: the potential role of fatty acid oxidation. Clin J Am Soc Nephrol. 2010;5(12):2373-2379. [3] KOJIMA H, KIM J, CHAN L. Emerging roles of hematopoietic cells in the pathobiology of diabetic complications. Trends Endocrinol Metab. 2014;25(4):178-187. [4] TOUSOULIS D, PAPAGEORGIOU N, ANDROULAKIS E, et al. Diabetes mellitus-associated vascular impairment: novel circulating biomarkers and therapeutic approaches. J Am Coll Cardiol. 2013;62(8):667-676. [5] ROBERTSON RP, HARMON J, TRAN PO, et al. Beta-cell glucose toxicity, lipotoxicity, and chronic oxidative stress in type 2 diabetes. Diabetes. 2004;53 Suppl 1:S119-124. [6] REBELATTO CK, AGUIAR AM, MORETÃO MP, et al. Dissimilar differentiation of mesenchymal stem cells from bone marrow, umbilical cord blood, and adipose tissue. Exp Biol Med (Maywood). 2008;233(7): 901-913. [7] RAO RR, STEGEMANN JP. Cell-based approaches to the engineering of vascularized bone tissue. Cytotherapy. 2013;15(11):1309-1322. [8] HAN Y, LI X, ZHANG Y, et al. Mesenchymal Stem Cells for Regenerative Medicine. Cells. 2019;8(8):886. [9] SCHMELZER E, MCKEEL DT, GERLACH JC. Characterization of Human Mesenchymal Stem Cells from Different Tissues and Their Membrane Encasement for Prospective Transplantation Therapies. Biomed Res Int. 2019;2019:6376271. [10] LIU X, ZHENG P, WANG X, et al. A preliminary evaluation of efficacy and safety of Wharton’s jelly mesenchymal stem cell transplantation in patients with type 2 diabetes mellitus. Stem Cell Res Ther. 2014;5(2):57. [11] XIE M, HAO HJ, CHENG Y, et al. Adipose-derived mesenchymal stem cells ameliorate hyperglycemia through regulating hepatic glucose metabolism in type 2 diabetic rats. Biochem Biophys Res Commun. 2017;483(1):435-441. [12] 赵一婷.脂肪间充质干细胞及其外泌体对炎症状态脂肪细胞及2型糖尿病大鼠的作用及机制[D].北京:北京协和医学院,2017. [13] BHONDE RR, SHESHADRI P, SHARMA S, et al. Making surrogate β-cells from mesenchymal stromal cells: perspectives and future endeavors. Int J Biochem Cell Biol. 2014;46:90-102. [14] MOON KC, SUH HS, KIM KB, et al. Potential of Allogeneic Adipose-Derived Stem Cell-Hydrogel Complex for Treating Diabetic Foot Ulcers. Diabetes. 2019;68(4):837-846. [15] MICHELHAUGH SA, CAMACHO A, IBRAHIM NE, et al. Proteomic Signatures During Treatment in Different Stages of Heart Failure. Circ Heart Fail. 2020;13(8):e006794. [16] LIANG X, DING Y, ZHANG Y, et al. Paracrine mechanisms of mesenchymal stem cell-based therapy: current status and perspectives. Cell Transplant. 2014;23(9):1045-1059. [17] ØVERBYE A, SKOTLAND T, KOEHLER CJ, et al. Identification of prostate cancer biomarkers in urinary exosomes. Oncotarget. 2015;6(30): 30357-30376. [18] CHEN B, LI Q, ZHAO B, et al. Stem Cell-Derived Extracellular Vesicles as a Novel Potential Therapeutic Tool for Tissue Repair. Stem Cells Transl Med. 2017;6(9):1753-1758. [19] LUDWIG N, HONG CS, LUDWIG S, et al. Isolation and Analysis of Tumor-Derived Exosomes. Curr Protoc Immunol. 2019;127(1):e91. [20] ZHOU N, CHEN X, XI J, et al. Genomic characterization reveals novel mechanisms underlying the valosin-containing protein-mediated cardiac protection against heart failure. Redox Biol. 2020;36:101662. [21] NAHID MA, SATOH M, CHAN EK. Interleukin 1β-Responsive MicroRNA-146a Is Critical for the Cytokine-Induced Tolerance and Cross-Tolerance to Toll-Like Receptor Ligands. J Innate Immun. 2015;7(4):428-440. [22] NAHID MA, SATOH M, CHAN EK. Mechanistic role of microRNA-146a in endotoxin-induced differential cross-regulation of TLR signaling. J Immunol. 2011;186(3):1723-1734. [23] 卢佩颖. 2型糖尿病患者血清miR-126表达的分析[D].杭州:浙江大学,2015. [24] 邓仁生,朱小琴,刘长召,等.血浆循环miRNA-126、miRNA-28-3p的表达与糖尿病的关系研究[J].局解手术学杂志,2017,26(6):400-405. [25] AN X, LI L, CHEN Y, et al. Mesenchymal Stem Cells Ameliorated Glucolipotoxicity in HUVECs through TSG-6. Int J Mol Sci. 2016;17(4): 483. [26] 张静,易阳艳,阳水发,等.脂肪干细胞来源外泌体对人脐静脉血管内皮细胞增殖、迁移及管样分化的影响[J]. 中国修复重建外科杂志,2018,32(10):1351-1357. [27] CHO BS, KIM JO, HA DH, et al. Exosomes derived from human adipose tissue-derived mesenchymal stem cells alleviate atopic dermatitis. Stem Cell Res Ther. 2018;9(1):187. [28] SHI Y, WANG Y, LI Q, et al. Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases. Nat Rev Nephrol. 2018;14(8):493-507. [29] RENDRA E, RIABOV V, MOSSEL DM, et al. Reactive oxygen species (ROS) in macrophage activation and function in diabetes. Immunobiology. 2019;224(2):242-253. [30] SHUKLA L, YUAN Y, SHAYAN R, et al. Fat Therapeutics: The Clinical Capacity of Adipose-Derived Stem Cells and Exosomes for Human Disease and Tissue Regeneration. Front Pharmacol. 2020;11:158. [31] DALIRFARDOUEI R, JAMIALAHMADI K, JAFARIAN AH, et al. Promising effects of exosomes isolated from menstrual blood-derived mesenchymal stem cell on wound-healing process in diabetic mouse model. J Tissue Eng Regen Med. 2019;13(4):555-568. [32] 陈子扬,蒲锐,邓爽,等.外泌体对运动介导胰岛素抵抗类疾病的调控作用[J].中国组织工程研究,2021,25(25):4089-4094. [33] GREENING DW, GOPAL SK, XU R, et al. Exosomes and their roles in immune regulation and cancer. Semin Cell Dev Biol. 2015;40:72-81. [34] ZHANG X, PEI Z, CHEN J, et al. Exosomes for Immunoregulation and Therapeutic Intervention in Cancer. J Cancer. 2016;7(9):1081-1087. [35] XUE WL, CHEN RQ, ZHANG QQ, et al. Hydrogen sulfide rescues high glucose-induced migration dysfunction in HUVECs by upregulating miR-126-3p. Am J Physiol Cell Physiol. 2020;318(5):C857-C869. [36] EISSA S, MATBOLI M, ABOUSHAHBA R, et al. Urinary exosomal microRNA panel unravels novel biomarkers for diagnosis of type 2 diabetic kidney disease. J Diabetes Complications. 2016;30(8): 1585-1592. [37] BRENTNALL M, RODRIGUEZ-MENOCAL L, DE GUEVARA RL, et al. Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol. 2013;14:32. [38] TOMITA T. Cleaved caspase-3 immunocytochemical staining for pancreatic islets and pancreatic endocrine tumors: A potential marker for biological malignancy. Islets. 2010;2(2):82-88. [39] RANI J, MITTAL I, PRAMANIK A, et al. T2DiACoD: A Gene Atlas of Type 2 Diabetes Mellitus Associated Complex Disorders. Sci Rep. 2017;7(1): 6892. [40] WEI F, WANG A, WANG Q, et al. Plasma endothelial cells-derived extracellular vesicles promote wound healing in diabetes through YAP and the PI3K/Akt/mTOR pathway. Aging (Albany NY). 2020;12(12): 12002-12018. [41] BATHINA S, DAS UN. Dysregulation of PI3K-Akt-mTOR pathway in brain of streptozotocin-induced type 2 diabetes mellitus in Wistar rats. Lipids Health Dis. 2018;17(1):168. |
| [1] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1045-1050. |
| [2] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [3] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [4] | Li Weiming, Xu Qingwen, Li Yijun, Sun Yanbo, Cui Jin, Xu Pengyuan . Deep seawater promotes wound healing in diabetic mice by activating PI3K/Akt pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 724-729. |
| [5] | Xu Manman, Ji Zhe, Ou Lingdong, Li Ang, Shen Caiqi, Jin Peisheng. Biological characteristics of adipose-derived mesenchymal stem cells in diabetic patients and their effect on promoting wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 3956-3960. |
| [6] | Tan Xu, Liang Yu, Liang Yan, Liao Jian. Hypoxia-treated dental pulp stem cell exosomes induce M2 macrophage polarization [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 3961-3967. |
| [7] | Wang Xianfeng, Ou Xin, Deng Biyong. Comparison of effects of exosomes secreted by different mesenchymal stem cells for the treatment of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 3980-3985. |
| [8] | Tang Jianhong, Zhang Nini, Huang Guilin, Lang Jiachan, Cui Tianning, Luo Qinliang. Role of mesenchymal stem cell-derived exosomes in tissue fibrosis repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4022-4027. |
| [9] | Wei Yufan, Fan Feiyan, Li Shuangli, Zhang Yunke. Future of exosomes and mesenchymal stem cell-derived exosomes in the diagnosis and treatment of Parkinson’s disease [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(25): 4076-4083. |
| [10] | Chen Na, Wang Xiaohan, Zhang Yunke. Effect and mechanism of mesenchymal stem cells on aging-related ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3914-3920. |
| [11] | Liu Haiqin, Ma Huagen, Tang Yuanyu. Expansion and identification of primary rat adipose-derived mesenchymal stem cells in vitro [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 2953-2957. |
| [12] | Shan Zhengming, Tao Shuchun, Hu Chunmei, Zhang Zhiyuan, Ding Yinan, He Mengcheng, Tang Qiusha. Extraction, identification and proteomic analysis of exosomes derived from human umbilical cord mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3036-3042. |
| [13] | Li Kun, An Jie, Dong Suge, Cui Lili, Liu Lei, Li Tingting. Effects of different fimA genotypes of Porphyromonas gingivalis on the secretion related proteins and cytokines in human umbilical vein endothelial cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3043-3047. |
| [14] | Wang Weiwei, Ou Zhixue, Zhang Xiaoyun, Li Shibin, Zhou Yi, Li Tong. Regulatory mechanism of exosomes in signal communication network of steroid induced avascular necrosis of the femoral head repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3056-3064. |
| [15] | Gu Xia, Wang Pingyi, Zhao Wanhua, Lin Yushi, Li Yimei, Li Wenhua. Important role of exosomes-mediated hypoxia-related signaling pathways in the occurrence and progression of diseases [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3065-3070. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||