Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (12): 1944-1949.doi: 10.12307/2022.521
Previous Articles Next Articles
Cell sheet technology applied in bone tissue engineering
Tan Xianyu, Liao Wenbo
- Department of Orthopedics, the Second Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2021-07-28Revised:2021-07-30Accepted:2021-08-24Online:2022-04-28Published:2021-12-15 -
Contact:Liao Wenbo, MD, Professor, Doctoral supervisor, Department of Orthopedics, the Second Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Tan Xianyu, Master, Physician, Department of Orthopedics, the Second Affiliated Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 82060415 (to LWB)
CLC Number:
Cite this article
Tan Xianyu, Liao Wenbo. Cell sheet technology applied in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(12): 1944-1949.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

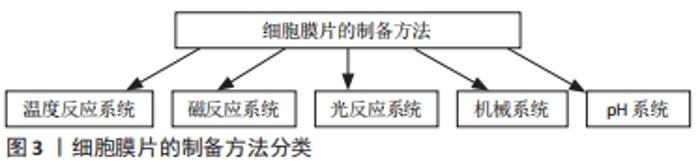
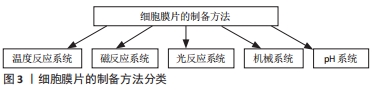
2.1.1 温度反应系统 温度反应系统是最先提出并用于制备细胞膜片的方法,也是目前应用最广泛的制备方法。温度敏感型聚异丙基丙烯酰胺(PIPAAm)通常以共价结合的方式结合到细胞培养皿的底部,该温敏型材料在不同的温度条件下表现出不同的性能。当环境温度为37 ℃时,温度敏感型聚异丙基丙烯酰胺具有疏水特性,可促进细胞黏附和增殖,从而有助于促进细胞膜片的形成;当温度低于32 ℃时,温度敏感型聚异丙基丙烯酰胺表现出亲水性,通过在培养皿表面和细胞膜片之间形成水合层,从而将培养的细胞膜片与培养皿完全分离[17]。 2.1.2 机械系统 机械系统是制备细胞膜片最简单的一种方法,只需向培养基中加入一定量的抗坏血酸即可,而不需要特定的培养基质或技术,但是其制备细胞膜片所需的时间较长,通常在2周左右可以观察到细胞膜片的形成。该方法的原理主要是通过抗坏血酸刺激细胞分泌大量的细胞外基质,通过连续培养从而形成膜片,当大量的细胞外基质沉积时,可观察到细胞膜片的形成。当细胞膜片形成后,通过细胞刮和镊子轻轻的将膜片从培养板里剥离并分离[18,22]。在骨组织工程中,细胞膜片还可通过将制备的细胞膜片成骨诱导从而制备成骨细胞膜片。最近,KIM等[23]提出了一种将明胶添加到成骨培养基中制备成骨细胞膜片的新型机械系统。与传统的成骨细胞膜片相比,明胶诱导的成骨细胞膜片具有极强的细胞增殖能力和丰富的细胞外基质,可形成较厚、机械性能较强的细胞膜片。 2.1.3 磁反应系统 磁反应系统在制备多层细胞膜片方面具有较大的优势。首先通过细胞内吞作用,将带有磁性的粒子内吞到细胞内,然后在外加磁场的作用下细胞可以快速地黏附在培养板上并形成细胞膜片。当单层细胞膜片形成后,可以继续向磁反应系统添加细胞,从而构建多层细胞膜片。此外,磁反应系统不仅能够制备单一的细胞膜片,同时还能制备不同类型细胞的复合细胞膜片[19]。 2.1.4 光反应系统 光反应系统通过将细胞培养在二氧化钛(TiO2)纳米涂层的培养板上,然后通过紫外光介导培养板亲水性和疏水性的变化,从而形成细胞膜片。用365 nm的紫外光照射20 min后,90%以上的细胞能从培养板上分离。在此基础上,研究者还通过在培养板上添加其他成分促进细胞的黏附从而来促进细胞膜片的形成,形成的细胞膜片在紫外光的照射下能够从培养板上分离[20,24]。 2.1.5 pH系统 GUILLAUME-GENTIL等[21]采用聚4-苯乙烯磺酸盐和聚烯丙胺盐酸盐两种聚合物制备电解质多层膜涂层,通过调节局部和整体的pH值来获取细胞膜片。当培养液缓冲基pH值达5.0以上时,细胞附着于表面;当pH值降低至4.0时,培养的间充质干细胞膜片则在两三分钟内完整脱离。由于该技术需要严格控制环境的pH值,否者会造成细胞的损伤,因此该技术的应用受到了一定的限制。同时,对于pH敏感的细胞,同样不适合用该方法来制备细胞膜片。 2.2 细胞膜片的类型 根据细胞来源的不同,细胞膜片可分为骨髓间充质干细胞膜片、脂肪间充质干细胞膜片、软骨细胞膜片等;根据细胞膜片层数和厚度的不同,细胞膜片可分为单层细胞膜片、双层细胞膜片和多层细胞膜片,其中,双层和多层细胞膜片根据细胞来源是否相同又可分为单一细胞膜片和混合细胞膜片。间充质干细胞是目前广泛应用的种子细胞之一,其具有广泛的来源和多系分化的能力,同时还具有潜在的抗炎、抗免疫的作用。通过对比骨髓、脂肪和脐带来源的间充质干细胞膜片,NAKAO等[25]发现不同来源的间充质干细胞膜片其转化生长因子β、白细胞介素10、前列素E2等细胞因子的表达不同,因此,在不同的疾病中,可选取不同细胞来源的细胞膜片来治疗相应的疾病。YOU等[18]采用单层人羊膜间充质干细胞膜片修复兔关节软骨缺损,取得了不错的效果。但是,单纯细胞膜片的机械强度较弱,在培养的过程中容易出现收缩,因此操作具有一定的难度。通过构建多层细胞膜片,可以促进细胞分泌更多的细胞外基质,加强细胞间的相互联系,从而提高膜片的机械强度。但是,多层细胞膜片由于其厚度的增加会导致营养物质渗透不足,容易导致细胞死亡的风险。SILVA等[19]通过磁反应系统制备了多层细胞膜片,该膜片分3层,最下面一层是脂肪间充质干细胞膜片层,中间层是人脐静脉内皮细胞膜片层,最上面一层为脂肪间充质干细胞层,体内外实验均证实该多层细胞膜片具有成骨能力,同时还具有成血管的特点,因此,在修复骨缺损方面具有不错的应用价值。 2.3 细胞膜片在骨组织工程中的应用 "

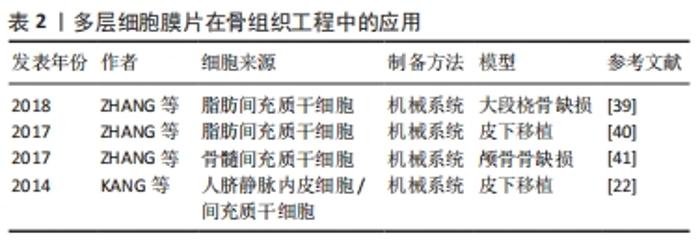
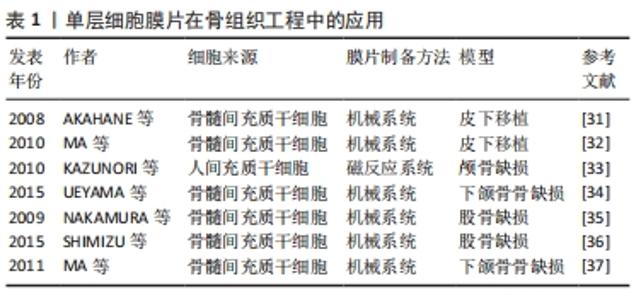
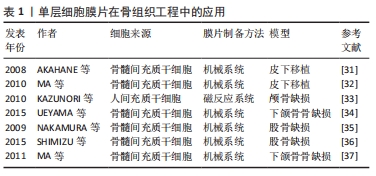
在骨组织工程领域的研究,首先是通过皮下移植观察其异位成骨的潜能。AKAHANE等[31]通过机械法成功制备了骨髓间充质干细胞膜片,成骨诱导后皮下移植到大鼠体内,在不使用其他支架的条件该膜片能明显促进新骨形成。此外,MA等[32]同样通过机械法制备了兔骨髓间充质干细胞膜片,将该膜片移植到裸鼠皮下可观察到新骨的形成。随后,研究者将研究的方向转向骨缺损的修复,通过建立骨缺损模型更好地模拟临床上的骨损伤,从而观察单层细胞膜片对骨缺损的修复效果。在骨缺损的研究中,常用的动物模型是颅骨缺损模型,其制备方法简单,在研究骨损伤修的复中具有重要的意义。KAZUNORI等[33]通过磁反应系统制备了细胞膜片,然后将其移植到大鼠颅骨缺损部位,可观察到缺损的部位几乎完全修复。UEYAMA等[34]用机械法制备了成骨细胞膜片,移植到兔下颌骨骨缺损部位,结果显示该膜片填充于缺损的部位能够明显促进缺损部位的骨再生。骨折的愈合是一个动态的过程,虽然骨折后大部分患者能够达到完全愈合,但是仍有一部分患者会发生骨折的延期愈合甚至不愈合,这可能与很多复杂的因素有关。因此,骨折后除了积极有效的复位和固定,还需要进行一定的干预加速骨折的愈合,避免骨折不愈合的发生。目前,大量的研究通过机械系统构建骨髓间充质干细胞膜片用于治疗骨折的治疗。NAKAMURA等[35]将成骨细胞膜片移植到骨折大鼠股骨中,可增强骨折部位的骨形成,并形成稳定的新生骨。SHIMIZU等[36]在大鼠股骨骨折愈合模型中注射成骨细胞膜片,可促进骨再生,最终导致骨愈合。MA等[37]包裹成骨分化的兔骨髓间充质干细胞膜片在下颌骨2 mm的骨折间隙处,可观察到骨折延迟愈合的改善并可见新生骨的形成。 2.3.2 多层细胞膜片在骨组织工程中的应用 骨膜是覆盖在骨外表面的一层薄膜,在骨的发育和再生中起着重要作用。骨膜主要由外层纤维层和内层成骨层组成,外层纤维层含有成纤维细胞、胶原纤维和血管,滋养内骨;内层成骨层含有间充质干细胞和骨祖细胞,负责骨的发育和再生[38]。因此,多层细胞膜片能够很好地模拟骨膜的结构和功能(外层膜片成血管、内层膜片成骨),通过构建多层细胞膜片来模拟骨膜的结构和功能为骨缺损的修复提供了新的思路,见表2。"
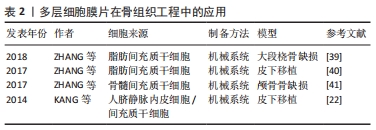
ZHANG等[39]通过构建双层细胞膜片制备了血管化的组织工程骨,首先提取兔脂肪间充质干细胞,然后将其分别诱导成骨和成血管,体内用于兔大段桡骨缺损的修复,实验结果显示,该复合细胞膜片能够促进大段骨缺损的修复。在另外一项研究中,ZHANG等[40]利用机械法成功构建了成骨和成血管膜片,然后将两者复合在一起制备复合细胞膜片,该复合细胞膜片体外具有明显的成骨和成血管潜能;然后将该复合膜片与包裹在羟基磷灰石表面,体内实验探究其成骨和成血管能力,结果同样证实了该复合细胞膜片具有成骨和成血管的潜力,因此其在骨再生方面具有很大的应用前景。ZHANG等[41]也通过机械法制备了多层细胞膜片从而来构建仿生骨膜,用于骨缺损的修复:首先制备大鼠骨髓间充质干细胞膜片,然后将其诱导为成骨细胞膜片,接着将骨髓干细胞诱导分化成内皮样细胞并将其接种到骨髓间充质干细胞膜片上,从而构建成血管细胞膜片;将成骨细胞膜片和成血管细胞膜片复合制备复合细胞膜片,模拟骨膜的结构;体内外实验均证实该仿生骨膜结构(复合细胞膜片)具有成血管和成骨能力,能够明显促进骨再生。KANG等[22]通过机械法制备了仿生骨膜结构的双层结构的细胞膜片,用于体内成骨的研究,外层构建了人来源的间充质干细胞膜片,并将人脐静脉内皮细胞种植到膜片上从而得到了具有血管化的细胞膜片;内层构建成骨细胞膜片并将其包裹在多孔磷酸三钙支架上,并将其移植到小鼠体内观察其成骨和成血管能力;体外结果显示血管化细胞膜片具有成血管的能力,体内实验证实该复合支架具有显著的成血管和成骨能力,是一种新的骨缺损修复的方法。 2.3.3 细胞膜片结合其他材料在骨组织工程中的应用 虽然细胞膜片具有丰富的细胞外基质,并且保留了完整的细胞间的连接,但是,由于其力学性能较差,单用细胞膜片无法起到骨支撑的作用,因此,许多研究者通过将制备的细胞膜片与骨组织工程支架相结合来修复骨缺损,充分发挥了骨组织工程支架和细胞膜片各自的优势,见表3。"
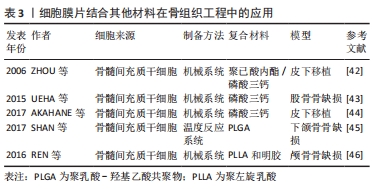
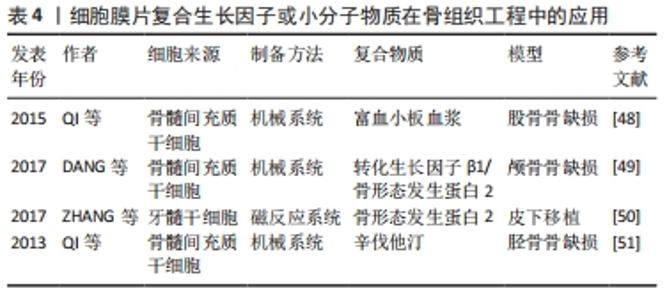
ZHOU等[42]首先将细胞膜片与聚已酸内酯和磷酸三钙复合材料复合用于骨缺损的修复,结果发现既有血管化的松质骨形成同时又有皮质骨的形成,并且新生的骨组织的抗压强度显著高于未与膜片复合的对照组。UEHA等[43]同样利用机械法制备了成骨细胞膜片,然后将该成骨膜片与磷酸三钙支架复合,体内外结果均证实该复合支架具有较强的成骨能力。AKAHANE等[44]比较了不同年龄阶段来源的骨髓间充质干细胞膜片在成骨方面的差异,通过提取7周龄和1岁龄的大鼠骨髓间充质干细胞并构建成骨细胞膜片,然后将膜片与磷酸三钙支架复合,结果显示:不管是7周龄和1岁龄来源的骨髓间充质干细胞膜片均能促进骨缺损的骨再生。除了磷酸三钙支架外,聚乳酸-羟基乙酸共聚物(PLGA)也常用于细胞膜片的复合材料。SHAN等[45]通过将骨髓间充质干细胞膜片成骨诱导后与聚乳酸-羟基乙酸共聚物复合构建复合支架材料,将其填充于犬下颌骨缺损处,结果显示具有良好的成骨能力,能够很好地促进缺损部位的骨再生。静电纺丝技术制备的纳米纤维膜对细胞的生长和增殖具有明显的促进作用,因此,也有研究以静电纺丝纳米纤维膜作为基质,将种子细胞种植在其表面,从而构建支架-细胞膜片的复合支架材料。静电纺纳米纤维膜除了具有可操作性外,还可以作为构建细胞膜片的支撑材料,通过纳米纤维特性调节细胞行为。REN等[46]将聚左旋乳酸(PLLA)和明胶(gelatin)以1∶1的比例混合,通过静电纺丝技术分别制备了无序结构和网状结构的静电纺丝纤维膜,然后将大鼠的骨髓间充质干细胞分别接种到培养板和不同结构的纳米纤维膜上并诱导成膜片。体外实验结果表明网状结构的纳米纤维膜能够明显促进大鼠的骨髓间充质干细胞成骨分化,而两种结构的纳米纤维膜对细胞的增殖无明显差异;随后将骨髓间充质干细胞膜片与纳米纤维膜复合用于大鼠颅骨缺损的修复,体内实验结果提示纳米纤维膜复合细胞膜片在促进骨缺损修复方面明显强于对照组,而不同结构的纳米纤维膜复合细胞膜片两者之间的差异不大。 2.3.4 细胞膜片复合生长因子或小分子物质在骨组织工程中的应用 生长因子在组织再生中起着关键作用,其在骨损伤修复的过程中同样发挥着重要的作用。因此,近年来越来越多的研究者向细胞膜片中加入适当的生长因子或小分子物质用于促进骨再生从而来修复骨缺损,见表4。"
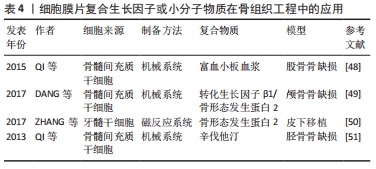

富血小板血浆(PRP)是自体血经过离心后而获得的血小板浓缩物,其富含有大量的生长因子,包括血小板衍生生长因子(PDGF)、转化生长因子(TGF-β1,TGF-β2)、胰岛素样生长因子(IGF-1,IGF-2)和血管内皮生长因子(VEGF)等[47]。QI等[48]通过机械法制备了兔骨髓间充质干细胞膜片,并将该膜片包裹富血小板血浆凝胶/CaP微粒,富血小板血浆能够持续释放细胞因子,有利于骨缺损的修复。体外研究表明包裹富血小板血浆/CaP的细胞膜片其成骨相关基因表达明显提高,体内实验也证实经富血小板血浆/CaP处理的细胞膜片较其他组能够明显的促进骨缺损的修复。DANG等[49]将人骨髓间充质干细胞膜片与纳米微粒相结合,该纳米微粒早期能够释放转化生长因子β1,促进软骨形成,晚期持续释放骨形态发生蛋白2,促进骨的形成和释放,体内影像学和组织学检测均提示该系统能够促进兔颅骨缺损的修复,持续的细胞因子释放对加速骨愈合起着重要的作用。ZHANG等[50]发现nGO@Fe3O4磁性纳米颗粒由于富含大量的羧基,因此能够很好地结合生长因子,并且能够被牙髓干细胞内吞而对细胞的增殖没有影响,因此,他们通过细胞内吞的作用将结合骨形态发生蛋白2的nGO@Fe3O4磁性纳米颗粒导入到牙髓干细胞内细胞内,然后在磁反应系统的作用下成功制备牙髓干细胞膜片,该膜片能够持续释放骨形态发生蛋白2,在成骨方面具有不错的应用前景,为以后构建骨组织工程细胞膜片提供了新的思路。QI等[51]将小分子物质辛伐他汀(simvastatin)加入到间充质干细胞膜片中,结果证实细胞膜片复合小分子物质simvastatin能够明显促进兔骨折愈合,其效果较单用细胞膜片更好。"

| [1] Zl A, XZD E, JIANG OC, et al. Ca2+ supplying black phosphorus-based scaffolds fabricated with microfluidic technology for osteogenesis. Bioact Mater. 2021;6(11):4053-4064. [2] ZHANG JZ, JIANG YB, SHANG ZZ, et al. Biodegradable metals for bone defect repair: A systematic review and meta-analysis based on animal studies. Bioact Mater. 2021;6(11):4027-4052. [3] 周思佳,姜文学,尤佳.骨缺损修复材料:现状与需求和未来[J].中国组织工程研究,2018,22(14):2251-2258. [4] GAO C, PENG S, PEI F, et al. Bone biomaterials and interactions with stem cells. Bone Res. 2017;5(4):1-33. [5] 胡金龙.组织工程学技术治疗骨缺损的最新研究进展[J].中国矫形外科杂志,2013,21(2):150-153. [6] MAO Y, ZHAO Y, GUAN J, et al. Electrospun fibers: an innovative delivery method for the treatment of bone diseases. Expert Opin Drug Del. 2020;17(7):993-1005. [7] QIAO W, XIE H, FANG J, et al. Sequential activation of heterogeneous macrophage phenotypes is essential for biomaterials-induced bone regeneration. Biomaterials. 2021;276:121038. . [8] ZHAI Y, SCHILLING K, WANG T, et al. Spatiotemporal blood vessel specification at the osteogenesis and angiogenesis interface of biomimetic nanofiber-enabled bone tissue engineering. Biomaterials. 2021;276:121041. [9] MA D, REN L, CAO Z, et al. Prefabrication of axially vascularized bone by combining β-tricalciumphosphate, arteriovenous loop, and cell sheet technique. Tissue Eng Regen Med. 2016;13(5):579-584. [10] REN LL, YUNQING KANG, BROWNE C, et al. Fabrication, vascularization and osteogenic properties of a novel synthetic biomimetic induced membrane for the treatment of large bone defects-ScienceDirect. Bone. 2014;64(7):173-182. [11] TANG Z, TERUO O. Recent development of temperature-responsive surfaces and their application for cell sheet engineering. Regen Biomater. 2014;(1):91-102. [12] GUO B, PAN G, GUO Q, et al. Saccharides and temperature dual-responsive hydrogel layers for harvesting cell sheets. Chem Commun. 2014;51(4):644-647. [13] PATEL NG, CAVICCHIA JP, ZHANG G, et al. Rapid cell sheet detachment using spin-coated pNIPAAm films retained on surfaces by an aminopropyltriethoxysilane network. Acta Biomater. 2012;8(7):2559-2567. [14] ENOMOTO J, MOCHIZUKI N, EBISAWA K, et al. Engineering thick cell sheets by electrochemical desorption of oligopeptides on membrane substrates. Regen Ther. 2016;3(C):24-31. [15] CHENG K, WANG T, YU M, et al. Effects of RGD immobilization on light-induced cell sheet detachment from TiO2 nanodots films. Mat Sci Eng C-Mater. 2016;63(1):240-246. [16] ITO A, HAYASHIDA M, HONDA H, et al. Construction and Harvest of Multilayered Keratinocyte Sheets Using Magnetite Nanoparticles and Magnetic Force. Tissue Eng. 2004;10(5):873-880. [17] KUMASHIRO, YAMATO, OKANO, et al. Cell attachment-detachment control on temperature-responsive thin surfaces for novel tissue engineering. Ann Biomed Eng. 2010;38:1977-1988. [18] YOU Q, LIU Z, ZHANG J, et al. Human Amniotic Mesenchymal Stem Cell Sheets Encapsulating Cartilage Particles Facilitate Repair of Rabbit Osteochondral Defects. Am J Sport Med. 2020;48(3):599-611. [19] SILVA AS, SANTOS LF, MENDES MC , et al. Multi-layer pre-vascularized magnetic cell sheets for bone regeneration. Biomaterials. 2020;231: 119664. [20] SHI D, XU X, YE Y, et al. Photo-cross-linked scaffold with kartogenin encapsulated nanoparticles for cartilage regeneration. Acs Nano. 2016;10(1):1292-1299. [21] GUILLAUME-GENTIL O, SEMENOV OV, ZISCH AH, et al. PH-controlled recovery of placenta-derived mesenchymal stem cell sheets. Biomaterials. 2011;32(19):4376-4384. [22] KANG Y, REN L, YANG Y. Engineering Vascularized Bone Grafts by Integrating a Biomimetic Periosteum and β-TCP Scaffold. Acs Appl Mater Inter. 2014;6(12):9622-9633. [23] KIM AY, KIM Y, LEE SH, et al. Effect of gelatin on osteogenic cell sheet formation using canine adipose-derived mesenchymal stem cells. Cell Transplant. 2016;26(1):115-123. [24] HONG Y, YU M, WENG W, et al. Light-induced cell detachment for cell sheet technology. Biomaterials. 2013;34(1):11-18. [25] NAKAO M, INANAGA D, NAGASA K, et al. Characteristic differences of cell sheets composed of mesenchymal stem cells with different tissue origins. Regen Ther. 2019;11:34-40. [26] SATO M, YAMATO M, MITANI G, et al. Combined surgery and chondrocyte cell-sheet transplantation improves clinical and structural outcomes in knee osteoarthritis. NPJ Regen Med. 2019;4:4. [27] TANI Y, SATO M, MAEHARA M, et al. The effects of using vitrified chondrocyte sheets on pain alleviation and articular cartilage repair. J Tissue Eng Regen Med. 2017;11(12):3437-3444. [28] WANG F, HU Y, HE D, et al. Scaffold-free cartilage cell sheet combined with bone-phase BMSCs-scaffold regenerate osteochondral construct in mini-pig model. Am J Transl Res. 2018;10(10):2997-3010. [29] KOBAVASHI T, KAN K, NISHIDA K, et al. Corneal regeneration by transplantation of corneal epithelial cell sheets fabricated with automated cell culture system in rabbit model. Biomaterials. 2013; 34(36):9010-9017. [30] LIU Q, YU Y, REISDORF RL, et al. Engineered tendon-fibrocartilage-bone composite and bone marrow-derived mesenchymal stem cell sheet augmentation promotes rotator cuff healing in a non-weight-bearing canine model. Biomaterials. 2019;192:189-198. [31] AKAHANE M, NAKAMURA A, OHGUSHI H, et al. Osteogenic matrix sheet-cell transplantation using osteoblastic cell sheet resulted in bone formation without scaffold at an ectopic site. J Tissue Eng Regen Med. 2010;2(4):196-201. [32] MA D, REN L, LIU Y, et al. Engineering scaffold-free bone tissue using bone marrow stromal cell sheets. J Orthop Res. 2010;28(5):697-702. [33] KAZUNORI SHIMIZU, AKIRA ITO, TATSURO YOSHIDA, et al. Bone tissue engineering with human mesenchymal stem cell sheets constructed using magnetite nanoparticles and magnetic force. J Biomed Mater Res B. 2010;82(2):471-480. [34] UEYAMA Y, YAGYUU T, MAEDA M, et al. Maxillofacial bone regeneration with osteogenic matrix cell sheets: An experimental study in rats. Arch Oral Biol. 2016;72:138-145. [35] NAKAMURA A, AKAHANE M, SHIGEMATSU H, et al. Cell sheet transplantation of cultured mesenchymal stem cells enhances bone formation in a rat nonunion model. Bone. 2010;46(2):418-424. [36] SHIMIZU T, AKAHANE M, MORITA Y, et al. The regeneration and augmentation of bone with injectable osteogenic cell sheet in a rat critical fracture healing model. Injury. 2015;34(8):1457-1464. [37] MA D, YAO H, TIAN W, et al. Enhancing bone formation by transplantation of a scaffold-free tissue-engineered periosteum in a rabbit model. Clin Oral Implants Res. 2011;22(10):1193-1199. [38] WU L, GU Y, LIU L, et al. Hierarchical micro/nanofibrous membranes of sustained releasing VEGF for periosteal regeneration. Biomaterials. 2019;227:119555. [39] ZHANG H, ZHOU Y, YU N, et al. Construction of vascularized tissue-engineered bone with polylysine-modified coral hydroxyapatite and a double cell-sheet complex to repair a large radius bone defect in rabbits. Acta Biomater. 2019:91:82-98. [40] ZHANG HL, ZHOU YL, ZHANG W , et al. Construction of vascularized tissue-engineered bone with a double-cell sheet complex-ScienceDirect. Acta Biomater. 2018;77:212-227. [41] ZHANG D, GAO P, LI Q, et al. Engineering biomimetic periosteum with β-TCP scaffolds to promote bone formation in calvarial defects of rats. Stem Cell Res Ther. 2017;8(1):134. [42] ZHOU Y, CHEN F, HO ST, et al. Combined marrow stromal cell-sheet techniques and high-strength biodegradable composite scaffolds for engineered functional bone grafts. Biomaterials. 2007;8(5):814-824. [43] UEHA T, AKAHANE M, SHIMIZU T, et al. Utility of tricalcium phosphate and osteogenic matrix cell sheet constructs for bone defect reconstruction. World J Stem Cells. 2015;(5):873-882. [44] AKAHANE M, SHIMIZU T, INAGAKI Y, et al. Implantation of bone marrow stromal cell sheets derived from old donors supports bone tissue formation. Tissue Eng Regen Med. 2017;15(1):89-100. [45] SHAN X, HU D. Bone engineering by cell sheet technology to repair mandibular defects. Exp Ther Med. 2017;14:5007-5011. [46] REN Z, MA S, JIN L, et al. Repairing a bone defect with a three-dimensional cellular construct composed of a multi-layered cell sheet on electrospun mesh. Biofabrication. 2017;9(2):025036. [47] ZHANG J, LIU Z, TANG J, et al. Fibroblast growth factor 2-induced human amniotic mesenchymal stem cells combined with autologous platelet rich plasma augmented tendon-to-bone healing. J Orthop Transl. 2020;24:155-165. [48] Qi YY, NIU L, ZHAO TF, et al. Combining mesenchymal stem cell sheets with platelet-rich plasma gel/calcium phosphate particles: a novel strategy to promote bone regeneration. Stem Cell Res Ther. 2015;6:256. [49] DANG PN, HERBERG S, VARGHAI D, et al. Endochondral Ossification in Critical-Sized Bone Defects via Readily Implantable Scaffold-Free Stem Cell Constructs. Stem Cell Transl Med. 2017;6(7):1644-1659. [50] ZHANG W, YANG G, WANG X, et al. Magnetically Controlled Growth-Factor-Immobilized Multilayer Cell Sheets for Complex Tissue. Adv Mater. 2017;29(43): doi: 10.1002/adma.201703795. [51] QI Y, ZHAO T, YAN W, et al. Mesenchymal stem cell sheet transplantation combined with locally released simvastatin enhances bone formation in a rat tibia osteotomy model. Cytotherapy. 2013;15(1):44-56. |
| [1] | Xue Yadong, Zhou Xinshe, Pei Lijia, Meng Fanyu, Li Jian, Wang Jinzi . Reconstruction of Paprosky III type acetabular defect by autogenous iliac bone block combined with titanium plate: providing a strong initial fixation for the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1424-1428. |
| [2] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [3] | Jiang Huanchang, Zhang Zhaofei, Liang De, Jiang Xiaobing, Yang Xiaodong, Liu Zhixiang. Comparison of advantages between unilateral multidirectional curved and straight vertebroplasty in the treatment of thoracolumbar osteoporotic vertebral compression fracture [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1407-1411. |
| [4] | Li Wei, Zhu Hanmin, Wang Xin, Gao Xue, Cui Jing, Liu Yuxin, Huang Shuming. Effect of Zuogui Wan on bone morphogenetic protein 2 signaling pathway in ovariectomized osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1173-1179. |
| [5] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [6] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [7] | Wu Bingshuang, Wang Zhi, Tang Yi, Tang Xiaoyu, Li Qi. Anterior cruciate ligament reconstruction: from enthesis to tendon-to-bone healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1293-1298. |
| [8] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [9] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| [10] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [11] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [12] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [13] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1020-1026. |
| [14] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [15] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||