Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (33): 5381-5387.doi: 10.12307/2021.331
Previous Articles Next Articles
Participation and regulation of long-chain non-coding RNA on pathogenesis of avascular necrosis of the femoral head
Wang Chuan1, Peng Wuxun2
- 1Clinical Medical College of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Emergency Department of Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2020-03-27Revised:2020-04-02Accepted:2020-12-09Online:2021-11-28Published:2021-08-06 -
Contact:Peng Wuxun, Chief physician, Professor, Doctoral supervisor, Emergency Department of Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Wang Chuan, Master candidate, Clinical Medical College of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 82060397, 81860387 (to PWX)
CLC Number:
Cite this article
Wang Chuan, Peng Wuxun. Participation and regulation of long-chain non-coding RNA on pathogenesis of avascular necrosis of the femoral head[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(33): 5381-5387.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
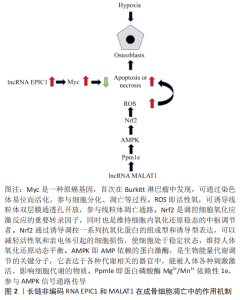
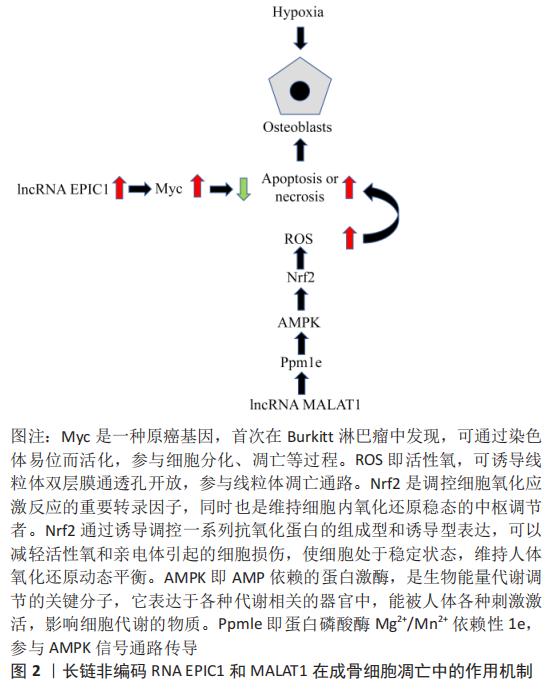
2.1.1 Lnc-MALAT1 转移相关肺腺癌转录子1(metastasis-associated lung adenocarcinoma tran-script 1,MALAT1) 因最早是从早期非小细胞肺癌组织中发现的,故以此命名。转移相关肺腺癌转录子1是一种在进化中高度保守的核内长链非编码RNA,广泛存在哺乳动物中且呈高丰度表达,是目前研究热门、调控范围较广的长链非编码RNA。相关研究表明在股骨头缺血性坏死患者的股骨头组织中转移相关肺腺癌转录子1表达显著降低,转移相关肺腺癌转录子1表达增加可下调蛋白磷酸酶Mg2+/Mn2+依赖性1e(protein phosphatase Mg2+/Mn2+dependent 1e,Ppm1e)的表达,进而激活AMP激活的蛋白激酶(AMP-activated protein kinase,AMPK)信号传导,并可通过磷酸化作用而激活核因子E2相关因子2(nuclear factor erythroid 2-related factor 2,Nrf2),减少活性氧产生[7]。活性氧可通过破坏细胞膜或者使线粒体DNA突变造成成骨细胞凋亡或坏死。 XIN等[8]研究发现,在骨微血管内皮细胞中敲除转移相关肺腺癌转录子1表达,检测细胞凋亡标志物基因半胱氨酸天冬氨酸蛋白酶3(Caspase-3)和半胱氨酸天冬氨酸蛋白酶8(Caspase-8)的表达显著上调,促凋亡基因Bax的表达显著上调,而抗凋亡标志物B细胞淋巴瘤-2(B-cell lymphoma-2,Bcl-2)基因表达明显下调,证实转移相关肺腺癌转录子1基因表达的下调与细胞凋亡成反向调节作用。ZHANG等[9]研究发现,抑制转移相关肺腺癌转录子1的表达,使细胞周期停滞在G2/M期,导致细胞发生凋亡,抑制细胞的增殖、迁移和分化。另外,ATM-CHK2 信号通路的磷酸化作用与转移相关肺腺癌转录子1的表达呈反向调节作用,即抑制转移相关肺腺癌转录子1基因表达会激活 ATM-CHK2 信号通路,阻滞细胞正常生长周期。LI等[10]研究发现,骨髓间充质干细胞来源的胞外小泡通过长链非编码RNA MALAT1/miR-143/NRSN2/Wnt/β-catenin轴促进骨肉瘤细胞的增殖、侵袭和迁移。 2.1.2 长链非编码RNA ANRIL 细胞周期素依赖性激酶抑制因子4b(INK4b)位点的长链非编码RNA ( antisense non-coding RNA in the INK4 locus,ANRIL)首次被发现是在患有家族遗传性黑色素瘤合并神经系统肿瘤患者的遗传分析中。ANRIL可以通过与多梳抑制复合物2(polycomb repressive complex-2,PRC-2)结合并激活2种细胞周期素依赖性激酶抑制剂(p15INK4b和p16INK4a),影响细胞周期中关键生化反应(凋亡、干细胞自我修复及复制性衰老)的调节因子发生基因沉默,抑制细胞凋亡[11]。WANG等[12]研究表明,ANRIL可通过miR-125a/APC轴激活Wnt/β-catenin通路,促进脂多糖诱导的骨髓间充质干细胞成骨分化及抑制凋亡。LIU等[13]在缺血缺氧性疾病研究表明ANRIL基因敲除可通过靶向转化生长因子βR1/Smad信号通路抑制血管内皮细胞活化和凋亡,加重缺氧程度。 2.1.3 长链非编码RNA EPIC1 长链非编码RNA EPIC1在人坏死骨组织和成骨细胞中高度表达,其可以调节成骨细胞中长链非编码RNA EPIC1的靶标Myc来减轻人成骨细胞对缺氧微环境的影响,且经过缺氧微环境后,长链非编码RNA EPIC1和Myc的表达均增强[8],进一步证实长链非编码RNA EPIC1对缺氧损伤的成骨细胞具有保护作用,见图2。 2.1.4 长链非编码RNA 00473 缺氧微环境诱导骨髓间充质干细胞凋亡,导致骨微血管内血栓形成,使股骨头血液供应减少,间接导致骨髓间充质干细胞凋亡。股骨头缺血性坏死患者骨髓间充质干细胞中长链非编码RNA 00473显著下调。长链非编码RNA 00473可以抑制缺氧微环境诱导的骨髓间充质干细胞凋亡过程中最重要的终末剪切酶caspase-3活化,可以减轻骨髓间充质干细胞凋亡[14] 。 2.2 长链非编码RNA与骨髓间充质干细胞异常分化的关系 骨髓间充质干细胞是一种多能干细胞,并且作为脂肪细胞和成骨细胞的共同祖细胞,受到许多生物活性的调控分化。研究表明,缺氧微环境可通过激活或者抑制成骨和成脂的相关转录调节因子影响骨髓间充质干细胞的分化,如PPARγ和C/EBP可调控成脂作用,而runt相关转录因子2 (runt-related transcription factor 2,Runx2)和具有PDZ结合基序的转录共激活子 (The transcriptional co-activator with PDZ binding motif,TAZ)可调控成骨作用[15]。缺氧微环境可诱导骨髓间充质干细胞中PPARγ和C/EBP表达,从而导致成脂分化增强和成骨分化降低。在缺氧微环境病变区域中PPARγ表达升高,并且PPARγ可通过抑制Wnt/LRP5/β-catenin信号传导通路进而抑制成骨基因表达和促进成脂基因表达。TAZ通过激活Runx2依赖的基因转录促进骨髓间充质干细胞的成骨分化,并通过抑制PPARγ依赖的基因转录来阻止骨髓间充质干细胞的成脂分化[16]。在缺氧的作用下,骨髓间充质干细胞衍生的成骨细胞将明显减少,但是脂肪细胞的大小和体积都显著的增加,并且破骨细胞的活性被激活。WANG等[17]研究发现,与对照组相比,在缺氧微环境性凋亡的骨髓间充质干细胞中,有1 878个长链非编码RNA被上调,其中最显著上调的长链非编码RNA是T189748,而有1 842个长链非编码RNA被下调,其中最显著下调的长链非编码RNA是ENST00000588041。证明长链非编码RNA与骨髓间充质干细胞的缺氧性凋亡紧密相连。长链非编码RNA在缺氧性凋亡的骨髓间充质干细胞异常分化中的作用机制,见图3。 "
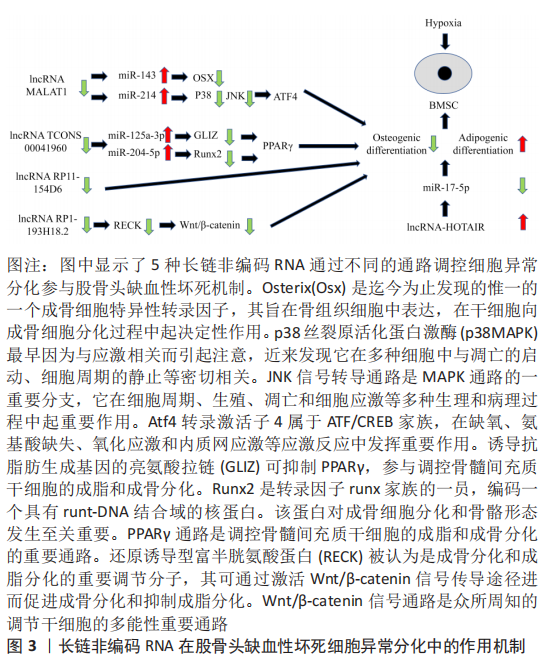
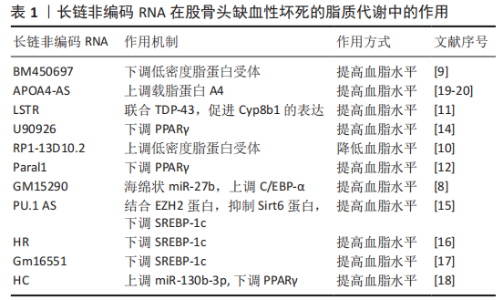
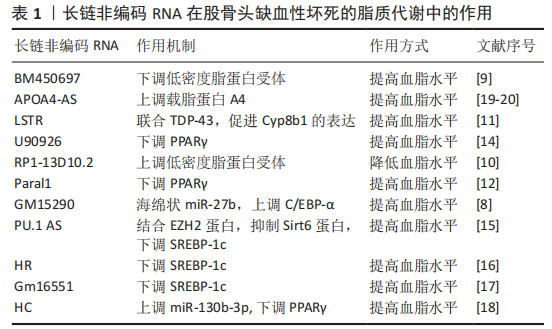
2.3 骨骼肌核糖体生物发生的信号调控 2.3.1 长链非编码RNA-MALAT1 缺氧微环境中,骨髓间充质干细胞的长链非编码RNA-MALAT1表达降低,而miR-214表达升高。相关研究表明长链非编码RNA-MALAT1可通过海绵作用结合miR-214来上调ATF4,减弱miR-143对Osx(Osterix)表达的抑制,从而促进骨髓间充质干细胞成骨分化,改善骨髓间充质干细胞缺氧性凋亡[18-19]。另外也有研究表明,miR-214可以与长链非编码RNA相互作用,通过抑制p38和JNK途径来抑制成骨细胞的分化[20],那么ATF4与p38和JNK途径之间是否有相互作用关系呢?研究表明,JNK失活会抑制ATF4的表达[21],并且 p38磷酸化被激活会导致ATF4活化[22]。这些因素相互作用,共同调节骨髓间充质干细胞的分化。 2.3.2 长链非编码RNA TCONS 00041960 缺氧微环境会使骨髓间充质干细胞中的长链非编码RNA TCONS 00041960下调。TCONS 00041960是位于14号染色体上长度为1 669 nt的长链非编码RNA,其可通过ceRNA作用与miR-125a-3p和 miR-204-5p相互调控,促进诱导抗脂肪生成基因的亮氨酸拉链(glucocorticoid-induced leucine zipper,GLIZ)和Runx2的表达,从而抑制PPARγ,参与调节缺氧微环境骨髓间充质干细胞的成脂和成骨分化[23]。 2.3.3 长链非编码RNA RP11-154D6 XIANG等[24]发现,在股骨头缺血性坏死患者的骨髓间充质干细胞中,有572个DE-长链非编码RNA的表达发生改变,并且长链非编码RNA RP11-154D6的表达显著降低。他们还证明长链非编码RNA RP11-154D6过表达可增强成骨分化,同时抑制成脂分化,但是其具体的机制尚不清楚。 2.4 长链非编码RNA促进骨髓间充质干细胞成脂分化,抑制成骨分化 2.4.1 长链非编码RNA-HOTAIR 长链非编码RNA-HOTAIR在股骨头缺血性坏死患者的骨髓间充质干细胞中异常增加,并且长链非编码RNA-HOTAIR可通过抑制miR-17-5p进而抑制股骨头缺血性坏死的成骨分化[25]。HOTAIR与Bmi1相关,Bmi1过表达可改善成骨微环境来刺激骨形成, HOTAIR与Bmi1的关系可能是股骨头缺血性坏死患者骨髓间充质干细胞分化异常的一种发病机制[26]。 2.4.2 长链非编码RNA RP1-193H18.2 长链非编码RNA RP1-193H18.2在股骨头缺血性坏死患者的骨髓间充质干细胞中显著下调,其过表达可作为几种miRNA的内源性海绵来下调具有Kazal基序的还原诱导型富半胱氨酸蛋白(reversion-inducing cysteine-rich protein with Kazal motifs,RECK)的表达。其被认为是成骨分化和成脂分化的重要调节分子,其可通过激活Wnt/β-catenin信号传导途径进而促进成骨分化和抑制成脂分化[27]。 2.5 长链非编码RNA与脂质代谢紊乱的关系 缺氧微环境使人体脂质代谢紊乱,导致脂质分布异常、骨微血管内脂肪栓塞、脂肪细胞体积增大和髓腔脂肪堆积。另外,还可以引起骨细胞脂质沉积,进而导致细胞死亡,加重骨微循环障碍。研究表明,长链非编码RNA对脂质代谢紊乱的调控是通过一些脂质代谢因素介导的,如XIANG等[28]研究发现过表达RP11-154D6抑制骨髓间充质干细胞在股骨头坏死的成脂分化。HUO等[29]研究发现通过高通量基因测序发现股骨头坏死组织中1 878个长链非编码RNAs和838个mRNAs显著上调,1 842个长链非编码RNAs和1 937个mRNAs显著下调,且存在显著性差异。因此,以下介绍一些长链非编码RNA通过调节脂质代谢因素进而参与脂质代谢的作用,见表1。 2.5.1 长链非编码RNA与脂质的摄取和清除 当脂质积聚在血液中时,低密度脂蛋白会发生氧化修饰,低密度脂蛋白受体(Low-density lipoprotein receptor,LDLR)会摄取和清除血液中的低密度脂蛋白颗粒。而长链非编码RNA BM450697是LDLR的调节因子,其沉默会激活LDLR[30]。长链非编码RNA BM450697可降低 LDLR mRNA 的水平,这可能是由于在LDLR启动子处阻断了与RNA聚合酶Ⅱ和SREBP1a的相互作用[30]。与长链非编码RNA BM450697相反,长链非编码RNA RP1-13D10.2被证明能激活LDLR的转录[31]。Li等[32]发现特异性敲除小鼠肝脏的长链非编码RNA LSTR显著降低血浆三酰甘油水平。他们还发现,长链非编码RNA LSTR基因敲除降低了与TDP-43的结合,抑制了Cyp8b1基因的表达,并增加了胆汁酸中多酚酸与胆酸的比例。胆汁酸成分的改变激活了FXRs,增加了载脂蛋白C2的水平,导致小鼠三酰甘油清除率更高,进而导致血脂水平的降低,减弱缺氧微环境对股骨头微循环的影响。 2.5.2 长链非编码RNA与皮下脂肪细胞脂肪形成 三酰甘油的储存由皮下脂肪细胞控制,其来源于白色脂肪组织(WAT),皮下脂肪动员增加,脂肪生成减少,血脂水平升高。许多长链非编码RNA已被证明通过关键转录因子如PPARγ和C/EBP调控脂肪生成。例如,在皮下脂肪组织中,长链非编码RNA Paral1通过增强PPARγ的转录活性来促进脂肪生成[33],而长链非编码RNA GM15290通过海绵miR-27b以减轻对miR-27b靶基因的C/EBP-α抑制作用,以上调C/EBP-α来正调控脂肪生成[29],从而使血脂降低。 与上述长链非编码RNA的功能不同,AdipoQ AS长链非编码RNA和长链非编码RNA U90926被证明与脂肪生成呈负相关[34-35]。AdipoQ AS长链非编码RNA是AdipoQ基因反向链转录而来长度为2 216 nt的反义长链非编码RNA,可通过形成AdipoQ AS长链非编码RNA/AdipoQ mRNA双链来抑制白色脂肪组织表达,抑制脂肪生成[34]。另外,过表达长链非编码RNA U90926,白色脂肪组织表达下调,进而影响PPARγ的转录活性抑制脂肪生成[37]。 2.5.3 长链非编码RNA与三酰甘油生物合成 三酰甘油的生物合成在调节脂质代谢中起着重要作用,而长链非编码RNA通过参与转录因子SREBP(SREBP-1a、SREBP-1c和SREBP-2)调节三酰甘油的生物合成进而调节脂质代谢。如在慢性砷饲养小鼠的肝脏中,长链非编码RNA PU.1 AS表达显著上调,并且显示血清三酰甘油水平降低。长链非编码RNA PU.1 AS通过与EZH2蛋白相互作用来抑制Sirt6蛋白的表达,从而降低SREBP-1c的表达和血清三酰甘油水平[36]。长链非编码RNA HR和长链非编码RNA Gm16551作为SREBP-1c的负性调节因子,其过表达可抑制三酰甘油的合成,导致血浆三酰甘油水平降低[37-38]。除了长链非编码RNA对SREBP-1c的影响外,长链非编码RNA HC过表达会通过上调miR-130b-3p的表达,抑制PPARγ表达,降低三酰甘油的生物合成[39]。 2.5.4 长链非编码RNA与脂蛋白形成 天然反义转录物 (natural antisense transcript,NAT)是调节脂质稳态的一类长链非编码RNA。天然反义转录物长链非编码RNA是从DNA的反义链转录而来的,且大多数长链非编码RNA属于天然反义转录物[40]。APOA4-AS作为天然反义转录物长链非编码RNA的一种,是载脂蛋白A-IV(Apolipoprotein A-IV,APOA4)的正调节因子。APOA4是高密度脂蛋白和富含三酰甘油的脂蛋白颗粒的主要成分,控制三酰甘油的分泌。APOA4-AS基因敲除降低了ob/ob小鼠APOA4的表达,导致血清三酰甘油水平降低[41]。 2.6 长链非编码RNA与血液供应不足的关系 缺氧微环境可抑制血管内皮细胞生长因子的表达,使血管修复和新生血管形成障碍,导致血栓形成,并且诱导股骨头血管内皮细胞凋亡,最终影响股骨头血液供应。除了血管内皮细胞生长因子,缺氧诱导因子1α、骨形态发生蛋白2和骨钙素等也可促进早期股骨头缺血性坏死患者的骨微血管生成[42]。相关研究表明,长链非编码RNA与血管内皮细胞凋亡密切相关,如YU等[43]对缺氧微环境处理前后基因表达差异分析,发现在骨微血管内皮细胞中有166个长链非编码RNA被下调和73个长链非编码RNA被上调,长链非编码RNA与股骨头微循环障碍密切相关,但是具体的股骨头缺血性坏死的骨微血管内皮细胞中的长链非编码RNA对凋亡的作用机制还需进一步的研究。 长链非编码RNA对人微血管内皮细胞凋亡的作用机制同样也提示文章长链非编码RNA在股骨头缺血性坏死的骨微血管内皮细胞凋亡中具有调控作用。同时,由于Bcl-2/Bax比率下调,caspase-3和caspase-9被切割,抑制HULC可导致人微血管内皮细胞凋亡。其机制可能是长链非编码RNA HULC通过结合miR-124,从而抑制MCL-1被miR-124结合,MCL-1激活PI3K/AKT和JAK/STAT信号通路进而抑制人微血管内皮细胞凋亡[44];另外,抑制长链非编码RNA-UCA1也可显著降低人微血管内皮细胞的生长和血管形成,促进人微血管内皮细胞凋亡,其机制为抑制UCA1可显著上调miR-195的表达,进而抑制MEK/ERK和mTOR信号通路,最终导致CCND1的表达下调,CCND1在促生长、促迁移和促血管生成方面具有重要调控作用,可降低人微血管内皮细胞的生长和血管形成[45]。 2.7 长链非编码RNA与基因多态性的关系 基因多态性是产生于基因水平的变异,一般发生于基因序列中无重要调节功能和非编码蛋白的区域,其主要分为3类:DNA重复序列多态性(repetitive sequence polymorphism,RSP),主要表现于重复序列拷贝数的变异;DNA片段长度多态性(fragment length polymorphism,FLP),即单个碱基的缺失、重复和插入所引起DNA片段长度的变化;单核苷酸多态性(single nucleotide polymorphism,SNP),即分散的单个碱基的不同,在缺氧微环境序列上频繁出现,是目前最受关注的一类多态性。 长链非编码RNA由DNA转录形成,并且DNA某些位点可以调控长链非编码RNA的表达,如位于1p36.12的骨质疏松症风险基因的单核苷酸多态性(rs6426749),作为一种远端等位基因特异性增强子,通过远端染色质环形成来调控长链非编码RNA LINC00339的表达,该环由CCCTC结合因子介导,CCCTC结合因子介导rs6426749和LINC00339之间的长程染色质相互作用[46];ZHOU等[47]发现,在HOTAIR基因单核苷酸多态性(rs7958904)中,与G等位基因相比,rs7958904的C等位基因与骨肉瘤风险显著降低有关。对长链非编码RNA HOTAIR表达的功能分析表明,骨肉瘤组织中长链非编码RNA HOTAIR的表达水平显著高于相应的正常组织,具有rs7958904 CC基因型的受试者的长链非编码RNA HOTAIR水平明显低于其他基因型。文章推测在股骨头缺血性坏死中,基因多态性与长链非编码RNA调控细胞凋亡、骨髓间充质干细胞分化、脂质代谢和血液供应等密切相关,但目前关于与基因多态性相关研究是通过大样本基因差异与疾病发生相关性分析,尚没有文献详细阐述基因多态性如何具体调控,因此值得未来进一步研究。 "
| [1] CHEN CY, DU W, RAO SS, et al. Extracellular vesicles from human urine-derived stem cells inhibit glucocorticoid-induced osteonecrosis of the femoral head by transporting and releasing pro-angiogenic DMBT1 and anti-apoptotic TIMP1. Acta Biomater. 2020;111:208-220. [2] LUO H, LAN W, LI Y, et al. Microarray analysis of long-noncoding RNAs and mRNA expression profiles in human steroid-induced avascular necrosis of the femoral head. J Cell Biochem. 2019;120(9):15800-15813. [3] WANG A, REN M, WANG J. The pathogenesis of steroid-induced osteonecrosis of the femoral head: a systematic review of the literature. Gene. 2018;671:103-109. [4] ZHANG Y, SUN R, ZHANG L, et al. Effect of blood biochemical factors on nontraumatic necrosis of the femoral head: Logistic regression analysis. Orthopade. 2017;46(9):737-743. [5] QI Y, WANG J, SUN M, et al. MMP-14 single-nucleotide polymorphisms are related to steroid-induced osteonecrosis of the femoral head in the population of northern China. Mol Genet Genomic Med. 2019; 7(2):510-519. [6] YU Y, ZHANG Y, WU J, et al. Genetic polymorphisms in IL1B predict susceptibility to steroid-induced osteonecrosis of the femoral head in Chinese Han population. Osteoporos Int. 2019;30(4):871-877. [7] FAN JB, ZHANG Y, LIU W, et al. Long Non-Coding RNA MALAT1 Protects Human Osteoblasts from Dexamethasone-Induced Injury via Activation of PPM1E-AMPK Signaling. Cell Physiol Biochem. 2018;51(1):31-45. [8] XIN JW, JIANG YG. Long noncoding RNA MALAT1 inhibits apoptosis induced by oxygen-glucose deprivation and reoxygenation in human brain microvascular endothelial cells. Exp Ther Med. 2017;13(4):1225-1234. [9] ZHANG Y, TANG X, SHI M, et al.MiR-216a decreases MALAT1 expression, induces G2/M arrest and apoptosis in pancreatic cancer cells. Biochem Biophys Res Commun. 2017;483(2):816-822. [10] Li F, Chen X, Shang C, et al. Bone Marrow Mesenchymal Stem Cells-Derived Extracellular Vesicles Promote Proliferation, Invasion and Migration of Osteosarcoma Cells via the lncRNA MALAT1/miR-143/NRSN2/Wnt/β-Catenin Axis. Onco Targets Ther. 2021;14:737-749. [11] HU X, JIANG H, JIANG X. Downregulation of lncRNA ANRIL inhibits proliferation, induces apoptosis, and enhances radiosensitivity in nasopharyngeal carcinoma cells through regulating miR-125a. Cancer Biol Ther. 2017;18(5):331-338. [12] WANG YL, LV FY, HUANG LT, et al. Human amnion-derived mesenchymal stem cells promote osteogenic differentiation of lipopolysaccharide-induced human bone marrow mesenchymal stem cells via ANRIL/miR-125a/APC axis. Stem Cell Res Ther. 2021;12:25-35. [13] LIU XL, LI SF, YANG YI, et al. The lncRNA ANRIL regulates endothelial dysfunction by targeting the let-7b/TGF-βR1 signalling pathway. J Cell Physiol. 2021;236:2058-2069. [14] XU Y, JIANG Y, WANG Y, et al. LINC00473 regulated apoptosis, proliferation and migration but could not reverse cell cycle arrest of human bone marrow mesenchymal stem cells induced by a high-dosage of dexamethasone. Stem Cell Res. 2020;48:1019-1027. [15] HAN L, WANG B, WANG R, et al. The shift in the balance between osteoblastogenesis and adipogenesis of mesenchymal stem cells mediated by glucocorticoid receptor. Stem Cell Res Ther. 2019;10(1): 377. [16] HONG JH, HWANG ES, MCMANUS MT, et al. TAZ, a transcriptional modulator of mesenchymal stem cell differentiation. Science. 2005; 309(5737):1074-1078. [17] WANG Q, YANG Q, CHEN G, et al. lncRNA expression profiling of BMSCs in osteonecrosis of the femoral head associated with increased adipogenic and decreased osteogenic differentiation. Sci Rep. 2018;8(1):117-127. [18] GAO Y, XIAO F, WANG C, et al. Long noncoding RNA MALAT1 promotes osterix expression to regulate osteogenic differentiation by targeting miRNA-143 in human bone marrow-derived mesenchymal stem cells. J Cell Biochem. 2018;119(8):6986-6996. [19] HUANG XZ, HUANG J, LI WZ, et al. lncRNA-MALAT1 promotes osteogenic differentiation through regulating ATF4 by sponging miR-214: implication of steroid-induced avascular necrosis of the femoral head. Steroids. 2020;154:108-117. [20] GUO Y, LI L, GAO J, et al. miR-214 suppresses the osteogenic differentiation of bone marrow-derived mesenchymal stem cells and these effects are mediated through the inhibition of the JNK and p38 pathways. Int J Mol Med. 2017;39(1):71-80. [21] HOCSAK E, SZABO V, KALMAN N, et al. PARP inhibition protects mitochondria and reduces ROS production via PARP-1-ATF4-MKP-1-MAPK retrograde pathway. Free Radic Biol Med. 2017;108:770-784. [22] SU Z, XU T, WANG Y, et al. Low‑intensity pulsed ultrasound promotes apoptosis and inhibits angiogenesis via p38 signaling‑mediated endoplasmic reticulum stress in human endothelial cells. Mol Med Rep. 2019;19(6):4645-4654. [23] SHANG G, WANG Y, XU Y, et al. Long non-coding RNA TCONS_00041960 enhances osteogenesis and inhibits adipogenesis of rat bone marrow mesenchymal stem cell by targeting miR-204-5p and miR-125a-3p. J Cell Physiol. 2018;233(8):6041-6051. [24] XIANG S, LI Z, WENG X. The role of lncRNA RP11-154D6 in steroid-induced osteonecrosis of the femoral head through BMSCs regulation. J Cell Biochem. 2019;120(10):18435-18445. [25] WEI B, WEI W, ZHAO B, et al. Long non-coding RNA HOTAIR inhibits miR-17-5p to regulate osteogenic differentiation and proliferation in non-traumatic osteonecrosis of femoral head. PLoS One. 2017;12(2):1016-1025. [26] ZHOU X, DAI X, WU X, et al. Overexpression of Bmi1 in Lymphocytes Stimulates Skeletogenesis by Improving the Osteogenic Microenvironment. Sci Rep. 2016;6:291-302. [27] MAHL C, EGEA V, MEGENS RT, et al. RECK (reversion-inducing cysteine-rich protein with Kazal motifs) regulates migration, differentiation and Wnt/β-catenin signaling in human mesenchymal stem cells. Cell Mol Life Sci. 2016;73(7):1489-1501. [28] XIANG S, LI Z, WENG XS. The role of lncRNA RP11-154D6 in steroid-induced osteonecrosis of the femoral head through BMSC regulation. J Cell Biochem, 2019;120:18435-18445. [29] HUO S, ZHOU Y, HE X, et al. Zhou X, Zheng L. Insight into the Role of Long Non-coding RNAs During Osteogenesis in Mesenchymal Stem Cells. Curr Stem Cell Res Ther. 2018;13(1):52-59. [30] RAY RM, HANSEN AH, SLOTT S, et al. Control of LDL Uptake in Human Cells by Targeting the LDLR Regulatory Long Non-coding RNA BM450697. Mol Ther Nucleic Acids. 2019;17:264-276. [31] MITCHEL K, THEUSCH E, CUBITT C, et al. RP1-13D10.2 Is a Novel Modulator of Statin-Induced Changes in Cholesterol. Circ Cardiovasc Genet. 2016;9(3):223-230. [32] LI P, RUAN X, YANG L, et al. A liver-enriched long non-coding RNA, lncLSTR, regulates systemic lipid metabolism in mice. Cell Metab. 2015;21(3):455-467. [33] NUERMAIMAITI N, LIU J, LIANG X, et al. Effect of lncRNA HOXA11-AS1 on adipocyte differentiation in human adipose-derived stem cells. Biochem Biophys Res Commun. 2018;495(2):1878-1884. [34] CAI R, SUN Y, QIMUGE N, et al. Adiponectin AS lncRNA inhibits adipogenesis by transferring from nucleus to cytoplasm and attenuating Adiponectin mRNA translation. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863(4):420-432. [35] CHEN J, LIU Y, LU S, et al.The role and possible mechanism of lncRNA U90926 in modulating 3T3-L1 preadipocyte differentiation. Int J Obes (Lond). 2017;41(2):299-308. [36] DONG Z, LI C, YIN C, et al. lncRNA PU.1 AS regulates arsenic-induced lipid metabolism through EZH2/Sirt6/SREBP-1c pathway. J Environ Sci (China). 2019;85:138-146. [37] YANG L, LI P, YANG W, et al. Integrative Transcriptome Analyses of Metabolic Responses in Mice Define Pivotal lncRNA Metabolic Regulators. Cell Metab. 2016;24(4):627-639. [38] LI D, CHENG M, NIU Y, et al. Identification of a novel human long non-coding RNA that regulates hepatic lipid metabolism by inhibiting SREBP-1c. Int J Biol Sci. 2017;13(3):349-357. [39] LAN X, WU L, WU N, et al. Long Noncoding RNA lnc-HC Regulates PPARγ-Mediated Hepatic Lipid Metabolism through miR-130b-3p. Mol Ther Nucleic Acids. 2019;18:954-965. [40] HALLEY P, KADAKKUZHA BM, FAGHIHI MA, et al. Regulation of the apolipoprotein gene cluster by a long noncoding RNA. Cell Rep. 2014; 6(1):222-230. [41] QIN W, LI X, XIE L, et al. A long non-coding RNA, APOA4-AS, regulates APOA4 expression depending on HuR in mice. Nucleic Acids Res. 2016; 44(13):6423-6433. [42] XIE Q, XIE J, ZHONG J, et al. Hypoxia enhances angiogenesis in an adipose-derived stromal cell/endothelial cell co-culture 3D gel model. Cell Prolif. 2016;49(2):236-245. [43] YU QS, GUO WS, CHENG LM, et al. Glucocorticoids Significantly Influence the Transcriptome of Bone Microvascular Endothelial Cells of Human Femoral Head. Chin Med J (Engl). 2015;128(14):1956-1963. [44] YIN D, LI Y, FU C, et al. Pro-Angiogenic Role of lncRNA HULC in Microvascular Endothelial Cells via Sequestrating miR-124. Cell Physiol Biochem. 2018;50(6):2188-2202. [45] YIN D, FU C, SUN D.Silence of lncRNA UCA1 Represses the Growth and Tube Formation of Human Microvascular Endothelial Cells Through miR-195. Cell Physiol Biochem. 2018;49(4):1499-1511. [46] CHEN XF, ZHU DL, YANG M, et al.An Osteoporosis Risk SNP at 1p36.12 Acts as an Allele-Specific Enhancer to Modulate LINC00339 Expression via Long-Range Loop Formation. Am J Hum Genet. 2018;102(5): 776-793. [47] ZHOU Q, CHEN F, FEI Z, et al. Genetic variants of lncRNA HOTAIR contribute to the risk of osteosarcoma. Oncotarget. 2016;7(15): 19928-19934. (责任编辑:WJ,ZN,ZH) |
| [1] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [2] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [3] | Chen Junming, Yue Chen, He Peilin, Zhang Juntao, Sun Moyuan, Liu Youwen. Hip arthroplasty versus proximal femoral nail antirotation for intertrochanteric fractures in older adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1452-1457. |
| [4] | Chen Jinping, Li Kui, Chen Qian, Guo Haoran, Zhang Yingbo, Wei Peng. Meta-analysis of the efficacy and safety of tranexamic acid in open spinal surgery [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1458-1464. |
| [5] | Hu Kai, Qiao Xiaohong, Zhang Yonghong, Wang Dong, Qin Sihe. Treatment of displaced intra-articular calcaneal fractures with cannulated screws and plates: a meta-analysis of 15 randomized controlled trials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1465-1470. |
| [6] | Huang Dengcheng, Wang Zhike, Cao Xuewei. Comparison of the short-term efficacy of extracorporeal shock wave therapy for middle-aged and elderly knee osteoarthritis: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1471-1476. |
| [7] | Xu Feng, Kang Hui, Wei Tanjun, Xi Jintao. Biomechanical analysis of different fixation methods of pedicle screws for thoracolumbar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1313-1317. |
| [8] | Jiang Yong, Luo Yi, Ding Yongli, Zhou Yong, Min Li, Tang Fan, Zhang Wenli, Duan Hong, Tu Chongqi. Von Mises stress on the influence of pelvic stability by precise sacral resection and clinical validation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1318-1323. |
| [9] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [10] | Zhang Yu, Tian Shaoqi, Zeng Guobo, Hu Chuan. Risk factors for myocardial infarction following primary total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1340-1345. |
| [11] | Wei Wei, Li Jian, Huang Linhai, Lan Mindong, Lu Xianwei, Huang Shaodong. Factors affecting fall fear in the first movement of elderly patients after total knee or hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1351-1355. |
| [12] | Wang Jinjun, Deng Zengfa, Liu Kang, He Zhiyong, Yu Xinping, Liang Jianji, Li Chen, Guo Zhouyang. Hemostatic effect and safety of intravenous drip of tranexamic acid combined with topical application of cocktail containing tranexamic acid in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1356-1361. |
| [13] | Xiao Guoqing, Liu Xuanze, Yan Yuhao, Zhong Xihong. Influencing factors of knee flexion limitation after total knee arthroplasty with posterior stabilized prostheses [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1362-1367. |
| [14] | Huang Zexiao, Yang Mei, Lin Shiwei, He Heyu. Correlation between the level of serum n-3 polyunsaturated fatty acids and quadriceps weakness in the early stage after total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1375-1380. |
| [15] | Zhang Chong, Liu Zhiang, Yao Shuaihui, Gao Junsheng, Jiang Yan, Zhang Lu. Safety and effectiveness of topical application of tranexamic acid to reduce drainage of elderly femoral neck fractures after total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1381-1386. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||