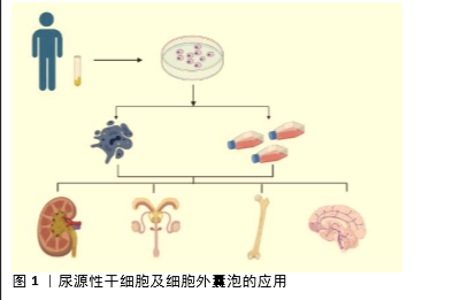
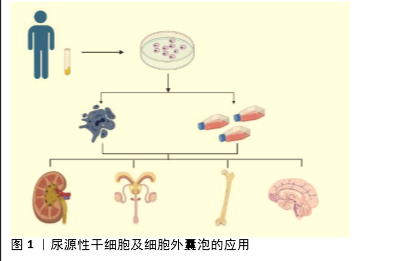
Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (31): 5046-5052.doi: 10.12307/2021.149
Previous Articles Next Articles
Effect of urinary-derived stem cells and extracellular vesicles in tissue damage repair
Cui Tianning1, Liu Tao1, Xiao Xiangyang1, Wang Shuai2, Zhang Nini2, Huang Guilin2
- 1Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Department of Dentofacial Surgery, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2020-11-24Revised:2020-11-30Accepted:2021-01-09Online:2021-11-08Published:2021-04-25 -
Contact:Huang Guilin, MD, Professor, Master’s supervisor, Department of Dentofacial Surgery, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Cui Tianning, Master candidate, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81760201, 81960204 (to HGL); the National Natural Science Foundation of China, No. 81860198 (to ZNN)
CLC Number:
Cite this article
Cui Tianning, Liu Tao, Xiao Xiangyang, Wang Shuai, Zhang Nini, Huang Guilin. Effect of urinary-derived stem cells and extracellular vesicles in tissue damage repair[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 5046-5052.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
| [1] MOAD M, PAL D, HEPBURN AC, et al. A novel model of urinary tract differentiation, tissue regeneration, and disease: reprogramming human prostate and bladder cells into induced pluripotent stem cells. Eur Urol. 2013;64(5):753-761. [2] SALEM SA, HWIE AN, SAIM A, et al. Human adipose tissue derived stem cells as a source of smooth muscle cells in the regeneration of muscular layer of urinary bladder wall. Malays J Med Sci. 2013;20(4):80-87. [3] DREWA T, JOACHIMIAK R, KAZNICA A, et al. Hair stem cells for bladder regeneration in rats: preliminary results. Transplant Proc. 2009;41(10): 4345-4351. [4] BHARADWAJ S, LIU G, SHI Y, et al. Characterization of urine-derived stem cells obtained from upper urinary tract for use in cell-based urological tissue engineering. Tissue Eng Part A. 2011;17(15-16): 2123-2132. [5] ZHANG Y, MCNEILL E, TIAN H, et al. Urine derived cells are a potential source for urological tissue reconstruction. J Urol. 2008;180(5): 2226-2233. [6] QIN D, LONG T, DENG J, et al. Urine-derived stem cells for potential use in bladder repair. Stem Cell Res Ther. 2014;5(3):69. [7] QIN H, ZHU C, AN Z, et al. Silver nanoparticles promote osteogenic differentiation of human urine-derived stem cells at noncytotoxic concentrations. Int J Nanomedicine. 2014;9:2469-2478. [8] LIU G, PARETA RA, WU R, et al. Skeletal myogenic differentiation of urine-derived stem cells and angiogenesis using microbeads loaded with growth factors. Biomaterials. 2013;34(4):1311-1326. [9] WU S, LIU Y, BHARADWAJ S, et al. Human urine-derived stem cells seeded in a modified 3D porous small intestinal submucosa scaffold for urethral tissue engineering. Biomaterials. 2011;32(5):1317-1326. [10] CHUN SY, KIM HT, LEE JS, et al. Characterization of urine-derived cells from upper urinary tract in patients with bladder cancer. Urology. 2012;79(5):1186.e1-1186.e7. [11] ZHU Q, LI Q, NIU X, et al. Extracellular Vesicles Secreted by Human Urine-Derived Stem Cells Promote Ischemia Repair in a Mouse Model of Hind-Limb Ischemia. Cell Physiol Biochem. 2018;47(3):1181-1192. [12] GURUNATHAN S, KANG MH, JEYARAJ M, et al. Review of the Isolation, Characterization, Biological Function, and Multifarious Therapeutic Approaches of Exosomes. Cells. 2019;8(4):307. [13] LEE Y, EL ANDALOUSSI S, WOOD MJ. Exosomes and microvesicles: extracellular vesicles for genetic information transfer and gene therapy. Hum Mol Genet. 2012;21(R1):R125-134. [14] KONALA VB, MAMIDI MK, BHONDE R, et al. The current landscape of the mesenchymal stromal cell secretome: A new paradigm for cell-free regeneration. Cytotherapy. 2016;18(1):13-24. [15] MADRIGAL M, RAO KS, RIORDAN NH. A review of therapeutic effects of mesenchymal stem cell secretions and induction of secretory modification by different culture methods. J Transl Med. 2014;12:260. [16] LIU Y, MA W, LIU B, et al. Urethral reconstruction with autologous urine-derived stem cells seeded in three-dimensional porous small intestinal submucosa in a rabbit model. Stem Cell Res Ther. 2017;8(1):63. [17] VERSTEEGDEN LRM, DE JONGE PKJD, INTHOUT J, et al. Tissue Engineering of the Urethra: A Systematic Review and Meta-analysis of Preclinical and Clinical Studies. Eur Urol. 2017;72(4):594-606. [18] BHARADWAJ S, LIU G, SHI Y, et al. Multipotential differentiation of human urine-derived stem cells: potential for therapeutic applications in urology. Stem Cells. 2013;31(9):1840-1856. [19] CHAN YY, SANDLIN SK, KURZROCK EA, et al. The Current Use of Stem Cells in Bladder Tissue Regeneration and Bioengineering. Biomedicines. 2017;5(1):4. [20] ZHAO Z, LIU D, CHEN Y, et al. Ureter tissue engineering with vessel extracellular matrix and differentiated urine-derived stem cells. Acta Biomater. 2019;88:266-279. [21] SINGH A, BIVALACQUA TJ, SOPKO N. Urinary Tissue Engineering: Challenges and Opportunities. Sex Med Rev. 2018;6(1):35-44. [22] DJOUAD F, BOUFFI C, GHANNAM S, et al. Mesenchymal stem cells: innovative therapeutic tools for rheumatic diseases. Nat Rev Rheumatol. 2009;5(7):392-399. [23] LIU G, WU R, YANG B, et al. Human Urine-Derived Stem Cell Differentiation to Endothelial Cells with Barrier Function and Nitric Oxide Production. Stem Cells Transl Med. 2018;7(9):686-698. [24] LONG T, WU R, LU X, et al. Urine-Derived Stem Cells for Tissue Repair in the Genitourinary System. J Stem Cell Res Ther. 2015;5:11. [25] YANG Q, CHEN X, ZHENG T, et al. Transplantation of Human Urine-Derived Stem Cells Transfected with Pigment Epithelium-Derived Factor to Protect Erectile Function in a Rat Model of Cavernous Nerve Injury. Cell Transplant. 2016;25(11):1987-2001. [26] CHEN WM, YANG QY, BIAN J, et al. Protective effect of urine-derived stem cells on erectile dysfunction in rats with cavernous nerve injury. Zhonghua Nan Ke Xue. 2018;24(6):483-490. [27] ZHANG C, LUO D, LI T, et al. Transplantation of Human Urine-Derived Stem Cells Ameliorates Erectile Function and Cavernosal Endothelial Function by Promoting Autophagy of Corpus Cavernosal Endothelial Cells in Diabetic Erectile Dysfunction Rats. Stem Cells Int. 2019;2019:2168709. [28] OUYANG B, XIE Y, ZHANG C, et al. Extracellular Vesicles From Human Urine-Derived Stem Cells Ameliorate Erectile Dysfunction in a Diabetic Rat Model by Delivering Proangiogenic MicroRNA. Sex Med. 2019; 7(2):241-250. [29] YANG L, XING G, WANG L, et al. Acute kidney injury in China: a cross-sectional survey. Lancet. 2015;386(10002):1465-1471. [30] CARONNI GM, KOMAROMI H, GUILLERMIN A, et al. Acute kidney injury in 2017 - Management in a secondary care hospital: an example of interdisciplinary collaboration. Rev Med Suisse. 2017;13(573):1502-1508. [31] OZTURK H, CETINKAYA A, DUZCU SE, et al. Carvacrol attenuates histopathogic and functional impairments induced by bilateral renal ischemia/reperfusion in rats. Biomed Pharmacother. 2018;98:656-661. [32] OLIVEIRA ARCOLINO F, TORT PIELLA A, PAPADIMITRIOU E, et al. Human Urine as a Noninvasive Source of Kidney Cells. Stem Cells Int. 2015; 2015:362562. [33] TIAN SF, JIANG ZZ, LIU YM, et al. Human urine-derived stem cells contribute to the repair of ischemic acute kidney injury in rats. Mol Med Rep. 2017;16(4):5541-5548. [34] ZHANG C, GEORGE SK, WU R, et al. Reno-protection of Urine-derived Stem Cells in A Chronic Kidney Disease Rat Model Induced by Renal Ischemia and Nephrotoxicity. Int J Biol Sci. 2020;16(3):435-446. [35] SUN B, LUO X, YANG C, et al. Therapeutic Effects of Human Urine-Derived Stem Cells in a Rat Model of Cisplatin-Induced Acute Kidney Injury In Vivo and In Vitro. Stem Cells Int. 2019;2019:8035076. [36] GONZALES PA, PISITKUN T, HOFFERT JD, et al. Large-scale proteomics and phosphoproteomics of urinary exosomes. J Am Soc Nephrol. 2009; 20(2):363-379. [37] KURO-O M, MATSUMURA Y, AIZAWA H, et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature. 1997; 390(6655):45-51. [38] GRANGE C, PAPADIMITRIOU E, DIMUCCIO V, et al. Urinary Extracellular Vesicles Carrying Klotho Improve the Recovery of Renal Function in an Acute Tubular Injury Model. Mol Ther. 2020;28(2):490-502. [39] HWANG Y, CHA SH, HONG Y, et al. Direct differentiation of insulin-producing cells from human urine-derived stem cells. Int J Med Sci. 2019;16(12):1668-1676. [40] XIONG G, TAO L, MA WJ, et al. Urine-derived stem cells for the therapy of diabetic nephropathy mouse model. Eur Rev Med Pharmacol Sci. 2020;24(3):1316-1324. [41] JIANG ZZ, LIU YM, NIU X, et al. Exosomes secreted by human urine-derived stem cells could prevent kidney complications from type I diabetes in rats. Stem Cell Res Ther. 2016;7:24. [42] DUAN YR, CHEN BP, CHEN F, et al. Exosomal microRNA-16-5p from human urine-derived stem cells ameliorates diabetic nephropathy through protection of podocyte. J Cell Mol Med. 2019 Sep 30. doi: 10.1111/jcmm.14558. Online ahead of print. [43] SUN G, DING B, WAN M, et al. Formation and optimization of three-dimensional organoids generated from urine-derived stem cells for renal function in vitro. Stem Cell Res Ther. 2020;11(1):309. [44] GUAN J, ZHANG J, LI H, et al. Human Urine Derived Stem Cells in Combination with β-TCP Can Be Applied for Bone Regeneration. PLoS One. 2015;10(5):e0125253. [45] SUN X, ZHENG W, QIAN C, et al. Focal adhesion kinase promotes BMP2-induced osteogenic differentiation of human urinary stem cells via AMPK and Wnt signaling pathways. J Cell Physiol. 2020;235(5):4954-4964. [46] SUN Z, MA Y, CHEN F, et al. miR-133b and miR-199b knockdown attenuate TGF-β1-induced epithelial to mesenchymal transition and renal fibrosis by targeting SIRT1 in diabetic nephropathy. Eur J Pharmacol. 2018;837:96-104. [47] CHEN L, LI L, XING F, et al. Human Urine-Derived Stem Cells: Potential for Cell-Based Therapy of Cartilage Defects. Stem Cells Int. 2018;2018: 4686259. [48] CHEN CY, RAO SS, TAN YJ, et al. Extracellular vesicles from human urine-derived stem cells prevent osteoporosis by transferring CTHRC1 and OPG. Bone Res. 2019;7:18. [49] 刘一飞,王宇辰,朱昱,等.尿源性干细胞的外泌体修复骨不连的作用研究[J].上海医学,2019,42(7):411-417. [50] CHEN CY, DU W, RAO SS, et al. Extracellular vesicles from human urine-derived stem cells inhibit glucocorticoid-induced osteonecrosis of the femoral head by transporting and releasing pro-angiogenic DMBT1 and anti-apoptotic TIMP1. Acta Biomater. 2020;111:208-220. [51] ZHANG J, LIU X, LI H, et al. Exosomes/tricalcium phosphate combination scaffolds can enhance bone regeneration by activating the PI3K/Akt signaling pathway. Stem Cell Res Ther. 2016;7(1):136. [52] CHEN W, XIE M, YANG B, et al. Skeletal myogenic differentiation of human urine-derived cells as a potential source for skeletal muscle regeneration. J Tissue Eng Regen Med. 2017;11(2):334-341. [53] WU R, HUANG C, WU Q, et al. Exosomes secreted by urine-derived stem cells improve stress urinary incontinence by promoting repair of pubococcygeus muscle injury in rats. Stem Cell Res Ther. 2019;10(1):80. [54] GUAN JJ, NIU X, GONG FX, et al. Biological characteristics of human-urine-derived stem cells: potential for cell-based therapy in neurology. Tissue Eng Part A. 2014;20(13-14):1794-1806. [55] KIM JY, CHUN SY, PARK JS, et al. Laminin and Platelet-Derived Growth Factor-BB Promote Neuronal Differentiation of Human Urine-Derived Stem Cells. Tissue Eng Regen Med. 2017;15(2):195-209. [56] LING X, ZHANG G, XIA Y, et al. Exosomes from human urine-derived stem cells enhanced neurogenesis via miR-26a/HDAC6 axis after ischaemic stroke. J Cell Mol Med. 2020;24(1):640-654. [57] BALASUBRAMANIYAN V, BODDEKE E, BAKELS R, et al. Effects of histone deacetylation inhibition on neuronal differentiation of embryonic mouse neural stem cells. Neuroscience. 2006;143(4):939-951. [58] SIEBZEHNRUBL FA, BUSLEI R, EYUPOGLU IY, et al. Histone deacetylase inhibitors increase neuronal differentiation in adult forebrain precursor cells. Exp Brain Res. 2007;176(4):672-678. [59] SUN G, FU C, SHEN C, et al. Histone deacetylases in neural stem cells and induced pluripotent stem cells. J Biomed Biotechnol. 2011; 2011:835968. [60] HSIEH J, NAKASHIMA K, KUWABARA T, et al. Histone deacetylase inhibition-mediated neuronal differentiation of multipotent adult neural progenitor cells. Proc Natl Acad Sci U S A. 2004;101(47): 16659-16664. [61] CAI J, YU X, XU R, et al. Maximum efficacy of mesenchymal stem cells in rat model of renal ischemia-reperfusion injury: renal artery administration with optimal numbers. PLoS One. 2014;9(3):e92347. [62] KIM SY, JOGLEKAR MV, HARDIKAR AA, et al. Placenta Stem/Stromal Cell-Derived Extracellular Vesicles for Potential Use in Lung Repair. Proteomics. 2019;19(17):e1800166. |
| [1] | Wang Menghan, Qi Han, Zhang Yuan, Chen Yanzhi. Three kinds of 3D printed models assisted in treatment of Robinson type II B2 clavicle fracture [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1403-1408. |
| [2] | Yang Cekai, Cai Zhuoyan, Chen Ming, Liu Hao, Weng Rui, Cui Jianchao, Zhang Shuncong, Yao Zhensong. Relationship between degeneration of paraspinal muscle and refractures in postmenopausal women treated by percutaneous vertebroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1414-1419. |
| [3] | Yu Weijie, Liu Aifeng, Chen Jixin, Guo Tianci, Jia Yizhen, Feng Huichuan, Yang Jialin. Advantages and application strategies of machine learning in diagnosis and treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1426-1435. |
| [4] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [5] | Chen Kaijia, Liu Jingyun, Cao Ning, Sun Jianbo, Zhou Yan, Mei Jianguo, Ren Qiang. Application and prospect of tissue engineering in treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1450-1456. |
| [6] | Xiaheida·Yilaerjiang, Nijiati·Tuerxun, Reyila·Kuerban, Baibujiafu·Yelisi, Chen Xin. Three-dimensional finite element analysis of the distribution pattern of stress in bone tissues with different characteristics [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1277-1282. |
| [7] | Lin Zeyu, Xu Lin. Research progress in gout-induced bone destruction mechanism [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1295-1300. |
| [8] | Zhang Xiaoyun, Liu Hua, Chai Yuan, Chen Feng, Zeng Hao, Gao Zhengang, Huang Yourong. Effect of Yishen Gushu Formula on bone metabolic markers and clinical efficacyn in patients with osteoporosis of kidney deficiency and blood stasis type [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1155-1160. |
| [9] | Huang Xiarong, Hu Lizhi, Sun Guanghua, Peng Xinke, Liao Ying, Liao Yuan, Liu Jing, Yin Linwei, Zhong Peirui, Peng Ting, Zhou Jun, Qu Mengjian. Effect of electroacupuncture on the expression of P53 and P21 in articular cartilage and subchondral bone of aged rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1174-1179. |
| [10] | Dai Yuexing, Zheng Liqin, Wu Minhui, Li Zhihong, Li Shaobin, Zheng Desheng, Lin Ziling. Effect of vessel number on computational fluid dynamics in vascular networks [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1206-1210. |
| [11] | Zhuang Xinyi, Peng Yuanhao, Yu Ting, Lyu Dongmei, Wen Xiujie, Cheng Qian. Cone-beam CT evaluation of bone mass in the external oblique line of the mandible in adolescents with different cervical vertebral bone ages [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1253-1258. |
| [12] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [13] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [14] | Huang Peizhen, Dong Hang, Cai Qunbin, Lin Ziling, Huang Feng. Finite element analysis of anterograde and retrograde intramedullary nail for different areas of femoral shaft fractures [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 868-872. |
| [15] | Wang Tihui, Wang Xu, Wu Jinqing, Chen Jiliang, Wang Xiaolu, Miao Juan. Application of three-dimensional simulated osteotomy of the distal femur in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 905-910. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||