Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (26): 4130-4136.doi: 10.12307/2021.110
Previous Articles Next Articles
Effect of proanthocyanidins on proliferation and differentiation of skeletal muscle satellite cells under hypoxic-ischemic condition
Chen Shi1, Du Chao1, He Xuemei1, 2, Zhou Xiangyu1
- 1Department of Thyroid Surgery, 2Medical Experimental Center, Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China
-
Received:2020-09-12Revised:2020-09-15Accepted:2020-10-22Online:2021-09-18Published:2021-04-26 -
Contact:Zhou Xiangyu, MD, Professor, Department of Thyroid Surgery, Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China E-mail:xiangyuzhou971@163.com -
About author:Chen Shi, Master candidate, Physician, Department of Thyroid Surgery, Affiliated Hospital of Southwest Medical University, Luzhou 646000, Sichuan Province, China -
Supported by:Sichuan Science and Technology Department Project, No. 2018JY0408 (to ZXY); Chinese Medicine Administration Project, No. 2018JC040 (to ZXY); National Natural Science Foundation of China (Youth Program), No. 81800421 (to HXM).
CLC Number:
Cite this article
Chen Shi, Du Chao, He Xuemei, Zhou Xiangyu. Effect of proanthocyanidins on proliferation and differentiation of skeletal muscle satellite cells under hypoxic-ischemic condition[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4130-4136.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
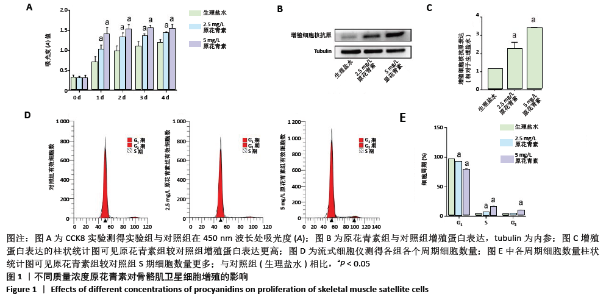
2.1 细胞实验结果 2.1.1 原花青素对缺血缺氧条件下人骨骼肌卫星细胞的影响 首先将人骨骼肌卫星细胞置于生理盐水或不同质量浓度原花青素(5,10,15,20 mg/L)的培养基中培养1 d。当原花青素质量浓度增加到5 mg/L以上时,人骨骼肌卫星细胞的细胞状态变差,提示原花青素质量浓度不超过5 mg/L可能是合适的诱导浓度。 2.1.2 原花青素对缺血缺氧条件下人骨骼肌卫星细胞增殖的影响 骨骼肌肌卫星细胞处于缺氧缺血状态时,细胞被激活,进入细胞周期,进行增殖。为了探索不同质量浓度原花青素对细胞增殖的影响,用生理盐水(对照组)、2.5 mg/L或5 mg/L原花青素(实验组)分别处理细胞,并置于缺血缺氧条件下培养,于不同时间点观察各组细胞增殖情况。 (1)细胞增殖实验:缺血缺氧第1,2,3,4天,原花青素组A值明显高于对照组,差异有显著性意义(P < 0.05);其中5 mg/L原花青素组A值高于2.5 mg/L原花青素组,但差异无显著性意义(P > 0.05),见图1A,说明原花青素组细胞增殖明显高于对照组。 (2)增殖蛋白的表达:缺血缺氧第1天,原花青素组增殖细胞核抗原蛋白表达明显高于对照组,差异有显著性意义(P < 0.05);其中5 mg/L原花青素组中蛋白表达高于2.5 mg/L原花青素组,但差异无显著性意义(P > 0.05),见图1B,C。 (3)细胞周期:缺血缺氧第1天,原花青素组S期所占细胞数量明显多于对照组,说明原花青素组DNA复制活跃;其中5 mg/L原花青素组中蛋白表达高于2.5 mg/L原花青素组,但差异无显著性意义(P > 0.05),见图1D,E,说明原花青素组细胞增殖较对照组活跃。 以上结果提示,原花青素可能促进骨骼肌卫星细胞增殖。"
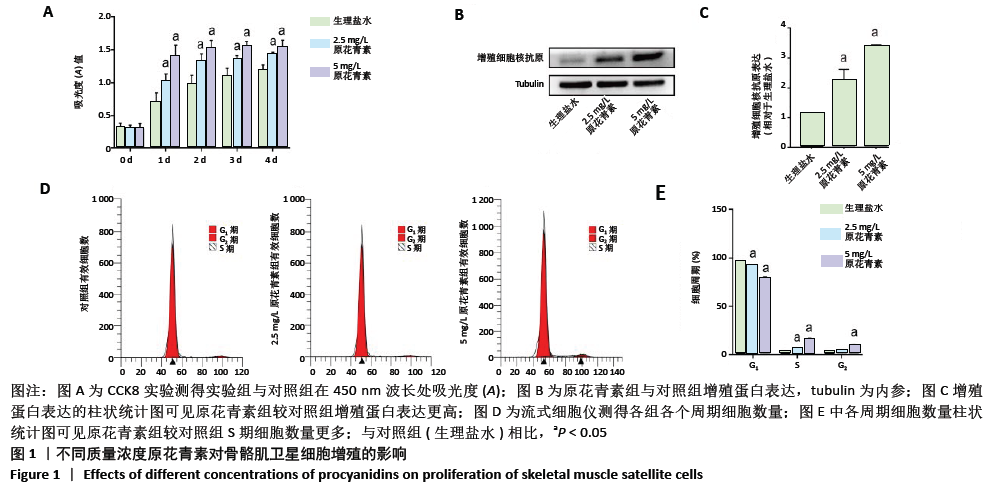
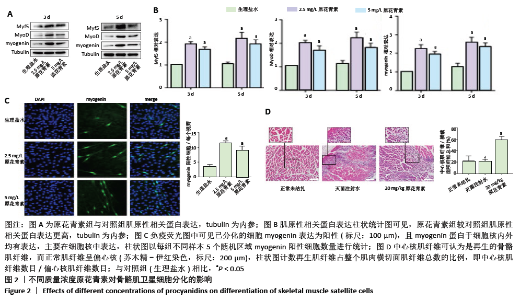
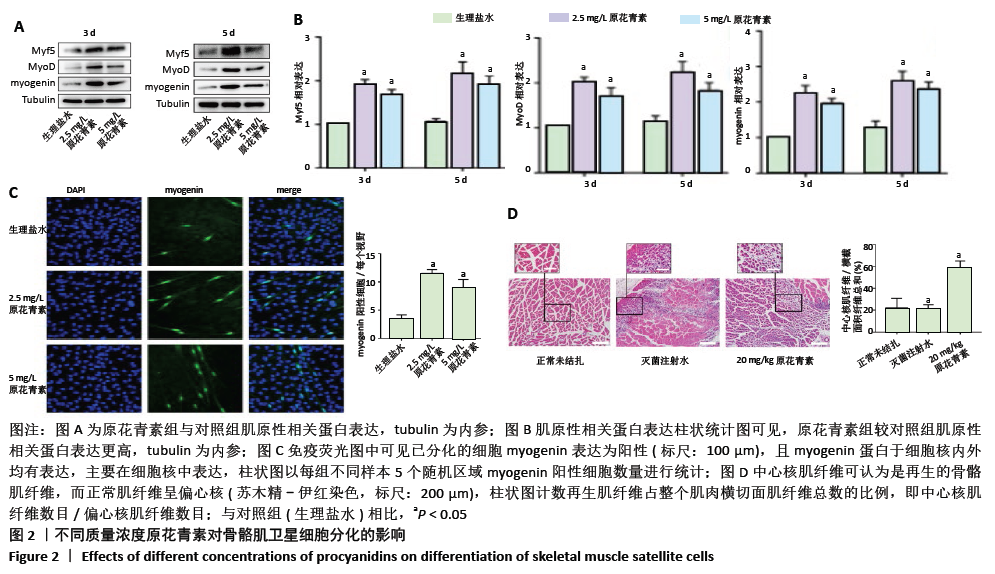
2.1.3 原花青素对缺血缺氧条件下人骨骼肌卫星细胞分化的影响 当人骨骼肌卫星细胞处于缺氧缺血状态时,细胞被激活,诱导细胞融合及多核肌管形成。为了探索不同质量浓度原花青素对缺血缺氧条件下人骨骼肌卫星细胞分化的影响,用生理盐水(对照组)、2.5 mg/L或5 mg/L原花青素(实验组)分别处理细胞,并且置于缺血缺氧条件下培养3-5 d,观察各组细胞分化程度。 (1)肌原性标志蛋白的表达:3组细胞均有肌原性标志蛋白的表达,且随时间增加表达增加。与对照组相比较,两种质量浓度的原花青素组肌原性相关蛋白MyF5、myoD和myogenin的表达更高(P <0.05);其中2.5 mg/L原花青素组中蛋白表达略高于5 mg/L原花青素组,但差异无显著性意义(P > 0.05),见图2A,B。 (2)细胞免疫荧光观察肌管的形成:Myogenin作为肌源性分化的标志主要于细胞核内表达,见图2C。在正常条件下,3组均无肌管形成。在缺血缺氧条件下,3组细胞均有肌管形成,肌管数目随时间增加而增加,但各组细胞分化程度不一致:与对照组相比,原花青素组多核肌管数量明显增加,生成肌管数目更多(P < 0.05);2.5 mg/L原花青素组较5 mg/L原花青素组肌管数目略多,但差异无显著性意义(P > 0.05)。 综上,说明在缺血缺氧条件下,原花青素可促进骨骼肌卫星细胞肌原性标志基因的表达,从而促进更多肌管形成。"
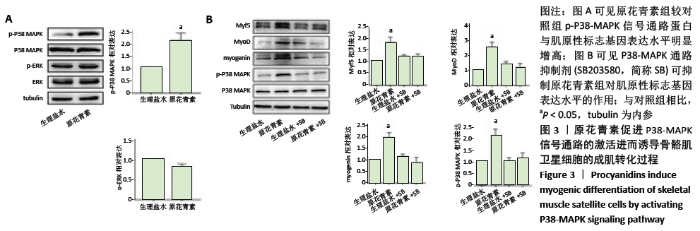
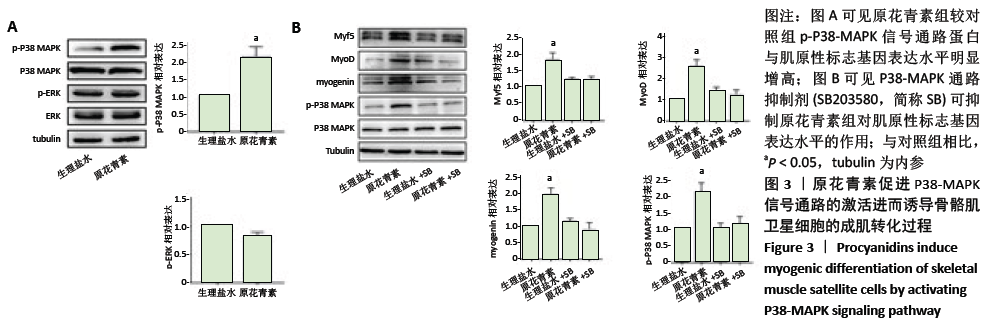
2.1.4 缺血缺氧条件下参与人骨骼肌卫星细胞分化的信号通路 结果显示,原花青素组p-P38-MAPK信号通路蛋白与肌原性标志基因表达水平较对照组明显增高,见图3A,说明p-P38-MAPK信号通路可能参与了原花青素促进人骨骼肌卫星细胞成肌分化过程。为进一步明确P38-MAPK信号通路在其间的作用,分别设置4组,分别为生理盐水组、原花青素组、生理盐水+P38-MAPK通路抑制剂组、原花青素+P38-MAPK通路抑制剂组,结果显示,原花青素组p-P38-MAPK信号通路蛋白与肌原性标志基因表达水平较生理盐水组明显增高 (P < 0.05),而后两组无明显差异(P > 0.05),见图3B,提示38-MAPK通路抑制剂不仅能抑制原花青素对P38-MAPK 信号通路的激活,还能抑制肌原性标志基因表达,进一步说明原花青素可能通过促进P38-MAPK信号通路的激活,进而诱导骨骼肌卫星细胞的成肌转化过程。"
| [1] WEITZ JI, BYRNE J, CLAGETT GP, et al. Diagnosis and treatment of chronic arterial insufficiency of the lower extremities: a critical review. Circulation. 1996;94(11):3026-3049. [2] FOWKES FG, RUDAN D, RUDAN I, et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: a systematic review and analysis. Lancet (London, England). 2013;382(9901):1329-1340. [3] GRAY BH, CONTE MS, DAKE MD, et al. Atherosclerotic Peripheral Vascular Disease Symposium II: lower-extremity revascularization: state of the art. Circulation. 2008;118(25):2864-2872. [4] LEJAY A, MEYER A, SCHLAGOWSKI AI, et al. Mitochondria: mitochondrial participation in ischemia-reperfusion injury in skeletal muscle. Int J Biochem Cell Biol. 2014;50:101-105. [5] BISCHOFF R, HEINTZ C. Enhancement of skeletal muscle regeneration. Dev Dyn. 1994;201(1):41-54. [6] CHANG NC, RUDNICKI MA. Satellite cells: the architects of skeletal muscle. Curr Top Dev Biol. 2014;107:161-181. [7] WANG YX, RUDNICKI MA. Satellite cells, the engines of muscle repair. Nat Rev Mol Cell Biol. 2011;13(2):127-133. [8] KUANG S, GILLESPIE MA, RUDNICKI MA. Niche regulation of muscle satellite cell self-renewal and differentiation. Cell Stem Cell. 2008; 2(1):22-31. [9] KADI F, CHARIFI N, DENIS C, et al. The behaviour of satellite cells in response to exercise: what have we learned from human studies? Pflugers Arch. 2005;451(2):319-327. [10] KIM AR, KIM KM, BYUN MR, et al. Catechins activate muscle stem cells by Myf5 induction and stimulate muscle regeneration. Biochem Biophys Res Commun. 2017;489(2):142-148. [11] RUDNICKI MA, SVHNEGELSBERG PN, STEAD RH, et al. MyoD or Myf-5 is required for the formation of skeletal muscle. Cell. 1993;75(7):1351-1359. [12] CORNELISON DD, WOLD BJ. Single-cell analysis of regulatory gene expression in quiescent and activated mouse skeletal muscle satellite cells. Dev Biol. 1997;191(2):270-283. [13] KUMAR R, DEEP G, WEMPE MF, et al. Procyanidin B2 3,3″-di-O-gallate inhibits endothelial cells growth and motility by targeting VEGFR2 and integrin signaling pathways. Curr Cancer Drug Targets. 2015;15(1):14-26. [14] LEE TM, CHARNG MJ, TSENG CD, et al. A Double-Blind, Randomized, Placebo-Controlled Study to Evaluate the Efficacy and Safety of STA-2 (Green Tea Polyphenols) in Patients with Chronic Stable Angina. Acta Cardiologica Sinica. 2016;32(4):439-449. [15] DAGLIA M. Polyphenols as antimicrobial agents. Curr Opin Biotechnol. 2012;23(2):174-181. [16] NAKAYAMA T. Suppression of hydroperoxide-induced cytotoxicity by polyphenols. Cancer Res. 1994;54(7 Suppl):1991s-1993s. [17] CHANG WT, SHAO ZH, YIN JJ, et al. Comparative effects of flavonoids on oxidant scavenging and ischemia-reperfusion injury in cardiomyocytes. Eur J Pharmacol. 2007;566(1-3):58-66. [18] QUINTIERI AM, BALDINO N, FILICE E, et al. Malvidin, a red wine polyphenol, modulates mammalian myocardial and coronary performance and protects the heart against ischemia/reperfusion injury. J Nutr Biochem. 2013;24(7):1221-1231. [19] MONTESANO A, LUZI L, SENESI P, et al. Resveratrol promotes myogenesis and hypertrophy in murine myoblasts. J Transl Med. 2013;11:310. [20] DING Y, DAI X, JIANG Y, et al. Grape seed proanthocyanidin extracts alleviate oxidative stress and ER stress in skeletal muscle of low-dose streptozotocin- and high-carbohydrate/high-fat diet-induced diabetic rats. Mol Nutr Food Res. 2013;57(2):365-369. [21] 杜超, 何雪梅, 施森,等.原花青素对小鼠后肢缺血肌肉的作用及miR-133b的表达变化[J]. 中国生物化学与分子生物学报,2018, 34(5):108-116. [22] TOGLIATTO G, TROMBETTA A, DENTELLI P, et al. Unacylated ghrelin promotes skeletal muscle regeneration following hindlimb ischemia via SOD-2-mediated miR-221/222 expression. J Am Heart Assoc. 2013; 2(6):e000376. [23] HELLINGMAN AA, BASTIAANSEN AJ, DE VRIES MR, et al. Variations in surgical procedures for hind limb ischaemia mouse models result in differences in collateral formation. Eur JVasc Endovasc Surg. 2010; 40(6):796-803. [24] LIMBOURG A, KORFF T, NAPP LC, et al. Evaluation of postnatal arteriogenesis and angiogenesis in a mouse model of hind-limb ischemia. Nat Protoc. 2009;4(12):1737-1746. [25] ZHAI L, WU R, HAN W, et al. miR-127 enhances myogenic cell differentiation by targeting S1PR3. Cell Death Dis. 2017;8(3):e2707. [26] CRIQUI MH, ABOYANS V. Epidemiology of peripheral artery disease. Circ Res. 2015;116(9):1509-1526. [27] TEDESCO FS, DELLAVALLE A, DIAZ-MANERA J, et al. Repairing skeletal muscle: regenerative potential of skeletal muscle stem cells. J Clin Invest. 2010;120(1):11-19. [28] TROY A, CADWALLADER AB, FEDOROV Y, et al. Coordination of satellite cell activation and self-renewal by Par-complex-dependent asymmetric activation of p38α/β MAPK. Cell Stem Cell. 2012;11(4):541-553. [29] ZAMMIT PS, GOLDING JP, NAGATA Y, et al. Muscle satellite cells adopt divergent fates: a mechanism for self-renewal? J Cell Biol. 2004;166(3): 347-357. [30] SABOURIN LA, GIRGIS-GABARDO A, SEALE P, et al. Reduced differentiation potential of primary MyoD-/- myogenic cells derived from adult skeletal muscle. J Cell Biol. 1999;144(4):631-643. [31] CHARGE SB, RUDNICKI MA. Cellular and molecular regulation of muscle regeneration. Physiol Rev. 2004;84(1):209-238. [32] POUSSARD S, PIRES-ALVES A, DIALLO R, et al. A natural antioxidant pine bark extract, Oligopin®, regulates the stress chaperone HSPB1 in human skeletal muscle cells: a proteomics approach. Phytother Res. 2013;27(10):1529-1535. [33] ZHANG YP, LIU SY, SUN QY, et al. Proanthocyanidin B2 attenuates high-glucose-induced neurotoxicity of dorsal root ganglion neurons through the PI3K/Akt signaling pathway. Neural Regen Res. 2018;13(9): 1628-1636. [34] GAO M, ZHAO Z, LV P, et al. Quantitative combination of natural anti-oxidants prevents metabolic syndrome by reducing oxidative stress. Redox Biol. 2015;6:206-217. [35] LI P, WANG J, LU S, et al. Protective effect of hawthorn leaf procyanidins on cardiomyocytes of neonatal rats subjected to simulated ischemia-reperfusion injury. Zhongguo Zhong yao za zhi. 2009;34(1):96-99. [36] MORENO-LUNA R, MUNOZ-HERNANDEZ R, MIRANDA ML, et al. Olive oil polyphenols decrease blood pressure and improve endothelial function in young women with mild hypertension. Am JHypertens. 2012;25(12):1299-1304. [37] KONG X, GUAN J, GONG S, et al. Neuroprotective Effects of Grape Seed Procyanidin Extract on Ischemia-Reperfusion Brain Injury. Chin Med Sci J. 2017;32(2):92-99. [38] KRUGER MJ, SMITH C. Postcontusion polyphenol treatment alters inflammation and muscle regeneration. Med Sci Sports Exerc. 2012; 44(5):872-880. [39] ZACCAGNINIG, MARTELLI F, MAGENTA A, et al. p66(ShcA) and oxidative stress modulate myogenic differentiation and skeletal muscle regeneration after hind limb ischemia. J Biol Chem. 2007;282(43): 31453-31459. [40] COLETTI D, TEODORI L, LIN Z, et al. Restoration versus reconstruction: cellular mechanisms of skin, nerve and muscle regeneration compared. Regen Med Res. 2013;1(1):4. [41] YIN H, PRICE F, RUDNICKI MA. Satellite cells and the muscle stem cell niche. Physiol Rev. 2013;93(1):23-67. [42] COUFFINHAL T, SILVER M, ZHENG LP, et al. Mouse model of angiogenesis. Am JPathol. 1998;152(6):1667-1679. [43] PERDIGUERO E, SOUSA-VICTOR P, RUIZ-BONILLA V, et al. p38/MKP-1-regulated AKT coordinates macrophage transitions and resolution of inflammation during tissue repair. J Cell Biol. 2011;195(2):307-322. [44] DUMONT NA, BENTZINGER CF, SINCENNES MC, et al. Satellite Cells and Skeletal Muscle Regeneration. Compr Physiol. 2015;5(3):1027-1059. [45] RIUZZI F, SORCI G, SAHGEDDU R, et al. HMGB1-RAGE regulates muscle satellite cell homeostasis through p38-MAPK- and myogenin-dependent repression of Pax7 transcription. J Cell Sci. 2012;125(Pt 6): 1440-1454. |
| [1] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [2] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [3] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [4] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [5] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [6] | Liu Bo, Chen Xianghe, Yang Kang, Yu Huilin, Lu Pengcheng. Mechanism of DNA methylation in exercise intervention for osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 791-797. |
| [7] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [8] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [9] | Zhou Jihui, Yao Meng, Wang Yansong, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Influence of novel nanoscaffolds on biological behaviors of neural stem cells and the related gene expression [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 532-536. |
| [10] | Ye Haimin, Ding Linghua, Kong Weihao, Huang Zutai, Xiong Long. Role and mechanism of hierarchical microchanneled bone scaffolds in promoting osteogenesis and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 621-625. |
| [11] | Li Jun, Zuo Xinhui, Liu Xiaoyuan, Zhang Kai, Han Xiangzhen, He Huiyu, . Effect of over expression of miR-378a on osteogenic and vascular differentiation of bone marrow mesenchymal stem cell sheet [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4939-4944. |
| [12] | Li Junqing, He Wenxi, Guo Qian, Wu Jiayuan. Effect of hydrostatic pressure on odontogenic/osteogenic differentiation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4976-4980. |
| [13] | Liu Lei, Di Haiping, Guo Haina, Cao Dayong, Niu Xihua, Xia Chengde. Changes in biological characteristics of platelet-rich fibrin by freeze-drying technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(31): 4995-4999. |
| [14] | Hou Jingzhao, Zhang Zhen. Extracellular matrix promotes fibroblast proliferation and post-operation knee arthrofibrosis through ERK signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4168-4174. |
| [15] | Zheng Feng, Zhang Fucai, Xu Zhe. MicroRNA-98-5p promotes osteoblast proliferation and differentiation: possibilities and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(26): 4112-4117. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

