中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (24): 6289-6296.doi: 10.12307/2026.200
• 组织构建综述 tissue construction review • 上一篇 下一篇
miRNA参与软骨发育的机制:新策略及新靶点
王正业,刘万林,赵振群
- 内蒙古医科大学第二附属医院儿童骨科医学中心,内蒙古自治区呼和浩特市 010090
-
收稿日期:2025-06-27修回日期:2025-09-26出版日期:2026-08-28发布日期:2026-02-03 -
通讯作者:刘万林,硕士,教授,内蒙古医科大学第二附属医院儿童骨科医学中心,内蒙古自治区呼和浩特市 010090 共同通讯作者:赵振群,博士,教授,内蒙古医科大学第二附属医院儿童骨科医学中心,内蒙古自治区呼和浩特市 010090 -
作者简介:王正业,男,1997年生,内蒙古自治区鄂尔多斯市准格尔旗人,汉族,在读硕士,主要从事髋关节的相关研究。 -
基金资助:国家自然科学基金项目(81960397,82260424),项目负责人:刘万林;国家自然科学基金项目(82160414,81760391),项目负责人:赵振群;内蒙古自治区成果转化项目(CGZH2018146),项目负责人:刘万林;内蒙古自治区自然科学基金杰出青年基金项目(2023SHZR1613),项目负责人:赵振群
Mechanisms of miRNAs involved in cartilage development: new strategies and targets
Wang Zhengye, Liu Wanlin, Zhao Zhenqun
- Center for Pediatric Orthopedics, Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010090, Inner Mongolia Autonomous Region, China
-
Received:2025-06-27Revised:2025-09-26Online:2026-08-28Published:2026-02-03 -
Contact:Liu Wanlin, MS, Professor, Center for Pediatric Orthopedics, Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010090, Inner Mongolia Autonomous Region, China Co-corresponding author: Zhao Zhenqun, PhD, Professor, Center for Pediatric Orthopedics, Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010090, Inner Mongolia Autonomous Region, China -
About author:Wang Zhengye, MS candidate, Center for Pediatric Orthopedics, Second Affiliated Hospital of Inner Mongolia Medical University, Hohhot 010090, Inner Mongolia Autonomous Region, China -
Supported by:National Natural Science Foundation of China, Nos. 81960397 and 82260424 (both to LWL); National Natural Science Foundation of China, Nos. 82160414 and 81760391 (both to ZZQ); Inner Mongolia Autonomous Region Achievement Transformation Project, No. CGZH2018146 (to LWL); Outstanding Young Scientist Project of Inner Mongolia Autonomous Region Natural Science Foundation, No. 2023SHZR1613 (to ZZQ)
摘要:
文题释义:
miRNA:是一类由17-25个核苷酸组成的单链非编码RNA分子,广泛存在于真核生物中。在软骨发育过程中,miRNA通过与靶mRNA的3’非翻译区特异性结合,调控基因的转录后沉默或翻译抑制。研究表明,miRNA在软骨细胞分化、增殖和凋亡中发挥关键作用。此外,miRNA的异常表达与多种软骨疾病密切相关,如骨关节炎和软骨发育不良。因此,miRNA不仅是软骨发育的重要调控因子,也是相关疾病潜在的治疗靶点。
软骨细胞分化:是指间充质干细胞或软骨祖细胞逐渐发育为成熟软骨细胞的过程,是软骨发育的核心环节。该过程涉及多种信号通路和转录因子的协同调控。在生长板区域,软骨细胞分化经历静止期、增殖期和肥大期,最终形成成熟的软骨细胞。miRNA通过精准调控关键转录因子和信号通路来影响软骨细胞分化的时序和方向。软骨细胞分化的调控机制不仅决定了软骨的正常发育,还与软骨相关疾病的发生发展密切相关。
背景:软骨发育的分子调控机制是骨科领域的关键科学问题之一。miRNA作为基因表达的重要调节因子,通过与靶mRNA的3’非翻译区结合,调控转录后沉默或翻译抑制,参与细胞分化、增殖及代谢稳态的调控。近年来,研究发现miRNA在软骨发育及多种软骨相关疾病中发挥着重要作用,其异常表达与骨骼发育不良、骨关节炎等疾病密切相关。深入研究miRNA在软骨发育中的作用机制,对于理解软骨细胞命运决定以及相关疾病的发病机制具有重要意义。
目的:旨在全面总结miRNA在软骨发育中的作用机制,探讨miRNA在软骨细胞分化、增殖、凋亡等过程中的调控作用,以及在骨关节炎、软骨发育不良等疾病状态下的功能,为相关疾病的防治提供理论依据。
方法:通过检索PubMed、中国知网、万方数据库及维普数据库(各数据库建库至2025年6月),结合手工查阅相关书籍,筛选与miRNA调控软骨发育相关的高质量文献。优先选择近10年内发表的研究,最终纳入99篇文献(英文95篇,中文4篇)进行系统分析与总结。
结果与结论:研究表明,miRNA通过精准调控关键转录因子、信号通路及表观遗传修饰,影响软骨细胞的分化、增殖和凋亡。在疾病状态下,miRNA的异常表达与多种软骨疾病密切相关。这些发现不仅揭示了miRNA在软骨发育中的直接调控作用,还为相关疾病的诊断、预后评估及治疗提供了新的策略和靶点。随着基因编辑、单细胞测序、生物信息学等前沿技术的不断发展,miRNA研究在软骨发育及疾病中的应用取得了显著进展,有望为骨骼疾病的早期诊断、精准治疗及预后评估提供新的手段。
https://orcid.org/0009-0006-6873-7422 (王正业)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
王正业, 刘万林, 赵振群. miRNA参与软骨发育的机制:新策略及新靶点[J]. 中国组织工程研究, 2026, 30(24): 6289-6296.
Wang Zhengye, Liu Wanlin, Zhao Zhenqun. Mechanisms of miRNAs involved in cartilage development: new strategies and targets[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(24): 6289-6296.
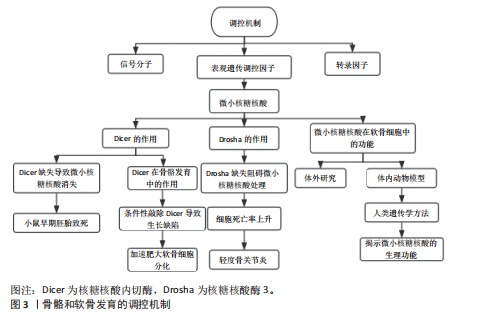
样,核糖核酸酶3(Drosha ribonuclease III,Drosha)缺失也会阻碍miRNA处理的第一步,致使大多数miRNA含量锐减,而在出生后小鼠关节软骨细胞中条件性敲除核糖核酸酶3会导致细胞死亡率上升以及轻度骨关节炎等症状[12]。这些研究发现凸显了miRNA在软骨细胞增殖、分化以及存活过程中发挥重要作用。目前,学界已在多种实验系统中广泛研究了个体miRNA在骨骼谱系细胞中的功能,相关研究成果在大量文献中有所报道。然而,多数研究集中于体外环境,采用间充质基质细胞和细胞系等不同类型的细胞进行实验。尽管这些研究在细胞与分子层面深化了对miRNA作用机制的理解,但在体内生理环境中的确切意义仍有待进一步明确。基于此,该研究聚焦于利用体内动物模型或人类遗传学方法,揭示个体miRNA在骨骼发育中的生理功能,力求为该领域研究提供更精准的视角与依据。
2.1 miRNA在软骨发育中的作用机制
2.1.1 软骨细胞分化 软骨细胞分化作为软骨发育的核心环节,其分子调控网络涉及miRNA对关键转录因子、信号通路及表观遗传修饰的精准调节。近年来研究表明,miRNA通过靶向性别决定区Y框蛋白9、Runt相关转录因子2等核心转录因子调控软骨细胞命运。研究发现,miRNA通过复杂的反馈环路来调控软骨细胞分化的关键阶段。在生长板区域,miR-203a
通过直接靶向Runt相关转录因子2,能够抑制软骨细胞的肥大化进程,这个机制是通过双荧光素酶报告基因实验得到验证的[13]。而且,不同区域的miRNA表达存在明显的差异:静息区、增殖区和肥大区分别富集着不同的miRNA谱系,这种空间特异性的分布与甲状旁腺激素相关蛋白/印度刺猬因子/骨形态发生蛋白(Parathyroid Hormone-related protein/indian hedgehog/Bone morphogenetic protein,PTHrP/IHH/BMP)信号轴的梯度激活关系密切[14]。例如,骨关节炎患者的受损软骨组织中miR-199a-5p表达明显下调,它通过负调控印度刺猬因子信号来影响软骨细胞分化的时序[15]。在细胞周期调控方面,小鼠piRNA hsa_piR_019949的过表达可以显著促进软骨细胞增殖,并且抑制细胞凋亡,这种效应和NOD样受体信号通路的激活有关[16]。相反,miR-451在生长板软骨中的异常表达会促进基质降解酶的产生,从而加剧白细胞介素1β对关节软骨的破坏作用[17]。这些发现揭示了miRNA在维持软骨细胞稳态中具有双向调节作用。在细胞外基质代谢方面,miRNA也有着重要的调控作用。Toll样受体4/核因子κB通路是miRNA调控细胞外基质代谢的核心枢纽。白花蛇舌草提取物通过上调miR-145-5p来抑制Toll样受体4的表达,进而阻断核因子κB信号转导,最终减轻白细胞介素1β诱导的基质降解[18]。同时,丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)信号通路也参与了这一过程:软骨源性细胞外囊泡携带的miRNA通过调控黏着斑和叉头转录因子O信号通路来影响基质的完整性[19]。在机械信号转导方面,初级纤毛作为力学感受器,其功能状态受到miR-140等的调控。当基质黏弹性发生改变时,纤毛依赖的机械信号会通过Wnt/β-连环蛋白通路影响聚集蛋白多糖和Ⅱ型胶原的表达[20]。特别值得注意的是,组蛋白去甲基化酶6a缺失导致的组蛋白修饰变化会同时影响烟酰胺腺嘌呤二核苷酸磷酸氧化酶的活性和Wnt信号的强度,从而形成了一个表观遗传-代谢-机械信号的三维调控网络[21]。在微环境重编程方面,炎症微环境和miRNA之间形成了一个正反馈调节环。白细胞介素6/Janus激酶/信号转导与转录激活因子3(Janus kinase/Signal transducer and activator of transcription 3,JAK/STAT3)通路被证实是miR-653-5p的关键靶点,这个miRNA在骨关节炎软骨中异常表达会导致衰老相关分泌表型增强[22];同时,M1/M2型巨噬细胞的极化也受到miRNA的调控:白藜芦醇处理通过改变miRNA谱系来促进M2型极化,从而改善滑膜微环境[23]。在组织工程应用中,富含抗坏血酸的细胞外基质(Ascorbate-enriched extracellular matrix,ACECM)支架联合miR-148a-3p转染可以显著提高Ⅱ型胶原的分泌,这种效应与基质金属蛋白酶13/Runt相关转录因子2的平衡调节有关[24]。而Ras同源基因家族成员A(Ras homolog gene family,member A,RhoA)过表达的软骨细胞和脂肪干细胞共培养时,特定miRNA谱变化会使工程化软骨的生物力学性能接近天然组织[25]。在机制差异和转化医学思考方面,不同研究之间存在一些值得关注的矛盾。例如,miR-138在软骨形成和细胞分解代谢中的作用存在明显分歧,这可能与实验模型(原代细胞系和组织切片)以及微环境刚度的差异有关[26-27]。在治疗策略上,基于CRISPR-Cas9的基质金属蛋白酶13靶向沉默和miRNA联合干预显示出协同效应[28],但需要注意的是,载体递送效率会受到软骨基质屏障的限制。
2.1.2 软骨细胞增殖 软骨细胞增殖是软骨发育的关键环节,其分子调控网络涉及miRNA对关键转录因子、信号通路及表观遗传修饰的精准调节(表1)。在软骨细胞增殖过程中,miRNA通过调控关键细胞周期蛋白和信号通路,影响软骨细胞的增殖和代谢稳态。磷酸酶和张力蛋白同源物(phosphatase and tensin homolog,PTEN)是磷脂酰肌醇3激酶/蛋白激酶B(phosphoinositide 3-kinase/protein kinase B,PI3K/AKT)信号通路的关键负调控因子[29],miR-21通过抑制磷酸酶和张力蛋白同源物的表达,激活磷脂酰肌醇3激酶/蛋白激酶B信号通路,促进软骨细胞增殖和存活。相反,miR-27b通过靶向细胞周期蛋白依赖激酶2(cyclin-dependent kinase 2,CDK2),抑制软骨细胞增殖。细胞周期蛋白依赖激酶2是丝裂原活化蛋白激酶信号通路的关键分子,miR-27b通过抑制细胞周期蛋白依赖激酶2的表达,抑制丝裂原活化蛋白激酶信号通路的激活,从而抑制软骨细胞增殖。此外,miR-374-5p、miR-379-5p和miR-503-5p在生长板的增殖区中高表达,通过调控甲状旁腺激素相关蛋白信号通路,促进软骨细胞增殖。这些miRNA通过与甲状旁腺激素相关蛋白信号通路的关键分子结合,抑制软骨细胞的肥大分

化,从而维持软骨细胞的增殖能力[30]。miRNA在软骨细胞增殖中的作用在动物实验中得到了广泛验证。例如,敲除小鼠体内由Ⅱ型胶原介导的核糖核酸内切酶会导致严重的骨生成障碍和小鼠死亡,表明miRNA在软骨细胞增殖中起着重要的作用。此外,骨质疏松症患者骨组织中miR-16-2-3p表达与骨形成相关基因(如Runt相关转录因子2、Osterix)呈负相关,表明miR-16-2-3p可能通过抑制Wnt信号通路,阻碍软骨细胞增殖[29]。通过体外实验和体内实验研究了人脐带间充质干细胞衍生小细胞外囊泡在软骨再生中的作用和机制,发现其通过传递miR-23a-3p抑制磷酸酶和张力蛋白同源物水平和提高蛋白激酶B表达来促进软骨再生。在体内实验中,含有甲基丙烯酰化明胶/纳米黏土水凝胶的人脐带间充质干细胞衍生小细胞外囊泡可有效促进软骨再生[31]。此外,研究还发现miR-146b-5p表达驱动间充质干细胞向脂肪分化,并在斑马鱼成熟和衰老过程中显著增加,且在女性中的表达高于男性并与人类的衰老有关。行走身体活动方式可下调人类女性循环miR-146b-5p水平并增加软骨祖细胞的数量[32]。
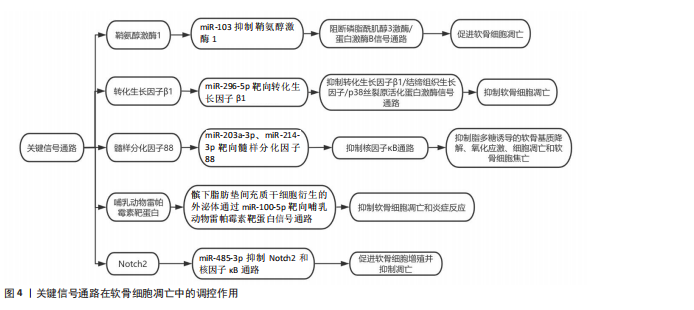
2.1.3 软骨细胞凋亡 miRNA通过与靶mRNA的3’非翻译区结合介导转录后沉默或翻译抑制,从而调控细胞凋亡、代谢和稳态。在分子机制层面,鞘氨醇激酶1(sphingosine kinase 1,SPHK1)是磷脂酰肌醇3激酶/蛋白激酶B信号通路的关键分子,miR-103通过抑制鞘氨醇激酶1的表达,阻断该信号通路的激活,进而促进软骨细胞凋亡[33]。此外,miR-296-5p通过靶向转化生长因子β1,抑制转化生长因子β1/结缔组织生长因子(connective tissue growth factor,CTGF)/p38丝裂原活化蛋白激酶(mitogen- activated protein kinase,p38MAPK)信号通路,从而抑制软骨细胞凋亡。转化生长因子β1是丝裂原活化蛋白激酶信号通路的关键分子,miR-296-5p通过抑制转化生长因子β1的表达,阻断丝裂原活化蛋白激酶信号通路的激活,进而抑制软骨细胞凋亡[34]。另有研究发现,骨关节炎患者滑液中外泌体的miR-214-3p表达显著下调,而软骨细胞和滑膜成纤维细胞来源的外泌体miR-214-3p可促进软骨细胞增殖、抑制细胞凋亡和炎症反应,且在大鼠模型中抑制骨赘形成和软骨退变[35]。miRNA通过精细调控凋亡相关信号通路(如线粒体途径、死亡受体途径及内质网应激途径),在软骨细胞凋亡中发挥关键作用,见图4。近期研究发现,环状RNA HECW2通过甲基化抑制miR-93的表达,增加脂多糖诱导的软骨细胞凋亡[36]。而miR-203a-3p、miR-214-3p在脂多糖处理的大鼠软骨细胞和骨关节炎软骨中表达下调,它们直接靶向髓样分化因子88(myeloid differentiation primary response 88,MYD88),通过抑制髓性分化因子88/核因子κB通路显著抑制脂多糖诱导的软骨基质降解、氧化应激、细胞凋亡和软骨细胞焦亡[37-38]。最新研究揭示,髌下脂肪垫间充质干细胞衍生的外泌体通过miR-100-5p靶向哺乳动物雷帕霉素靶蛋白信号通路,抑制软骨细胞凋亡和炎症反应。在体外实验中,髌下脂肪垫间充质干细胞衍生的外泌体显著抑制了软骨细胞凋亡,增强了基质合成,并降低了分解代谢因子的表达[39]。还有研究发现,miR-485-3p在骨关节炎患者和脂多糖处理的软骨细胞中表达降低,其过表达可抑制Notch同源蛋白2(neurogenic locus notch homolog protein 2,Notch2)和核因子κB通路,促进软骨细胞增殖并抑制凋亡,减轻软骨基质降解、炎症和氧化应激[40]。
2.2 miRNA在疾病状态下的作用
2.2.1 骨关节炎 骨关节炎是一种常见的退行性关节疾病,其特征是软骨细胞凋亡、细胞外基质降解及炎症反应加剧。近年来,miRNA在骨关节炎中的作用机制受到广泛关注。在骨关节炎中,miRNA主要通过调控炎症因子、基质金属蛋白酶、凋亡相关蛋白以及细胞外基质成分的表达,影响软骨细胞的存活、增殖和代谢[41]。在炎症信号通路方面的研究发现,miR-149可通过调控肿瘤坏死因子α、白细胞介素1β和白细胞介素6水平发挥抗炎作用。此外,miR-146a通过靶向NUMB蛋白,调节炎症、自噬和软骨细胞凋亡[42]。在凋亡调控方面,miRNA通过调控凋亡相关基因影响软骨细胞存活。近期研究表明,miR-34a通过靶向Ⅱ型胶原α1促进软骨细胞凋亡[43]。在软骨细胞外基质调控方面,miRNA通过调控外基质成分的表达影响软骨稳态[44]。在体内实验中,miR-26b-5p agomir改善了骨关节炎小鼠的步态异常和机械异常性痛,减轻滑膜炎和软骨退变,延缓骨关节炎进展[45]。一项研究显示,使用5 μg(每2周注射1次,共3次)的miR-34a-5p锁定核酸反义寡核苷酸(locked nucleic acid antisense oligonucleotide,LNA-ASO)注射到小鼠膝关节,能显著减少软骨细胞凋亡和基质降解,此外,miR-21-5p通过靶向成纤维细胞生长因子18促进软骨基质降解,其抑制剂也显示出治疗潜力[46]。此外,系统性给药一种新的针对骨关节炎的外泌体抑制剂(Osteoarthritis Exosome Inhibitor,OCExoInhib)可以减轻手术诱导的小鼠骨关节炎进展[47]。研究显示,miR-34a-5p锁定核酸反义寡核苷酸能显著减少软骨细胞凋亡和基质降解,改善软骨结构和功能[48]。miR-21-5p抑制剂的剂量和效果也在研究中得到验证,显示出减少软骨基质降解和软骨细胞凋亡的潜力[49]。miRNA治疗适用于早期和中期骨关节炎患者,尤其是对传统治疗方法反应不佳的患者,这些患者通常表现为软骨损伤较轻、炎症反应较弱,适合通过miRNA调节来改善软骨细胞的存活和功能。miRNA治疗在动物模型和临床试验中均显示出良好效果。例如,miR-34a-5p锁定核酸反义寡核苷酸治疗的小鼠模型中,软骨细胞凋亡减少了30%,基质降解减少了40%[46]。临床试验中,接受miR-34a-5p锁定核酸反义寡核苷酸治疗的患者软骨损伤评分显著下降,关节疼痛减轻,功能改善[50]。最新发现miR-214-3p在炎症软骨细胞和骨关节炎软骨中下调,可调节软骨中的细胞外基质代谢和细胞凋亡。在机制上,miR-214-3p的保护作用通过下调IκB激酶β(IκB kinase β,IKK-β)表达使核因子κB信号通路出现功能障碍。最新研究揭示了miRNA在骨关节炎治疗中的精准调控机制及临床转化潜力。研究表明,miR-181a-5p在腰椎小关节和膝骨关节炎软骨中表达升高,与分解代谢标志物相关,而LNA-miR-181a-5p反义寡核苷酸在体外和体内均显示出减轻软骨破坏和保护软骨的作用,表明其可能成为治疗骨关节炎的新疗法[51]。在动物实验中,以25 nmol的剂量关节内注射miR-140 agomir,每周2次,持续8周,显著减轻了大鼠膝关节软骨损伤,减少了关节炎症和软骨细胞凋亡,显示出更好的关节组织形态和更低的国际骨关节炎研究协会(Osteoarthritis Research Society International,OARSI)评分[52];miR-1 agomir单剂量注射到大鼠膝关节后,通过抑制印度刺猬因子信号通路,显著减少基质金属蛋白酶水平,减轻骨关节炎进展;miR-17 agomir单剂量注射到大鼠膝关节后,通过抑制基质金属蛋白酶3、基质金属蛋白酶13、解整合素金属蛋白酶样蛋白酶5和诱导型一氧化氮合酶的表达,显著减轻骨关节炎严重程度 [53];miR-210 agomir单剂量注射到大鼠膝关节后,显著促进部分撕裂前交叉韧带和受损半月板的愈合[54]。这些研究表明,miRNA在骨关节炎治疗中具有显著潜力。通过靶向特定miRNA,可有效减轻关节炎症、减少软骨损伤并改善关节功能。未来,随着技术不断发展,miRNA治疗有望成为骨关节炎治疗的新选择。
2.2.2 软骨发育不良 软骨发育不良是一种常见的遗传性骨骼发育障碍,主要特征为四肢短小、大头畸形及特殊面容。该病由成纤维细胞生长因子受体3基因突变引起,导致成纤维细胞生长因子受体3信号通路异常激活,抑制软骨细胞增殖和分化,进而影响骨骼正常生长。在软骨发育不良小鼠模型中,miR-365-5p通过负调控丝裂原活化蛋白激酶/细胞外调节蛋白激酶通路,可逆转成纤维细胞生长因子受体3突变导致的增殖抑制,过表达miR-365-5p的小鼠模型显示胫骨长度增加28.7%,且生长板组织结构评分改善3.2倍[55]。近年来,miRNA在软骨发育不良中的调控机制及治疗潜力受到越来越多的关注。在分子机制层面,miRNA主要通过调控成纤维细胞生长因子受体3信号通路及其下游的丝裂原活化蛋白激酶、信号转导与转录激活因子1等信号通路,影响软骨细胞的生长和发育[56-58]。在治疗药物方面,Vosoritide是一种重组C型利钠肽(C-type natriuretic peptide,CNP)类似物,通过抑制成纤维细胞生长因子受体3信号通路,促进软骨细胞的正常分化和增殖。Ⅱ期临床试验显示,每日1次剂量为15 μg/kg的Vosoritide可显著提高患者年生长速度,该药物已在多个地区获批用于改善软骨发育不良[59-61]。Infigratinib作为一种口服小分子成纤维细胞生长因子受体3抑制剂,通过直接靶向成纤维细胞生长因子受体3信号通路促进骨骼生长,Ⅱ期临床试验表明以每日1次0.016-0.25 mg/kg剂量给药可显著提高患者年生长速度[62]。持续释放型C型利钠肽是一种C型利钠肽前体药物,每周1次给药可有效避免高剂量C型利钠肽的心血管不良反应,Ⅱ期临床试验显示治疗效果良好,患者年生长速度显著提高[63]。miRNA在软骨发育不良治疗中具有显著潜力,能有效改善软骨结构和功能。未来,随着技术不断进步,miRNA在软骨发育不良中的研究将更深入,为疾病治疗带来新希望。
2.2.3 椎间盘退变 miRNA在椎间盘退变中发挥着复杂而关键的作用,通过调控细胞增殖、凋亡、炎症反应及细胞外基质代谢等多种机制影响椎间盘退变的进程。在细胞增殖方面,miR-10b在椎间盘退变组织中显著上调,通过靶向同源框D10,刺激Rho C小GTP酶(Rho C GTPase,RhoC)表达和磷酸化,进而促进髓核细胞的异常增殖,而敲除Rho C小GTP酶或抑制蛋白激酶B可消除miR-10b对髓核细胞增殖的影响;miR-96在人类退变髓核组织中上调,通过靶向富含AT的相互作用域2激活蛋白激酶B信号通路,促进髓核细胞增殖[64]。在细胞凋亡方面,miR-21在椎间盘退变组织中上调,通过靶向磷酸酶和张力蛋白同源物抑制髓核细胞凋亡;miR-222模拟物促进髓核细胞凋亡,其通过影响肿瘤坏死因子α、白细胞介素1β和白细胞介素6等炎症因子以及Ⅱ型胶原蛋白和蛋白多糖的表达来发挥作用[65]。在炎症反应方面,miR-146a可抑制白细胞介素1介导的炎症基因的mRNA表达以及基质金属蛋白酶和蛋白多糖的蛋白表达,其在椎间盘退变患者的外周血单核细胞中显著下调,且过表达miR-146a可显著下调脂多糖刺激的髓核细胞中促炎细胞因子水平,这一过程依赖于肿瘤坏死因子受体相关因子6/核因子κB通路;miR-194-5p在椎间盘退变患者中显著下调,其通过调控Cullin 4A和Cullin 4B影响炎症反应,白细胞介素6和肿瘤坏死因子α抑制剂可减少髓核细胞和纤维环细胞中Cullin 4A和Cullin 4B的表达。在细胞外基质代谢方面,miR-25-3p可通过靶向金属反应转录因子1蛋白表达抑制白细胞介素1β诱导的效应,从而减少细胞外基质降解酶(基质金属蛋白酶3、基质金属蛋白酶13、血小板反应蛋白解整合素金属肽酶5等)的表达,并恢复细胞外基质蛋白(如Ⅱ型胶原蛋白、蛋白多糖)的表达。在动物实验中,热响应载体递送的miR-25-3p模拟物可延缓椎间盘退变的进展;miR-665在椎间盘退变组织中高表达,通过靶向生长分化因子5促进髓核细胞的异常增殖,同时降低蛋白多糖和Ⅱ型胶原蛋白的表达,增加基质金属蛋白酶3和基质金属蛋白酶13的表达。此外,miRNA还参与纤维环退变。例如,miR-221在退变纤维环细胞中的基础水平显著降低,其可通过激活骨形态发生蛋白-Smad信号通路抑制退变纤维环细胞的成骨分化;miR-194-5p低表达可导致纤维环细胞中Cullin 4A、Cullin 4B和促炎细胞因子增加,从而促进纤维环细胞退变;miR-106a-5p上调可抑制自噬相关蛋白7的表达,导致纤维环细胞增殖、自噬和凋亡异常。这些研究表明,miRNA通过多种机制和通路参与椎间盘退变的发生发展,有望成为椎间盘退变早期诊断和治疗的新靶点。在椎间盘退变中miR-140-3p低表达,其过表达可缓解椎间盘退变。研究发现miR-140-3p通过靶向Kruppel样因子5并调控Kruppel样因子5/N-钙黏蛋白/MDM2/Slug轴,增强退化椎间盘退变髓核来源间充质干细胞的增殖、迁移和分化,抑制其凋亡,从而发挥再生作用,为椎间盘退变治疗提供了潜在靶点[66]。有研究通过荧光素酶和RNA下拉实验验证了长链非编码RNA JPX与miR-18a-5p的直接结合,发现miR-18a-5p与缺氧诱导因子1α存在负相关关系。通过靶向缺氧诱导因子1α抑制Hippo-Yes相关蛋白1转录共激活因子通路,影响体外培养的人髓核细胞的增殖和凋亡。在体内实验中,长链非编码RNA JPX过表达通过抑制miR-18a-5p上调缺氧诱导因子1α,促进人髓核细胞增殖并减少凋亡,改善骨关节炎小鼠的病理表现,表明长链非编码RNA JPX是椎间盘退变治疗的潜在新靶点。外泌体疗法是治疗椎间盘退变有前景的方法,通过释放miRNA、蛋白质和转录因子调节代谢、微环境和细胞稳态,但面临外泌体快速清除和破坏的挑战。有研究制备了热敏脱细胞外基质水凝胶与脂肪间充质干细胞外泌体复合物,兼具髓核组织和脂肪间充质干细胞的优越特性。该水凝胶体系能原位凝胶补充髓核细胞外基质渗漏,为细胞生长提供环境,还可持续释放脂肪间充质干细胞衍生外泌体,调节基质金属蛋白酶以调控基质合成降解,并减轻体外炎症反应抑制焦亡。动物实验显示,该水凝胶体系可维持椎间盘退变早期微环境稳态、改善椎间盘退变,有望成为椎间盘退变药物传递和生物治疗的有力平台及椎间盘退变替代疗法[67-68]。
2.2.4 Feingold综合征与软骨-毛发发育不良 研究表明,miR-17-92簇基因(在人类中为Mir17HG,小鼠中为Mirc1)是首个被发现与人类骨骼发育缺陷相关的miRNA编码基因,该基因的缺失会导致短指综合征(Feingold综合征2型)。Feingold综合征是一种骨骼发育不良疾病,其特征包括身材矮小、短指畸形、小头畸形、面部畸形、胃肠道闭锁以及轻度至中度学习障碍。大多数Feingold综合征病例(1型)是由MYCN基因的失功能突变引起的。在小鼠模型中,针对miR-17-92簇基因的敲除实验成功复制了Feingold综合征患者的关键特征,证实了Feingold综合征2型是由于miR-17-92基因的单倍剂量不足所致[69-70]。尽管Feingold综合征1型和2型的临床表现相似,但它们的分子病理机制存在显著差异。
此外,人类软骨-毛发发育不良(Cartilage-hair hypoplasia,CHH)是一种罕见的常染色体隐性遗传综合征,该综合征的主要特征是由于干骺端发育不良导致的短肢侏儒症,以及多种系统性异常,包括毛发细小、稀疏、呈浅色等低色素性表现和免疫缺陷。软骨-毛发发育不良是由非编码RNA RMRP的点突变引起的。RMRP是一种含有268个核苷酸的非编码RNA,是线粒体RNA处理核糖核酸酶MRP (Ribonuclease MRP,RNase MRP)的组成成分,该酶主要负责处理线粒体核糖体RNA。在软骨-毛发发育不良患者中发现了多种突变,研究表明RNase MRP内切酶活性的丧失是该疾病发生的主要原因。近期研究发现,RMRP可产生2个较短的RNA分子(约20个核苷酸),即RMRP-S1和RMRP-S2,它们能够发挥miRNA的作用[71]。由于多个致病突变被定位到RMRP-S1和S2,这些miRNA的异常可能对软骨-毛发发育不良的表型多样性产生影响。通路分析进一步提示,许多受RMRP-S1/S2调控的基因与骨骼发育、毛囊发育和造血过程密切相关,从而揭示了RMRP-S1和S2的潜在生理功能[72]。
2.3 相关技术路径分析
2.3.1 基因编辑技术 基因编辑技术在miRNA相关软骨发育研究中主要分为规律成簇间隔短回文重复序列相关核酸酶系统、锌指核酸酶和转录激活因子样效应核酸酶三大类,其原理、操作及特点存在显著差异。规律成簇间隔短回文重复序列相关核酸酶9系统通过单导向RNA引导Cas9蛋白靶向切割miRNA前体或宿主基因,在软骨发育研究中应用最广,例如通过双腺相关病毒载体递送金黄色葡萄球菌Cas9可高效敲除生长板软骨细胞中的miR-140簇,敲除效率较高且脱靶率低[73]。
2.3.2 单细胞测序技术 单细胞测序技术主要包括基于微流控和液滴分选的两大技术体系。基于微流控的平台如Fluidigm C1系统[74],通过物理隔离单细胞实现低通量但高灵敏度的miRNA-mRNA共测序,其原理依赖于微腔室芯片捕获单个细胞并完成反转录扩增,适用于miRNA与靶基因互作网络的单细胞分辨率解析[75]。而液滴分选技术如10x Genomics Chromium系统[76],通过油包水乳滴将细胞与带有独特分子标识符的磁珠随机包裹,实现高通量(数千细胞/次)但灵敏度较低的miRNA捕获,其核心优势在于大规模单细胞转录组数据与miRNA调控变异性分析[76]。此外,基于微孔板的Smart-seq2技术采用全转录组扩增策略,虽通量低但覆盖miRNA异构体检测,可揭示白细胞介素1β等因子对软骨细胞miRISC复合体动态重编程的影响[77]。上述技术均需结合靶向富集策略(如miRNA-specific RT引物)解决低丰度miRNA检测难题,并在软骨分化研究中成功应用于鉴定诱导多能干细胞向软骨细胞分化过程中miR-525-5p等关键调控因子[78],以及骨关节炎软骨细胞亚群特异的miRNA表达特征[79]。
2.3.3 生物信息学分析技术 生物信息学分析技术主要可分为3类:①miRNA靶基因预测技术,如靶标扫描[80]、miRNA分析[81],通过序列互补性算法预测miRNA-mRNA相互作用,其中靶标扫描侧重种子区匹配而miRNA分析整合自由能计算;②差异表达分析技术,如差异表达分析2[82]、边缘发现率[83],基于RNA测序数据通过负二项分布模型识别软骨发育中差异miRNA,前者更适合小样本而后者处理批次效应更优;③网络构建技术,如细胞景观[83-84]、串扰网络[85],将miRNA-靶基因关系可视化并富集通路,细胞景观支持多组学数据整合,而串扰网络侧重蛋白互作网络验证。这些技术已成功应用于鉴定性别决定区Y框蛋白9、Ⅱ型胶原等软骨形成关键基因的调控miRNA(如miR-140[84]、miR-145[86]),并通过实验验证了生物信息学预测的可靠性[87-88]。最新进展显示,多组学整合分析(如长链非编码RNA-miRNA-mRNA网络[89])和动力学建模正成为揭示软骨发育表观遗传调控网络的有力工具[87]。
2.3.4 其他技术 最近,外泌体递送系统和类器官模型这些新技术,给研究miRNA调控软骨发育带来了新思路。研究发现,外泌体能当作天然的纳米载体,把治疗用的miRNA(如miR-140和miR-92a-3p)高效递送到软骨细胞中。这些miRNA通过作用于组蛋白去乙酰化酶2/8、Wnt5A这些通路,促进软骨基质的合成,还能阻止软骨退化[90-91]。特别是从骨髓间充质干细胞中提取的外泌体,通过miR-126-3p和miR-874-3p这些成分,能发挥抗炎和抗细胞凋亡的作用[92-93]。在骨关节炎模型中,这些外泌体能显著减轻软骨的退化。研究人员成功用人类细胞培养出软骨类器官,它能模拟天然软骨的分层结构,比如浅表层、增殖区和肥大区[94]。研究还发现,miR-322-5p通过RAF/丝裂原活化蛋白激酶激酶/细胞外调节蛋白激酶通路调控软骨分化[95]。通过比较新生关节软骨和软骨类器官的miRNA组学,研究人员发现miR-107和miR-140能诱导性别决定区Y框蛋白9和Ⅱ型胶原的表达[96]。而且,用工程化外泌体装载miR-23a-3p,能显著促进软骨再生[97]。这些技术不仅解决了传统二维培养无法模拟细胞和基质相互作用的问题,还为开发靶向外泌体药物递送系统和个性化治疗策略提供了实验基础。
2.4 展望 在软骨发育和再生领域,miRNA的作用机制研究已经取得了不少进展。如miR-140、miR-455、miR-199-5p这些miRNA,它们通过调控甲状旁腺激素相关蛋白/印度刺猬因子/骨形态发生蛋白信号通路,能影响软骨细胞的增殖、分化和基质合成,在动物模型中也显示出促进软骨再生的潜力[14,98]。不过,临床转化还是遇到了不少难题。首先,实验设计有问题,另外招募志愿者困难,导致研究进展不顺利[99];其次,miR-138这类miRNA在调控软骨代谢方面的结论还存在矛盾,没有一个标准化的评估体系;此外,递送系统的靶向性不足,现有的技术很难实现软骨亲和肽介导的精准递送。针对这些问题,有以下建议:一是建立类器官模型,例如用诱导多能干细胞来源软骨来替代传统的动物实验,这样可以优化临床前的数据;二是开发多组学整合分析平台,通过丝裂原活化蛋白激酶/叉头转录因子O这些通路网络,筛选出具有双向调控能力的miRNA组合;三是结合单细胞测序技术,明确miR-203a/Runt相关转录因子2这些靶点在不同软骨分区(静息区和肥大区)的表达差异。最新的研究发现,miR-206a-3p在胫骨软骨发育异常中表达特别高,这为转化医学提供了一个疾病特异性的标志物[13]。不过,还需要通过液态活检等技术来验证它在临床上的可行性。总的来说,这些进展不仅给软骨发育机制的研究带来了新的视角,也为未来的临床应用打下了基础。未来的研究应该进一步优化递送系统,提高miRNA治疗的精准性和有效性。同时,加强多学科合作,推动miRNA在软骨疾病治疗中的临床转化。

| [1] 王正业,刘万林,赵振群.miRNA在激素诱导股骨头坏死机制中的研究进展[J].中国组织工程研究,2026,30(5):1207-1214. [2] 李晓龙,孔清泉.微小RNA在MSCs成骨分化中的调控作用及在骨科疾病中作为治疗靶点和诊断工具的研究进展[J].中国修复重建外科杂志,2020,34(10):1332-1340. [3] YAO W, KULYAR MF, DING Y, et al. The Effect of miR-140-5p with HDAC4 towards Growth and Differentiation Signaling of Chondrocytes in Thiram-Induced Tibial Dyschondroplasia. Int J Mol Sci. 2023;24(13):10975. [4] GUO C, ZHANG M, SU W, et al. miR-199a-5p Relieves Obstructive Sleep Apnea Syndrome-Related Hypertension by Targeting HIF-1α. J Immunol Res. 2022;2022:7236647. [5] STEINLE H, WEBER J, STOPPELKAMP S, et al. Delivery of synthetic mRNAs for tissue regeneration. Adv Drug Deliv Rev. 2021;179: 114007. [6] GROGAN SP, CHEN X, SOVANI S, et al. Influence of cartilage extracellular matrix molecules on cell phenotype and neocartilage formation. Tissue Eng Part A. 2014;20(1-2):264-274. [7] KRISHNAN Y, GRODZINSKY AJ. Cartilage diseases. Matrix Biol. 2018;71-72:51-69. [8] PREIN C, BEIER F. ECM signaling in cartilage development and endochondral ossification. Curr Top Dev Biol. 2019;133:25-47. [9] KOZHEMYAKINA E, LASSAR AB, ZELZER E. A pathway to bone: signaling molecules and transcription factors involved in chondrocyte development and maturation. Development. 2015;142(5):817-831. [10] BERNSTEIN E, KIM SY, CARMELL MA, et al. Dicer is essential for mouse development. Nat Genet. 2003;35(3):215-217. [11] KOBAYASHI T, LU J, COBB BS, et al. Dicer-dependent pathways regulate chondrocyte proliferation and differentiation. Proc Natl Acad Sci U S A. 2008;105(6):1949-1954. [12] KOBAYASHI T, PAPAIOANNOU G, MIRZAMOHAMMADI F, et al. Early postnatal ablation of the microRNA-processing enzyme, Drosha, causes chondrocyte death and impairs the structural integrity of the articular cartilage. Osteoarthritis Cartilage. 2015;23(7):1214-1220. [13] WU S, LIU K, HUANG X, et al. Molecular mechanism of miR-203a targeting Runx2 to regulate thiram induced-chondrocyte development. Pestic Biochem Physiol. 2024;200:105817. [14] THAKORE P, DELANY AM. miRNA-based regulation in growth plate cartilage: mechanisms, targets, and therapeutic potential. Front Endocrinol (Lausanne). 2025;16:1530374. [15] HUANG L, JIN M, GU R, et al. miR-199a-5p Reduces Chondrocyte Hypertrophy and Attenuates Osteoarthritis Progression via the Indian Hedgehog Signal Pathway. J Clin Med. 2023;12(4):1313. [16] ZHANG X, WANG X, YU F, et al. PiRNA hsa_piR_019949 promotes chondrocyte anabolic metabolism by inhibiting the expression of lncRNA NEAT1. J Orthop Surg Res. 2024;19(1):31. [17] SCOTT KM, COHEN DJ, NIELSON DW, et al. Prophylactic administration of miR-451 inhibitor decreases osteoarthritis severity in rats. Sci Rep. 2022;12(1):16068. [18] LIN J, HUANG Y, LIN X, et al. Bauhinia championii alleviates extracellular matrix degradation in IL-1β induced chondrocytes via miRNA-145-5p/TLR4/NF-κB axis. Heliyon. 2023;9(8):e19138. [19] NING Y, ZHANG F, LI S, et al. Integrative analysis of miRNA in cartilage-derived extracellular vesicles and single-cell RNA-seq profiles in knee osteoarthritis. Arch Biochem Biophys. 2023;748:109785. [20] SONG J, ZENG X, LI C, et al. Alteration in cartilage matrix stiffness as an indicator and modulator of osteoarthritis. Biosci Rep. 2024;44(1): BSR20231730. [21] LIAN WS, WU RW, KO JY, et al. Inhibition of histone lysine demethylase 6A promotes chondrocytic activity and attenuates osteoarthritis development through repressing H3K27me3 enhancement of Wnt10a. Int J Biochem Cell Biol. 2023;158:106394. [22] LIN Y, ZHANG L, JI M, et al. MiR-653-5p drives osteoarthritis pathogenesis by modulating chondrocyte senescence. Arthritis Res Ther. 2024; 26(1):111. [23] YU H, DING C, HU Z, et al. Biocompatible ionized air alleviates rat osteoarthritis by modulating polarization from M1 to M2 macrophages. Sci Rep. 2024;14(1):31901. [24] WU M, WANG X, SHUAI J, et al. Identification of key miRNAs in unilateral mastication-induced disruption of cartilage homeostasis. Oral Dis. 2024;30(2):551-561. [25] WU Y, WANG J, LI X, et al. Co-culture of RhoA-overexpressed microtia chondrocytes and adipose-derived stem cells in the construction of tissue-engineered ear-shaped cartilage. Stem Cells. 2024;42(6):554-566. [26] BRITO VGB, BELL-HENSLEY A, MCALINDEN A. MicroRNA-138: an emerging regulator of skeletal development, homeostasis, and disease. Am J Physiol Cell Physiol. 2023;325(6):C1387-C1400. [27] LAN M, LIU Y, LIU J, et al. Matrix Viscoelasticity Tunes the Mechanobiological Behavior of Chondrocytes. Cell Biochem Funct. 2024;42(7): e4126. [28] LIANG Y, XU X, XU L, et al. Chondrocyte-specific genomic editing enabled by hybrid exosomes for osteoarthritis treatment. Theranostics. 2022; 12(11):4866-4878. [29] WANG C, WANG L, GUAN X, et al. MiR-4303 relieves chondrocyte inflammation by targeting ASPN in osteoarthritis. J Orthop Surg Res. 2021; 16(1):618. [30] JEE YH, WANG J, YUE S, et al. mir-374-5p, mir-379-5p, and mir-503-5p Regulate Proliferation and Hypertrophic Differentiation of Growth Plate Chondrocytes in Male Rats. Endocrinology. 2018;159(3):1469-1478. [31] HU H, DONG L, BU Z, et al. miR-23a-3p-abundant small extracellular vesicles released from Gelma/nanoclay hydrogel for cartilage regeneration. J Extracell Vesicles. 2020;9(1):1778883. [32] DALLE CARBONARE L, MINOIA A, BRAGGIO M, et al. Modulation of miR-146b Expression during Aging and the Impact of Physical Activity on Its Expression and Chondrogenic Progenitors. Int J Mol Sci. 2023;24(17):13163. [33] LI F, YAO J, HAO Q, et al. miRNA-103 promotes chondrocyte apoptosis by down-regulation of Sphingosine kinase-1 and ameliorates PI3K/AKT pathway in osteoarthritis. Biosci Rep. 2019;39(10): BSR20191255. [34] CAO Z, LIU W, QU X, et al. miR-296-5p inhibits IL-1β-induced apoptosis and cartilage degradation in human chondrocytes by directly targeting TGF-β1/CTGF/p38MAPK pathway. Cell Cycle. 2020; 19(12):1443-1453. [35] LAI C, LIAO B, PENG S, et al. Synovial fibroblast-miR-214-3p-derived exosomes inhibit inflammation and degeneration of cartilage tissues of osteoarthritis rats. Mol Cell Biochem. 2023;478(3):637-649. [36] ZUO J, CHEN C, ZHANG X, et al. Circ_HECW2 regulates LPS-induced apoptosis of chondrocytes via miR-93 methylation. Immun Inflamm Dis. 2021;9(3):943-949. [37] CHEN J, LIU Z, SUN H, et al. MiR-203a-3p attenuates apoptosis and pyroptosis of chondrocytes by regulating the MYD88/NF-κB pathway to alleviate osteoarthritis progression. Aging (Albany NY). 2023;15(23):14457-14472. [38] CAO Y, TANG S, NIE X, et al. Decreased miR-214-3p activates NF-κB pathway and aggravates osteoarthritis progression. EBioMedicine. 2021;65: 103283. [39] WU J, KUANG L, CHEN C, et al. miR-100-5p-abundant exosomes derived from infrapatellar fat pad MSCs protect articular cartilage and ameliorate gait abnormalities via inhibition of mTOR in osteoarthritis. Biomaterials. 2019;206: 87-100. [40] ZHOU Y, ZHAO Z, YAN L, et al. MiR-485-3p promotes proliferation of osteoarthritis chondrocytes and inhibits apoptosis via Notch2 and the NF-κB pathway. Immunopharmacol Immunotoxicol. 2021;43(3):370-379. [41] FELEKKIS K, PIERI M, PAPANEOPHYTOU C. Exploring the Feasibility of Circulating miRNAs as Diagnostic and Prognostic Biomarkers in Osteoarthritis: Challenges and Opportunities. Int J Mol Sci. 2023;24(17):13144. [42] RIGG E, WANG J, XUE Z, et al. Inhibition of extracellular vesicle-derived miR-146a-5p decreases progression of melanoma brain metastasis via Notch pathway dysregulation in astrocytes. J Extracell Vesicles. 2023;12(10): e12363. [43] ZHOU M, LIU B, YE HM, et al. ROS-induced imbalance of the miR-34a-5p/SIRT1/p53 axis triggers chronic chondrocyte injury and inflammation. Heliyon. 2024;10(11):e31654. [44] ZHENG D, CHEN T, YANG K, et al. Microfluidic Synthesis of miR-200c-3p Lipid Nanoparticles: Targeting ZEB2 to Alleviate Chondrocyte Damage in Osteoarthritis. Int J Nanomedicine. 2025;20:505-521. [45] QIAN Y, CHU G, ZHANG L, et al. M2 macrophage-derived exosomal miR-26b-5p regulates macrophage polarization and chondrocyte hypertrophy by targeting TLR3 and COL10A1 to alleviate osteoarthritis. J Nanobiotechnology. 2024;22(1):72. [46] SZALA D, KOPAŃSKA M, TROJNIAK J, et al. The Role of MicroRNAs in the Pathophysiology of Osteoarthritis. Int J Mol Sci. 2024;25(12):6352. [47] LIU J, WU X, LU J, et al. Exosomal transfer of osteoclast-derived miRNAs to chondrocytes contributes to osteoarthritis progression. Nat Aging. 2021;1(4):368-384. [48] ENDISHA H, DATTA P, SHARMA A, et al. MicroRNA-34a-5p Promotes Joint Destruction During Osteoarthritis. Arthritis Rheumatol. 2021;73(3): 426-439. [49] ZHU H, YAN X, ZHANG M, et al. miR-21-5p protects IL-1β-induced human chondrocytes from degradation. J Orthop Surg Res. 2019;14(1):118. [50] 周绪昌,陈妍,华蔚中,等.miR-21在骨关节炎中作用机制的研究进展[J].中国细胞生物学学报,2022,44(5):940-947. [51] NAKAMURA A, RAMPERSAUD YR, NAKAMURA S, et al. microRNA-181a-5p antisense oligonucleotides attenuate osteoarthritis in facet and knee joints. Ann Rheum Dis. 2019;78(1):111-121. [52] HE K, HUANG X, SHAN R, et al. Intra-articular Injection of Lornoxicam and MicroRNA-140 Co-loaded Cationic Liposomes Enhanced the Therapeutic Treatment of Experimental Osteoarthritis. AAPS PharmSciTech. 2021;23(1):9. [53] LI P, GAO Y, ZHOU R, et al. Intra-articular injection of miRNA-1 agomir, a novel chemically modified miRNA agonists alleviates osteoarthritis (OA) progression by downregulating Indian hedgehog in rats. Sci Rep. 2024;14(1):8101. [54] ZHANG Y, LI S, JIN P, et al. Dual functions of microRNA-17 in maintaining cartilage homeostasis and protection against osteoarthritis. Nat Commun. 2022;13(1):2447. [55] 余永国.软骨发育不全诊断及治疗[J].中华儿科杂志,2023,61(6):573-575. [56] HÖGLER W, WARD LM. New developments in the management of achondroplasia. Wien Med Wochenschr. 2020;170(5-6):104-111. [57] WANG Z, RAO Z, WANG X, et al. circPhc3 sponging microRNA‑93‑3p is involved in the regulation of chondrocyte function by mechanical instability in osteoarthritis. Int J Mol Med. 2022;49(1):6. [58] LIU X, YUAN J, WU Z, et al. Plasma exosome miRNA-26b-3p derived from idiopathic short stature impairs longitudinal bone growth via the AKAP2/ERK1/2 axis. J Nanobiotechnology. 2023;21(1):94. [59] SAVARIRAYAN R, IRVING M, WILCOX WR, et al. Sustained growth-promoting effects of vosoritide in children with achondroplasia from an ongoing phase 3 extension study. Med. 2025;6(5):100566. [60] WROBEL W, PACH E, BEN-SKOWRONEK I. Advantages and Disadvantages of Different Treatment Methods in Achondroplasia: A Review. Int J Mol Sci. 2021;22(11):5573. [61] SAVARIRAYAN R, TOFTS L, IRVING M, et al. Once-daily, subcutaneous vosoritide therapy in children with achondroplasia: a randomised, double-blind, phase 3, placebo-controlled, multicentre trial. Lancet. 2020;396(10252):684-692. [62] SAVARIRAYAN R, DE BERGUA JM, ARUNDEL P, et al. Oral Infigratinib Therapy in Children with Achondroplasia. N Engl J Med. 2025; 392(9):865-874. [63] SAVARIRAYAN R, HOERNSCHEMEYER DG, LJUNGBERG M, et al. Once-weekly TransCon CNP (navepegritide) in children with achondroplasia (ACcomplisH): a phase 2, multicentre, randomised, double-blind, placebo-controlled, dose-escalation trial. EClinicalMedicine. 2023; 65:102258. [64] WANG C, CUI L, GU Q, et al. The Mechanism and Function of miRNA in Intervertebral Disc Degeneration. Orthop Surg. 2022;14(3):463-471. [65] YANG F, WANG J, CHEN Z, et al. Role of microRNAs in intervertebral disc degeneration (Review). Exp Ther Med. 2021;22(2):860. [66] WANG Z, ZHANG S, ZHAO Y, et al. MicroRNA-140-3p alleviates intervertebral disc degeneration via KLF5/N-cadherin/MDM2/Slug axis. RNA Biol. 2021;18(12):2247-2260. [67] XING H, ZHANG Z, MAO Q, et al. Injectable exosome-functionalized extracellular matrix hydrogel for metabolism balance and pyroptosis regulation in intervertebral disc degeneration. J Nanobiotechnology. 2021;19(1):264. [68] LI Z, WU Y, TAN G, et al. Exosomes and exosomal miRNAs: A new therapy for intervertebral disc degeneration. Front Pharmacol. 2022;13:992476. [69] DE PONTUAL L, YAO E, CALLIER P, et al. Germline deletion of the miR-17∼92 cluster causes skeletal and growth defects in humans. Nat Genet. 2011; 43(10):1026-1030. [70] TASSANO E, DI ROCCO M, SIGNA S, et al. De novo 13q31.1-q32.1 interstitial deletion encompassing the miR-17-92 cluster in a patient with Feingold syndrome-2. Am J Med Genet A. 2013;161A(4):894-896. [71] ROGLER LE, KOSMYNA B, MOSKOWITZ D, et al. Small RNAs derived from lncRNA RNase MRP have gene-silencing activity relevant to human cartilage-hair hypoplasia. Hum Mol Genet. 2014; 23(2):368-382. [72] SHVEDOVA M, KOBAYASHI T. MicroRNAs in cartilage development and dysplasia. Bone. 2020; 140:115564. [73] TORELLA L, KLERMUND J, BILBAO-ARRIBAS M, et al. Efficient and safe therapeutic use of paired Cas9-nickases for primary hyperoxaluria type 1. EMBO Mol Med. 2024;16(1):112-131. [74] CHEN Y, WANG X, NA X, et al. Highly Multiplexed, Efficient, and Automated Single-Cell MicroRNA Sequencing with Digital Microfluidics. Small Methods. 2024;8(3):e2301250. [75] ZHANG J, LIU L, XU T, et al. Exploring cell-specific miRNA regulation with single-cell miRNA-mRNA co-sequencing data. BMC Bioinformatics. 2021;22(1):578. [76] LIU W, SHOMRON N. Analysis of MicroRNA Regulation and Gene Expression Variability in Single Cell Data. J Pers Med. 2022;12(10):1750. [77] ENGEL A, RISHIK S, HIRSCH P, et al. SingmiR: a single-cell miRNA alignment and analysis tool. Nucleic Acids Res. 2024;52(W1):W374-W380. [78] STELCER E, KULCENTY K, RUCINSKI M, et al. The Role of MicroRNAs in Early Chondrogenesis of Human Induced Pluripotent Stem Cells (hiPSCs). Int J Mol Sci. 2019;20(18):4371. [79] GAO H, DI J, YIN M, et al. Identification of chondrocyte subpopulations in osteoarthritis using single-cell sequencing analysis. Gene. 2023; 852:147063. [80] CHEN L, HEIKKINEN L, WANG C, et al. Trends in the development of miRNA bioinformatics tools. Brief Bioinform. 2019;20(5):1836-1852. [81] JIANG L, ZHANG J, XUAN P, et al. BP Neural Network Could Help Improve Pre-miRNA Identification in Various Species. Biomed Res Int. 2016;2016:9565689. [82] MAEDA Y, FARINA NH, MATZELLE MM, et al. Synovium-Derived MicroRNAs Regulate Bone Pathways in Rheumatoid Arthritis. J Bone Miner Res. 2017;32(3):461-472. [83] WANG G, HE L, XIANG Y, et al. Long noncoding and micro-RNA expression in a model of articular chondrocyte degeneration induced by stromal cell-derived factor-1. Asian Biomed (Res Rev News). 2022;16(4):169-179. [84] PROCTOR CJ, SMITH GR. Computer simulation models as a tool to investigate the role of microRNAs in osteoarthritis. PLoS One. 2017; 12(11):e0187568. [85] SHORTER E, AVELAR R, ZACHARIOU M, et al. Identifying Novel Osteoarthritis-Associated Genes in Human Cartilage Using a Systematic Meta-Analysis and a Multi-Source Integrated Network. Int J Mol Sci. 2022;23(8):4395. [86] JIA B, ZHANG L, ZHANG Y, et al. Integrated analysis of miRNA and mRNA transcriptomic reveals antler growth regulatory network. Mol Genet Genomics. 2021;296(3):689-703. [87] PATEL K, BARTER M, SOUL J, et al. Systems analysis of miR-199a/b-5p and multiple miR-199a/b-5p targets during chondrogenesis. Elife. 2024;12:RP89701. [88] CHEN J, WU X. Cyclic tensile strain promotes chondrogenesis of bone marrow-derived mesenchymal stem cells by increasing miR-365 expression. Life Sci. 2019;232:116625. [89] SHU T, HE L, WANG X, et al. Long noncoding RNA UCA1 promotes chondrogenic differentiation of human bone marrow mesenchymal stem cells via miRNA-145-5p/SMAD5 and miRNA-124-3p/SMAD4 axis. Biochem Biophys Res Commun. 2019;514(1):316-322. [90] MAO G, ZHANG Z, HU S, et al. Exosomes derived from miR-92a-3p-overexpressing human mesenchymal stem cells enhance chondrogenesis and suppress cartilage degradation via targeting WNT5A. Stem Cell Res Ther. 2018;9(1):247. [91] DUAN L, LIANG Y, XU X, et al. Recent progress on the role of miR-140 in cartilage matrix remodelling and its implications for osteoarthritis treatment. Arthritis Res Ther. 2020;22(1):194. [92] ZHOU Y, MING J, LI Y, et al. Exosomes derived from miR-126-3p-overexpressing synovial fibroblasts suppress chondrocyte inflammation and cartilage degradation in a rat model of osteoarthritis. Cell Death Discov. 2021;7(1):37. [93] WU Y, FENG Y, HU F, et al. Engineered Stem Cell Clusters for Extracellular Vesicles-Mediated Gene Delivery to Rejuvenate Chondrocytes and Facilitate Chondrogenesis in Osteoarthritis Therapy. Adv Sci (Weinh). 2025;12(25):e2500964. [94] LIU Z, ZHOU H, WU Q, et al. Constructing condylar cartilage organoid to explore primary cilia functions. Heliyon. 2024;10(17):e35972. [95] BLUHM B, EHLEN HWA, HOLZER T, et al. miR-322 stabilizes MEK1 expression to inhibit RAF/MEK/ERK pathway activation in cartilage. Development. 2017;144(19):3562-3577. [96] GANESH V, HE R, KEEN HL, et al. Profiles of Exosomal microRNAs in Joint Cells and Candidate microRNAs for Cartilage Regeneration. Tissue Eng Part A. 2025. doi: 10.1089/ten.tea.2024.0299. [97] CHEN J, HUANG T, LIU R, et al. Congenital microtia patients: the genetically engineered exosomes released from porous gelatin methacryloyl hydrogel for downstream small RNA profiling, functional modulation of microtia chondrocytes and tissue-engineered ear cartilage regeneration. J Nanobiotechnology. 2022;20(1):164. [98] FU L, WU J, LI P, et al. A novel mesenchymal stem cell-targeting dual-miRNA delivery system based on aptamer-functionalized tetrahedral framework nucleic acids: Application to endogenous regeneration of articular cartilage. Bioact Mater. 2024;40:634-648. [99] RAMIREZ-GOMEZ J, DALAL S, DEVARA D, et al. MicroRNA-based recent research developments in Alzheimer’s disease. J Alzheimers Dis. 2025; 104(1):14-31. |
| [1] | 于晨锜, 刘洋, 余建锋, 康康, 邓垚歌, 夏小伟, 张一健, 朱雪松. 仿生黑磷纳米系统调控滑膜巨噬细胞极化治疗骨关节炎[J]. 中国组织工程研究, 2026, 30(在线): 1-13. |
| [2] | 张 楠, 孟庆华, 鲍春雨. 踝关节有限元模型的特性及临床应用[J]. 中国组织工程研究, 2026, 30(9): 2343-2349. |
| [3] | 陈秋函, 杨 龙, 袁代柱, 吴展羽, 邹梓豪, 叶 川. 膝关节周围截骨治疗膝骨关节炎:治疗策略的优化[J]. 中国组织工程研究, 2026, 30(9): 2303-2312. |
| [4] | 张子峥, 罗 旺, 刘长路. 膝内侧间室骨关节炎单髁置换中有限元分析的应用价值[J]. 中国组织工程研究, 2026, 30(9): 2313-2322. |
| [5] | 王奇飒, 卢雨征, 韩秀峰, 赵文玲, 石海涛, 徐 哲. 3D打印甲基丙烯酰化透明质酸/脱细胞皮肤水凝胶支架的细胞相容性[J]. 中国组织工程研究, 2026, 30(8): 1912-1920. |
| [6] | 孙 蕾, 张 琦, 张 宇. 绿原酸蛋白微球/聚己内酯静电纺丝膜的促成骨效应[J]. 中国组织工程研究, 2026, 30(8): 1877-1884. |
| [7] | 黎清斌, 林建辉, 黄文杰, 王明爽, 杜间开, 劳永锵. 膝关节周围骨巨细胞瘤病灶扩大刮除后填充骨水泥:软骨下植骨与不植骨的比较[J]. 中国组织工程研究, 2026, 30(8): 1896-1902. |
| [8] | 宋浦蓁, 马贺宾, 陈宏广, 章亚东. 骨髓间充质干细胞外泌体联合转化生长因子β1对巨噬细胞的作用[J]. 中国组织工程研究, 2026, 30(7): 1616-1623. |
| [9] | 金东升, 赵张红, 朱子银, 张 森, 孙祖延, 邓 江. 淫羊藿苷缓释微球三维支架对兔骨髓间充质干细胞成骨分化的影响[J]. 中国组织工程研究, 2026, 30(7): 1658-1668. |
| [10] | 李林臻, 焦泓焯, 陈伟南, 张铭哲, 王建龙, 张君涛. 淫羊藿苷含药血清对脂多糖诱导人软骨细胞炎症损伤的影响[J]. 中国组织工程研究, 2026, 30(6): 1368-1374. |
| [11] | 吕国庆, 艾孜麦提江·肉孜, 熊道海. 鸢尾素抑制人关节软骨细胞中铁死亡的作用及其机制[J]. 中国组织工程研究, 2026, 30(6): 1359-1367. |
| [12] | 陈 驹, 郑锦畅, 梁 振, 黄成硕, 林 颢, 曾 莉. β-石竹烯对小鼠膝骨关节炎的作用及机制[J]. 中国组织工程研究, 2026, 30(6): 1341-1347. |
| [13] | 李 豪, 陶红成, 曾 平, 刘金富, 丁 强, 牛驰程, 黄 凯, 康宏誉. 丝裂原活化蛋白激酶信号通路调控骨关节炎的发生发展:指导中药靶点治疗[J]. 中国组织工程研究, 2026, 30(6): 1476-1485. |
| [14] | 张 倩, 黄东锋. 加权基因共表达网络分析结合机器学习筛选及验证骨关节炎生物标记物[J]. 中国组织工程研究, 2026, 30(5): 1096-1105. |
| [15] | 黄柳艳, 张文烯, 陈淑雯, 余诗美, 戴 忠, 左长清. 毛喉素调控ERK和Akt信号通路促进C2C12成肌细胞分化[J]. 中国组织工程研究, 2026, 30(5): 1114-1121. |
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 由第一作者在2025年6月进行检索。
1.1.2 文献检索时限 各数据库建库至2025年6月。
1.1.3 检索数据库 PubMed、中国知网、万方数据库及维普数据库。
1.1.4 检索词 英文检索词为“MicroRNAs/miRNAs/miRNA,Cartilage,Joint,Chondrogenesis,Joint development,Regulation,Signal pathway,Biomarker,Therapeutic Target”,中文检索词为“MicroRNAs/miRNAs/miRNA,软骨,关节,软骨发育,关节发育,调控,信号通路,生物标志物,治疗靶点”。
1.1.5 检索文献类型 研究原著及综述。
1.1.6 手工检索情况 手工查阅相关书籍《骨科学》(第二版)、《运动系统与疾病》(第二版)、《实用骨科学》(第二版)。
1.1.7 检索策略 以中国知网与PubMed数据库检索策略为例,见图1。
1.2 入组标准
1.2.1 纳入标准 ①与miRNA在关节软骨发育中的调控机制和研究进展有关;②文献质量、相关性、可靠性高或创新性较为突出的文献;③优先选择最近10年内发表的文献。
1.2.2 排除标准 ①与研究主题无关的文献;②重复性文献;③观点陈旧或存在争议的文献。
1.3 文献质量评价和筛选 共检索到3 411篇文献,初筛剔除重复文献后,通过泛读对剩余文献的标题、摘要进行筛选,无法判别时精读全文,选取与主题更为相符的文献,最终纳入符合要求的文献99篇,包括英文文献95篇、中文文献4篇。文献筛选流程见图2。
miRNA在软骨发育和疾病中的研究面临诸多挑战,如现有技术在动态监测和靶向干预方面存在瓶颈,临床转化研究缺乏标准化评估体系。此综述系统梳理了近10年的关键研究成果和技术突破,结合前沿技术的应用进展,为骨科领域miRNA研究的范式革新提供了理论依据和实践指引;还详细讨论了miRNA在软骨发育和疾病中的双向调节作用,强调了miRNA在维持软骨稳态和促进骨骼发育中的关键功能,局限性体现在部分miRNA的具体作用机制尚未完全明确,尤其是在体内生理环境中的确切意义仍需进一步探索。临床转化研究还处于初步阶段,多数实验停留在动物模型阶段,miRNA治疗的临床应用面临递送系统靶向性和效率等挑战。该文章系统总结了miRNA在软骨发育和疾病中的作用机制,为后续研究提供了理论依据和参考方向。随着基因编辑、单细胞测序、生物信息学等前沿技术的发展,miRNA研究取得了显著进展,有望为骨骼疾病的早期诊断、精准治疗和预后评估提供新手段。
课题组专家对未来研究方向提出建议:优化递送系统,提高miRNA治疗的精准性和有效性;加强多学科合作,推动临床转化;结合单细胞测序技术,明确miRNA在不同软骨分区的表达差异;建立类器官模型,优化临床前数据。未来研究应深化对miRNA作用机制的理解,推动miRNA在软骨修复与再生医学领域取得新突破,为骨骼疾病防治提供新策略。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
近年来,miRNA在软骨发育及疾病中的研究热点主要集中在miRNA如何通过调控关键信号通路和转录因子影响软骨细胞的分化、增殖和凋亡。此外,单细胞测序技术的应用使得研究者能够解析miRNA在软骨发育过程中的时空特异性表达谱,揭示了miRNA在不同软骨分区的差异表达模式。临床前研究显示,通过基因编辑技术或外泌体递送系统靶向调控miRNA,能够有效改善软骨损伤和疾病进展。然而,miRNA治疗的临床转化仍面临诸多挑战,如递送系统的靶向性和效率问题,以及miRNA功能的组织特异性悖论。本综述系统总结了miRNA在软骨发育及疾病中的作用机制,结合前沿技术的应用进展,为该领域研究提供了全面的视角。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||