1.1 设计 体外细胞学实验,两组之间比较采用t检验,多组间比较采用单因素方差分析,组间两两比较采用Tukey检验。
1.2 时间及地点 实验于2023年1月至2024年6月在川北医学院第二临床学院组织工程与干细胞研究所完成。
1.3 材料 小鼠来源的成骨前体细胞(MC3T3-E1 Subclone 14)购自中国科学院细胞库,实验所采用的细胞皆为生长状态优良的第4代细胞。柠檬酸铁铵(美国sigma-Aldrich公司);α-MEM培养基、青霉素/链霉素、胎牛血清、0.25%胰蛋白酶(Gibco公司);去铁胺和抗氧化剂ferrostatin-1(fer-1,MCE公司);CCK-8试剂盒(凯基生物公司);β-甘油磷酸钠、L-抗坏血酸-2-磷酸酯、PBS、Hoechst33342、BCA蛋白浓度测定试剂盒(北京索莱宝科技有限公司);地塞米松(美国Sigma-Aldrich公司);40 g/L
多聚甲醛、茜素红染色液、碱性磷酸酶定量检测试剂盒(上海碧云天生物技术有限公司);天狼星红(美国Sigma公司);TRIzol试剂、反转录试剂盒、SYBR green qPCR试剂盒(中国诺唯赞生物公司);PCR引物(上海生工生物科技有限公司);线粒体膜电位(JC-1)荧光探针、谷胱甘肽、丙二醛检测试剂盒、活性氧荧光探针DCFH-DA、亚铁离子比色法试剂盒(Fe2+)(中国Elabscience公司);BCIP/NBT碱性磷酸酶显色试剂盒(中国碧云天公司);戊二醛、缓冲液、乙醇、丙酮、柠檬酸铅、亚甲基蓝(国药集团化学试剂公司);RIPA细胞裂解液、5×上样缓冲液、十二烷基硫酸钠聚丙烯酰胺凝胶电泳(SDS-PAGE)凝胶制备试剂盒(中国雅酶公司);锇酸、Spurr包埋剂、醋酸双氧铀(美国SPI-CHEM公司);骨桥蛋白抗体、SCL7A11抗体、β-actin抗体(中国华安生物公司);Leica UC7超薄切片机(德国徕卡公司);ChemiDoc XRS+化学发光凝胶成像系统、CFX96实时荧光定量PCR仪(Bio-Rad);Multiskan Go酶标仪、NanoDrop2000超微量分光光度计(美国Thermo公司);HT7800透射电子显微镜(日本日立公司);倒置荧光显微镜(日本尼康公司)。
1.4 实验方法
1.4.1 细胞培养 MC3T3-E1细胞用α-MEM培养基(含1%青霉素/链霉素+体积分数10%胎牛血清),在37 ℃、体积分数5% CO2条件下培养,当细胞融合度达到80%-90%时消化传代。
1.4.2 细胞活力检测 采用CCK-8试剂盒测定细胞的增殖活力。将第4代MC3T3-E1细胞以5 000个/孔密度接种于96孔板中,培养24 h后吸去旧培养基,PBS洗涤3次,加入含有10,100,300,500 μmol/L柠檬酸铁铵的α-MEM培养基,培养24,48,72 h后,弃去培养基,PBS洗涤2次,加入含1% CCK-8溶液的无血清α-MEM培养基,37 ℃孵育1 h,用酶标仪在450 nm波长处检测吸光度值,确定柠檬酸铁铵的最佳浓度和干预时间。
将第4代MC3T3-E1细胞以5 000个/孔密度接种于96孔板中,培养24 h后吸去旧培养基,PBS洗涤3次,加入含1,5,10 μmol/L fer-1的α-MEM培养基预处理8 h或者含1,5,10 μmol/L去铁胺的α-MEM培养基预处理8 h后更换为含300 μmol/L柠檬酸铁胺的α-MEM培养基培养48 h,弃去培养基,PBS洗涤2次,加入含1% CCK-8溶液的无血清α-MEM培养基,37 ℃孵育1 h,用酶标仪在450 nm波长处检测吸光度值,确定fer-1和去铁胺的最佳浓度。
1.4.3 细胞分组 将第4代MC3T3-E1细胞按一定的密度种植在培养板中,培养24 h后吸去旧培养基,PBS洗涤3次,加入含不同药物的培养基,每2 d换液1次。实验分为4组:①空白组:只加入α-MEM培养基;②铁过载组:加入含有300 μmol/L柠檬酸铁铵的α-MEM培养基;③fer-1组:加入含5 μmol/L fer-1的α-MEM培养基预处理8 h后更换为含有300 μmol/L柠檬酸铁胺的α-MEM培养基;④去铁胺组:加入含5 μmol/L去铁胺的α-MEM培养基预处理8 h后更换为含有300 μmol/L柠檬酸铁胺的α-MEM培养基。
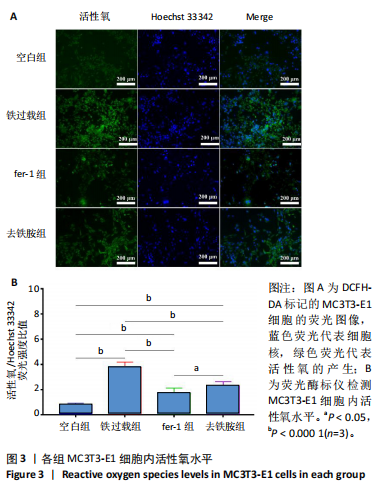
1.4.4 细胞内活性氧水平检测 利用荧光探针DCFH-DA检测细胞内活性氧水平。将MC3T3-E1细胞以1×104个/孔密度接种于24孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后用无血清α-MEM培养基清洗2次,加入α-MEM稀释的DCFH-DA(10 μmol/L)避光孵育20 min,用无血清培养基清洗2次,加入Hoechst 33342孵育10 min染色细胞核,将染色液更换为新鲜的α-MEM培养基,置于荧光显微镜下观察。此外,采用荧光酶标仪在488 nm波长处检测吸光度值,定量检测细胞内的荧光强度。
1.4.5 细胞内谷胱甘肽含量检测 采用谷胱甘肽试剂盒检测细胞内还原型谷胱甘肽含量。将MC3T3-E1细胞以1×105个/孔密度接种于6孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后用PBS洗涤细胞2次,加入0.25%胰酶蛋白酶消化2 min,300×g离心5 min,使用RIPA裂解液冰上裂解细胞30 min,4 ℃、10 000×g离心10 min,取上清液0.1 mL,加入显色液孵育5 min,酶标仪振板1 min,静置5 min,在405 nm波长处检测吸光度值,计算细胞内谷胱甘肽含量。
1.4.6 细胞内丙二醛含量检测 采用丙二醛试剂盒检测细胞内丙二醛含量。将MC3T3-E1细胞以1×105个/孔密度接种于6孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后用PBS洗涤细胞2次,加入0.25%胰酶蛋白酶消化2 min,300×g离心5 min,使用RIPA裂解液冰上裂解细胞30 min,4 ℃、10 000×g离心10 min,取上清液100 μL加入200 μL 丙二醛检测工作液,沸水浴加热15 min后水浴冷却至室温,1 000×g室温离心10 min,取200 μL上清加入到96孔板中,采用酶标仪在532 nm波长处测定吸光度值,计算细胞内丙二醛含量。
1.4.7 细胞内亚铁离子含量检测 采用亚铁离子比色法试剂盒检测细胞内二价铁含量。将MC3T3-E1细胞以1×105个/孔密度接种于6孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后用PBS洗涤细胞2次,加入0.25%胰酶蛋白酶消化2 min,300×g离心5 min,使用RIPA裂解液冰上裂解细胞30 min,4 ℃、10 000×g离心10 min,取上清液200 μL与亚铁离子检测工作液 37 ℃孵育10 min,12 000×g离心10 min,取300 μL上清液加入酶标板各对应孔中,采用酶标仪在593 nm波长处检测吸光度值,计算细胞内亚铁离子含量。
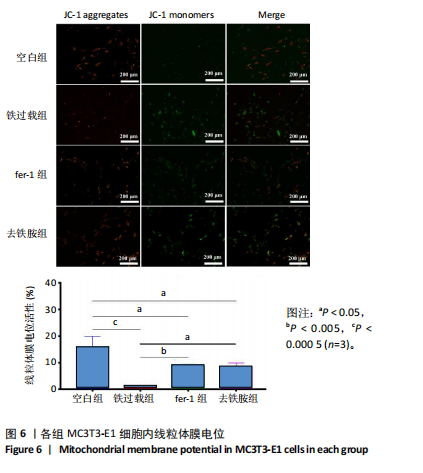
1.4.8 线粒体膜电位检测 利用JC-1荧光探针对线粒体膜电位进行检测。将MC3T3-E1细胞以1×104个/孔密度接种于24孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后弃去培养基,用无血清培养基洗涤2次,每孔加入1 mL JC-1染色工作液,充分混匀后37 ℃孵育20 min,吸除上清,用JC-1染色缓冲液洗涤2次,加入2 mL无血清细胞培养基,荧光显微镜下观察。
1.4.9 线粒体形态观察 用透射电子显微观察线粒体的超微结构。将MC3T3-E1细胞以1×105个/孔密度接种于6孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后PBS洗涤2次,2.5%戊二醛室温固定5 min,收集细胞,用2.5%戊二醛4 ℃重悬过夜,倒掉固定液,PBS漂洗3次,用1%锇酸溶液固定样品1.0-2.0 h,PBS漂洗3次;用梯度体积分数(30%,50%,70%,80%)的乙醇溶液对样品进行脱水处理,每种体积分数处理15 min,然后过渡到90%,95%的丙酮溶液中对样品进行脱水处理,每种浓度处理15 min,最后用纯丙酮处理2次,每次20 min,用Spurr包埋剂与丙酮的混合液(体积比为1∶1)对样品渗透1 h,用Spurr包埋剂与丙酮的混合液(体积比为3∶1)对样品渗透3 h,用Spurr包埋剂对样品渗透过夜,将渗透处理的样品放入装有包埋剂的包埋模具中,70 ℃加热过夜,在超薄切片机中切片,厚度为70-90 nm,切片经柠檬酸铅溶液和醋酸双氧铀50%乙醇饱和溶液各染色5-10 min,用透射电子显微镜观察线粒体的超微结构。
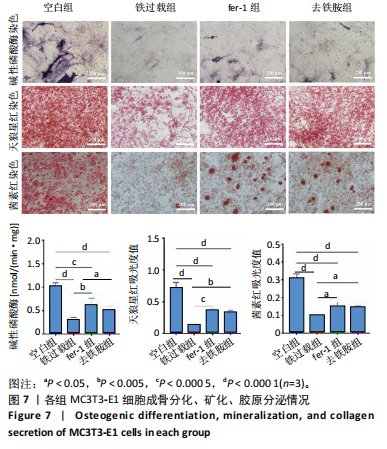
1.4.10 碱性磷酸酶染色及活性检测 采用BCIP/NBT碱性磷酸酶显色试剂盒及碱性磷酸酶活性试剂盒评估细胞的成骨分化能力。将MC3T3-E1细胞以1×104个/孔密度接种于24孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后用无血清α-MEM培养基清洗,加入成骨诱导培养基(含1%青霉素/链霉素+体积分数10%胎牛血清+10 mmol/L β-甘油磷酸钠+50 μg/mL L-抗坏血酸-2-磷酸酯+10 nmol/L 地塞米松),成骨诱导7 d后用40 g/L多聚甲醛固定20 min,PBS洗3次,每孔加入1 mL BCIP/NBT染色工作液,室温避光反应30 min;吸弃BCIP/NBT染色工作液终止反应后加入PBS洗2次,倒置光学显微镜观察。
使用碱性磷酸酶活性试剂盒定量检测MC3T3-E1细胞碱性磷酸酶活性。将MC3T3-E1细胞以1×104个/孔密度接种于24孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后用无血清α-MEM培养基清洗,加入成骨诱导培养基,成骨诱导7 d后,使用RIPA裂解液裂解细胞,3 000 r/min离心20 min,将上清液转移到96孔板中,每孔加入20 μL显色底物,37 ℃孵育30 min,利用酶标仪测定405 nm波长处的吸光度值。同时,取4 µL细胞裂解上清液加入216 µL总蛋白工作液,室温避光反应30 min,利用酶标仪测定562 nm波长处的吸光度值,得到总蛋白浓度,对样品进行归一化处理,计算细胞的碱性磷酸酶活性。
1.4.11 茜素红S染色 采用茜素红S染色表征细胞的钙结节生成情况。将MC3T3-E1细胞以1×104个/孔密度接种于24孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后弃去培养基,PBS清洗2次,每孔加入1 mL成骨诱导培养基,培养14 d,PBS清洗2次,用多聚甲醛避光固定15 min,用茜素红染液室温避光染色10 min,显微镜观察各组钙结节沉积情况,然后每孔加入0.5 mL 10%氯化十六烷基吡啶,振荡15 min溶解钙结节,采用酶标仪测定562 nm波长处的吸光度值。
1.4.12 天狼星红染色 采用天狼星红染色表征细胞的胶原分泌情况。将MC3T3-E1细胞以1×104个/孔密度接种于24孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后弃去培养基,PBS清洗2次,每孔加入1 mL成骨诱导培养基,培养14 d,PBS清洗2次,多聚甲醛避光固定15 min,PBS清洗3次,用含0.1%天狼星红的苦味酸溶液室温避光染色18 h,用0.1 mol/L乙酸反复漂洗样品,至无红色析出,显微镜观察细胞的胶原分泌情况,然后每孔加入溶解液(0.2 mol/L NaOH∶甲醇=
1∶1) 0.5 mL,振荡15 min溶解样品表面染料,采用酶标仪测定492 nm波长处的吸光度值。
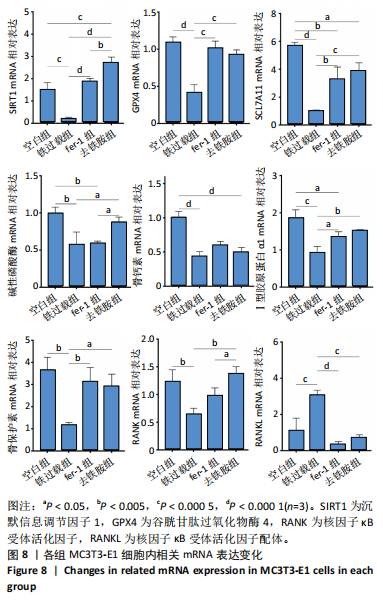
1.4.13 RT-qPCR 利用RT-qPCR检测细胞内相关mRNA的表达。将MC3T3-E1细胞以1×105个/孔密度接种于6孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后弃去培养基,PBS清洗3次,使用RNA提取试剂盒提取细胞总RNA,对提取的RNA进行定量分析,然后反转录RNA获得cDNA。以cDNA为模板,使用SYBR Green法检测沉默信息调节因子1(sirtuin 1,SIRT1)、Ⅰ型胶原蛋白α1、谷胱甘肽过氧化物酶4(glutathione peroxidase 4,GPX4)、SCL7A11、骨保护素、核因子κ B受体活化因子(receptor activator of nuclear factor-κB,RANK)、核因子κB受体活化因子配体(receptor activator of nuclear factor-κB ligand,RANKL)基因表达量。
将MC3T3-E1细胞以1×105个/孔密度接种于6孔板中,培养24 h后按照实验分组换液并加入相应培养基,培养48 h后用无血清α-MEM培养基清洗,加入成骨诱导培养基,成骨诱导7 d后弃去培养基,PBS清洗3次,按照上述方法检测骨钙素、Ⅰ型胶原蛋白α1、碱性磷酸酶基因表达量。
采用2-ΔΔCt法进行定量分析。每个样品至少重复3次。引物序列见表1。

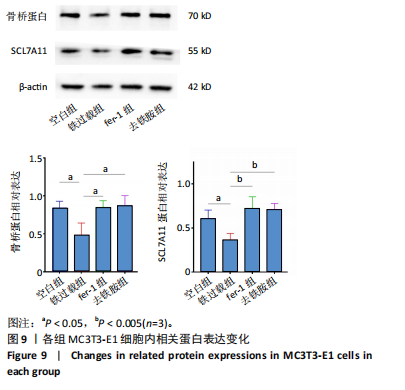
1.4.14 Western blot检测 将MC3T3-E1细胞以1×105个/孔密度接种于6孔板中,培养24 h后按照实验分组加入相应培养基,48 h后弃去培养基,用RIPA 裂解液于冰上裂解30 min,使用BCA蛋白浓度测定试剂盒测定各组蛋白浓度并进行归一化处理。将各组样品等量等体积进行SDS-PAGE凝胶电泳分离蛋白后,将蛋白转移至PVDF膜上,用5%的脱脂奶粉4 ℃封闭1 h,用TBST清洗4次;采用稀释后的一抗在4 ℃过夜孵育PVDF 膜。一抗稀释浓度分别为:骨桥蛋白(稀释比1∶1 000)、SCL7A11(稀释比1∶1 000),内参β-actin(稀释比1∶100 000)。用PBST清洗PVDF膜3次;室温下用山羊抗兔二抗(稀释比1∶10 000)孵育1 h,用PBST清洗4次,使用ChemiDoc XRS+化学发光凝胶成像系统显影。使用Image J软件对各条带灰度值进行半定量分析,最终结果以目的蛋白与内参的灰度值比值表示。
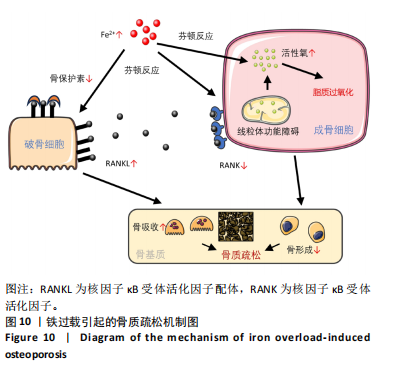
1.5 主要观察指标 各组 MC3T3-E1 细胞活性氧含量,亚铁离子、谷胱甘肽、丙二醛水平,线粒体电位与形态,碱性磷酸酶染色、茜素红染色、天狼星红染色结果,成骨与铁死亡相关基因的 mRNA 与蛋白表达。
1.6 统计学分析 采用SPSS 25.0统计软件进行统计分析。所有实验均重复3次,实验数据以 x±s表示,两组之间比较采用独立样本t检验,多组间比较采用单因素方差分析,P < 0.05 为差异有显著性意义。统计数据用GraphPad prism 9.5绘图。文章统计学方法已经通过张成栋、罗栩伟专家审核。