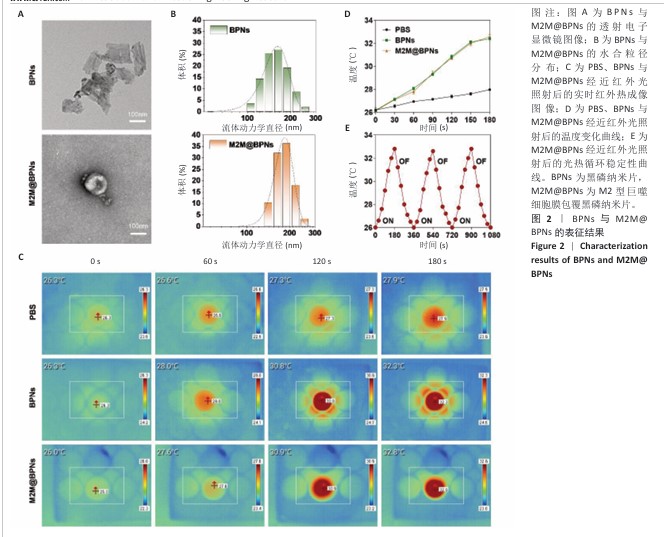
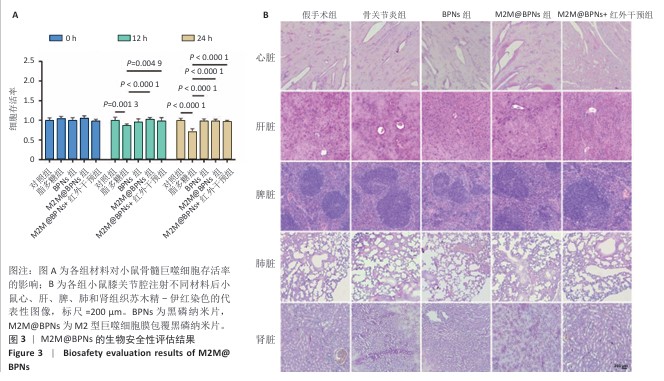
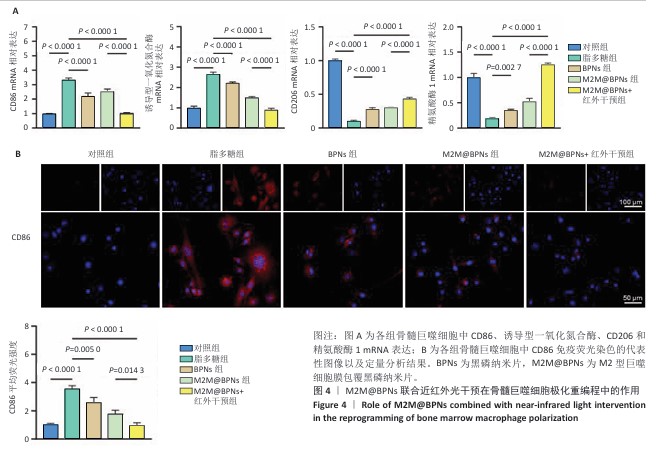
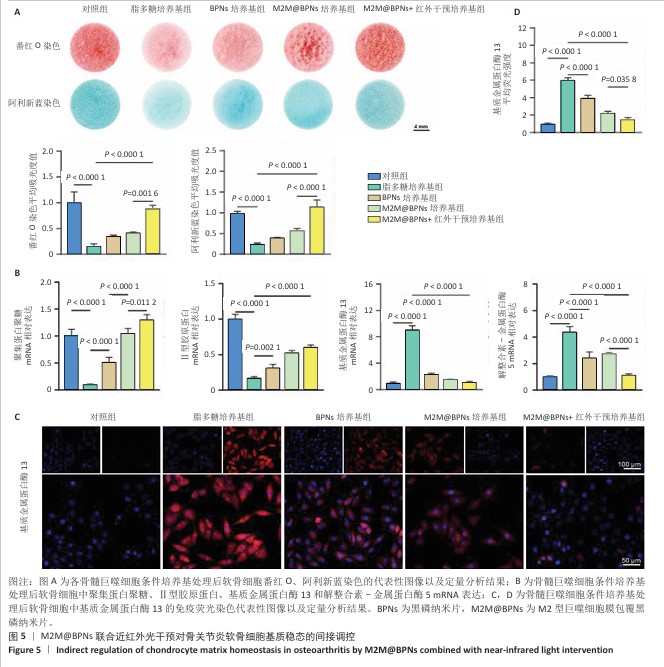
[1] KLOPPENBURG M, NAMANE M, CICUTTINI F. Osteoarthritis. Lancet. 2025; 405(10472):71-85.
[2] ZHANG X, HUANG C, HU Z, et al. Global, regional, and country-specific lifetime risks of osteoarthritis, 1990–2021: a systematic analysis for the global burden of disease study 2021. Glob Health Res Policy. 2025;10(1):29.
[3] SAFIRI S, KOLAHI AA, SMITH E, et al. Global, regional and national burden of osteoarthritis 1990-2017: a systematic analysis of the Global Burden of Disease Study 2017. Ann Rheum Dis. 2020;79(6):819-828.
[4] ZHAO R, LIANG B, SHI Y, et al. Immunomodulatory biomaterials for osteoarthritis: Targeting inflammation and enhancing cartilage regeneration. Mater Today Bio. 2025;34:102100.
[5] THOMSON A, HILKENS MU. Synovial Macrophages in Osteoarthritis: The Key to Understanding Pathogenesis? Front Immunol. 2021;12:678757.
[6] GRIFFIN TM, SCANZELLO CR. Innate Inflammation and Synovial Macrophages in Osteoarthritis Pathophysiology. Clin Exp Rheumatol. 2020;120(5):57-63.
[7] ZHANG Y, JI Q. Macrophage polarization in osteoarthritis progression: a promising therapeutic target. Front Cell Dev Biol. 2023;11:1269724.
[8] YAO Q, WU X, TAO C, et al. Osteoarthritis: pathogenic signaling pathways and therapeutic targets. Signal Transduct Target Ther. 2023;8(1):56.
[9] ZHANG Z, WANG R, XUE H, et al. Phototherapy techniques for the management of musculoskeletal disorders: strategies and recent advances. Biomater Res. 2023;27(1):123.
[10] XIA S, LIU D, JIANG K, et al. Photothermal driven BMSCs osteogenesis and M2 macrophage polarization on polydopamine-coated Ti3C2 nanosheets/poly(vinylidene fluoride trifluoroethylene) nanocomposite coatings. Mater Today Bio. 2024;27:101156.
[11] HSU Y, HE Y, ZHAO X, et al. Photothermal Coating on Zinc Alloy for Controlled Biodegradation and Improved Osseointegration. Adv Sci. 2025;12(9):2409051.
[12] XUE Y, ZENG G, CHENG J, et al. Engineered macrophage membrane‐enveloped nanomedicine for ameliorating myocardial infarction in a mouse model. Bioeng Transl Med. 2021;6(2):e10197.
[13] DENG W, WANG T, LI L, et al. A review of nanomaterials in osteoarthritis treatment and immune modulation. Regen Biomater. 2025;12:rbaf048.
[14] THURAKKAL S, ZHANG X. Recent Advances in Chemical Functionalization of 2D Black Phosphorous Nanosheets. Adv Sci. 2020;7(2):1902359.
[15] TONG L, LIAO Q, ZHAO Y, et al. Near-infrared light control of bone regeneration with biodegradable photothermal osteoimplant. Biomaterials. 2019;193:1-11.
[16] SUN T, LI C, LUAN J, et al. Black phosphorus for bone regeneration: Mechanisms involved and influencing factors. Mater Today Bio. 2024;28:101211.
[17] MIAO Y, CHEN Y, LUO J, et al. Black phosphorus nanosheets-enabled DNA hydrogel integrating 3D-printed scaffold for promoting vascularized bone regeneration. Bioact Mater. 2023;21:97-109.
[18] QIU M, TULUFU N, TANG G, et al. Black Phosphorus Accelerates Bone Regeneration Based on Immunoregulation. Adv Sci. 2024;11(1):2304824.
[19] LI H, XUE Y, ZHANG T, et al. Black Phosphorus Nanosheets Incorporated Mesenchymal Stem Cell Spheroids for Accelerated Bone Regeneration. ACS Appl Nano Mater. 2025;8(17):8963-8976.
[20] QIU S, ZHU F, TONG L. Application of targeted drug delivery by cell membrane-based biomimetic nanoparticles for inflammatory diseases and cancers. Eur J Med Res. 2024;29(1):523.
[21] LIU H, SU YY, JIANG XC, et al. Cell membrane-coated nanoparticles: a novel multifunctional biomimetic drug delivery system. Drug Deliv Transl Res. 2023; 13(3):716-737.
[22] ZHOU K, YANG C, SHI K, et al. Activated macrophage membrane-coated nanoparticles relieve osteoarthritis-induced synovitis and joint damage. Biomaterials. 2023;295:122036.
[23] TEO KYW, SEVENCAN C, CHEOW YA, et al. Macrophage Polarization as a Facile Strategy to Enhance Efficacy of Macrophage Membrane‐Coated Nanoparticles in Osteoarthritis. Small Sci. 2022;2(4):2100116.
[24] LIAO J, ZHU Z, ZOU J, et al. Macrophage Membrane‐Biomimetic Multi‐Layered Nanoparticles Targeting Synovial Angiogenesis for Osteoarthritis Therapy. Adv Healthc Mater. 2025;14(2):2401985.
[25] WU Y, WAN S, YANG S, et al. Macrophage cell membrane-based nanoparticles: a new promising biomimetic platform for targeted delivery and treatmen. J Nanobiotechnol. 2022;20(1):542.
[26] TODA G, YAMAUCHI T, KADOWAKI T, et al. Preparation and culture of bone marrow-derived macrophages from mice for functional analysis. STAR Protoc. 2021;2(1):100246.
[27] GOSSET M, BERENBAUM F, THIRION S, et al. Primary culture and phenotyping of murine chondrocytes. Nat Protoc. 2008;3(8):1253-1260.
[28] TAO W, ZHU X, YU X, et al. Black Phosphorus Nanosheets as a Robust Delivery Platform for Cancer Theranostics. Adv Mater. 2017;29(1):1603276.
[29] LIU Y, HAO R, LV J, et al. Targeted knockdown of PGAM5 in synovial macrophages efficiently alleviates osteoarthritis. Bone Res. 2024;12(1):15.
[30] YU T, GAN S, ZHU Q, et al. Modulation of M2 macrophage polarization by the crosstalk between Stat6 and Trim24. Nat Commun. 2019;10(1):4353.
[31] MERKESTEIN M, LABER S, MCMURRAY F, et al. FTO influences adipogenesis by regulating mitotic clonal expansion. Nat Commun. 2015;6(1):6792.
[32] HOU J, WANG H, GE Z, et al. Treating Acute Kidney Injury with Antioxidative Black Phosphorus Nanosheets. Nano Lett. 2020;20(2):1447-1454.
[33] GLASSON SS. The OARSI histopathology initiative - recommendations for histological assessments of osteoarthritis in the mouse. Osteoarthritis Cartilage. 2010;18 Suppl 3:S17-23.
[34] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182): 1745-1759.
[35] SCANZELLO CR, GOLDRING SR. The role of synovitis in osteoarthritis pathogenesis. Bone. 2012;51(2):249-257.
[36] BONDESON J, WAINWRIGHT SD, LAUDER S, et al. The role of synovial macrophages and macrophage-produced cytokines in driving aggrecanases, matrix metalloproteinases, and other destructive and inflammatory responses in osteoarthritis. Arthritis Res Ther. 2006;8(6):R187.
[37] SANCHEZ-LOPEZ E, CORAS R, TORRES A, et al. Synovial inflammation in osteoarthritis progression. Nat Rev Rheumatol. 2022;18(5):258-275.
[38] ZHANG H, CAI D, BAI X. Macrophages regulate the progression of osteoarthritis. Osteoarthritis Cartilage. 2020;28(5):555-561.
[39] LI B, LIU F, YE J, et al. Regulation of Macrophage Polarization Through Periodic Photo‐Thermal Treatment to Facilitate Osteogenesis. Small. 2022;18(38): 2202691.
[40] ZHAO J, LUO Y, ZHANG L, et al. Mild Hyperthermia Accelerates Bone Repair by Dynamically Regulating iNOS/Arg1 Balance in the Early Stage. Adv Sci. 2025; 12(8):2409882.
[41] YUE H, YUAN L, ZHANG W, et al. Progress on therapeutic applications of polymer decorated black phosphorus and black phosphorus analogues nanomaterials in biomedicine. J Mater Chem B. 2018;6(3):393-400.
[42] WANG Z, LIU Z, SU C, et al. Biodegradable Black Phosphorus-based Nanomaterials in Biomedicine: Theranostic Applications. Curr Med Chem. 2019;26(10): 1788-1805.
[43] ZHANG X, ZHAO M, XIAO X, et al. Inorganic Nanomaterials for Osteoarthritis: From Delivery Vehicles to Therapeutic Agents. ACS Appl Mater Interfaces. 2025; 17(28):39845-39862.
[44] ZHANG Y, SONG J, LI Y, et al. Diagnostic and Therapeutic Potential of Photo-Responsive Nanomaterials in Osteoarthritis. Aging Dis. 2025. doi: 10.14336/AD.2025.0166.
[45] ZENG J, GU C, GENG X, et al. Combined photothermal and sonodynamic therapy using a 2D black phosphorus nanosheets loaded coating for efficient bacterial inhibition and bone-implant integration. Biomaterials. 2023;297:122122.
[46] MURRAY PJ, ALLEN JE, BISWAS SK, et al. Macrophage Activation and Polarization: Nomenclature and Experimental Guidelines. Immunity. 2014;41(1):14-20.
[47] LOCATI M, CURTALE G, MANTOVANI A. Diversity, Mechanisms, and Significance of Macrophage Plasticity. Annu Rev Pathol. 2020;15(1):123-147.
[48] WANG H, ZHANG J, LI Z, et al. NIR-programmable 3D-printed shape-memory scaffold with dual-thermal responsiveness for precision bone regeneration and bone tumor management. J Nanobiotechnol. 2025;23(1):300.
[49] ZHANG L, CHEN X, CAI P, et al. Reprogramming Mitochondrial Metabolism in Synovial Macrophages of Early Osteoarthritis by a Camouflaged Meta‐Defensome. Adv Mater. 2022;34(30):2202715.
[50] GOLDRING MB, GOLDRING SR. Articular cartilage and subchondral bone in the pathogenesis of osteoarthritis. Ann N Y Acad Sci. 2010;1192(1):230-237.
[51] KAPOOR M, MARTEL-PELLETIER J, LAJEUNESSE D, et al. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011; 7(1):33-42.
[52] LU H, WEI J, LIU K, et al. Radical-Scavenging and Subchondral Bone-Regenerating Nanomedicine for Osteoarthritis Treatment. ACS Nano. 2023;17(6):6131-6146.
[53] LOTZ M, MARTEL-PELLETIER J, CHRISTIANSEN C, et al. Value of biomarkers in osteoarthritis: current status and perspectives. Ann Rheum Dis. 2013;72(11): 1756-1763.
[54] SHEN Y, LIANG L, ZHANG S, et al. A core-shell structure QRu-PLGA-RES-DS NP nanocomposite with photothermal response-induced M2 macrophage polarization for rheumatoid arthritis therapy. Nanoscale. 2018;10(4):1622-1630.
[55] YIN W, SUN S, YAO H, et al. Black Phosphorus Nanosheet-Based Composite Biomaterials for the Enhanced Repair of Infectious Bone Defects. ACS Biomater Sci Eng. 2025;11(3):1317-1337.
[56] LIAO J, GU Q, LIU Z, et al. Edge advances in nanodrug therapies for osteoarthritis treatment. Front Pharmacol. 2024;15:1402825.
[57] MO J, XIE Q, WEI W, et al. Revealing the immune perturbation of black phosphorus nanomaterials to macrophages by understanding the protein corona. Nat Commun. 2018;9(1): 2480.
|