[1] HUANG J, FU X, CHEN X, et al. Promising Therapeutic Targets for Treatment of Rheumatoid Arthritis. Front Immunol. 2021;12:686155.
[2] PRASAD P, VERMA S, SURBHI, et al. Rheumatoid arthritis: advances in treatment strategies. Mol Cell Biochem. 2023;478(1):69-88.
[3] BRZUSTEWICZ E, HENC I, DACA A, et al. Autoantibodies, C-reactive protein, erythrocyte sedimentation rate and serum cytokine profiling in monitoring of early treatment. Cent Eur J Immunol. 2017;42(3):259-268.
[4] ALETAHA D, SMOLEN JS. Diagnosis and Management of Rheumatoid Arthritis: A Review. JAMA. 2018;320(13):1360-1372.
[5] SMOLEN JS, ALETAHA D, BARTON A, et al. Rheumatoid arthritis. Nat Rev Dis Primers. 2018; 4:18001.
[6] 田新平,李梦涛,曾小峰.我国类风湿关节炎诊治现状与挑战:来自中国类风湿关节炎2019年年度报告[J].中华内科杂志,2021,60(7):593-598.
[7] 池里群,周彬,高文远,等.治疗类风湿性关节炎常用药物的研究进展[J].中国中药杂志, 2014,39(15):2851-2858.
[8] MIN HK, KIM SH, KIM HR, et al. Therapeutic Utility and Adverse Effects of Biologic Disease-Modifying Anti-Rheumatic Drugs in Inflammatory Arthritis. Int J Mol Sci. 2022;23(22):13913.
[9] TAN Y, BUCH MH. ‘Difficult to treat’ rheumatoid arthritis: current position and considerations for next steps. RMD Open. 2022;8(2):e002387.
[10] 李洋,刘健,胡月迪,等.基于《黄帝内经》营卫理论探讨类风湿关节炎的中医辨治思路[J].山西中医药大学学报,2024,25(12):1405-1411.
[11] 《中成药治疗优势病种临床应用指南》标准化项目组.中成药治疗类风湿关节炎临床应用指南(2022年)[J].中国中西医结合杂志,2023, 43(3):261-273.
[12] SHEN J, FANG Y, XU N, et al. Exploring the mechanism of triptolide inhibiting the motility of fibroblast-like synoviocytes in rheumatoid arthritis via RhoA/Rho-associated kinase axis, based on network pharmacology, molecular docking and molecular dynamics simulations. Front Pharmacol. 2025;16:1545514.
[13] 杨洁,石英杰,舒峻,等.雷公藤甲素靶向FASN调控类风湿关节炎脂代谢的机制研究[J].中国中医基础医学杂志,2024,30(12):2053-2059.
[14] BAO J, DAI SM. A Chinese herb Tripterygium wilfordii Hook F in the treatment of rheumatoid arthritis: mechanism, efficacy, and safety. Rheumatol Int. 2011;31(9):1123-1129.
[15] 许植方,李珠莲,宋涛能.民间杀虫药雷公藤根成分研究:雷公藤甲素[J].化学世界,1957(3): 102-105.
[16] KUPCHAN SM, COURT WA, DAILEY RG Jr, et al. Triptolide and tripdiolide, novel antileukemic diterpenoid triepoxides from Tripterygium wilfordii. J Am Chem Soc. 1972;94(20):7194-7205.
[17] ZENG XJ, ZHANG NZ. Effects of tripchlorolide (T4) of Tripterygium Wilfordii Hook on the production of immunoglobulins by peripheral blood mononuclear cells and by synovial cells of rheumatoid arthritis patients in vitro. Yao Xue Xue Bao. 1997;32(3):171-173.
[18] GUO Y, YU M, JIANG Y, et al. Effect of Tripterygium Wilfordii Hook T4 monomer on proliferation and interleukin-6 production of synovial fibroblasts of patients with rheumatoid arthritis. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2000;22(2):190-202.
[19] LIN N, LIU C, XIAO C, et al. Triptolide, a diterpenoid triepoxide, suppresses inflammation and cartilage destruction in collagen-induced arthritis mice. Biochem Pharmacol. 2007;73(1):136-146.
[20] HU J, NI J, ZHENG J, et al. Tripterygium hypoglaucum extract ameliorates adjuvant-induced arthritis in mice through the gut microbiota. Chin J Nat Med. 2023;21(10):730-744.
[21] WANG Y, YAN J, ZHANG Z, et al. Immunosuppressive Sesquiterpene Pyridine Alkaloids from Tripterygium wilfordii Hook. f. Molecules. 2022;27(21):7274.
[22] TIAN Y, WEI Y, LUO H, et al. Diterpenoids and lignans from the stems of Tripterygium wilfordii and their anti-inflammatory activities. Fitoterapia. 2025;180:106306.
[23] CHEN J, XUE Y, SHUAI X, et al. Effect of major components of Tripterygium wilfordii Hook. f on the uptake function of organic anion transporting polypeptide 1B1. Toxicol Appl Pharmacol. 2022; 435:115848.
[24] LONG C, YANG Y, YANG Y, et al. The Exploration of Novel Pharmacophore Characteristics and Multidirectional Elucidation of Structure-Activity Relationship and Mechanism of Sesquiterpene Pyridine Alkaloids from Tripterygium Based on Computational Approaches. Evid Based Complement Alternat Med. 2021;2021:6676470.
[25] MOON M, PYEON M, YANG J, et al. Subtype-selective effect and molecular regulation of celastrol and triptolide at human nicotinic acetylcholine receptors. Chem Biol Interact. 2025;408:111412.
[26] LI S, CHEN Q, ZHANG Y, et al. Hyaluronic acid dissolving microneedle patch-assisted acupoint transdermal delivery of triptolide for effective rheumatoid arthritis treatment. Sci Rep. 2024; 14(1):25256.
[27] BHAMBHANI S, KONDHARE KR, GIRI AP. Diversity in Chemical Structures and Biological Properties of Plant Alkaloids. Molecules. 2021;26(11):3374.
[28] 高淑红,韦玲.雷公藤的化学成分及其提取物检验方法的分析[J].生物技术世界,2015(11):39.
[29] DUAN H, TAKAISHI Y, MOMOTA H, et al. Immunosuppressive diterpenoids from Tripterygium wilfordii. J Nat Prod. 1999;62(11): 1522-1535.
[30] ZHANG Y, MAO X, LI W, et al. Tripterygium wilfordii: An inspiring resource for rheumatoid arthritis treatment. Med Res Rev. 2021;41(3):1337-1374.
[31] WANG YJ, YAN JG, ZHANG ZM, et al. Structure Characterization of Four New Sesquiterpene Pyridine Alkaloids from Tripterygium wilfordii Hook. f. and Anti-Inflammatory Activity Evaluations. Molecules. 2024;29(22):5284.
[32] GAO C, HUANG XX, BAI M, et al. Anti-inflammatory sesquiterpene pyridine alkaloids from Tripterygium wilfordii. Fitoterapia. 2015;105:49-54.
[33] LI D, JIA Q, ZHAO Q, et al. Macrolide sesquiterpene pyridine alkaloids from the roots of Tripterygium regelii and their anti-inflammatory activity. Bioorg Chem. 2025;158:108330.
[34] ZHANG Y, XU W, LI H, et al. Therapeutic effects of total alkaloids of Tripterygium wilfordii Hook f. on collagen-induced arthritis in rats. J Ethnopharmacol. 2013;145(3):699-705.
[35] GANG W, HAO H, YONG H, et al. Therapeutic Potential of Triptolide in Treating Bone-Related Disorders. Front Pharmacol. 2022;13:905576.
[36] 康艳慧,穆萍萍,张海雷.雷公藤甲素对类风湿关节炎成纤维样滑膜细胞线粒体自噬、NLRP3炎症小体活化和细胞焦亡的影响[J].现代药物与临床,2024,39(2):290-295.
[37] TOREQUL ISLAM M, QUISPE C, HERRERA-BRAVO J, et al. Activities and Molecular Mechanisms of Diterpenes, Diterpenoids, and Their Derivatives in Rheumatoid Arthritis. Evid Based Complement Alternat Med. 2022;2022:4787643.
[38] CUI J, LI X, WANG S, et al. Triptolide prevents bone loss via suppressing osteoclastogenesis through inhibiting PI3K-AKT-NFATc1 pathway. J Cell Mol Med. 2020;24(11):6149-6161.
[39] LIU C, ZHANG Y, KONG X, et al. Triptolide Prevents Bone Destruction in the Collagen-Induced Arthritis Model of Rheumatoid Arthritis by Targeting RANKL/RANK/OPG Signal Pathway. Evid Based Complement Alternat Med. 2013;2013:626038.
[40] BRINKER AM, MA J, LIPSKY PE, et al. Medicinal chemistry and pharmacology of genus Tripterygium (Celastraceae). Phytochemistry. 2007;68(6):732-766.
[41] 邹联银,廖群英.雷公藤多苷治疗类风湿关节炎对免疫球蛋白的影响[J].吉林医学,2023, 44(2):444-446.
[42] QIAN K, ZHANG L, SHI K. Triptolide prevents osteoarthritis via inhibiting hsa-miR-20b. Inflammopharmacology. 2019;27(1):109-119.
[43] PIAO X, ZHOU J, XUE L. Triptolide decreases rheumatoid arthritis fibroblast-like synoviocyte proliferation, invasion, inflammation and presents a therapeutic effect in collagen-induced arthritis rats via inactivating lncRNA RP11-83J16.1 mediated URI1 and β-catenin signaling. Int Immunopharmacol. 2021;99:108010.
[44] QIU HB, YANG Y, ZHOU SX. Effects of tripcholorolide on inflammatory reaction of mouse alveolar macrophages in vitro. Acta Pharmacol Sin. 2000;21(12):1197-1201.
[45] ZHU Y, ZHANG L, ZHANG X, et al. Tripterygium wilfordii glycosides ameliorates collagen-induced arthritis and aberrant lipid metabolism in rats. Front Pharmacol. 2022;13:938849.
[46] LIU P, LIU H, SANG Y, et al. Triptolide regulates neutrophil function through the Hippo signaling pathway to alleviate rheumatoid arthritis disease progression. J Transl Autoimmun. 2024;8:100242.
[47] 管咏梅,万志艳,王舒慧,等.基于NF-κB、Nrf2/HO-1信号通路及Bcl-2/Caspase-3凋亡蛋白表达探讨雷公藤-川芎组分配伍对类风湿关节炎成纤维样滑膜细胞的影响[J].中国实验方剂学杂志,2025,31(2):17-26.
[48] QIN A, CHENG TS, LIN Z, et al. Prevention of wear particle-induced osteolysis by a novel V-ATPase inhibitor saliphenylhalamide through inhibition of osteoclast bone resorption. PLoS One. 2012; 7(4):e34132.
[49] HU Y, XU T, YIN W, et al. Anti-inflammatory sesquiterpene polyol esters from the stem and branch of Tripterygium wilfordii. Chin J Nat Med. 2023;21(3):233-240.
[50] ZHU Y, WAN N, SHAN X, et al. Celastrol targets adenylyl cyclase-associated protein 1 to reduce macrophages-mediated inflammation and ameliorates high fat diet-induced metabolic syndrome in mice. Acta Pharm Sin B. 2021;11(5): 1200-1212.
[51] WANG B, SHEN J, WANG X, et al. Biomimetic nanoparticles for effective Celastrol delivery to targeted treatment of rheumatoid arthritis through the ROS-NF-κB inflammasome axis. Int Immunopharmacol. 2024;131:111822.
[52] HUANG CL, CHEN DY, TZANG CC, et al. Celastrol attenuates human parvovirus B19 NS1induced NLRP3 inflammasome activation in macrophages. Mol Med Rep. 2023;28(4):193.
[53] YANG J, HE B, DANG L, et al. Celastrol Regulates the Hsp90-NLRP3 Interaction to Alleviate Rheumatoid Arthritis. Inflammation. 2025;48(1):346-360.
[54] CAO Y, LIU J, HUANG C, et al. Wilforlide A ameliorates the progression of rheumatoid arthritis by inhibiting M1 macrophage polarization. J Pharmacol Sci. 2022;148(1):116-124.
[55] XUE M, JIANG ZZ, LIU JP, et al. Comparative study on the anti-inflammatory and immune suppressive effect of Wilforlide A. Fitoterapia. 2010;81(8):1109-112.
[56] QIU Q, FENG Q, TAN X, et al. JAK3-selective inhibitor peficitinib for the treatment of rheumatoid arthritis. Expert Rev Clin Pharmacol. 2019;12(6):547-554.
[57] XIN P, XU X, DENG C, et al. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int Immunopharmacol. 2020;80:106210.
[58] ZHENG Y, WEI K, JIANG P, et al. Macrophage polarization in rheumatoid arthritis: signaling pathways, metabolic reprogramming, and crosstalk with synovial fibroblasts. Front Immunol. 2024;15:1394108.
[59] EMORI T, KASAHARA M, SUGAHARA S, et al. Role of JAK-STAT signaling in the pathogenic behavior of fibroblast-like synoviocytes in rheumatoid arthritis: Effect of the novel JAK inhibitor peficitinib. Eur J Pharmacol. 2020;882:173238.
[60] HU L, LIU R, ZHANG L. Advance in bone destruction participated by JAK/STAT in rheumatoid arthritis and therapeutic effect of JAK/STAT inhibitors. Int Immunopharmacol. 2022;111:109095.
[61] HU Q, BIAN Q, RONG D, et al. JAK/STAT pathway: Extracellular signals, diseases, immunity, and therapeutic regimens. Front Bioeng Biotechnol. 2023;11:1110765.
[62] BALDINI C, MORICONI FR, GALIMBERTI S, et al. The JAK-STAT pathway: an emerging target for cardiovascular disease in rheumatoid arthritis and myeloproliferative neoplasms. Eur Heart J. 2021;42(42):4389-4400.
[63] LIN JJ, TAO K, GAO N, et al. Triptolide Inhibits Expression of Inflammatory Cytokines and Proliferation of Fibroblast-like Synoviocytes Induced by IL-6/sIL-6R-Mediated JAK2/STAT3 Signaling Pathway. Curr Med Sci. 2021;41(1):133-139.
[64] 王杰,刘健,文建庭,等.雷公藤甲素抑制类风湿关节炎患者的成纤维样滑膜细胞的炎症和迁移:基于circRNA 0003353/JAK2/STAT3信号通路[J].南方医科大学学报,2022,42(3):367-374.
[65] WANG Z, JIN H, XU R, et al. Triptolide downregulates Rac1 and the JAK/STAT3 pathway and inhibits colitis-related colon cancer progression. Exp Mol Med. 2009;41(10):717-727.
[66] GUO Q, JIN Y, CHEN X, et al. NF-κB in biology and targeted therapy: new insights and translational implications. Signal Transduct Target Ther. 2024; 9(1):53.
[67] SUN SC. The non-canonical NF-κB pathway in immunity and inflammation. Nat Rev Immunol. 2017;17(9):545-558.
[68] TANG Y, LIU Q, FENG Y, et al. Tripterygium Ingredients for Pathogenicity Cells in Rheumatoid Arthritis. Front Pharmacol. 2020;11:583171.
[69] ROMAN-BLAS JA, JIMENEZ SA. NF-kappaB as a potential therapeutic target in osteoarthritis and rheumatoid arthritis. Osteoarthritis Cartilage. 2006;14(9):839-848.
[70] ROBERTI A, CHAFFEY LE, GREAVES DR. NF-κB Signaling and Inflammation-Drug Repurposing to Treat Inflammatory Disorders? Biology (Basel). 2022;11(3):372.
[71] HUANG J, ZHOU L, WU H, et al. Triptolide inhibits osteoclast formation, bone resorption, RANKL-mediated NF-қB activation and titanium particle-induced osteolysis in a mouse model. Mol Cell Endocrinol. 2015;399:346-353.
[72] JING M, YANG J, ZHANG L, et al. Celastrol inhibits rheumatoid arthritis through the ROS-NF-κB-NLRP3 inflammasome axis. Int Immunopharmacol. 2021;98:107879.
[73] ZHANG C, WENG Y, WANG H, et al. A synergistic effect of triptolide and curcumin on rheumatoid arthritis by improving cell proliferation and inducing cell apoptosis via inhibition of the IL-17/NF-κB signaling pathway. Int Immunopharmacol. 2024;142(Pt A):112953.
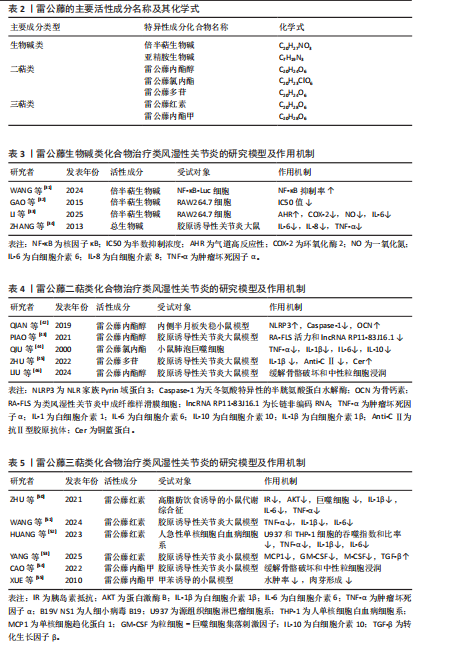
[74] ZHU Q, ZHOU H. The role of cGAS-STING signaling in rheumatoid arthritis: from pathogenesis to therapeutic targets. Front Immunol. 2024;15: 1466023.
[75] CHAUVIN SD, STINSON WA, PLATT DJ, et al. Regulation of cGAS and STING signaling during inflammation and infection. J Biol Chem. 2023; 299(7):104866.
[76] YANG X, ZHAO L, PANG Y. cGAS-STING pathway in pathogenesis and treatment of osteoarthritis and rheumatoid arthritis. Front Immunol. 2024; 15:1384372.
[77] KALINKOVICH A, LIVSHITS G. The cross-talk between the cGAS-STING signaling pathway and chronic inflammation in the development of musculoskeletal disorders. Ageing Res Rev. 2025;104:102602.
[78] 许阿兰,龙瑛婕,王祥,等.雷公藤甲素对类风湿性关节炎患者滑膜成纤维细胞cGAS-STING信号通路的影响[J].中华中医药杂志, 2022,37(2):1087-1090.
[79] LU J, ZHANG Y, DONG H, et al. New mechanism of nephrotoxicity of triptolide: Oxidative stress promotes cGAS-STING signaling pathway. Free Radic Biol Med. 2022;188:26-34.
[80] YU L, WEI J, LIU P. Attacking the PI3K/Akt/mTOR signaling pathway for targeted therapeutic treatment in human cancer. Semin Cancer Biol. 2022;85:69-94.
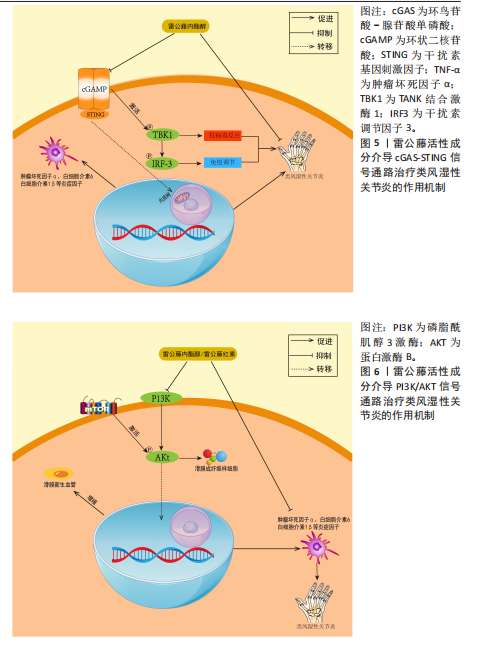
[81] PASKEH MDA, GHADYANI F, HASHEMI M, et al. Biological impact and therapeutic perspective of targeting PI3K/Akt signaling in hepatocellular carcinoma: Promises and Challenges. Pharmacol Res. 2023;187:106553.
[82] LIU S, MA H, ZHANG H, et al. Recent advances on signaling pathways and their inhibitors in rheumatoid arthritis. Clin Immunol. 2021;230: 108793.
[83] 刘静,燕丽君.雷公藤内酯醇对类风湿性关节炎模型大鼠血管新生和PTEN/PI3K/AKT通路的影响[J].吉林大学学报(医学版),2020, 46(6):1227-1233+1351.
[84] YANG J, LIU J, LI J, et al. Celastrol inhibits rheumatoid arthritis by inducing autophagy via inhibition of the PI3K/AKT/mTOR signaling pathway. Int Immunopharmacol. 2022;112: 109241.
[85] LI Y, QI W, YAN L, et al. Tripterygium wilfordii derivative LLDT-8 targets CD2 in the treatment of rheumatoid arthritis. Biomed Rep. 2021;15(4):81.
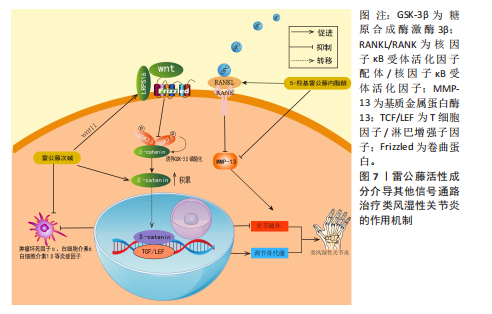
[86] ZENG JZ, MA LF, MENG H, et al. (5R)-5-hydroxytriptolide (LLDT-8) prevents collagen-induced arthritis through OPG/RANK/RANKL signaling in a rat model of rheumatoid arthritis. Exp Ther Med. 2016;12(5):3101-3106.
[87] HUANG Y, PENG Y, LI H, et al. Wilforine inhibits rheumatoid arthritis pathology through the Wnt11/β-catenin signaling pathway axis. Arthritis Res Ther. 2023;25(1):243.
|