Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (9): 1461-1467.doi: 10.3969/j.issn.2095-4344.1098
Previous Articles Next Articles
Effects of autophagy on tendinopathy
Zhu Qianzheng1, Gao Fuqiang2, Lin Peng1, Sun Wei2
- 1Third Department of Orthopedics, 2First Department of Orthopedics, China-Japan Friendship Hospital, Beijing 100029, China
-
Revised:2018-10-07Online:2019-03-28Published:2019-03-28 -
Contact:Sun Wei, MD, Professor, First Department of Orthopedics, China-Japan Friendship Hospital, Beijing 100029, China -
About author:Zhu Qianzheng, MD, Third Department of Orthopedics, China-Japan Friendship Hospital, Beijing 100029, China -
Supported by:the National Natural Science Foundation of China (General Program), No. 81672236 and 81372013 (to SW); the Natural Science Foundation of Beijing (General Program), No. 7182146 (to SW); the Beijing University of Chemical Technology & China-Japan Friendship Hospital Joint Foundation, No. PYBZ1828 (to SW)
CLC Number:
Cite this article
Zhu Qianzheng, Gao Fuqiang, Lin Peng, Sun Wei. Effects of autophagy on tendinopathy[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(9): 1461-1467.
share this article
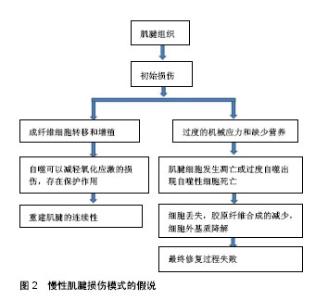
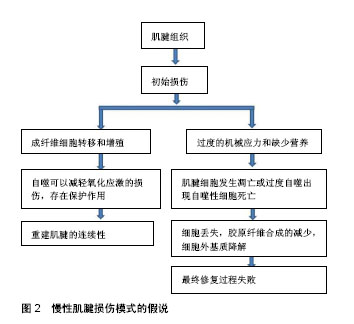
2.1 自噬的发生 自噬是真核生物在进化中高度保守的细胞代谢活动,是一种自我分解的代谢现象,细胞内生物大分子和细胞器得以降解回收,细胞的稳态和存活得以维持[2]。自噬分为3种:大、小自噬和分子伴侣介导自噬[2-3]。大自噬分为4个阶段:首先形成自噬前体,进一步自噬前体延长并包裹自噬底物,进而形成自噬泡,最终自噬泡与溶酶体融合底物降解完成,可将其概括为起始、延长、成熟、融合4个阶段[4]。 自噬启动的关键蛋白激酶是ATG1/ULK1 (AuTophaGy related,自噬相关基因,Unc-51 like kinase-1),属于丝氨酸/苏氨酸类蛋白激酶,在哺乳动物的同源蛋白复合物有ULK1(同酵母中Atg1),FIP200(同酵母中Atg17)和mAtg13[8]。自噬发生初始阶段,主要是自噬诱导和自噬膜的形成,自噬膜的形成首先需要自噬前体的形成,自噬前体是自噬调控的重要节点,Beclin1-Vps34复合体是哺乳动物自噬前提形成的核心复合物。Atg14参与自噬泡的形成[9],紫外线抗性相关基因作用于自噬泡成熟及其运输过程[10],Rubicon负调节其功能[11]。自噬被诱导后,在Atg14-Vps15- mVps34复合物作用下膜泡的成核反应启动,进一步结合Atg21和Atg24后,前自噬体形成[12]。自噬膜泡进一步扩张包绕底物,自噬体最终形成。两种种泛素化系统参与自噬体的形成,即Atg12-Atg5复合物系统和LC3-Ⅱ(Light chain,轻链蛋白,Atg8同系物)-磷脂酰乙醇胺(phosphatidylethanolamine,PE) 复合物系统。 细胞内的微管骨架将自噬体运输到溶酶体,二者融合后水解酶降解自噬体内容物,Rab7,溶酶体膜蛋白2(Lysosomal membrane protein 2,Lamp2)?紫外线放射抗性相关蛋白等因子参与此过程。Rab7与膜泡表面的脂分子尾部作用进行定位,紫外线放射抗性相关蛋白能活化Rab7将囊泡运送到靶位点[13]。 2.2 自噬的调控机制 诱发自噬的因素包括能量缺乏、氧化应激、运动刺激或细胞器和蛋白质累积。自噬诱导的经典途径是哺乳动物雷帕霉素靶蛋白(mammalian target of rapamycin,mTOR)与ULK1复合物作用。细胞的能量充足时激活mTOR,使mAtg13和ULK1高度磷酸化,显著降低ULK1的活性,细胞只保持基础的自噬水平。细胞营养缺乏时mTOR被抑制,ULK1通过磷酸化并激活mAtg13,FIP200和其自身[8],自噬开始发生,与此同时AMP活化蛋白激酶(AMP-activated protein kinase,AMPK)也可以激活ULK1进一步促进自噬[14]。雷帕霉素的发现已经有30多年,已被证明其抑制mTOR的激活,并诱导自噬。 Beclin1是一种哺乳动物特异性的自噬基因,Vps34/PI3K-Beclin1复合物促进磷脂酰肌醇-3-磷酸(PI3P)产生,含有PX和FYVE结构域的蛋白质在PI3P的组装下到自噬泡产生的早期位置,促使自噬前体的形成,如DFCP1(double FYVE-containing protein 1)和WIPI家族蛋白(WD-repeat domain protein interacting with phosphoinositides)[15]。Bcl-2是一种凋亡抑制蛋白可以抑制Beclin1诱导的细胞自噬。P53对细胞自噬具有双重调节作用,在细胞核中上调细胞自噬;但在细胞质中抑制细胞自噬的发生。 近年来,研究者报道了细胞自噬与细胞周期的关系,微管相关蛋白1A/1B轻链3B(LC3B)和细胞周期双免疫荧光染色显示,细胞周期G1期和S期优先诱导自噬,在G2期和M期自噬被抑制[16]。饥饿过程中,G1/G0静止状态下有丝分裂的出口和停滞需要自噬参与[17]。类似地,ATG5过表达通过抑制G2/M细胞周期阻滞来抑制胶原合成[18]。 2.3 自噬与细胞死亡 自噬可以作为细胞的生存机制,抑制坏死和PARP来调节的细胞死亡[19]。相反,自噬也可能作为细胞的死亡机制,可能有2个独立的功能——自噬的促凋亡功能和引起自噬性细胞死亡[20]。自噬囊泡可出现在在发生程序性坏死的细胞内,程序性坏死信号能活化自噬,提示自噬可能参与程序性坏死的执行,二者形成正反馈环促进细胞死亡[21-23]。自噬性细胞死亡并不意味着细胞死亡是由自噬作用的,但简单地解释细胞死亡过程中伴随自噬。药理和生化研究提示,自噬介导的细胞生存或死亡,可以通过调节通路的类型和程度进行区分。 许多研究表明,持续自噬可能导致自噬型Ⅱ型程序性细胞死亡[24-25]。自噬性细胞死亡是形态学上定义的一种细胞死亡,它发生在没有染色质缩合的情况下,但伴随着细胞质的大规模自噬空泡化[26]。在哺乳动物中,没有明确的证据表明在生理环境下存在这种自噬性细胞死亡。然而,许多报道显示自噬的死亡功能和细胞以“自噬性细胞死亡”的形式死亡的情况大多来自体外细胞培养模型[20]。在人类癌症细胞中,有一种报道显示了一种新的抗癌作用,即自噬性细胞死亡对化疗的反应[27]。这一发现很重要,因为它为癌症的治疗目的提供了诱导自噬细胞死亡的可能探索的证据[25]。 2.4 慢性肌腱损伤模式的假说 初始损伤发生后,成纤维细胞由周围组织或腱鞘和腱内膜进入损伤部位[28],肌腱细胞也分化为成肌纤维细胞,试图重建肌腱的连续性。持续的负面因素,如过度的机械应力和缺少营养,肌腱细胞发生凋亡或自噬[29]。细胞凋亡和自噬的作用最终超过早期的细胞转移和增殖,导致细胞缺失和减少。细胞丢失进一步导致胶原纤维合成的减少,细胞外基质降解最终修复过程失败[30-31]。慢性肌腱损伤模式的假说,见图2。"

2.5 自噬参与骨科疾病的病理过程 自噬可以维持软骨细胞的内稳态。在老年和骨关节炎患者中可以观察到自噬表达的减少,同时伴随细胞凋亡的增加。表层的软骨细胞高水平表达自噬蛋白,如Beclin-1,ATG5和MAP1LC3。在轻度骨关节炎的细胞中,ULK1,Beclin-1和LC3的表达水平下降。伴随组织衰老,自噬的水平减低,细胞器损伤逐渐积累,自噬的抑制对于软骨细胞的存活和分化有负面影响[32]。骨关节炎的发生中,退变组织的溶酶体水解障碍,使自噬能力减退,导致细胞质内失活大分子蛋白增多及细胞自噬活性减低。软骨细胞数量减少、细胞因子和生长因子免疫应答及蛋白和基因表达障碍、抗氧化能力下降等都可使软骨退变,进而发展成为骨关节炎[33]。 对于椎间盘退变,自噬体在不同月龄SD大鼠椎间盘的髓核细胞中均有表达,同时自噬体数量随年龄呈增加趋势;LC3-Ⅱ/LC3-Ⅰ比值也随月龄增加而上升,提示大鼠的髓核细胞自噬水平随着年龄的增加而上升[34]。人体颈椎退变的终板细胞中,细胞自噬水平表达更低[35]。而椎间盘退变患者的纤维环细胞中自噬水平表达更高[36]。 骨质疏松症是以骨量减少、骨组织微结构破坏、骨脆性增加和易于骨折为特征的代谢性骨病,其特征是骨量和骨质量降低。大鼠ATG7基因敲除后,骨体积和骨皮质厚度明显降低,同时骨密度下降。骨细胞内自噬调控机制伴随年龄增加发生老化。细胞自噬同骨质疏松症关系密切[37]。 在神经系统,某些神经退行性疾病的发生与溶酶体的降解能力出现障碍所导致的神经元死亡有关[38]。细胞内蛋白质积聚和主要蛋白质降解系统的改变,是一些神经退行性疾病受累神经元的共同特点。在阿尔茨海默病和帕金森病的实验动物模型中观察到了自噬的信号转导发生了改变。 由此可见,自噬广泛参与包括骨性关节炎、神经退行性疾病和椎间盘退变在内的许多疾病的发展。 2.6 自噬与肌腱病的关系 2.6.1 自噬可以减轻氧化应激对人肌腱干细胞的损害 肌腱承受超负荷应力作用,会导致肌腱细胞应激增加,如氧化应激和内质网应激[39-40]。学者在体外培养人体肌腱干细胞,使用H2O2建立氧化应激模型[41],体外实验结果显示H2O2抑制人体肌腱干细胞细胞集落和细胞增殖,干细胞标记物的表达被抑制,干细胞向脂肪细胞、软骨细胞和骨细胞的分化能力也被抑制。使用雷帕霉素或细胞饥饿预处理后再加入H2O2,可以观察到这两种预处理可以保护干细胞,LC3-Ⅱ/LC3-Ⅰ和Beclin-1代表的细胞自噬水平升高,GFP-LC3标记的自噬体数量增加,同时前述的干细胞分化和增殖能力比单纯使用H2O2的显著提高。细胞内活性氧水平在H2O2处理后显著增加,活性氧水平可以被抗氧化剂NAC减轻,同时雷帕霉素和饥饿预处理也可以降低细胞内活性氧水平,提示自噬对于氧化应激有保护保护作用。反之,使用化学干预抑制自噬和基因技术使Atg-7,Beclin-1基因静默后,细胞自噬水平降低,前述各项细胞增殖分化和干细胞自我更新能力均被抑制。细胞自噬可以保护氧化应激对人体肌腱干细胞的损伤,降低细胞内活性氧水平。 2.6.2 细胞外基质降解与自噬程序性细胞死亡 肌腱组织的细胞外基质主要由Ⅰ型胶原纤维组成,提供肌腱的结构完整性和机械强度。损伤的胶原基质特征表现为正常的纵向纤维排列消失,胶原束断裂为碎片,最终导致肌腱断裂,肌腱细胞死亡在肌腱病中扮演主要角色[42]。文献证实人类退变肌腱内观察到细胞凋亡水平增高[43],伴随肌腱细胞成分化为成肌纤维细胞,这不会发生在正常肌腱组织内。 学者使用慢性肩袖损伤手术患者的肌腱组织,从破损的肌腱边缘取材。使用免疫组化技术,抗泛素抗体可以检测出自噬性细胞死亡[44];使用TUNEL试验检测细胞凋亡。研究结果显示,随着细胞外基质降解程度的增加,超过一半的肌腱细胞由于过度的细胞自噬出现自噬性细胞死亡,同样细胞凋亡随着细胞外基质降解分级的提高而增加,即便在细胞外基质0级的肌腱中,细胞凋亡水平也高于正常肌腱。细胞凋亡的增加导致细胞数量的减少,进一步影响胶原的合成以及组织的愈合。此外,肌腱细胞已分化为成肌纤维细胞,也与细胞外基质降解分级有关[45]。可能的原因是正常胶原纤维对肌腱细胞的应力遮挡随着肌腱损伤程度的增加而减弱,机械应力的刺激作用在肌腱细胞上引起细胞异常分化。EDM降解0级至1级,肌腱细胞密度增加,2,3级的细胞密度持续减少。细胞增生被认为是对早期损伤的修复反应,然而到肌腱疾病晚期,自噬性细胞死亡、细胞凋亡和成肌纤维异常分化会导致肌腱细胞的显著减少。 在慢性肱骨外上髁炎的患者中也观察到相似的结果,桡侧腕短伸肌肌腱的细胞凋亡和自噬性细胞死亡在退变程度高的肌腱内明显增加[46]。 2.6.3 前列腺素E2与自噬性细胞死亡 除了其作为促进细胞稳态和存活的代谢循环系统的功能外,自噬调节诸如程序性细胞死亡、炎症和适应性免疫机制等重要过程[47]。自噬性细胞死亡与肌腱病的病理生理学密切相关。前列腺素E2是导致肌腱炎的炎症递质,在体外可抑制成纤维细胞的增殖[48]。学者在体外培养人体髌腱成纤维细胞,给予外源性前列腺素E2或循环重复机械牵张刺激[49],外源性前列腺素E2以剂量依赖的方式显著诱导人体髌腱成纤维细胞细胞死亡和自噬。使用抑制剂3-甲基腺嘌呤和氯喹阻断自噬,或小干扰RNA阻断细胞自噬基因Becn-1和Atg-5可阻止前列腺素E2诱导的细胞死亡。研究表明24 h的周期性机械拉伸在8%和12%强度下显著刺激人体髌腱成纤维细胞释放前列腺素E2,呈强度依赖性。此外,机械牵张可导致自噬和细胞死亡。用环氧化酶抑制剂吲哚美辛和塞来昔布阻断前列腺素E2的产生,可显著减少牵张诱导的自噬和细胞死亡。说明周期性机械拉伸通过前列腺素E2产生活化来诱导肌腱成纤维细胞的自噬性细胞死亡[49]。 2.6.4 肌腱损伤后肌肉脂肪变性和自噬 肩袖损伤是引起肩关节慢性疼痛和功能受限的常见疾病,慢性肩袖损伤常见的病理改变是肌肉萎缩和脂肪变性[50]。巨大肩袖撕裂后肌肉回缩,肩胛上神经在肩胛切迹内卡压,神经卡压会进一步加重肩袖肌肉萎缩和退变[51]。大鼠肩袖切断或肩胛上神经切断后2周,肩袖肌肉肌肉萎缩,肌肉蛋白降解。肩袖切断组的肌肉萎缩受自噬调控,肩袖切断组内观察到LC3BⅡ显著上调,LC3BⅡ/LC3BⅠ比例显著升高,mRNA水平中LC3B和ATG12表达水平明显升高;去神经组的肌肉萎缩主要受泛素蛋白酶路径调控,其20 S蛋白酶活性显著升高,而mTOR信号通路上调抑制ULK1/Atg13/FIP200复合体的形成[52]。 2.6.5 雷帕霉素通过自噬的激活抑制腱周纤维化 腱鞘纤维化是骨科领域的一个重要问题,其结果是肌腱滑动功能障碍和日常生活中的局限性。不同组织中的纤维形成具有相似的病理特征,包括炎症反应、肌成纤维细胞活化和随后的过度细胞外基质沉积,以及功能性组织变性[53]。腱周纤维化是一种独特的病理组织纤维化,主要发生在肌腱损伤、肌腱手术和关节固定后。研究表明,腱鞘粘连形成是一种异常的愈合过程,其特征在于细胞增殖的激活和过度的细胞外基质沉积。肌纤维母细胞是纤维变性疾病的主要影响因素,有助于细胞外基质沉积、组织挛缩和功能受损。 在大鼠肌腱损伤模型中,有学者观察到过度的细胞外基质沉积过程中的自噬失调[54]。雷帕霉素对自噬的诱导作用明显减轻了体内周围纤维化的严重程度。在NIH/3T3成纤维细胞和腱细胞中,转化生长因子β1显著激活肌成纤维细胞,胶原合成增加。增加雷帕霉素激活的自噬,减少胶原合成,抑制肌成纤维细胞活化。体外实验还表明,雷帕霉素降低细胞增殖,增加细胞数量停滞在G0/G1期。然而,在用自噬抑制剂3-甲基腺嘌呤进行预处理后,雷帕霉素不能抑制转化生长因子β1诱导的纤维化改变。在成纤维细胞中干扰自噬相关蛋白5 RNA也消除了雷帕霉素在体外的保护作用。总之,自噬可能作为预防肌腱损伤后腱周纤维化的潜在治疗策略。细胞自噬与肌腱病的相关研究见表1。"

| [1] Nourissat G, Berenbaum F, Duprez D. Tendon injury: from biology to tendon repair. Nat Rev Rheumatol. 2015;11(4): 223-233. [2] Takeshige K, Baba M, Tsuboi S, et al. Autophagy in yeast demonstrated with proteinase-deficient mutants and conditions for its induction. J Cell Biol. 1992;119(2):301-311. [3] Kim KH, Lee MS. Autophagy-a key player in cellular and body metabolism. Nat Rev Endocrinol. 2014;10(6):322-337. [4] Feng Y, He D, Yao Z, et al. The machinery of macroautophagy. Cell Res. 2014;24(1):24-41. [5] Duarte JH. Osteoarthritis: autophagy prevents age-related OA. Nat Rev Rheumatol. 2015;11(12):683. [6] Genevini P, Papiani G, Ruggiano A, et al. Amyotrophic lateral sclerosis-linked mutant VAPB inclusions do not interfere with protein degradation pathways or intracellular transport in a cultured cell model. PLoS One. 2014;9(11): e113416. [7] Kadow T, Sowa G, Vo N, et al. Molecular basis of intervertebral disc degeneration and herniations: what are the important translational questions? Clin Orthop Relat Res. 2015;473(6):1903-1912. [8] Hosokawa N, Hara T, Kaizuka T, et al. Nutrient-dependent mTORC1 association with the ULK1-Atg13-FIP200 complex required for autophagy. Mol Biol Cell. 2009;20(7): 1981-1991. [9] Itakura E, Kishi C, Inoue K, et al. Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol Biol Cell. 2008;19(12):5360-5372. [10] Liang C, Lee J S, Inn K S, et al. Beclin1-binding UVRAG targets the class C Vps complex to coordinate autophagosome maturation and endocytic trafficking. Nat Cell Biol. 2008;10(7):776-787. [11] Matsunaga K, Saitoh T, Tabata K, et al. Two Beclin 1-binding proteins, Atg14L and Rubicon, reciprocally regulate autophagy at different stages. Nat Cell Biol. 2009;11(4): 385-396. [12] He C, Klionsky DJ. Regulation mechanisms and signaling pathways of autophagy. Annu Rev Genet. 2009;43:67-93. [13] Luo P, Gao F, Han J, et al. The role of autophagy in steroid necrosis of the femoral head: a comprehensive research review. Int Orthop. 2018;42(7):1747-1753. [14] Kim J, Kundu M, Viollet B, et al. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol. 2011;13(2):132-141. [15] Simonsen A, Tooze S A. Coordination of membrane events during autophagy by multiple class III PI3-kinase complexes. J Cell Biol. 2009;186(6):773-782. [16] Tasdemir E, Maiuri M C, Tajeddine N, et al. Cell cycle-dependent induction of autophagy, mitophagy and reticulophagy. Cell Cycle. 2007;6(18):2263-2267. [17] An Z, Tassa A, Thomas C, et al. Autophagy is required for G(1)/G(0) quiescence in response to nitrogen starvation in Saccharomyces cerevisiae. Autophagy. 2014;10(10): 1702-1711. [18] Li H, Peng X, Wang Y, et al. Atg5-mediated autophagy deficiency in proximal tubules promotes cell cycle G2/M arrest and renal fibrosis. Autophagy. 2016;12(9):1472-1486. [19] Martinet W, Agostinis P, Vanhoecke B, et al. Autophagy in disease: a double-edged sword with therapeutic potential. Clin Sci (Lond). 2009;116(9):697-712. [20] Shen HM, Codogno P. Autophagic cell death: loch ness monster or endangered species? Autophagy. 2011;7(5): 457-465. [21] Ch'En IL, Beisner DR, Degterev A, et al. Antigen-mediated T cell expansion regulated by parallel pathways of death. Proc Natl Acad Sci U S A. 2008;105(45):17463-17468. [22] Bell BD, Leverrier S, Weist BM, et al. FADD and caspase-8 control the outcome of autophagic signaling in proliferating T cells. Proc Natl Acad Sci U S A. 2008;105(43): 16677-16682. [23] Jain MV, Paczulla AM, Klonisch T, et al. Interconnections between apoptotic, autophagic and necrotic pathways: implications for cancer therapy development. J Cell Mol Med. 2013;17 (1):12-29. [24] Yu L, Alva A, Su H, et al. Regulation of an ATG7-beclin 1 program of autophagic cell death by caspase-8. Science. 2004;304(5676):1500-1502. [25] Gozuacik D, Kimchi A. Autophagy as a cell death and tumor suppressor mechanism. Oncogene. 2004, 23(16): 2891-2906. [26] Kroemer G, Levine B. Autophagic cell death: the story of a misnomer. Nat Rev Mol Cell Biol. 2008;9(12):1004-1010. [27] Xiong HY, Guo XL, Bu XX, et al. Autophagic cell death induced by 5-FU in Bax or PUMA deficient human colon cancer cell. Cancer Lett. 2010;288(1):68-74. [28] Russell J E, Manske PR. Collagen synthesis during primate flexor tendon repair in vitro. J Orthop Res. 1990;8 (1):13-20. [29] Ferraro E, Cecconi F. Autophagic and apoptotic response to stress signals in mammalian cells. Arch Biochem Biophys. 2007;462(2):210-219. [30] Wu B, Chen J, Dela R T, et al. Cellular response and extracellular matrix breakdown in rotator cuff tendon rupture. Arch Orthop Trauma Surg. 2011;131(3):405-411. [31] 桂金鹏,江静怡,张灵玉,等.力学因素对细胞自噬的影响[J].中国组织工程研究,2017,21(12):1946-1952.[32] Jeon H, Im GI. Autophagy in osteoarthritis. Connect Tissue Res. 2017;58(6):497-508. [33] 刘利国,徐超,伊力哈木•托合提.自噬基因在骨关节炎软骨细胞凋亡过程中的保护和平衡效应[J].中国组织工程研究, 2015, 19(20):3231-3235.[34] Ye W, Xu K, Huang D, et al. Age-related increases of macroautophagy and chaperone-mediated autophagy in rat nucleus pulposus. Connect Tissue Res. 2011;52(6):472-478. [35] Xu H, Xiong S, Wang H, et al. The evidence and the possible significance of autophagy in degeneration model of human cervical end-plate cartilage. Exp Ther Med. 2014;7(3): 537-542. [36] Gruber HE, Hoelscher GL, Ingram JA, et al. Autophagy in the degenerating human intervertebral disc: in vivo molecular and morphological evidence, and induction of autophagy in cultured annulus cells exposed to proinflammatory cytokines-implications for disc degeneration. Spine (Phila Pa 1976). 2015;40(11):773-782. [37] Onal M, Piemontese M, Xiong J, et al. Suppression of autophagy in osteocytes mimics skeletal aging. J Biol Chem. 2013;288(24):17432-17440. [38] Komatsu M, Waguri S, Chiba T, et al. Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature. 2006;441(7095):880-884. [39] Archambault JM, Wiley JP, Bray RC. Exercise loading of tendons and the development of overuse injuries. A review of current literature. Sports Med. 1995;20(2):77-89. [40] Yuan J, Murrell G A, Trickett A, et al. Involvement of cytochrome c release and caspase-3 activation in the oxidative stress-induced apoptosis in human tendon fibroblasts. Biochim Biophys Acta. 2003;1641(1):35-41. [41] Chen H, Ge HA, Wu GB, et al. Autophagy prevents oxidative stress-induced loss of self-renewal capacity and stemness in human tendon stem cells by reducing ROS accumulation. Cell Physiol Biochem. 2016;39(6):2227-2238.[42] Osti L, Buda M, Del BA, et al. Apoptosis and rotator cuff tears: scientific evidence from basic science to clinical findings. Br Med Bull. 2017;122(1):123-133. [43] Tuoheti Y, Itoi E, Pradhan R L, et al. Apoptosis in the supraspinatus tendon with stage II subacromial impingement. J Shoulder Elbow Surg. 2005;14(5):535-541. [44] Somers P, Knaapen M, Kockx M, et al. Histological evaluation of autophagic cell death in calcified aortic valve stenosis. J Heart Valve Dis. 2006;15(1):43-48. [45] Premdas J, Tang JB, Warner JP, et al. The presence of smooth muscle actin in fibroblasts in the torn human rotator cuff. J Orthop Res. 2001;19(2):221-228. [46] Chen J, Wang A, Xu J, et al. In chronic lateral epicondylitis, apoptosis and autophagic cell death occur in the extensor carpi radialis brevis tendon. J Shoulder Elbow Surg. 2010; 19(3):355-362. [47] Meijer AJ, Codogno P. Signalling and autophagy regulation in health, aging and disease. Mol Aspects Med. 2006;27(5-6): 411-425. [48] Khan MH, Li Z, Wang JH. Repeated exposure of tendon to prostaglandin-E2 leads to localized tendon degeneration. Clin J Sport Med. 2005;15(1):27-33. [49] Chen H, Chen L, Cheng B, et al. Cyclic mechanical stretching induces autophagic cell death in tenofibroblasts through activation of prostaglandin E2 production. Cell Physiol Biochem. 2015;36(1):24-33. [50] Yamaguchi K, Tetro AM, Blam O, et al. Natural history of asymptomatic rotator cuff tears: a longitudinal analysis of asymptomatic tears detected sonographically. J Shoulder Elbow Surg. 2001;10(3):199-203. [51] Albritton MJ, Graham RD, Richards RN, et al. An anatomic study of the effects on the suprascapular nerve due to retraction of the supraspinatus muscle after a rotator cuff tear. J Shoulder Elbow Surg. 2003;12(5):497-500. [52] Joshi SK, Kim HT, Feeley BT, et al. Differential ubiquitin-proteasome and autophagy signaling following rotator cuff tears and suprascapular nerve injury. J Orthop Res. 2014;32(1):138-144. [53] Wynn TA, Ramalingam TR. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 2012;18 (7):1028-1040. [54] Zheng W, Qian Y, Chen S, et al. Rapamycin protects against peritendinous fibrosis through activation of autophagy. Front Pharmacol. 2018;9:402. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [5] | Xie Yang, Zhang Shujiang, Liu Menglan, Luo Ying, Yang Yang, Li Zuoxiao. Mechanism by which rapamycin protects spinal cord neurons in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 695-700. |
| [6] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [7] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [8] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [9] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [10] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [11] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [12] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [13] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [14] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [15] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||