Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (29): 4675-4682.doi: 10.3969/j.issn.2095-4344.2014.29.015
Previous Articles Next Articles
Construction and identification of pIRES2-GDNF-VEGF165 bicistronic eukaryotic expression vector
Li Bing-nan, Li Wei-dong, Lin Jun-tang, Feng Hui-gen
- Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China
-
Revised:2014-05-01Online:2014-07-09Published:2014-07-09 -
Contact:Li Bing-nan, Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China -
About author:Li Bing-nan, Ph.D., Lecturer, Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China Li Wei-dong, Department of Life Sciences and Technology, Xinxiang Medical University, Xinxiang 453003, Henan Province, China Li Bing-nan and Li Wei-dong contributed equally to this work. -
Supported by:the Tender Subject of Key Research Areas of Xinxiang Medical University in 2011, No. ZD2011-16; Key Projects in Scientific Research of Henan Provincial Education Department, No. 13A180850.
CLC Number:
Cite this article
Li Bing-nan, Li Wei-dong, Lin Jun-tang, Feng Hui-gen. Construction and identification of pIRES2-GDNF-VEGF165 bicistronic eukaryotic expression vector[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(29): 4675-4682.
share this article
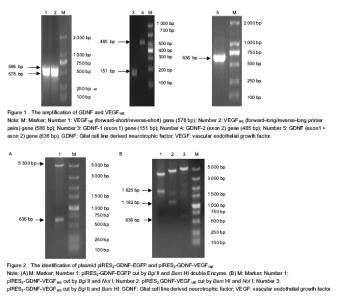
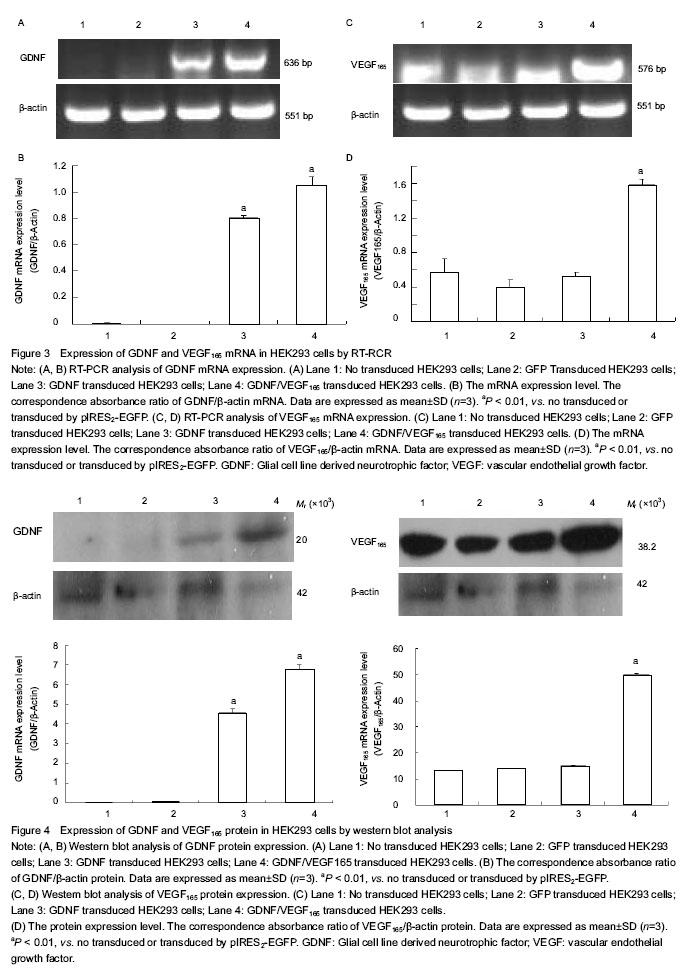
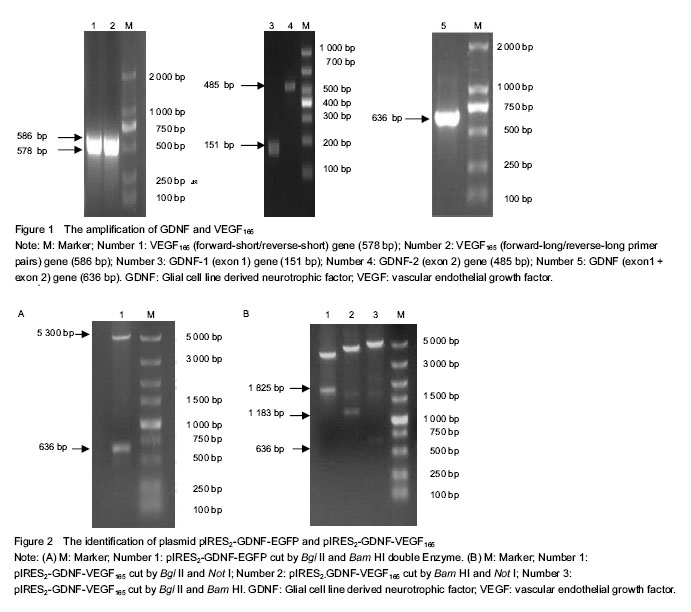
Amplification of GDNF and VEGF165 genes GDNF gene was obtained from the genomic DNA of human peripheral blood mononuclear cells by overlap PCR. The size of GDNF gene was 636 bp. The VEGF165 gene was obtained from pIRES2-VEGF165-EGFP plasmid by twin PCR, and the size of VEGF165 gene was 576 bp (Figure 1). Identification of plasmid pIRES2-GDNF-EGFP The plasmid pIRES2-GDNF-EGFP was cut by Bgl II and Bam HI double enzyme. A gene fragment with 636 bp was obtained, which was in full agreed with GDNF gene (Figure 2A). Identification of plasmid pIRES2-GDNF-VEGF165 The plasmid pIRES2-GDNF-VEGF165 was cut by Bgl II and Bam HI double enzyme. And a fragment about 636 bp would be obtained. This indicated GDNF gene inserted into the plasmid pIRES2-GDNF-VEGF165. The sequence of the plasmid pIRES2-GDNF-VEGF165 was in accordance with gene sequence in Gene Bank. The plasmid pIRES2-GDNF-VEGF165 was cut by Bam HI and Not I double enzyme. And a fragment about 1 183 bp would be obtained. This indicated IRES-VEGF165 gene inserted into the plasmid pIRES2-GDNF-VEGF165. The plasmid pIRES2-GDNF- VEGF165 was cut by Bgl II and Not I double enzyme. And a fragment about 1 825 bp would be obtained. This indicated BDNF-IRES-VEGF165 gene inserted into the plasmid pIRES2-GDNF-VEGF165. The sequence of the plasmid pIRES2-BDNF-VEGF165 was in accordance with gene sequence in Gene Bank (Figure 2B). RT-PCR analysis of the expression of GDNF and VEGF165 in HEK293 cells To illustrate mRNA expression by pIRES2-EGFP, pIRES2-GDNF/EGFP, and pIRES2-GDNF/VEGF165 transduced HEK293 cells, we evaluated the expression of GDNF and VEGF165 by RT-PCR analysis. RT-PCR was performed using GDNF-specific primers and the β-actin sequence as an internal standard. GFP expression was monitored in pIRES2-EGFP and pIRES2-GDNF/EGFP transduced HEK293 cells by inverted fluorescence microscopy. Expression of GDNF mRNA was higher in either pIRES2-GDNF/EGFP or pIRES2-GDNF/VEGF165 transduced HEK293 cells than that pIRES2-EGFP transduced HEK293 cells or negative control (Figure 3A, B). As shown above, expression of VEGF165 mRNA was higher in pIRES2-GDNF/VEGF165-transduced HEK293 cells than the other three (Figure 3C, D). These results demonstrated that the GDNF and VEGF165 had been introduced successfully into HEK293 cells by pIRES2-GDNF/EGFP and pIRES2-GDNF/VEGF165. After the HEK293 cells were transduced by pIRES2-GDNF/EGFP, we passaged them continually and then monitored the mean percentage of expression of GFP under fluorescence microscopy. There was no decrease in GFP fluorescence, illustrating the maintenance of transgenic expression in the transduced cells. Western blot analysis of the expression of GDNF and VEGF165 in HEK293 cells To illustrate protein expression by pIRES2-EGFP, pIRES2-GDNF/EGFP, and pIRES2-GDNF/VEGF165 transduced HEK293 cells, we evaluated the expression of GDNF and VEGF165 by western blot assay. After 72 hours of transfection, we collected the culture supernatants of infected HEK293 cells without serums which were processed for western blot analysis using an anti-GDNF and anti-VEGF165 antibody. β-actin served as an internal standard. The results suggested that exogenous GDNF protein was strongly expressed in pIRES2-GDNF/EGFP and pIRES2-GDNF/VEGF165 transduced HEK293 cells, but the pIRES2-EGFP transduced HEK293 cells and no transduced cells, the expression of endogenous GDNF was very low (Figures 4A, B). As shown above, western blot analysis also revealed that exogenous VEGF165 protein was strongly expressed in pIRES2-GDNF/VEGF165 transduced HEK293 cells, and expression of endogenous VEGF165 was very low in the pIRES2-EGFP, pIRES2-GDNF/EGFP transduced HEK293 cells (Figures 4C, D). These results also demonstrated that GDNF and VEGF165 had been introduced successfully into HEK293 cells by pIRES2-GDNF/VEGF165. "
| [1] Zurita M, Aguayo C, Bonilla C, et al. The pig model of chronic paraplegia: a challenge for experimental studies in spinal cord injury. Prog Neurobiol. 2012;97(3):288-303. [2] Shah AM, Mann DL. In search of new therapeutic targets and strategies for heart failure: recent advances in basic science. Lancet. 2011;378(9792):704-712. [3] Potente M, Gerhardt H, Carmeliet P. Basic and therapeutic aspects of angiogenesis. Cell. 2011;146(6):873-887. [4] Fournier NM1, Duman RS. Role of vascular endothelial growth factor in adult hippocampal neurogenesis: implications for thepathophysiology and treatment of depression. Behav Brain Res. 2012;227(2):440-449. [5] Gu X, Ding F, Yang Y, et al. Construction of tissue engineered nerve grafts and their application in peripheral nerve regeneration. Prog Neurobiol. 2011;93(2):204-230. [6] Warner-Schmidt JL, Duman RS. VEGF as a potential target for therapeutic intervention in depression. Curr Opin Pharmacol. 2008;8(1):14-19. [7] Kubota H, Wu X, Goodyear SM, et al. Glial cell line-derived neurotrophic factor and endothelial cells promote self-renewal of rabbit germ cells withspermatogonial stem cell properties. FASEB J. 2011;25(8):2604-2614. [8] Savitt J, Singh D, Zhang C, et al. The in vivo response of stem and other undifferentiated spermatogonia to the reversible inhibition of glial cell line-derived neurotrophic factor signaling in the adult. Stem Cells. 2012;30(4): 732-740. [9] May F, Buchner A, Schlenker B, et al. Schwann cell-mediated delivery of glial cell line-derived neurotrophic factor restores erectile function aftercavernous nerve injury. Int J Urol. 2013;20(3):344-348. [10] Rodrigues DM, Li AY, Nair DG, et al. Glial cell line-derived neurotrophic factor is a key neurotrophin in the postnatal enteric nervous system. Neurogastroenterol Motil. 2011; 23(2):e44-56. [11] Stahl K, Mylonakou MN, Skare O, et al. Cytoprotective effects of growth factors: BDNF more potent than GDNF in an organotypic culture model of Parkinson's disease. Brain Res. 2011;1378(1):105-118. [12] Madduri S1, Feldman K, Tervoort T, et al. Collagen nerve conduits releasing the neurotrophic factors GDNF and NGF. J Control Release. 2010;143(2):168-174. [13] Lin W, Li M, Li Y, et al. Bone marrow stromal cells promote neurite outgrowth of spinal motor neurons by means of neurotrophic factorsin vitro. Neurol Sci. 2014;35(3): 449-457. [14] Deng LX, Deng P, Ruan Y, et al. A novel growth-promoting pathway formed by GDNF-overexpressing Schwann cells promotes propriospinalaxonal regeneration, synapse formation, and partial recovery of function after spinal cord injury. J Neurosci. 2013;33(13):5655-5667. [15] Gyorkos AM, McCullough MJ, Spitsbergen JM. Glial cell line derived neurotrophic factor (GDNF) expression and NMJ plasticity in skeletal muscle followingendurance exercise. Neuroscience. 2014;257(1):111-118. [16] Newland B, Abu-Rub M, Naughton M, et al. GDNF gene delivery via a 2-(dimethylamino) ethyl methacrylate based cyclized knot polymer for neuronal cellapplications. ACS Chem Neurosci. 2013;4(4):540-546. [17] Nikkhah G. Restorative strategies for the dopaminergic nigrostriatal projection pathway. Acta Neurochir Suppl. 2013;117:79-85. [18] Yu ZQ, Zhang BL, Ren QX, Changes in transcriptional factor binding capacity resulting from promoter region methylation induce aberrantlyhigh GDNF expression in human glioma. Mol Neurobiol. 2013;48(3):571-580. [19] Xie BH, Xie YF. Twin PCRs: a simple and efficient method for directional cloning of PCR products. World J Microbiol Biotechnol. 2011;27(9):2223-2225. [20] Chou AK, Yang MC, Tsai HP, et al. Adenoviral-mediated glial cell line-derived neurotrophic factor gene transfer has a protective effect on sciatic nerve following constriction-induced spinal cord injury. PLoS One. 2014; 9(3): e92264. [21] Kubota H, Wu X, Goodyear SM, et al. Glial cell line-derived neurotrophic factor and endothelial cells promote self-renewal of rabbit germ cells with spermatogonial stem cell properties. FASEB J. 2011;25(8):2604-2614. [22] Savitt J, Singh D, Zhang C, et al. The in vivo response of stem and other undifferentiated spermatogonia to the reversible inhibition of glial cell line-derived neurotrophic factor signaling in the adult. Stem Cells. 2012;30(4): 732-740. [23] May F, Buchner A, Schlenker B, et al. Schwann cell-mediated delivery of glial cell line-derived neurotrophic factor restores erectile function after cavernous nerve injury. Int J Urol. 2013;20(3):344-348. [24] Fletcher AM, Kowalczyk TH, Padegimas L, et al. Transgene expression in the striatum following intracerebral injections of DNA nanoparticles encoding for human glial cell line-derived neurotrophic factor. Neuroscience. 2011; 194: 220-226. [25] Rodrigues DM, Li AY, Nair DG, et al. Glial cell line-derived neurotrophic factor is a key neurotrophin in the postnatal enteric nervous system. Neurogastroenterol Motil. 2011; 23(2):e44-56. [26] Xu P, Rosen KM, Hedstrom K, et al. Nerve injury induces glial cell line-derived neurotrophic factor (GDNF) expression in Schwann cells throughpurinergic signaling and the PKC-PKD pathway. Glia. 2013;61(7):1029-1040. [27] Nowacka MM, Obuchowicz E. Vascular endothelial growth factor (VEGF) and its role in the central nervous system: a new element in the neurotrophic hypothesis of antidepressant drug action. Neuropeptides. 2012; 46(1): 1-10. [28] Luo H, Zhang Y, Zhang Z, et al. The protection of MSCs from apoptosis in nerve regeneration by TGFβ1 through reducing inflammation andpromoting VEGF-dependent angiogenesis. Biomaterials. 2012;33(17):4277-4287. [29] Mackenzie F, Ruhrberg C. Diverse roles for VEGF-A in the nervous system. Development. 2012;139(8):1371-1380. [30] Li Z, Burns AR, Han L, et al. IL-17 and VEGF are necessary for efficient corneal nerve regeneration. Am J Pathol. 2011; 178(3):1106-1116. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [3] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [4] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [7] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [8] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [9] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [10] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [11] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||