Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (21): 3420-3424.doi: 10.3969/j.issn.2095-4344.2014.21.024
Previous Articles Next Articles
Modification of cardiovascular grafts with peptides to promote endothelializaion
Liu Zhen, Xiong Shao-hu, Zhang Chuan-sen
- Regenerative Medicine Center of Second Military Medical University, Shanghai 200433, China
-
Online:2014-05-21Published:2014-05-21 -
Contact:Zhang Chuan-sen, Professor, Regenerative Medicine Center of Second Military Medical University, Shanghai 200433, China -
About author:Liu Zhen, Studying for master’s degree, Regenerative Medicine Center of Second Military Medical University, Shanghai 200433, China -
Supported by:the Science and Technology Key Projects for the Natural Science in Shanghai, No. 08441900600; the Graduate Innovative Experimental Subjects of the Second Military Medical University, No. SCMRC1210
CLC Number:
Cite this article
Liu Zhen, Xiong Shao-hu, Zhang Chuan-sen. Modification of cardiovascular grafts with peptides to promote endothelializaion[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(21): 3420-3424.
share this article
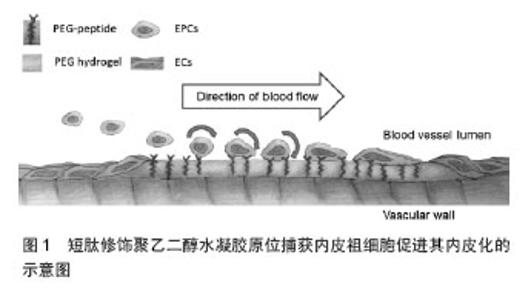
2.1 多肽修饰心血管材料的基本考量 在体内,细胞生活于以细胞外基质为主要组分的微环境中。细胞外基质除为细胞提供结构支撑以外,还与细胞因子等一道调控细胞的行为。对于血管而言,含有内皮细胞的内膜可生理性地阻止血栓形成相关蛋白和细胞的累积。另一方面,含有平滑肌细胞的中膜为血管提供强度和弹性。二者的平衡为血管生理性功能的实施提供保证。 正是考虑到细胞外基质的重要作用,因而最初的材料功能化多采用完整细胞外基质的蛋白来进行。研究也证明体外培养的内皮细胞能够很好地在胶原[5-6]、纤维粘连蛋白[7]、层粘连蛋白[8]、纤维蛋白等多种细胞外基质蛋白上黏附和扩展[9]。尽管如此,在心血管植入材料中直接引入天然细胞外基质成分的应用前景并不乐观。因为,这些蛋白质大分子需要从其他有机体分离、纯化,这增加了感染风险且易受到免疫排斥反应;而且,体内蛋白水解酶会造成其降解;此外,蛋白质分子的空间构象也会影响其效 果[10]。 研究表明,细胞外基质蛋白中的特定氨基酸序列主要通过与跨膜蛋白整合素家族受体或其他受体相结合,进而介导细胞的黏附、增殖和分化等。这些氨基酸序列成分固定、已知且易于合成。因此,近年来,越来越多的研究着眼于提取或合成细胞外基质功能蛋白中发挥关键作用的氨基酸序列,并将这些多肽类生物信息分子应用于心血管植入材料的表面修饰,人工模拟细胞外基质功能蛋白,促进此类材料的内皮化。经过多年的研究,目前的重点已由非特异性多肽转向特异性多肽,尤其是既能特异性吸附内皮细胞或内皮祖细胞,又能阻止其他类型细胞(特别是血小板或粒细胞)吸附的多肽。 2.2 常用的多肽修饰方案 2.2.1 非特异性多肽 精氨酸-甘氨酸-天冬氨酸(Arg-Gly-Asp,RGD):1986年,Ruoslahti等[11]首先在纤维连接蛋白中发现了RGD。此序列同样广泛存在于其他细胞外基质蛋白中,如玻连蛋白、层粘连蛋白等。多年的研究证实,RGD可被多种整合素受体所识别,如α3β1,α5β1,α8β1,αvβ1,αvβ3,αvβ5,αvβ6,αII bβ3等[12], 其中以α5β1和αvβ3最为突出。整合素α5β1与FN的结合恰是内皮细胞与细胞外基质黏附最主要的方式[13]。因此,RGD对心血管材料的修饰有助于内皮细胞的黏附与生长。事实上这一结论已通过众多的体外及体内实验得以验证。例如,有文献报道将含有RGD序列的多肽链负载于不同类型的心血管材料表面,均可有效促进内皮细胞的黏附和扩展[14-15]。而且,此种策略扩展的内皮细胞可承受相当于生理条件下的动脉壁切应力的冲击[16]。进一步的研究表明,RGD三肽与内皮祖细胞胞膜上αvβ5及α5β1整合素结合,从而使心血管材料具有捕获外周血中内皮祖细胞的能力,进而完成内皮化。RGD的这一特点使其具备运用于血管原位再生的能力。 除了从不同层面验证RGD序列促内皮细胞和内皮祖细胞黏附及生长的能力以外,学者们还着重探讨了RGD序列构型与其活性间的关系上。目前已明确的是,在RGD的N端增加不同氨基酸残基并不影响细胞的黏附,仅在促进细胞黏附过程中的作用有所差异[17]。另外,只有在C端天冬氨酸的羧基封闭时,RGD才能保留其活性,因此实际应用时常把C端羧基转变为酰胺基或在C端连接侧氨基,如RGDS、GRGDS、GRGDY和GRGDSPC等[13]。尤其值得一提的是,环形多肽与线性多肽相比,不仅能具有同样的生物学功能,而且其空间结构更稳定,半衰期更长,生物活性更持久[18]。环形RGD的优势,使得作者在实际操作中更倾向于选择环形多肽。 尽管RGD可以支持内皮细胞和内皮祖细胞的黏附和生长,但RGD同样具有识别血小板表面整合素受体αII bβ3的能力,进而促进血小板的聚集[19]。理论上,RGD能与血小板整合素受体αII bβ3结合的特性对于体外预内皮化的血管材料影响不大。但对借助原位再生策略重建受损血管尤为不利。这是因为,外周血中血小板的数量远远比多于内皮祖细胞的数量,众多的血小板可以在很短的时间内和RGD结合并覆盖于血管材料的内表面,进而竞争性抑制血管材料捕获内皮祖细胞。尤其重要的是血小板聚集将导致血栓的形成。 酪氨酸-异亮氨酸-甘氨酸-丝氨酸-精氨酸:(Tyr-Ile-Gly-Ser-Arg,YIGSR)来源于层粘连蛋白的B1链。与RGD不同,YIGSR作用的主要受体并非整合素受体家族成员,而是被称为67 kDa层粘连蛋白受体,共定位于肌动蛋白微丝束及钮蛋白[20],可促进肌细胞、成纤维细胞、内皮细胞等多种细胞类型的黏附[21- 22]。 Massia和Hubbell[23]报道,用YIGSR修饰玻璃、聚四氟乙烯、聚对苯二甲酸乙二醇酯(polyethylene terephthalate, PET)等多种材料,均可明显提高内皮细胞的黏附和扩展。他们还发现,原本无附着力的玻璃经YIGSR饰修后,在其上黏附和生长的内皮细胞可抵抗 2 Pa的切应力达8 h之久[24]。Jun等[25]报道将内皮细胞接种至经YIGSR修饰的聚氨酯薄膜后培养48 h,可见黏附的内皮细胞的数量较对照组明显增多,而且,这些细胞中表达增殖细胞核抗原的细胞比例也由60%提高至78%,显示YIGSR修饰不但能促进更多内皮细胞的黏附,更能促进其增殖。 2.2.2 特异性多肽 精氨酸-谷氨酸-天冬氨酸-缬氨酸(Arg-Glu-Asp-Val, REDV):REDV是纤维连接蛋白ⅢCS位点上的一个活性氨基酸序列,可被整合素α4β1特异性识别[26],而整合素α4β1主要表达于内皮细胞/内皮祖细胞胞膜,因而REDV可在内皮细胞、成纤维细胞、平滑肌细胞和血小板中选择性地促进内皮细胞的黏附和扩展[27- 28]。REDV的这一特性,使其越来越多地被用于生物材料的表面修饰,以建立内皮细胞高选择性的功能界面。Plouffe等[29]报道在微流体设备上负载REDV后,加入含内皮细胞、成纤维细胞和平滑肌细胞的等比例混合细胞悬液灌流,发现0.19 Pa切应力条件下,设备捕获的细胞中内皮细胞的比例为86%,显示出用此方法表面修饰心血管植入材料,可促进其原位内皮化。Veiseh等[30]通过微阵列技术比较了物理涂覆REDVY和共价结合KREDVY对内皮细胞黏附和活性的影响,发现两种方法均能提高内皮细胞的黏附,而且共价结合KREDVY还能减少细胞凋亡和坏死。在此实验中所用的多肽-REDVY和KREDVY,其发挥作用的核心均为REDV,多肽N端增加赖氨酸的主要作用是利用其所含的氨基发生缩合反应进而将多肽共价结合至基质上,多肽C端增加酪氨酸的主要目的是便于量化检测,两者均不会影响REDV的作用。Seeto等[31]发现用REDV修饰聚乙二醇水凝胶除了能在静态条件下促进内皮克隆形成细胞-晚期内皮祖细胞在其表面的黏附,更能显著降低内皮克隆形成细胞与其接触时的滚动速度,从而支持其动态黏附(图1)。该研究为REDV修饰促进心血管材料原位内皮化提供了新的理论依据。 基于以上认识,有研究者应用REDV修饰各种心血管植入材料以促进其内皮化。在计剑课题组的文献报道 中[32],体外实验证明了REDV修饰的PET片在与内皮细胞和平滑肌细胞共培养时,内皮细胞能更好地黏附和生长。体内实验采用REDV联合PMSN(一种2-甲基丙烯酰羟乙基磷酰胆碱衍生的高分子聚合物)修饰316 L不锈钢动脉支架后植入兔髂动脉,结果也显示内皮细胞更多地黏附和生长,实验组新生内膜厚度约为0.3 mm,而对照组约为 0.5 mm,实验组管腔狭窄率较对照组显著降低。 半胱氨酸-丙氨酸-甘氨酸(Cys-Ala-Gly,CAG):2012年,Kanie和同事[33]从IV型胶原中筛选出内皮细胞特异性三肽CAG。之所以选择IV型胶原,是因为该种胶原为血管基底膜中细胞外基质的主要组分。生理状态下,基底膜分隔了内膜的内皮细胞和中膜的平滑肌细胞,对于维持和控制内皮细胞和平滑肌细胞的平衡起着关键作用。体外实验证实,CAG与内皮细胞具有很高的亲和性,而与平滑肌细胞的亲和性较低[33]。该结果提示,如应用CAG功能化心血管材料将有助于快速内皮化,有效抑制内膜的过度增生。同年,日本学者Kuwabara等[34]将含有CAG三肽的聚羧基乙酸内酯(poly-ε-caprolactone,PCL)制备成直径为0.7 mm,长为7 mm的血管支架,进而以此替代SD大鼠的颈动脉,以此验证运用CAG在修饰或功能化小口径血管材料中的应用价值。实验结果表明,CAG的存在使PCL材料在移植后1周即可达到64.4%的内皮化。移植后6周,内皮化比例达到97.4%。在各个时相点,内皮化比例远较单纯PCL支架要高。而且,实验组内皮细胞中的内皮型一氧化氮合酶及血栓调节蛋白的表达也较对照组要高,该部分实验结果表明实验组不但内皮细胞的数量要高于对照组,细胞功能也要优于对照组。借助于平滑肌细胞特异性表型的检测,作者发现,α-平滑肌肌动蛋白在对照组中的表达要显著高于实验组,钙调蛋白的表达虽然在两组间无统计学的差异,但其在对照组中的表达依然要高于实验组,显示CAG修饰的血管材料具有抑制内膜过度增生的潜能。 丝氨酸-缬氨酸-缬氨酸-酪氨酸-亮氨酸-精氨酸(Ser-Val-Val-Tyr-Gly-Leu-Arg, SVVYGLR):骨桥蛋白中包含两种重要的活性序列,一是RGD,另一个是SVVYGLR。与RGD不同,SVVYGLR与整合素α4β1、α9β1及α4β7相互作用[35-36]。Hamada等[37-38]报道,虽然SVVYGLR不能促进内皮细胞的增殖,但能提高内皮细胞黏附与迁移的活性并诱导其分化,而且其促进血管生成的活性可和血管内皮生长因子相媲美,甚至更强[37]。Lei等[39]实验证明SVVYGLR修饰PET后,能更好地促进其内皮化[39]。 2.2.3 联合修饰 细胞外基质的成分较为复杂,其对细胞的支持也是多途径、多方面的。应用多肽修饰,模拟细胞外基质的功能,目前已显示出一定的应用前景,但要模拟天然细胞外基质对内皮细胞的支持,可能需要联合使用几种多肽甚至其他一些生物学信息分子。 Jun等[40]发现,联合YIGSR和PEG修饰聚氨酯后,除了能促进内皮细胞的黏附外,还能有效降低血小板的黏附,其中,YIGSR主要起到促进内皮细胞黏附的作用,而降低血小板的黏附主要与PEG的作用密切相关。该实验通过联合修饰,结合了两者的优势,较单因素修饰效果更好。Lei等[39]在比较了文章前述几种多肽修饰PET的效果后,提出RGD在促进内皮细胞的黏附方面更具优势,而YIGSR则主要促进内皮细胞的迁移和扩展,联合两种或多种多肽修饰则能达到强强联合的作用。尽管如此,这些多肽之间在促进内皮化方面到底是协同作用,还是相互拮抗,目前尚有争论。例如,Fittkau等[41]认为联合RGD与YIGSR表面修饰较单独使用RGD更能促进内皮细胞迁移;而Jung等[42]提出RGD与YIGSR在介导人脐静脉内皮细胞黏附和生长方面相互拮抗。 在联合应用多肽类与其他生物信息分子修饰材料以促进其内皮化方面,计剑课题组及WEST课题组均做出了有益的尝试[32,43]。在这些报道中,多肽通常选择特异性多肽,而其他生物信息分子主要包括PEG、PMC等。 "
| [1]Urban P,De Benedetti E.Thrombosis: the last frontier of coronary stenting?Lancet.2007;369(9562):619-621.
[2]Kannan RY,Salacinski HJ,Butler PE,et al.Current status of prosthetic bypass grafts: a review.J Biomed Mater Res B Appl Biomater.2005;74(1):570-581.
[3]Avci-Adali M,Stoll H,Wilhelm N,et al.In vivo tissue engineering: Mimicry of homing factors for self-endothelialization of blood-contacting materials. Pathobiology.2013;80(4):176-181.
[4]Sethi R,LEE CH.Endothelial progenitor cell capture stent: safety and effectiveness.J Interv Cardiol.2012;25(5):493-500.
[5]Bell E,Ehrlich HP,Buttle DJ,et al.Living tissue formed in vitro and accepted as skin-equivalent tissue of full thickness. Science.1981;211(4486):1052-1054.
[6]Patino MG,Neiders ME,Andreana S,et al.Collagen as an implantable material in medicine and dentistry.J Oral Implantol.2002;28(5):220-225.
[7]Clark RA,Lanigan JM,DellaPelle P,et al.Fibronectin and fibrin provide a provisional matrix for epidermal cell migration during wound reepithelialization. J Invest Dermatol. 1982; 79(5):264-269.
[8]Chen ZL,Indyk JA,Strickland S.The hippocampal laminin matrix is dynamic and critical for neuronal survival.Mol Biol Cell.2003;14(7):2665-2676.
[9]Itosaka H,Kuroda S,Shichinohe H,et al.Fibrin matrix provides a suitable scaffold for bone marrow stromal cells transplanted into injured spinal cord: a novel material for CNS tissue engineering.Neuropathology.2009;29(3):248-257.
[10]Hersel U,Dahmen C,Kessler H.RGD modified polymers: biomaterials for stimulated cell adhesion and beyond. Biomaterials.2003;24(24):4385-4415.
[11]Ruoslahti E,Pierschbacher MD.New perspectives in cell adhesion: RGD and integrins. Science. 1987;238(4826): 491-497.
[12]Pfaff M.Recognition sites of RGD-dependent integrins. Integrin-ligand interaction.Springer.1997:101-121.
[13]Hwang R, arner J.The role of integrins in tumor angiogenesis. Hematol Oncol Clin North Am.2004;18(5):991-1006.
[14]Meinhart JG,Schense JC,Schima H,et al.Enhanced endothelial cell retention on shear-stressed synthetic vascular grafts precoated with RGD-cross-linked fibrin. Tissue Eng. 2005;11(5-6):887-895.
[15]Larsen CC,Kligman F,Kottke-Marchant K,et al.The effect of RGD fluorosurfactant polymer modification of ePTFE on endothelial cell adhesion, growth, and function. Biomaterials. 2006;27(28):4846-4855.
[16]Tugulu S,Silacci P,Stergiopulos N,et al.RGD-Functionalized polymer brushes as substrates for the integrin specific adhesion of human umbilical vein endothelial cells. Biomaterials.2007;28(16):2536-2546.
[17]Verrier S,Pallu S,Bareille R,et al.Function of linear and cyclic RGD-containing peptides in osteoprogenitor cells adhesion process.Biomaterials.2002;23(2):585-596.
[18]Jeschke B,Meyer J,Jonczyk A,et al.RGD-peptides for tissue engineering of articular cartilage. Biomaterials. 2002;23(16): 3455-3463.
[19]Sánchez-Cortés J,Mrksich M.The platelet integrin αIIbβ3 binds to the RGD and AGD motifs in fibrinogen.Chem Biol. 2009;16(9):990-1000.
[20]Massia S,Rao S,Hubbell J.Covalently immobilized laminin peptide Tyr-Ile-Gly-Ser-Arg (YIGSR) supports cell spreading and co-localization of the 67-kilodalton laminin receptor with alpha-actinin and vinculin.J Biol Chem. 1993;268(11): 8053-8059.
[21]Graf J,Iwamoto Y,Sasaki M,et al.Identification of an amino acid sequence in laminin mediating cell attachment, chemotaxis, and receptor binding.Cell.1987;48(6): 989-996.
[22]Hubbel JA,Massia SP,Drumheller PD.Surface‐grafted Cell‐binding Peptides in Tissue Engineering of the Vascular Grafta. Ann N Y Acad Sci.1992;665(1):253-258.
[23]Massia SP,Hubbell JA.Convalent surface immobilization of Arg-Gly-Asp-and Tyr-Ile-Gly-Ser-Arg-containing peptides to obtain well-defined cell-adhesive substrates.Anal Biochem. 1990;187(2):292-301.
[24]Massia SP,Hubbell JA.Human endothelial cell interactions with surface-coupled adhesion peptides on a nonadhesive glass substrate and two polymeric biomaterials. J Biomed Mater Res.1991;25(2):223-242.
[25]Jun HW,West J.Development of a YIGSR-peptide-modified polyurethaneurea to enhance endothelialization.J Biomater Sci Polym Ed.2004;15(1):73-94.
[26]Mould A,Komoriya A,Yamada K,et al.The CS5 peptide is a second site in the IIICS region of fibronectin recognized by the integrin alpha 4 beta 1. Inhibition of alpha 4 beta 1 function by RGD peptide homologues.J Biol Chem. 1991; 266(6):3579-3585.
[27]Hubbell JA,Massia SP,Desai NP,et al.Endothelial cell-selective materials for tissue engineering in the vascular graft via a new receptor.Biotechnology (N Y). 1991;9(6): 568-572.
[28]Massia S,Hubbell J.Vascular endothelial cell adhesion and spreading promoted by the peptide REDV of the IIICS region of plasma fibronectin is mediated by integrin alpha 4 beta 1.J Biol Chem.1992;267(20):14019-14026.
[29]Plouffe BD,Njoka DN,Harris J,et al.Peptide-mediated selective adhesion of smooth muscle and endothelial cells in microfluidic shear flow.Langmuir.2007;23(9): 5050-5055.
[30]Veiseh M,Veiseh O,Martin MC,et al.Short peptides enhance single cell adhesion and viability on microarrays. Langmuir. 2007;23(8):4472-4479.
[31]Seeto WJ,Tian Y,Lipke EA.Peptide-grafted Poly (ethylene glycol) Hydrogels Support Dynamic Adhesion of Endothelial Progenitor Cells.Acta Biomater.2013; 9(8279-8289.
[32]Wei Y,Ji Y,Xiao LL,et al.Surface engineering of cardiovascular stent with endothelial cell selectivity for in vivo re-endothelialisation.Biomaterials.2013;34(2588-2599.
[33]Kanie K,Narita Y,Zhao Y,et al.Collagen type IV‐specific tripeptides for selective adhesion of endothelial and smooth muscle cells.Biotechnol Bioeng.2012; 109(7):1808-1816.
[34]Kuwabara F,Narita Y,Yamawaki-Ogata A,et al.Novel small-caliber vascular grafts with trimeric peptide for acceleration of endothelialization.Ann Thorac Surg. 2012; 93(1):156-163.
[35]Yokosaki Y,Matsuura N,Sasaki T,et al.The integrin α9β1 binds to a novel recognition sequence (SVVYGLR) in the thrombin-cleaved amino-terminal fragment of osteopontin.J Biol Chem.1999;274(51):36328-36334.
[36]Green PM,Ludbrook SB,Miller DD,et al.Structural elements of the osteopontin SVVYGLR motif important for the interaction with α< sub> 4</sub> integrins.FEBS Lett.2001;503(1):75-79.
[37]Hamada Y,Nokihara K,Okazaki M,et al.Angiogenic activity of osteopontin-derived peptide SVVYGLR.Biochem Biophys Res Commun.2003;310(1):153-157.
[38]Hamada Y,Egusa H,Kaneda Y,et al.Synthetic osteopontin-derived peptide SVVYGLR can induce neovascularization in artificial bone marrow scaffold biomaterials.Dent Mater J.2007;26(4):487.
[39]Lei Y,Rémy M,Labrugère C,et al.Peptide immobilization on polyethylene terephthalate surfaces to study specific endothelial cell adhesion, spreading and migration.J Mater Sci: Mater Med.2012;23(11):2761-2772.
[40]Jun HW,West JL.Modification of polyurethaneurea with PEG and YIGSR peptide to enhance endothelialization without platelet adhesion.J Biomed Mater Res B Appl Biomater. 2005;72(1):131-139.
[41]Fittkau M, Zilla P, Bezuidenhout D, et al. The selective modulation of endothelial cell mobility on RGD peptide containing surfaces by YIGSR peptides. Biomaterials. 2005; 26(2):167-174.
[42]Jung JP,Moyano JV,Collier JH.Multifactorial optimization of endothelial cell growth using modular synthetic extracellular matrices.Integr Biol (Camb).2011; 3(3):185-196.
[43]Taite LJ,Yang P,Jun HW,et al.Nitric oxide-releasing polyurethane-PEG copolymer containing the YIGSR peptide promotes endothelialization with decreased platelet adhesion.J Biomed Mater Res B Appl Biomater. 2008;84(1): 108-116. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||