Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (10): 1547-1553.doi: 10.3969/j.issn.2095-4344.2014.10.011
Previous Articles Next Articles
Effects of Corning® CellBIND® Surface medium on growth of human umbilical cord mesenchymal stem cells
Wan Ying1, Ben Liang1, Guan Zi-qiu 2, Li Chao1, Zhang Shi-dong1, Nie De-zhi1
- 1 TuoHua Biological Technology Company, Siping 136000, Jilin Province, China
2 Corning Incorporated, Shanghai 201206, China
-
Online:2014-03-05Published:2014-03-05 -
Contact:Nie De-zhi, M.D., Technician in charge, TuoHua Biological Technology Company, Siping 136000, Jilin Province, China -
About author:Wan Ying, M.D., TuoHua Biological Technology Company, Siping 136000, Jilin Province, China
CLC Number:
Cite this article
Wan Ying, Ben Liang, Guan Zi-qiu, Li Chao, Zhang Shi-dong, Nie De-zhi. Effects of Corning® CellBIND® Surface medium on growth of human umbilical cord mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(10): 1547-1553.
share this article
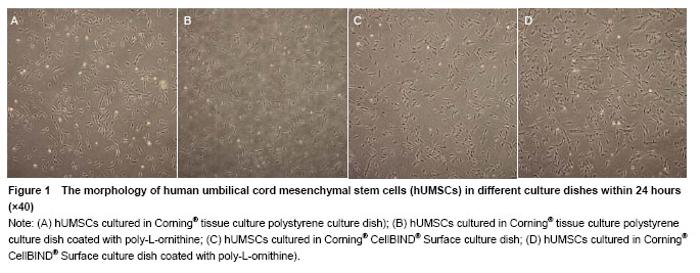
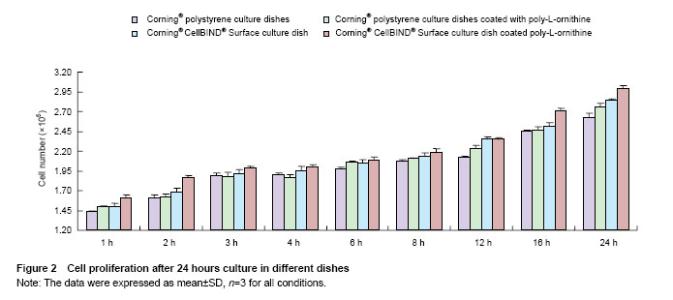
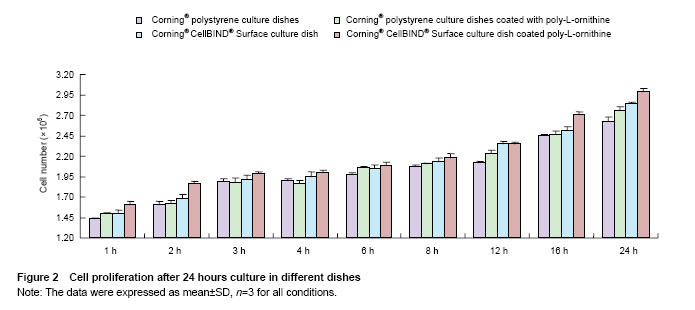
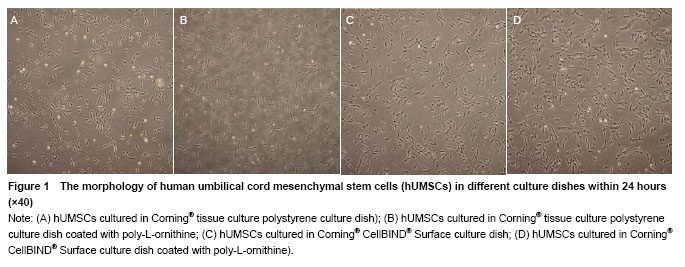
Effects of Corning® CellBIND® Surface dish coated with poly-L-ornithine on cell adhesion Cell numbers and morphology were observed in different culture dishes within 24 hours as shown in Figure 1. Cells cultured in the Corning® culture dish were most adhered and spindle-shaped, resembling fibroblasts. Corning® CellBIND® Surface culture dish also exhibited the powerful effect on cell growth[15-22], and the number of adherent cells was increased with the time prolonged within 24 hours. Depending on the treatment of microwave plasma process for treating the culture surface, Corning® CellBIND® Surface culture dish coated with poly-L-ornithine exhibited a better effect on cell attachment than Corning® tissue culture polystyrene dish coated with poly-L-ornithine (Figure 2)."
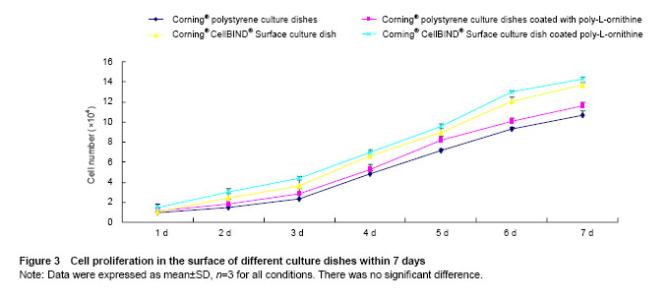
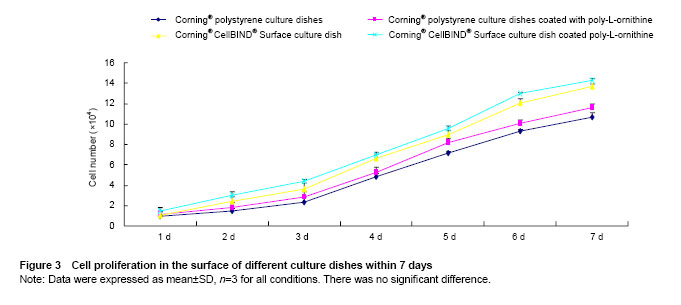
Effects of Corning® CellBIND® Surface dish coated with poly-L-ornithine on cell proliferation Cells proliferating in the different culture surfaces were observed in 7 days as shown in Figure 3. The results showed the growth curve of hUMSCs presented inverted “S”, and cells exhibited logarithmic growth on days 3, 4, 5 and 6. Owing to the effect of poly-L-ornithine on cell adhesion, Corning® tissue culture polystyrene dish and Corning® CellBIND® Surface dish coated with poly-L-ornithine improved cell growth at 7 days compared with the culture dishes that coated without poly-L-ornithine. However, Corning® CellBIND® Surface dish exerted the better effect on cell proliferation under any treatment on the surface of dish compared with Corning® tissue culture polystyrene dish (Figure 3)."
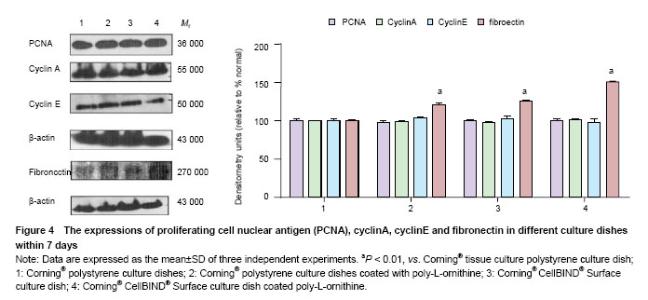
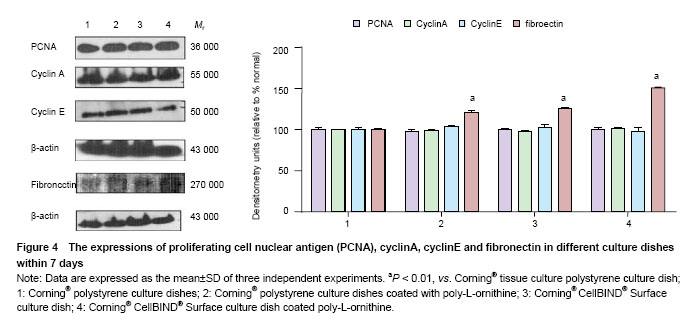
Effects of Corning® CellBIND® Surface dish coated with poly-L-ornithine on cell adhesion and cell proliferative protein expressions To test the effect of Corning®CellBIND® Surface culture dish on cell attachment and cell proliferative protein expressions, first, we detected cell proliferative protein including cyclinA, cyclinE and PCNA expressions. The results showed that neither Corning® CellBIND® Surface culture dish, Corning® tissue culture polystyrene dish nor those coated with poly-L-ornithine influence the expressions of PCNA, cyclinA or cyclinE by western blot analysis (P > 0.05). Furthermore, the expressions of adhesion cytokines were examined and the expression of fibronectin was significant increased in Corning® tissue culture polystyrene dish coated with poly-L-ornithine and Corning® CellBIND® Surface culture dish coated with or without poly-L-ornithine (P < 0.05) (Figure 4)."
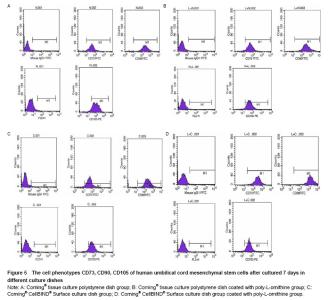
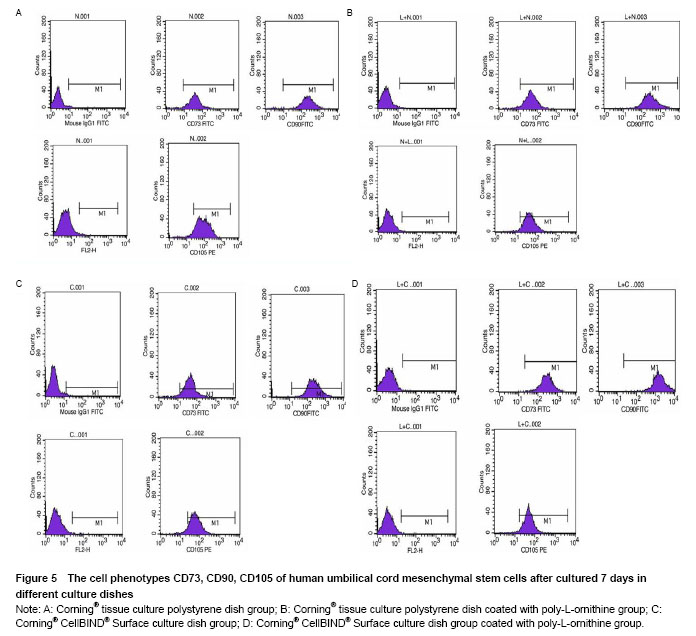
Effects of Corning® CellBIND® Surface dish coated with poly-L-ornithine on expression of cell surface markers We previously demonstrated that similarly processed hUMSCs expressed high levels of matrix receptors (CD73, CD90 and CD105) as assessed by flow cytometry. In addition, all adherent hUMSCs did not express CD45, CD34, CD14, CD19 or HLA-DR (data was not shown). In order to determine the effect of Corning® CellBIND® Surface culture dish on hUMSCs phenotype, we examined the levels of CD73, CD90 and CD105 in the four different groups. As our predicted, hUMSCs highly expressed CD73, CD90 and CD105 in the four groups as same as our isolated cells from human umbilical cords before. It implied that Corning® CellBIND® Surface culture dish coated with or without poly-L-ornithine did not affect the cell phenotype (Figure 5)."
| [1]Ding DC, Shyu WC, Lin SZ. Mesenchymal stem cells. Cell Transplant. 2011;20(1):5-14. [2]Ma L, Feng XY, Cui BL, et al. Human umbilical cord Wharton's Jelly-derived mesenchymal stem cells differentiation into nerve-like cells. Chin Med J (Engl). 2005;118(23):1987-1993.[3]Liu S, Yuan M, Hou K, et al. Immune characterization of mesenchymal stem cells in human umbilical cord Wharton's jelly and derived cartilage cells. Cell Immunol. 2012; 278(1-2):35-44. [4]Song WW, Bai H, Wang CB, et al. Effects of hypoxia on the proliferation of human bone marrow mesenchymal stem cells. Zhonghua Yi Xue Za Zhi. 2010;90(30):2149- 2152.[5]Mitchell KE, Weiss ML, Mitchell BM, et al. Matrix cells from Wharton's jelly form neurons and glia. Stem Cells. 2003; 21(1):50-60.[6]Poulsson AH, Mitchell SA, Davidson MR, et al. Attachment of human primary osteoblast cells to modified polyethylene surfaces. Langmuir. 2009;25(6):3718-3727. [7]Shen M, Horbett TA. The effects of surface chemistry and adsorbed proteins on monocyte/macrophage adhesion to chemically modified polystyrene surfaces. J Biomed Mater Res. 2001;57(3):336-345.[8]Pon-On W, Charoenphandhu N, Teerapornpuntakit J, et al. In vitro study of vancomycin release and osteoblast-like cell growth on structured calcium phosphate-collagen. Mater Sci Eng C Mater Biol Appl. 2013;33(3):1423-1431.[9]Rumian L, Wojak I, Scharnweber D, et al. Resorbable scaffolds modified with collagen type I or hydroxyapatite: in vitro studies on human mesenchymal stem cells. Acta Bioeng Biomech. 2013;15(1):61-67.[10]Gotman I, Ben-David D, Unger RE, et al. Mesenchymal stem cell proliferation and differentiation on load-bearing trabecular Nitinol scaffolds. Acta Biomater. 2013;9(9): 8440-8448. [11]Long X, Matsumoto R, Yang P, et al. Effect of human mesenchymal stem cells on the growth of HepG2 and Hela cells. Cell Struct Funct. 2013;38(1):109-121. [12]Dolley-Sonneville PJ, Romeo LE, Melkoumian ZK. Synthetic surface for expansion of human mesenchymal stem cells in xeno-free, chemically defined culture conditions. PLoS One. 2013;8(8):e70263. [13]Estrada JC, Torres Y, Benguría A, et al. Human mesenchymal stem cell-replicative senescence and oxidative stress are closely linked to aneuploidy. Cell Death Dis. 2013;4:e691. [14]Capra E, Beretta R, Parazzi V, et al. Changes in the proteomic profile of adipose tissue-derived mesenchymal stem cells during passages. Proteome Sci. 2012;10(1):46. [15]Harnett EM, Alderman J, Wood T. The surface energy of various biomaterials coated with adhesion molecules used in cell culture. Colloids Surf B Biointerfaces. 2007;55(1): 90-97.[16]Favi PM, Benson RS, Neilsen NR, et al. Cell proliferation, viability, and in vitro differentiation of equine mesenchymal stem cells seeded on bacterial cellulose hydrogel scaffolds. Mater Sci Eng C Mater Biol Appl. 2013;33(4):1935-1944. [17]Daley WP, Peters SB, Larsen M. Extracellular matrix dynamics in development and regenerative medicine. J Cell Sci. 2008;121(Pt 3):255-264. [18]Page-McCaw A, Ewald AJ, Werb Z. Matrix metalloproteinases and the regulation of tissue remodelling. Nat Rev Mol Cell Biol. 2007;8(3):221-233.[19]van Kooten TG, Spijker HT, Busscher HJ. Plasma-treated polystyrene surfaces: model surfaces for studying cell-biomaterial interactions. Biomaterials. 2004;25(10): 1735-1747.[20]Spoerke ED, Stupp SI. Synthesis of a poly(L-lysine)-calcium phosphate hybrid on titanium surfaces for enhanced bioactivity. Biomaterials. 2005; 26(25):5120-5129.[21]Lu H, Guo L, Kawazoe N, et al. Effects of poly(L-lysine), poly(acrylic acid) and poly(ethylene glycol) on the adhesion, proliferation and chondrogenic differentiation of human mesenchymal stem cells. J Biomater Sci Polym Ed. 2009; 20(5-6):577-589. [22]Steele JG, Dalton BA, Johnson G, et al. Polystyrene chemistry affects vitronectin activity: an explanation for cell attachment to tissue culture polystyrene but not to unmodified polystyrene. J Biomed Mater Res. 1993;27(7): 927-940.[23]Xu Y, Xiao Q, Tian Y, et al. Biological effects of the extracellular matrix on rat bone marrow mesenchymal stem cells. US Chin J Lymphology Oncol. 2007;10(1):26-28.[24]Shadpour H, Sims CE, Thresher RJ, et al. Sorting and expansion of murine embryonic stem cell colonies using micropallet arrays. Cytometry A. 2009;75(2):121-129. [25]Wang Y, Young G, Bachman M, et al. Collection and expansion of single cells and colonies released from a micropallet array. Anal Chem. 2007;79(6):2359-2366. [26]Xu W, Luikart AM, Sims CE, et al. Contact printing of arrayed microstructures. Anal Bioanal Chem. 2010;397(8): 3377-3385.[27]Gunn NM, Bachman M, Li GP, et al. Fabrication and biological evaluation of uniform extracellular matrix coatings on discontinuous photolithography generated micropallet arrays. J Biomed Mater Res A. 2010;95(2):401-412. [28]Shadpour H, Sims CE, Allbritton NL. Enrichment and expansion of cells using antibody-coated micropallet arrays. Cytometry A. 2009;75(7):609-618. [29]Beaulieu I, Geissler M, Mauzeroll J. Oxygen plasma treatment of polystyrene and Zeonor: substrates for adhesion of patterned cells. Langmuir. 2009;25(12): 7169-7176.[30]Kleinhans C, Barz J, Wurster S, et al. Ammonia plasma treatment of polystyrene surfaces enhances proliferation of primary human mesenchymal stem cells and human endothelial cells. Biotechnol J. 2013;8(3):327-337. [31]Lee JH, Jung HW, Kang IK, et al. Cell behaviour on polymer surfaces with different functional groups. Biomaterials. 1994;15(9):705-711.[32]Jankowski RJ, Haluszczak C, Trucco M, et al. Flow cytometric characterization of myogenic cell populations obtained via the preplate technique: potential for rapid isolation of muscle-derived stem cells. Hum Gene Ther. 2001;12(6):619-628.[33]Yun JK, DeFife K, Colton E, et al. Human monocyte/macrophage adhesion and cytokine production on surface-modified poly(tetrafluoroethylene/ hexafluoropropylene) polymers with and without protein preadsorption. J Biomed Mater Res. 1995;29(2):257-268.[34]DeFife KM, Yun JK, Azeez A, et al. Adhesion and cytokine production by monocytes on poly(2-methacryloyloxyethyl phosphorylcholine-co-alkyl methacrylate)-coated polymers. J Biomed Mater Res. 1995;29(4):431-439.[35]Lee JH, Lee HB. A wettability gradient as a tool to study protein adsorption and cell adhesion on polymer surfaces. J Biomater Sci Polym Ed. 1993;4(5):467-481.[36]Fisher RP. The CDK network: linking cycles of cell division and gene expression. Genes Cancer. 2012;3(11-12):731- 738. [37]Opas M, Dziak E. Adhesion, spreading, and proliferation of cells on protein carpets: effects of stability of a carpet. In Vitro Cell Dev Biol. 1991;27A(11):878-885.[38]Jenney CR, Anderson JM. Adsorbed serum proteins responsible for surface dependent human macrophage behavior. J Biomed Mater Res. 2000;49(4):435-447.[39]Flanagan LA, Rebaza LM, Derzic S, et al. Regulation of human neural precursor cells by laminin and integrins. J Neurosci Res. 2006;83(5):845-856. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [4] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [5] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [6] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [7] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [8] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [9] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [10] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [11] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [12] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [13] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [14] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [15] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||