Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (24): 3831-3837.doi: 10.12307/2023.194
Previous Articles Next Articles
Effects of high-mobility group box 1 on different subtypes of rat spinal reactive astrocytes after oxygen-glucose deprivation/restoration in vitro
Wang Liping1, Li Jisheng2, Deng Li2, Wang Zhiqiang2, Liu Jinming1, Deng Chen2, Sun Lin2
- 1College of Basic Medicine, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China; 2Department of Orthopedics, Third Hospital of Shanxi Medical University (Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Tongji Shanxi Hospital), Taiyuan 030032, Shanxi Province, China
-
Received:2022-04-01Accepted:2022-06-06Online:2023-08-28Published:2023-01-19 -
Contact:Sun Lin, MD, Chief physician, Department of Orthopedics, Third Hospital of Shanxi Medical University (Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Tongji Shanxi Hospital), Taiyuan 030032, Shanxi Province, China -
About author:Wang Liping, Master candidate, College of Basic Medicine, Shanxi Medical University, Taiyuan 030001, Shanxi Province, China -
Supported by:National Natural Science Foundation of China (General Program), No. 81870976 (to SL); Key Medical Project of Shanxi Province, No. 2020XM27 (to SL)
CLC Number:
Cite this article
Wang Liping, Li Jisheng, Deng Li, Wang Zhiqiang, Liu Jinming, Deng Chen, Sun Lin. Effects of high-mobility group box 1 on different subtypes of rat spinal reactive astrocytes after oxygen-glucose deprivation/restoration in vitro[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3831-3837.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
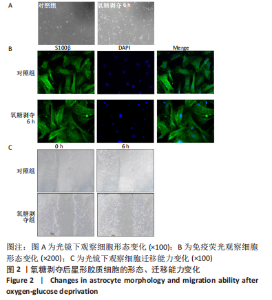
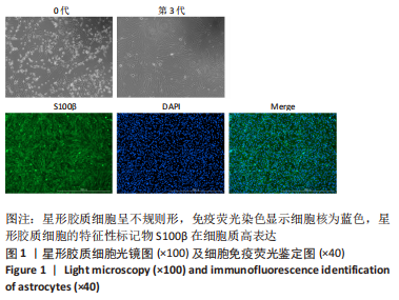
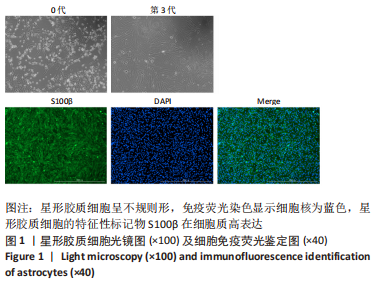
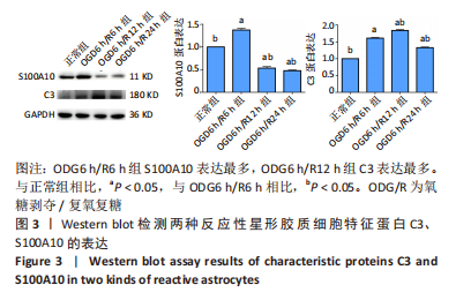
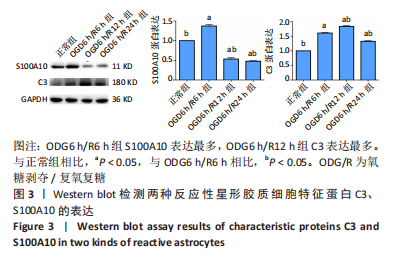
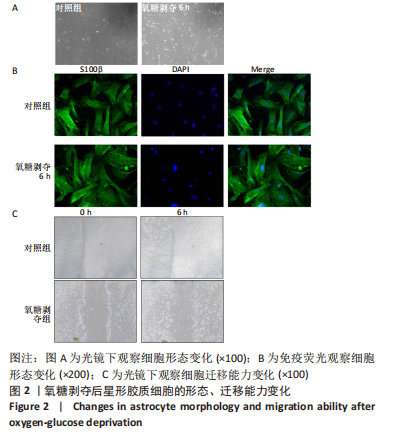
2.2 OGD/R损伤后星形胶质细胞活化为反应性星形胶质细胞 光镜下和细胞免疫荧光染色观察显示星形胶质细胞体积增大,划痕实验显示星形胶质细胞向损伤中心迁移,与对照组相比,氧糖剥夺后细胞的迁移能力增强,表明星形胶质细胞在损伤后成为一种更为活跃的状态,细胞的迁移能力增强,见图2。Western blot结果表明,OGD6 h/R6 h开始C3和S100A10表达增多,C3在OGD6 h/R12 h表达最多(P < 0.05),而S100A10在OGD6 h/R6 h表达最多(P < 0.05),表明星形胶质细胞在损伤早期活化为A2型反应性星形胶质细胞,在损伤后期活化为A1型反应性星形胶质细胞,见图3。为了使A2型反应性星形胶质细胞在整体的丰度增加,选择A2型反应性星形胶质细胞最多的时间点OGD6 h/R6 h作为后续实验的模型组。"
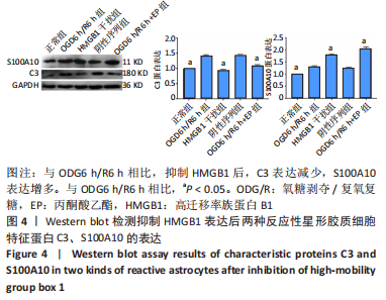
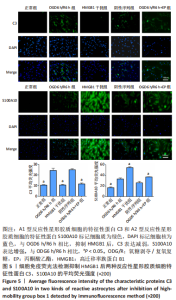
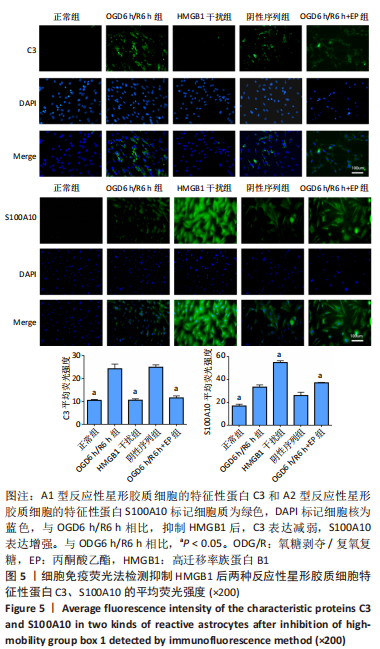
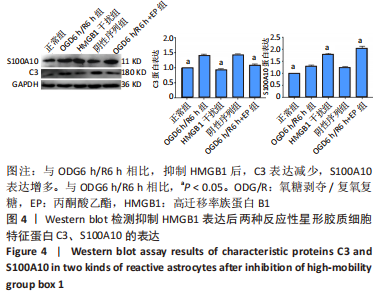
2.3 HMGB1对反应性星形胶质细胞的影响 以往研究已经证明OGD6 h/R2 h即可产生HMGB1,随着复氧复糖时间的增加HMGB1逐渐增多[14]。为了研究HMGB1对反应性星形胶质细胞的影响,实验选取慢病毒转染沉默HMGB1表达后进行OGD6 h/R6 h,以及氧糖剥夺6 h后加入丙酮酸乙酯继续复氧复糖6 h抑制HMGB1的表达,Western blot结果显示HMGB1干扰组及丙酮酸乙酯组的C3表达低于OGD6 h/R6 h组(P < 0.05),S100A10表达高于OGD6 h/R6 h组(P < 0.05),见图4。C3和S100A10在细胞质表达,DAPI染细胞核呈蓝色。细胞免疫荧光结果显示,与OGD6 h/R6 h组相比,HMGB1干扰组及丙酮酸乙酯组的C3平均荧光强度明显减弱(P < 0.05),S100A10平均荧光强度明显增强(P < 0.05),见图5。 结果表明抑制HMGB1后A1型反应性星形胶质细胞减少,A2型反应性星形胶质细胞增多。"
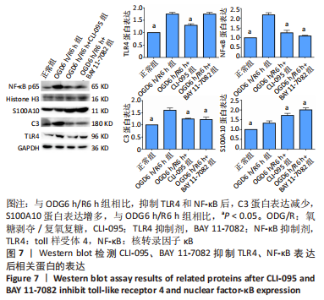
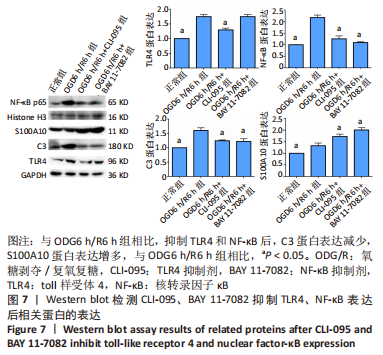
2.5 HMGB1对反应性星形胶质细胞影响的机制 为探究HMGB1对反应性星形胶质细胞影响的机制,氧糖剥夺6 h后加入CLI-095(TLR4抑制剂)和BAY 11-7082(NF-κB抑制剂)再复氧复糖6 h,Western blot结果显示与OGD6 h/R6 h组相比,CLI-095组TLR4表达减少(P < 0.05),CLI-095组和BAY 11-7082组NF-κB表达减少(P < 0.05),CLI-095组和BAY 11-7082组C3表达减少(P < 0.05),S100A10表达增多(P < 0.05),见图7。结果说明CLI-095可以有效抑制TLR4的表达,BAY 11-7082可以有效抑制NF-κB的表达,抑制TLR4可以降低NF-κB的表达,抑制TLR4和NF-κB可以使C3表达减少,S100A10表达增多。结果表明HMGB1可以与TLR4受体结合进一步激活NF-κB减少A1型反应性星形胶质细胞,增多A2型反应性星形胶质细胞。"
| [1] DAVID G, MOHAMMADI S, MARTIN AR, et al. Traumatic and nontraumatic spinal cord injury: pathological insights from neuroimaging. Nat Rev Neurol. 2019;15(12):718-731. [2] THURET S, MOON LD, GAGE FH. Therapeutic interventions after spinal cord injury. Nat Rev Neurosci. 2006;7(8):628-643. [3] ALIZADEH A, DYCK SM, KARIMI-ABDOLREZAEE S. Traumatic Spinal Cord Injury: An Overview of Pathophysiology, Models and Acute Injury Mechanisms. Front Neurol. 2019;10:282. [4] MONJE M. Spinal Cord Injury - Healing from Within. N Engl J Med. 2021; 384(2):182-184. [5] VON LEDEN RE, YAUGER YJ, KHAYRULLINA G, et al. Central Nervous System Injury and Nicotinamide Adenine Dinucleotide Phosphate Oxidase: Oxidative Stress and Therapeutic Targets. J Neurotrauma. 2017;34(4):755-764. [6] ESCARTIN C, GALEA E, LAKATOS A, et al. Reactive astrocyte nomenclature, definitions, and future directions. Nat Neurosci. 2021; 24(3):312-325. [7] LIDDELOW SA, GUTTENPLAN KA, CLARKE LE, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541(7638): 481-487. [8] CARTER SF, HERHOLZ K, ROSA-NETO P, et al. Astrocyte Biomarkers in Alzheimer’s Disease. Trends Mol Med. 2019;25(2):77-95. [9] LI L,LI Y,HE B, et al. HSF1 is involved in suppressing A1 phenotype conversion of astrocytes following spinal cord injury in rats.J Neuroinflammation. 2021;18:205. [10] ZAMANIAN JL, XU L, FOO LC, et al. Genomic analysis of reactive astrogliosis. J Neurosci. 2012;32(18):6391-6410. [11] TAKAHASHI S, MASHIMA K. Neuroprotection and Disease Modification by Astrocytes and Microglia in Parkinson Disease. Antioxidants (Basel). 2022;11(1):170. [12] LI X, LI M, TIAN L, et al. Reactive Astrogliosis: Implications in Spinal Cord Injury Progression and Therapy. Oxid Med Cell Longev. 2020; 2020:9494352. [13] HARA M, KOBAYAKAWA K, OHKAWA Y, et al. Interaction of reactive astrocytes with type I collagen induces astrocytic scar formation through the integrin-N-cadherin pathway after spinal cord injury. Nat Med. 2017;23(7):818-828. [14] GUO H,FAN Z,WANG S, et al. Astrocytic A1/A2 paradigm participates in glycogen mobilization mediated neuroprotection on reperfusion injury after ischemic stroke.J Neuroinflammation. 2021;18:230. [15] PAPATHEODOROU A, STEIN A, BANK M, et al. High-Mobility Group Box 1 (HMGB1) Is Elevated Systemically in Persons with Acute or Chronic Traumatic Spinal Cord Injury. J Neurotrauma. 2017;34(3):746-754. [16] SCAFFIDI P, MISTELI T, BIANCHI ME. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature. 2002;418(6894): 191-195. [17] FAN H, TANG HB, CHEN Z, et al. Inhibiting HMGB1-RAGE axis prevents pro-inflammatory macrophages/microglia polarization and affords neuroprotection after spinal cord injury. J Neuroinflammation. 2020; 17(1):295. [18] SUN L, LI M, MA X, et al. Inhibition of HMGB1 reduces rat spinal cord astrocytic swelling and AQP4 expression after oxygen-glucose deprivation and reoxygenation via TLR4 and NF-κB signaling in an IL-6-dependent manner. J Neuroinflammation. 2017;14(1):231. [19] BRAMBILLA R, BRACCHI-RICARD V, HU WH, et al. Inhibition of astroglial nuclear factor kappaB reduces inflammation and improves functional recovery after spinal cord injury. J Exp Med. 2005;202(1):145-156. [20] LIU J, ZHANG S, FAN X, et al. Dexmedetomidine Preconditioning Ameliorates Inflammation and Blood-Spinal Cord Barrier Damage After Spinal Cord Ischemia-Reperfusion Injury by Down-Regulation High Mobility Group Box 1-Toll-Like Receptor 4-Nuclear Factor κB Signaling Pathway. Spine (Phila Pa 1976). 2019;44(2):E74-E81. [21] ZHANG Y, MENG T, CHEN J, et al. miR-21a-5p Promotes Inflammation following Traumatic Spinal Cord Injury through Upregulation of Neurotoxic Reactive Astrocyte (A1) Polarization by Inhibiting the CNTF/STAT3/Nkrf Pathway. Int J Biol Sci. 2021;17(11):2795-2810. [22] KISUCKÁ A, BIMBOVÁ K, BAČOVÁ M, et al. Activation of Neuroprotective Microglia and Astrocytes at the Lesion Site and in the Adjacent Segments Is Crucial for Spontaneous Locomotor Recovery after Spinal Cord Injury. Cells. 2021;10(8):1943. [23] 吕聪,孙麟,冯皓宇,等.减少氧糖剥夺/复氧后脊髓星形胶质细胞凋亡:抑制高迁移率族蛋白B1/核转录因子κB通路的作用[J].中国组织工程研究,2019,23(33):5353-5359. [24] 宋君来,李满,孙麟,等.抑制高迁移率族蛋白1减轻大鼠脊髓星形胶质细胞氧糖剥夺/复氧后的损伤[J].中国组织工程研究,2018, 22(28):4537-4543. [25] WANG X,ZHANG Z,ZHU Z, et al. Photobiomodulation Promotes Repair Following Spinal Cord Injury by Regulating the Transformation of A1/A2 Reactive Astrocytes.Front Neurosci. 2021;15:768262. [26] ZHAO HD, HUANG SQ, TANG CL, et al. Influence of electroacupuncture on locomotor function and expression of spinal HMGB1 and TLR4 in mice with spinal cord injury. Zhen Ci Yan Jiu. 2021;46(4):259-265. [27] WANG Q, DING Q, ZHOU Y, et al. Ethyl pyruvate attenuates spinal cord ischemic injury with a wide therapeutic window through inhibiting high-mobility group box 1 release in rabbits. Anesthesiology. 2009;110(6):1279-1286. [28] SONG HH, SONG TC, YANG T, et al. High mobility group box 1 mediates inflammatory response of astrocytes via cyclooxygenase 2/prostaglandin E2 signaling following spinal cord injury. Neural Regen Res. 2021;16(9):1848-1855. [29] PAUDEL YN, SHAIKH MF, CHAKRABORTI A, et al. HMGB1: A Common Biomarker and Potential Target for TBI, Neuroinflammation, Epilepsy, and Cognitive Dysfunction. Front Neurosci. 2018;12:628. [30] NI B, CAO Z, LIU Y. Glycyrrhizin protects spinal cord and reduces inflammation in spinal cord ischemia-reperfusion injury. Int J Neurosci. 2013;123(11):745-751. [31] BI Y, ZHU Y, ZHANG M, et al. Effect of Shikonin on Spinal Cord Injury in Rats Via Regulation of HMGB1/TLR4/NF-kB Signaling Pathway. Cell Physiol Biochem. 2017;43(2):481-491. [32] YUAN Y, SU Z, PU Y, et al. Ethyl pyruvate promotes spinal cord repair by ameliorating the glial microenvironment. Br J Pharmacol. 2012; 166(2):749-763. [33] FILOUS AR, SILVER J. “Targeting astrocytes in CNS injury and disease: A translational research approach”. Prog Neurobiol. 2016;144:173-187. [34] ESCARTIN C, GUILLEMAUD O, CARRILLO-DE SAUVAGE MA. Questions and (some) answers on reactive astrocytes. Glia. 2019;67(12):2221-2247. [35] FAN YY, HUO J. A1/A2 astrocytes in central nervous system injuries and diseases: Angels or devils?Neurochem Int. 2021;148:105080. [36] WANG P, LI C, LIAO G, et al. Vanillin attenuates proinflammatory factors in a tMCAO mouse model via inhibition of TLR4/NF-kB signaling pathway. Neuroscience. 2022;491:65-74. [37] ZOU HJ, GUO SW, ZHU L, et al. Methylprednisolone Induces Neuro-Protective Effects via the Inhibition of A1 Astrocyte Activation in Traumatic Spinal Cord Injury Mouse Models. Front Neurosci. 2021;15:628917. [38] HOU Y, WANG K, WAN W, et al. Resveratrol provides neuroprotection by regulating the JAK2/STAT3/PI3K/AKT/mTOR pathway after stroke in rats. Genes Dis. 2018;5(3):245-255. [39] NING SL, ZHU H, SHAO J, et al. MiR-21 inhibitor improves locomotor function recovery by inhibiting IL-6R/JAK-STAT pathway-mediated inflammation after spinal cord injury in model of rat. Eur Rev Med Pharmacol Sci. 2019;23(2):433-440. [40] MORIZAWA YM, HIRAYAMA Y, OHNO N, et al. Author Correction: Reactive astrocytes function as phagocytes after brain ischemia via ABCA1-mediated pathway. Nat Commun. 2017;8(1):1598. [41] MA M, LI H, WU J, et al. Roles of Prokineticin 2 in Subarachnoid Hemorrhage-Induced Early Brain Injury via Regulation of Phenotype Polarization in Astrocytes. Mol Neurobiol. 2020;57(9):3744-3758. [42] ZHANG L, GUO K, ZHOU J, et al. Ponesimod protects against neuronal death by suppressing the activation of A1 astrocytes in early brain injury after experimental subarachnoid hemorrhage. J Neurochem. 2021;158(4):880-897. |
| [1] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [2] | Long Guiyue, Li Dongdong, Liao Hongbing. Calcium phosphate cement/poly(lactic-co-glycolic acid) degradation products promote osteoclast differentiation of mouse monocytes [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1193-1198. |
| [3] | Yang Jiujie, Li Zhi, Wang Shujie, Tian Ye, Zhao Wei. Intraoperative neurophysiological monitoring of functional changes following durotomy with decompression for acute spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1232-1236. |
| [4] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [5] | Li Wenjie, You Aijia, Zhou Junli, Fang Sujuan, Li Chun. Effects of different dressings in the treatment of burn wounds: a network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1141-1148. |
| [6] | Hao Liufang, Duan Hongmei, Wang Zijue, Hao Fei, Hao Peng, Zhao Wen, Gao Yudan, Yang Zhaoyang, Li Xiaoguang. Spatiotemporal dynamic changes of ependymal cells after spinal cord injury in transgenic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 883-889. |
| [7] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [8] | Zhang Qijian, Xu Ximing. Acquisition and application of ectodermal mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 928-934. |
| [9] | Chen Guodong, Zheng Meiyan, Zhang Peng, Wang Zhenchao, Jin Lixin. Changes in sensory neurons and astrocytes and the expression of interleukin 1beta and glial fibrillary acidic protein in the rat spinal cord after selective dorsal rhizotomy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 726-731. |
| [10] | Yang Heng, Zheng Liying, Liu Zhili, Wang Qinzhang, Hao Zhiqiang, Wang Jingshen, Wang Yuan, Li Yongle, Tan Minghui, Zou Xiaofeng, Zhang Guoxi, Huang Ruohui, Jiang Bo, Qian Biao. Physiological function of the testis in rats undergoing different parts of vasectomy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 720-725. |
| [11] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| [12] | Tao Xin, Xu Yi, Song Zhiwen, Liu Jinbo. Hippo signaling pathway in the regulation of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 619-625. |
| [13] | Guo Feng, Zhang Zhifeng, Sun Zhiwen, Lyu Xin, Piao Junjie, Wang Di, Niu Shuang, Chen Xiaowei, Zhang Yibo. Preoperative planning-based navigation system-assisted total hip arthroplasty: a randomized controlled clinical study [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(27): 4324-4331. |
| [14] | Wang Dongxu, Ren Jing, Li Jiping, Wang Yusu, Hu Pengfei, Zhang Guokun, Li Chunyi. Deer antler stem cell-derived exosomes prevent alcoholic liver injury via modulating nuclear factor kappa B signaling pathway in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3824-3830. |
| [15] | Cai Chuang, Huang Yonghui, Cao Xingbing, Sun Haitao, Guan Shihao, Huang Wenkang, Han Bo. Astrocyte-derived extracellular vesicles promote the differentiation of rat neural stem cells into neurons [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(24): 3845-3851. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||