Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (23): 3714-3720.doi: 10.12307/2023.487
Previous Articles Next Articles
Mechanism by which aerobic endurance exercise regulates cardiomyocyte autophagy in rats
Liu Jihuan1, Wang Peng1, Yuan Shunling1, Jian Ye1, Huang Shaoze1, Yao Sisi1, Liu Wenfeng1, 2
- 1Hunan Provincial Key Laboratory of Physical Fitness and Exercise Rehabilitation, Hunan Normal University, Changsha 410012, Hunan Province, China; 2Key Laboratory of Protein Chemistry and Developmental Biology, Ministry of Education, Hunan Normal University, Changsha 410081, Hunan Province, China
-
Received:2022-02-23Accepted:2022-07-18Online:2023-08-18Published:2023-01-16 -
Contact:Liu Wenfeng, MD, Professor, Hunan Provincial Key Laboratory of Physical Fitness and Exercise Rehabilitation, Hunan Normal University, Changsha 410012, Hunan Province, China; Key Laboratory of Protein Chemistry and Developmental Biology, Ministry of Education, Hunan Normal University, Changsha 410081, Hunan Province, China -
About author:Liu Jihuan, Master candidate, Hunan Provincial Key Laboratory of Physical Fitness and Exercise Rehabilitation, Hunan Normal University, Changsha 410012, Hunan Province, China -
Supported by:the National Natural Science Foundation of China, No. 81702236 (to LWF); Key Project of Hunan Provincial Department of Education, No. 20A333 (to LWF); Natural Science Foundation of Hunan Province, No. 2018JJ3363 (to LWF); Hunan Province College Young Backbone Teacher Project, No. [2020]43 (to LWF); Hunan Provincial Postgraduate Research and Innovation Project, No. [2021]267 (to LJH); Young Talents Training Program of Hunan Normal University, No. ET1507 (to LWF)
CLC Number:
Cite this article
Liu Jihuan, Wang Peng, Yuan Shunling, Jian Ye, Huang Shaoze, Yao Sisi, Liu Wenfeng. Mechanism by which aerobic endurance exercise regulates cardiomyocyte autophagy in rats[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(23): 3714-3720.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

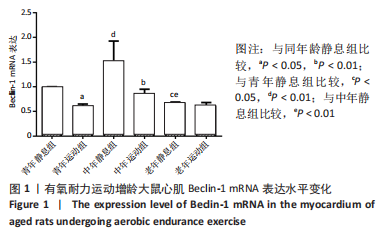
2.1 实验动物数量分析 纳入SD大鼠68只,3月龄(青年)20只,13月龄 (中年)和22月龄(老年)各24只,每个年龄组再随机分为静息、运动组,全部进入结果分析,无脱失。 2.2 有氧耐力运动对增龄大鼠心肌细胞自噬Beclin-1表达水平的影响 双因素方差分析结果显示,心肌细胞自噬因子Beclin-1运动与年龄之间存在交互作用[F(5,12)=10.160,P=0.004],主要作用因素为运动[F(1,12)=9.518,P=0.006]。由图1可见,3个年龄组中的静息大鼠心肌细胞自噬Beclin-1的表达水平没有出现衰老趋势;与青年静息组相比,中年静息组Beclin-1 mRNA的表达显著增加(P < 0.01),老年静息组Beclin-1 mRNA表达显著降低(P < 0.05)。定期有氧运动后,3个年龄组大鼠心肌细胞自噬Beclin-1的表达均较静息时降低;与青年静息组相比,青年运动组的Beclin-1 mRNA表达显著降低(P < 0.05);与中年静息组相比,中年运动组Beclin-1 mRNA表达显著降低(P < 0.01);而老年组的两组间Beclin-1 mRNA表达无明显变化(P > 0.05)。从图中可以看出中年静息组的Beclin-1 mRNA表达最高,青年运动组的Beclin-1 mRNA表达最低。 "
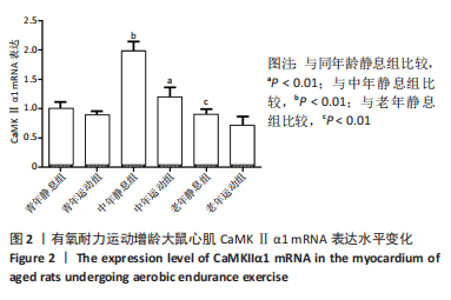
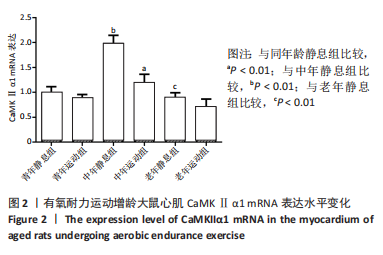
2.3 有氧耐力运动对增龄大鼠心肌CaMKⅡα1的表达水平的影响 双因素方差分析结果显示,心肌CaMKⅡα1的表达水平运动与年龄之间存在交互作用[F(5,12)=36.345,P < 0.001],主要作用因素为运动[F(1,12)=39.078,P < 0.001]。由图2可见,3组大鼠静息状态下心肌CaMKⅡα1 mRNA的表达与年龄无关,中年静息组大鼠心肌CaMKⅡα1 mRNA表达较青年静息组显著增加(P < 0.01)。老年静息组略低于青年静息组,但两组间CaMKⅡα1 mRNA表达量无显著差异(P > 0.05)。3个年龄组大鼠在有氧运动后,大鼠心肌CaMKⅡα1 mRNA表达均有所下降,与青年静息组相比,青年运动组CaMKⅡα1 mRNA表达低于青年静息组,但两组比较无显著差异(P > 0.05):与中年静息组相比,中年运动组CaMKⅡα1 mRNA表达明显低于中年静息组,差异有显著性意义(P < 0.01);老年静息组与老年运动组的CaMKⅡα1 mRNA表达无显著差异(P > 0.05)。从整体来看中年静息组的CaMKⅡα1 mRNA表达最高,而老年运动组的CaMKⅡα1 mRNA表达最少。"
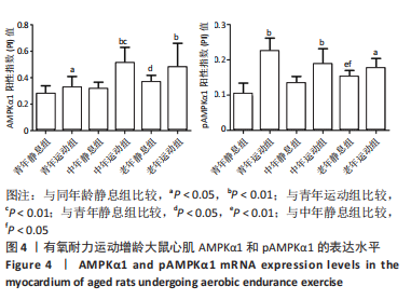
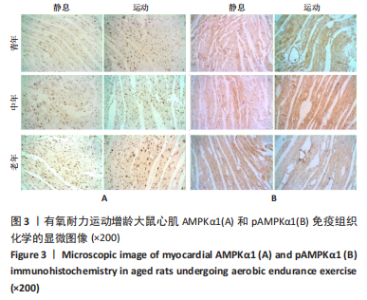
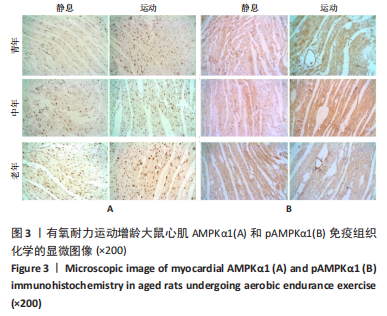
双因素方差分析结果显示,心肌细胞AMPKα1表达水平在运动与年龄之间没有交互作用[F(5,12)=1.571,P=0.222],单独作用因素为运动为[F(1,12)=12.008,P=0.001]和年龄[F(2,12)=5.820,P=0.007]。由图4可见,3个安静组大鼠的AMPKα1蛋白表达情况随着年龄的增长呈现出递增现象,其中青年静息组和中年静息组相比无显著差异(P > 0.05);而老年静息组的AMPKα1表达明显高于青年静息组(P < 0.05);中年静息组和老年静息组AMPKα1的表达无显著差异(P > 0.05)。常规有氧运动后,运动组大鼠心肌AMPKα1表达明显升高,青年运动组、中年运动组和老年运动组的AMPKα1表达水平分别上调16.50%(P < 0.05)、63.09%(P < 0.01)和31.12%(P < 0.01),从图中整体来看,中年运动组的AMPKα1表达最高,青年安静组的AMPKα1表达最少。"
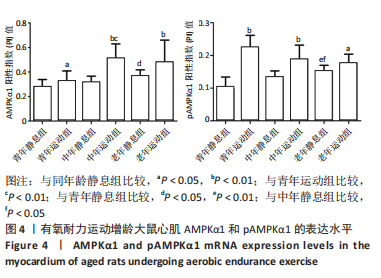
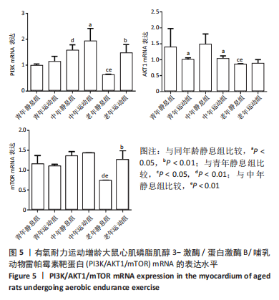
双因素ANOVA方差分析结果显示,心肌细胞pAMPKα1表达水平在运动与年龄之间存在交互作用[F(5,12)=9.698,P < 0.001],主要作用因素为运动[F(1,12)=46.680,P < 0.001]。从图4可以得出,3个安静组大鼠的pAMPKα1蛋白表达随着年龄的增长呈现出递增现象,定期有氧运动后,与安静组相比,3个运动组大鼠心肌pAMPKα1表达水平均显著升高,老年运动组增加16.77%(P < 0.05),中年运动组增加40.55%(P < 0.01),青年运动组增加131.29%(P < 0.01)。从图中还可以看出,青年运动组的pAMPKα1表达最高,而青年静息组的pAMPKα1表达最少。随着年龄增长,有氧运动对pAMPKα1的影响程度逐渐缩小。 2.5 有氧耐力运动对增龄大鼠心肌PI3K/AKT/mTOR mRNA表达水平的影响 双因素方差分析结果显示,心肌PI3K和mTOR表达水平运动与年龄之间存在交互作用 [F(5,12)= 10.101,P=0.003]和[F(5,12)=9.785,P=0.005],而心肌AKT表达水平运动与年龄之间不存在交互作用[F(5,12)=2.385,P=0.144]。图5表明,PI3K、AKT1、mTOR mRNA的表达水平在3个年龄静息组间并没有显示出衰老的趋势,AKT1 mRNA的表达在中年静息组表达最高,而PI3K和mTOR mRNA的表达在中年运动组最高,老年组PI3K、AKT1和mTOR mRNA的表达水平及变化趋势一样。与青年静息组比较,中年静息组PI3K mRNA表达显著增加(P < 0.01),而AKT1、mTOR mRNA表达无显著差异(P > 0.05);老年静息组PI3K、AKT1、mTOR mRNA表达明显降低(P < 0.01);其次与中年静息组相比,老年静息组PI3K、AKT1、mTOR mRNA表达水平也是明显降低(P < 0.01)。"
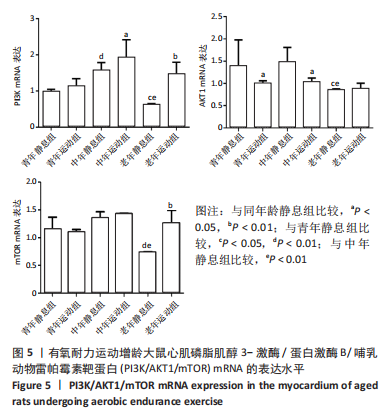

定期的有氧运动后,发现只有PI3K mRNA的表达相较于静息组均增加,而AKT1 mRNA的表达只有老年组表达略微增加,mTOR mRNA也是老年组表达显著上调。与青年静息组相比,青年运动组AKT1 mRNA表达水平明显下降(P < 0.05)。与中年静息组相比,中年运动组PI3K mRNA和AKT1 mRNA的表达都有明显差异,其中PI3K mRNA明显增加(P < 0.05),AKT1 mRNA明显降低(P < 0.05);与老年静息组相比,老年运动组PI3K mRNA和mTOR mRNA都明显上调(P < 0.01)。从图中整体来看,中年运动组PI3K mRNA的表达最高,老年安静组PI3K mRNA的表达最低;中年安静组AKT1 mRNA的表达最高,老年安静组的AKT1 mRNA表达最低;中年运动组的mTOR mRNA表达最高,同样mTOR mRNA表达最低的也是老年静息组。PI3K、AKT1、mTOR mRNA的表达在3个静息组和3个运动组趋势都一样,在静息状态下都是中年最高,在运动状态下同样也是中年最高。"

| [1] NARASIMHAN M, RAJASEKARAN NS. Cardiac Aging - Benefits of Exercise, Nrf2 Activation and Antioxidant Signaling. Adv Exp Med Biol. 2017;999:231-255. [2] NARASIMHAN M, RAJASEKARAN NS. Exercise, Nrf2 and Antioxidant Signaling in Cardiac Aging. Front Physiol. 2016;7:241. [3] NICCOLI T, PARTRIDGE L. Ageing as a risk factor for disease. Curr Biol. 2012; 22(17):R741-R752. [4] ABDELBASSET WK, ALQAHTANI BA, ALRAWAILI SM, et al. Similar effects of low to moderate-intensity exercise program vs moderate-intensity continuous exercise program on depressive disorder in heart failure patients: A 12-week randomized controlled trial. Medicine. 2019;98(32):e16820. [5] ABAD C, DO NA, DE SOUZA LE, et al. Interval and continuous aerobic exercise training similarly increase cardiac function and autonomic modulation in infarcted mice. J Exerc Rehabil. 2017;13(3):257-265. [6] PLOUMI C, PAPANDREOU ME, TAVERNARAKIS N. The complex interplay between autophagy and cell death pathways. Biochem J. 2022;479(1):75-90. [7] MADEO F, ZIMMERMANN A, MAIURI MC, et al. Essential role for autophagy in life span extension. J Clin Invest. 2015;125(1):85-93. [8] 何帆,来帅威,张沙沙,等. 自噬相关基因Rubicon缺失可抑制心脏细胞衰老[J]. 中国组织工程研究,2022,26(24):3897-3902. [9] KIM J, KIM YC, FANG C, et al. Differential regulation of distinct Vps34 complexes by AMPK in nutrient stress and autophagy. Cell. 2013;152(1-2):290-303. [10] 张欣,尚画雨,王瑞元. Omi/HtrA2介导的细胞自噬在运动性骨骼肌损伤中的作用[J]. 西安体育学院学报,2022,39(1):104-111. [11] HANDS SL, PROUD CG, WYTTENBACH A. mTOR’s role in ageing: protein synthesis or autophagy? Aging (Albany NY). 2009;1(7):586-597. [12] RUBINSZTEIN DC, MARINO G, KROEMER G. Autophagy and aging. Cell. 2011; 146(5):682-695. [13] ZHANG SL, LI ZY, WANG DS, et al. Aggravated ulcerative colitis caused by intestinal Metrnl deficiency is associated with reduced autophagy in epithelial cells. Acta Pharmacol Sin. 2020;41(6):763-770. [14] SU S, WANG X, XI X, et al. Phellodendrine promotes autophagy by regulating the AMPK/mTOR pathway and treats ulcerative colitis. J Cell Mol Med. 2021;25(12):5707-5720. [15] CHEN P, WANG Y, CHEN L, et al. Apelin-13 Protects Dopaminergic Neurons against Rotenone-Induced Neurotoxicity through the AMPK/mTOR/ULK-1 Mediated Autophagy Activation. Int J Mol Sci. 2020;21(21):8376. [16] STAHMANN N, WOODS A, CARLING D, et al. Thrombin activates AMP-activated protein kinase in endothelial cells via a pathway involving Ca2+/calmodulin-dependent protein kinase kinase beta. Mol Cell Biol. 2006; 26(16):5933-5945. [17] LIU W, CHEN G, LI F, et al. Calcineurin-NFAT Signaling and Neurotrophins Control Transformation of Myosin Heavy Chain Isoforms in Rat Soleus Muscle in Response to Aerobic Treadmill Training. J Sports Sci Med. 2014;13(4):934-944. [18] KOLTAI E, HART N, TAYLOR AW, et al. Age-associated declines in mitochondrial biogenesis and protein quality control factors are minimized by exercise training. Am J Physiol Regul Integr Comp Physiol. 2012;303(2):R127-R134. [19] 韩雨梅, 刘子泉, 常永霞, 等. 有氧耐力训练对增龄大鼠骨骼肌线粒体生物合成的影响[J]. 中国运动医学杂志,2010,29(4):425-429. [20] BEDFORD TG, TIPTON CM, WILSON NC, et al. Maximum oxygen consumption of rats and its changes with various experimental procedures. J Appl Physiol Respir Environ Exerc Physiol. 1979;47(6):1278-1283. [21] GATICA D, CHIONG M, LAVANDERO S, et al. Molecular mechanisms of autophagy in the cardiovascular system. Circ Res. 2015;116(3):456-467. [22] MAEJIMA Y, ZABLOCKI D, NAH J, et al. The role of the Hippo pathway in autophagy in the heart. Cardiovasc Res. 2022:cvac014. [23] CHEN CY, HSU HC, LEE BC, et al. Exercise training improves cardiac function in infarcted rabbits: involvement of autophagic function and fatty acid utilization. Eur J Heart Fail. 2010;12(4):323-330. [24] 马晓雯, 常芸, 王世强, 等. 不同强度不同时间耐力训练对于大鼠心肌细胞自噬发生程度的影响[J]. 中国运动医学杂志,2016,35(1):27-31. [25] 马晓雯, 常芸, 王世强. 长期耐力训练对大鼠心肌细胞自噬相关因子Beclin1和LC3的影响[J]. 体育科学,2016,36(2):66-71. [26] 贾绍辉, 刘君, 寇现娟, 等. 运动诱导的细胞自噬对小鼠心肌的保护作用[J]. 武汉体育学院学报,2014,48(10):53-56. [27] 刘文锋, 魏霞, 张冰侠, 等. 规律运动对增龄大鼠比目鱼肌细胞凋亡与自噬及PGC-1α信号调控的影响[J]. 中国运动医学杂志,2017,36(2):128-135. [28] WOHLGEMUTH SE, SEO AY, MARZETTI E, et al. Skeletal muscle autophagy and apoptosis during aging: effects of calorie restriction and life-long exercise. Exp Gerontol. 2010;45(2):138-148. [29] HARIHARAN N, MAEJIMA Y, NAKAE J, et al. Deacetylation of FoxO by Sirt1 Plays an Essential Role in Mediating Starvation-Induced Autophagy in Cardiac Myocytes. Circ Res. 2010;107(12):1470-1482. [30] NISHIDA K, YAMAGUCHI O, OTSU K. Crosstalk between autophagy and apoptosis in heart disease. Circ Res. 2008;103(4):343-351. [31] CHEN L, LIU D, ZHANG Y, et al. The autophagy molecule Beclin 1 maintains persistent activity of NF-kappaB and Stat3 in HTLV-1-transformed T lymphocytes. Biochem Biophys Res Commun. 2015;465(4):739-745. [32] LIANG XH, KLEEMAN LK, JIANG HH, et al. Protection against fatal Sindbis virus encephalitis by beclin, a novel Bcl-2-interacting protein. J Virol. 1998; 72(11):8586-8596. [33] RYAN F, KHODAGHOLI F, DARGAHI L, et al. Temporal Pattern and Crosstalk of Necroptosis Markers with Autophagy and Apoptosis Associated Proteins in Ischemic Hippocampus. Neurotox Res. 2018;34(1):79-92. [34] POLIVKA JJ, JANKU F. Molecular targets for cancer therapy in the PI3K/AKT/mTOR pathway. Pharmacol Ther. 2014;142(2):164-175. [35] FUJITA N, SATO S, KATAYAMA K, et al. Akt-dependent phosphorylation of p27Kip1 promotes binding to 14-3-3 and cytoplasmic localization. J Biol Chem. 2002;277(32):28706-28713. [36] 张嫒, 漆正堂, 丁树哲. Akt对骨骼肌质量的作用及运动训练对其影响的分子机制[J]. 体育科学,2008(3):73-78. [37] 田振军, 郝美丽, 席悦. 抗阻运动激活FSTL1-Akt-mTOR信号通路促进心梗大鼠心肌细胞增殖[J]. 体育科学,2018,38(3):40-47. [38] 谭支内, 蔡梦昕, 陈婷, 等. 间歇运动激活心梗大鼠心肌NRG1-PI3K/Akt通路抑制心肌细胞凋亡[J]. 北京体育大学学报,2016,39(6):69-76. [39] 李欣, 丁树哲, 卢健. 耐力训练对衰老小鼠心肌Akt/mTOR信号通路的影响[J]. 中国运动医学杂志,2010,29(1):38-41. [40] SCIARRETTA S, ZHAI P, SHAO D, et al. Rheb is a critical regulator of autophagy during myocardial ischemia: pathophysiological implications in obesity and metabolic syndrome. Circulation. 2012;125(9):1134-1146. [41] ZHANG J, ZHAO P, QUAN N, et al. The endotoxemia cardiac dysfunction is attenuated by AMPK/mTOR signaling pathway regulating autophagy. Biochem Biophys Res Commun. 2017;492(3):520-527. [42] LI Q, DONG QT, YANG YJ, et al. AMPK-mediated cardioprotection of atorvastatin relates to the reduction of apoptosis and activation of autophagy in infarcted rat hearts. Am J Transl Res. 2016;8(10):4160-4171. [43] HAWLEY SA, PAN DA, MUSTARD KJ, et al. Calmodulin-dependent protein kinase kinase-beta is an alternative upstream kinase for AMP-activated protein kinase. Cell Metab. 2005;2(1):9-19. [44] FERRARO E, GIAMMARIOLI AM, CHIANDOTTO S, et al. Exercise-induced skeletal muscle remodeling and metabolic adaptation: redox signaling and role of autophagy. Antioxid Redox Signal. 2014;21(1):154-176. [45] LIRA VA, BENTON CR, YAN Z, et al. PGC-1alpha regulation by exercise training and its influences on muscle function and insulin sensitivity. Am J Physiol Endocrinol Metab. 2010;299(2):E145-E161. [46] LIAO M, XIE Q, ZHAO Y, et al. Main active components of Si-Miao-Yong-An decoction (SMYAD) attenuate autophagy and apoptosis via the PDE5A-AKT and TLR4-NOX4 pathways in isoproterenol (ISO)-induced heart failure models. Pharmacol Res. 2022;176:106077. [47] LI X, LIU L, JIANG W, et al. SIRT6 Protects Against Myocardial Ischemia-Reperfusion Injury by Attenuating Aging-Related CHMP2B Accumulation. J Cardiovasc Transl Res. 2022;15(4):740-753. [48] PAN XC, XIONG YL, HONG JH, et al. Cardiomyocytic FoxP3 is involved in Parkin-mediated mitophagy during cardiac remodeling and the regulatory role of triptolide. Theranostics. 2022;12(5):2483-2501. [49] CUI XB, WANG C, LI L, et al. Insulin decreases myocardial adiponectin receptor 1 expression via PI3K/Akt and FoxO1 pathway. Cardiovasc Res. 2012;93(1):69-78. |
| [1] | Du Xueting, Zhang Xiaodong, Chen Yanjun, Wang Mei, Chen Wubiao, Huang Wenhua. Application of compressed sensing technology in two-dimensional magnetic resonance imaging of the ankle joint [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1396-1402. |
| [2] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [3] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [4] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [5] | Xiong Juan, Guan Yalin, Yang Yutong, Wang Fan, Liu Zhongshan. Application of stem cells to skin anti-aging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 948-954. |
| [6] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [7] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| [8] | Liu Guangluan, Guo Zonglei, Ge Jin, Huang Dong, Wang Yehua. Anatomic risk factors for medial meniscus posterior root tears combined with anterior cruciate ligament injuries [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 663-668. |
| [9] | Wu Yujie, Wan Xiaofang, Wei Mianxing, Peng Shiyuan, Xu Xiaomei. Correlation between autophagy and the Hippo-YAP protein pathway in periodental ligament cells on the pressure side of a mouse model of orthodontic tooth movement [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 683-689. |
| [10] | Zhao Siqi, Du Juan, Qu Haifeng, Li Jianmin, Zhang Yuxin, Liu Junjie. Effects of enriched environment combined with melatonin on learning and memory function and brain neuron apoptosis in SAMP8 mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 701-706. |
| [11] | Gu Mingxi, Wang Bo, Tian Fengde, An Ning, Hao Ruihu, Wang Changcheng, Guo Lin. Comparison of early efficacy and safety of simultaneous and staged bilateral total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 565-571. |
| [12] | Guo Yingqi, Gong Xianxu, Zhang Yan, Xiao Han, Wang Ye, Gu Wenguang. Meniscus extrusion and patellofemoral joint cartilage injury and bone marrow lesions: MRI semi-quantitative score [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 600-605. |
| [13] | Liu Hao, Yang Hongsheng, Zeng Zhimou, Wang Liping, Yang Kunhai, Hu Yongrong, Qu Bo. Lumbar MRI vertebral bone quality score to evaluate the severity of osteoporosis in postmenopausal women [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 606-611. |
| [14] | Wu Tong, Yin Caiyun, Zhao Mingzhe, Zhu Yishen. Application of functional peptides for biomedical diagnosis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 478-485. |
| [15] | Chen Chao, Wang Xuenan, Zhan Enyu, Lyu Zhengpin, Zhang Fan. Mechanism underlying the mutual regulation between circadian rhythm and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(29): 4696-4703. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||