Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (21): 3349-3355.doi: 10.12307/2023.170
Previous Articles Next Articles
Physical and chemical properties of porcine dermis matrix affected by process integration for decellularization, viral inactivation and sterilization
Huang Senli1, Sun Haigang1, Sun Wenquan2
- 1Ruijian Gaoke Biotechnology Co., Ltd., Beijing 102200, China; 2School of Health Science and Engineering, Shanghai University for Science and Technology, Shanghai 200093, China
-
Received:2022-03-24Accepted:2022-05-13Online:2023-07-28Published:2022-11-24 -
Contact:Sun Wenquan, PhD, Professor, Doctoral supervisor, School of Health Science and Engineering, Shanghai University for Science and Technology, Shanghai 200093, China -
About author:Huang Senli, Master, Ruijian Gaoke Biotechnology Co., Ltd., Beijing 102200, China
CLC Number:
Cite this article
Huang Senli, Sun Haigang, Sun Wenquan. Physical and chemical properties of porcine dermis matrix affected by process integration for decellularization, viral inactivation and sterilization[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3349-3355.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
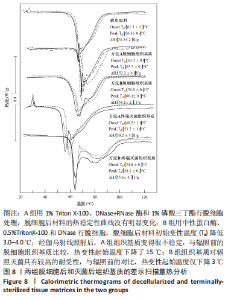
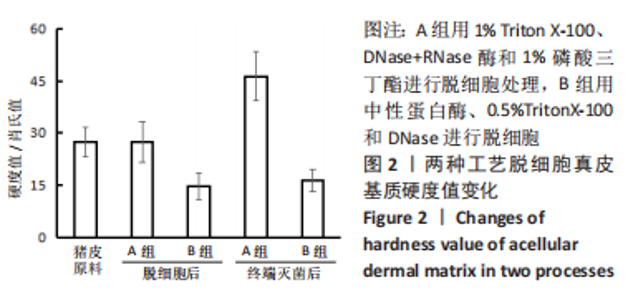
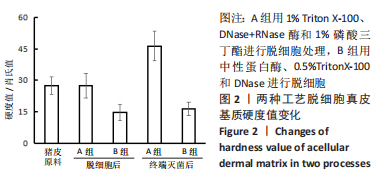
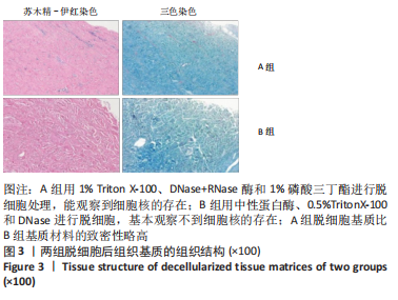
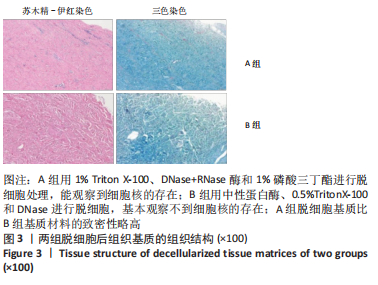
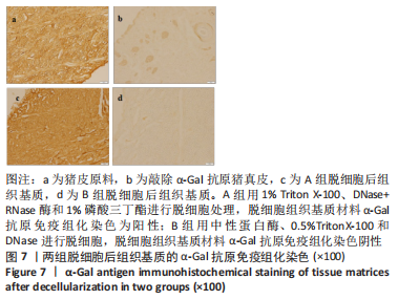
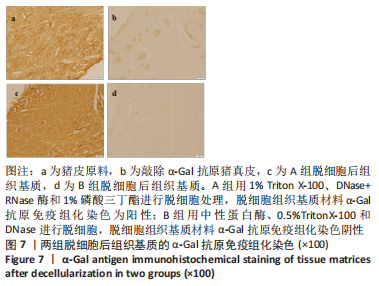
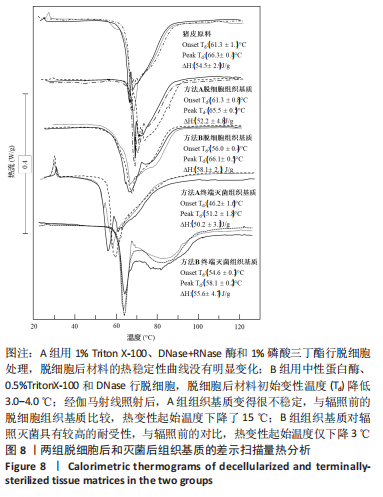
A组脱细胞组织基质材料与未处理猪真皮样品相似,染色很深,说明方法A对降低α-Gal抗原没有效果。方法B制备的脱细胞组织基质材料免疫组化染色为阴性,α-Gal抗原表位去除彻底。 2.5 两组基质材料的热稳定性 图8比较了用方法A和方法B制备的组织基质的热稳定性。与未处理的猪真皮相比,A组脱细胞后材料的热稳定性曲线没有明显变化,如初始(Td)或峰值变性温度、蛋白质变性的热焓值及热图的形状都没有变化。B组脱细胞后材料初始变性温度(Td)降低3.0-4.0 ℃。然而,经伽马射线照射后,A组组织基质变得很不稳定,起始变性温度Td下降到(46.2±1.6) ℃,变性峰值温度下降到(51.2±1.8) ℃,与辐照前的脱细胞组织基质比较,热变性起始温度下降了15 ℃;B组组织基质对辐照灭菌具有较高的耐受性,起始Td大于53 ℃,与辐照前的对比,热变性起始温度仅下降了3 ℃。因此,采用不同的脱细胞制备工艺,材料经过与病毒灭活和灭菌工艺的整合影响材料的稳定性。实验采用的这两种方法制备的组织基质材料热稳定性有显著差异。"
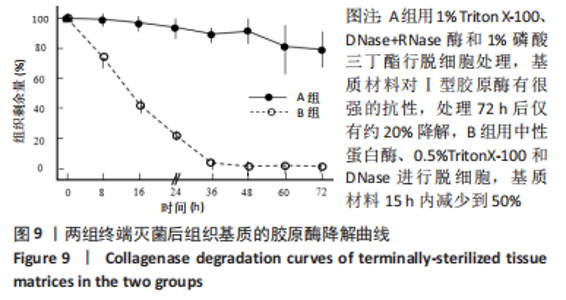
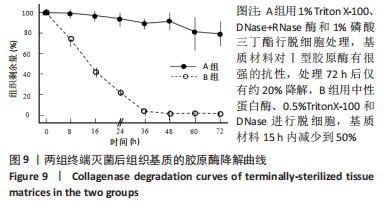
2.6 两组基质材料的力学性能 两组组织基质材料力学性能存在差异。结果显示,伽马射线终端灭菌后,A组基质材料在拉伸至应力为5 N时的低应变回弹区间,应变值为(14.8±3.1)%,B组基质材料相对应的应变值为(24.0±5.7)%;与B组相比,A组基质材料延展性较差。A组基质材料最大载荷为(150±47) N,B组基质材料最大载荷为(210±59) N。与B组相比,A组基质材料的强度略有下降。A、B组材料的断裂伸长率分别为(69±12)%和(65±12)%,没有明显差异。A、B组材料的杨氏模量分别为(24.6±8.8) MPa和(44.8±5.4) MPa。与B组相比,A组基质材料的抵抗形变能力或刚度较差。 2.7 两组基质材料的体外酶解性能 方法A和方法B制备的组织基质材料抗蛋白酶降解的能力不同。在伽马射线终端灭菌后,A组基质材料对Ⅰ型胶原酶具有很强的抗性,处理72 h后仅有约20%的降解,B组基质材料15 h内减少到50%,见图9。不过,A组基质材料对胰蛋白酶的抗性下降,胰蛋白酶可以降解80%以上的基质材料,B组基质材料只能降解小于35%的材料,见图10。对两个酶的抗性,方法B制备的基质材料与没有经过脱细胞处理的猪真皮材料极为相似。"
| [1] ZHANG X, CHEN X, HONG H, et al. Decellularized extracellular matrix scaffolds: recent trends and emerging strategies in tissue engineering. Bioact Mater. 2021;10:15-31. [2] AMIRAZAD H, DADASHPOUR M, ZARGHAMI N. Application of decellularized bone matrix as a bioscaffold in bone tissue engineering. J Biol Eng. 2022; 16(1):1-18. [3] LI T, JAVED R, AO Q. Xenogeneic Decellularized extracellular matrix-based biomaterials for peripheral nerve repair and regeneration. Curr Neuropharmacol. 2021;19(12):2152-2163. [4] YAO Q, ZHENG YW, LAN QH, et al. Recent development and biomedical applications of decellularized extracellular matrix biomaterials. Mater Sci Eng C Mater Biol Appl. 2019;104:109942. [5] CAPELLA-MONSONÍS H, ZEUGOLIS DI. Decellularized xenografts in regenerative medicine: From processing to clinical application. Xenotransplantation. 2021;28(4):e12683. [6] MENDIBIL U, RUIZ-HERNANDEZ R, RETEGI-CARRION S, et al. Tissue-Specific Decellularization Methods: Rationale and Strategies to Achieve Regenerative Compounds. Int J Mol Sci. 2020;21(15):5447. [7] EBRAHIMI SADRABADI A, BAEI P, HOSSEINI S, et al. Decellularized Extracellular Matrix as a Potent Natural Biomaterial for Regenerative Medicine. Adv Exp Med Biol. 2021;1341:27-43. [8] SAFDARI M, BIBAK B, SOLTANI H, et al. Recent advancements in decellularized matrix technology for bone tissue engineering. Differentiation. 2021;121: 25-34. [9] BUCKENMEYER MJ, MEDER TJ, PREST TA, et al. Decellularization techniques and their applications for the repair and regeneration of the nervous system. Methods. 2020;171:41-61. [10] CHENG J, LI J, CAI Z, et al. Decellularization of porcine carotid arteries using low- concentration sodium dodecyl sulfate. Int J Artif Organs. 2021;44(7): 497-508. [11] CRAPO PM, GILBERT TW, BADYLAK SF. An overview of tissue and whole organ decellularization processes. Biomaterials. 2011;32(12):3233-3243. [12] NONAKA PN, CAMPILLO N, URIARTE JJ, et al. Effects of freezing/thawing on the mechanical properties of decellularized lungs. J Biomed Mater Res A. 2013;102(2):413-419. [13] HUANG YH, TSENG FW, CHANG WH, et al. Preparation of acellular scaffold for corneal tissue engineering by supercritical carbon dioxide extraction technology. Acta Biomater. 2017;58:238-243. [14] 张国安,宁方刚,钟京鸣,等.反复冻融配合超声振荡洗涤制备猪脱细胞真皮基质[J].中国组织工程研究与临床康复,2007,11(41):8280-8284. [15] CHENG J, WANG C, GU Y. Combination of freeze-thaw with detergents: A promising approach to the decellularization of porcine carotid arteries. Biomed Mater Eng. 2019;30(2):191-205. [16] TAYLOR DA, SAMPAIO LC, FERDOUS Z, et al. Decellularized matrices in regenerative medicine. Acta Biomater. 2018;74:74-89. [17] CAI Z, GU Y, XIAO Y, et al. Porcine carotid arteries decellularized with a suitable concentration combination of Triton X-100 and sodium dodecyl sulfate for tissue engineering vascular grafts. Cell Tissue Bank. 2021;22(2): 277-286. [18] HREBIKOVA H, DIAZ D, MOKRY J. Chemical decellularization: a promising approach for preparation of extracellular matrix. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2015;159(1):12-17. [19] KEANE TJ, SWINEHART IT, BADYLAK SF. Methods of tissue decellularization used for preparation of biologic scaffolds and in vivo relevance. Methods. 2015;84:25-34. [20] WILSON SL, SIDNEY LE, DUNPHY SE, et al. Corneal decellularization: a method of recycling unsuitable donor tissue for clinical translation? Curr Eye Res. 2016;41(6):769-782. [21] HUSSEIN KH, PARK KM, KANG KS, et al. Biocompatibility evaluation of tissue-engineered decellularized scaffolds for biomedical application. Mater Sci Eng C Mater Biol Appl. 2016;67:766-778. [22] FERNÁNDEZ-PÉREZ J, AHEARNE M. The impact of decellularization methods on extracellular matrix derived hydrogels. Sci Rep. 2019;9(1):1-12. [23] GOKTAS S, MATUSKA AM, PIERRE N, et al. Decellularization method influences early remodeling of an allogenic tissue scaffold. J Biomed Mater Res A. 2013;102(1):8-16. [24] WANG F, ZHANG J, WANG R, et al. Triton X-100 combines with chymotrypsin: A more promising protocol to prepare decellularized porcine carotid arteries. Biomed Mater Eng. 2017;28(5):531-543. [25] LIAO J, JOYCE EM, SACKS MS. Effects of decellularization on the mechanical and structural properties of the porcine aortic valve leaflet. Biomaterials. 2008;29(8):1065-1074. [26] BROWN BN, FREUND JM, HAN L, et al. Comparison of three methods for the derivation of a biologic scaffold composed of adipose tissue extracellular matrix. Tissue Eng Part C Methods. 2011;17(4):411-421. [27] SAJITH S. Comparative study of two decellularization protocols on a biomaterial for tissue engineering. J Clin Exp Cardiolog. 2017;8(523):1-3. [28] RIEDER E, KASIMIR MT, SILBERHUMER G, et al. Decellularization protocols of porcine heart valves differ importantly in efficiency of cell removal and susceptibility of the matrix to recellularization with human vascular cells. J Thorac Cardiovasc Surg. 2004;127(2):399-405. [29] 史新立,张东刚,陈冰.动物源性医疗器械的病毒灭活进展[J].中华损伤与修复杂志(电子版),2009,4(3):340-345. [30] 李彩艳,王志超,刘灵飞,等.环氧乙烷灭菌和辐照灭菌在医疗器械灭菌中的应用[J].中国医疗器械信息,2020,26(17):175-176. [31] AEBERHARD PA, GROGNUZ A, PENEVEYRE C, et al. Efficient decellularization of equine tendon with preserved biomechanical properties and cytocompatibility for human tendon surgery indications. Artif Organs. 2020;44(4):E161-E171. [32] XU H, WAN H, ZUO W, et al. A porcine-derived acellular dermal scaffold that supports soft tissue regeneration: removal of terminal galactose-α-(1, 3)-galactose and retention of matrix structure. Tissue Eng Part A. 2009; 15(7):1807-1819. [33] 刘明,张旗,王蕊,等.过氧乙酸-乙醇对同种骨植入材料中病毒的灭活效果研究[J].中国消毒学杂志,2010,27(3):241-243. [34] 刘思扬,庄道民,董如华,等.60^钴辐照法对生物羊膜中人类免疫缺陷病毒灭活效果的研究[J].中国消毒学杂志,2015(3):217-218. [35] URIARTE JJ, NONAKA PN, CAMPILLO N, et al. Mechanical properties of acellular mouse lungs after sterilization by gamma irradiation. J Mech Behav Biomed Mater. 2014;40:168-177. [36] HODDE J, JANIS A, ERNST D, et al. Effects of sterilization on an extracellular matrix scaffold: Part I. Composition and matrix architecture. J Mater Sci Mater Med. 2007;18(4):537-543. [37] 周沫,李幼忱,张乃丽,等.过氧乙酸-乙醇联合辐照灭菌对同种肌腱力学强度的影响[J].中国矫形外科杂志,2011,19(22):1898-1900. [38] KANG HV, IM JH, CHUNG YG, et al. Comparison of two different decellularization methods for processed nerve allograft. Cell Tissue Bank. 2021;22(4):575-585. [39] MORADI L, JOBANIA BM, JAFARNEZHAD-ANSARIHA F, et al. Evaluation of different sterilization methods for decellularized kidney tissue. Tissue Cell. 2020;66: 101396. [40] Frank GP. Methods for tissue decellularization, US09566369B2 [P]. 2013. [41] ZHIGANG L, XINHUA L. Method for preparing an animal decellularized tissue matrix material and a decellularized tissue matrix material prepared thereby, US20160199540A1[P]. 2014. [42] BOONE M, DRAYE JP, VERWEEN G, et al. Real‐time three‐dimensional imaging of epidermal splitting and removal by high‐definition optical coherence tomography. Exp Dermatol. 2014;23(10):725-730. [43] 许银峰,胡军,肖良宝,等.制备去细胞人体肌腱支架材料的实验研究[J].中华显微外科杂志,2013,36(2):144-148. [44] CARTMELL JS, DUNN MG. Effect of chemical treatments on tendon cellularity and mechanical properties. J Biomed Mater Res. 2000;49(1):134-140. [45] XING S, LIU C, XU B, et al. Effects of various decellularization methods on histological and biomechanical properties of rabbit tendons. Exp Ther Med. 2014;8(2):628-634. [46] 程海明,陈敏,李志强.胶原及皮革等电点的表征方法综述[J].皮革科学与工程,2012,22(4):20-24. [47] 白玉龙,高玉凤,衷鸿宾,等.同种异体及异种组织修复材料: 如何选用适宜的病毒灭活工艺[J].中国组织工程研究,2019,23(14):2261-2268. [48] 金洹宇,银华,安艳.辐射技术在医用生物材料领域中的应用[J].辐射研究与辐射工艺学报,2011,29(5):261-265. [49] FIDALGO C, IOP L, SCIRO M, et al. A sterilization method for decellularized xenogeneic cardiovascular scaffolds. Acta Biomater. 2018;67:282-294. [50] HODDE J, HILES M. Virus safety of a porcine‐derived medical device: Evaluation of a viral inactivation method. Biotechnol Bioeng. 2002;79(2): 211-216. [51] LIN Q, LIM JYC, XUE K, et al. Sanitizing agents for virus inactivation and disinfection. View. 2020;1(2):e16. [52] 胡凯,陈维明,骆井万,等.过氧乙酸/乙醇对异体皮性能的影响[J].中国组织工程研究,2022,26(10):1537-1543. [53] UNI EN ISO 11137-2-2015,医疗保健产品灭菌.辐射.第2部分:建立灭菌剂量[S]. [54] SUN WQ, LEUNG P. Calorimetric study of extracellular tissue matrix degradation and instability after gamma irradiation. Acta Biomater. 2008; 4(4):817-826. [55] GOUK SS, LIM TM, TEOH SH, et al. Alterations of human acellular tissue matrix by gamma irradiation: Histology, biomechanical property, stability, in vitro cell repopulation, and remodeling. J Biomed Mater Res B Appl Biomater. 2010;84B(1):205-217. [56] EDWARDS JH, HERBERT A, JONES GL, et al. The effects of irradiation on the biological and biomechanical properties of an acellular porcine superflexor tendon graft for cruciate ligament repair. J Biomed Mater Res B Appl Biomater. 2017;105(8):2477-2486. [57] MA T, SUN W Q, WANG J. Thermophysical stability and biodegradability of regenerative tissue scaffolds. J Therm Anal Calorim. 2022:1-8. |
| [1] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [2] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [3] | Li Long, Li Guangdi, Shi Hao, Deng Keqi. Circular RNA as a competing endogenous RNA is involved in the regulation of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 751-757. |
| [4] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [5] | Jiang Haifang, Liu Rong, Hu Peng, Chen Wei, Wei Zairong, Yang Chenglan, Nie Kaiyu. Application of 3D printing technology in the precise and personalized treatment of cleft lip and palate [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 413-419. |
| [6] | Dong Shiwu, Zhou Lanxi, Shao Lu, Yu Zhengwen. Effect of magnesium alloy biomaterial degradation on endothelialized cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3398-3406. |
| [7] | Qi Junqiang, Guo Chao, Niu Dongyang, Wang Haotian, Xiao Bing, Xu Guohua. Characteristics and application of bone repair materials of metal ion doped hydroxyapatite [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3415-3422. |
| [8] | Xu Xin, Liu Yaowei, Mu Yunping, Wang Jianying, Li Fanghong, Zhao Zijian. Preparation and biological evaluation of decellularized dermal matrix hydrogel [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3325-3331. |
| [9] | Niu Lin, Mei Yukun, Zou Rui, Zhang Yuwei, Zhang Yifei, Hao Yaqi, Dong Shaojie. Application of inorganic nonmetal biomaterials in the treatment of osteosarcoma and the regeneration of tumor-related bone defects [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3368-3374. |
| [10] | Liu Peng, Ma Gang, He Ruibo, Peng Peng. Effect of aerobic exercise on renal fibrosis in rats with chronic renal failure [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3209-3215. |
| [11] | Wang Yanyang, Xu Pu. hFOB1.19 application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(20): 3266-3272. |
| [12] | Yang Yong, Xu Shunen, Gu Xianyang, Hua Dawei, Teng Jianxiang, Wang Zhen, Ye Chuan. Preparation and property analysis of electrospinning composite sustained-release antibacterial microspheres [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2534-2541. |
| [13] | Jia Yuanyuan, Duan Mianmian, Tang Zhenglong. Research and application of local drug delivery system in promoting fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2587-2594. |
| [14] | Hu Kai, Li Miao, Shao Yiran, Wang Jingjing, Chen Jinfa. Efficient removal of a decellularized reagent sodium dodecyl sulfate in allogeneic tissue [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(15): 2311-2317. |
| [15] | Ling Huajun, Cui Ruiwen, Wang Qiyou. 3D extracellular matrix hydrogel loaded with exosomes promotes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1900-1905. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||