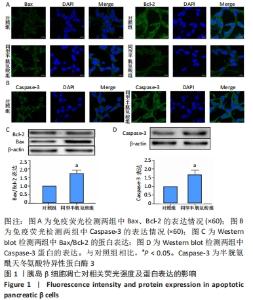
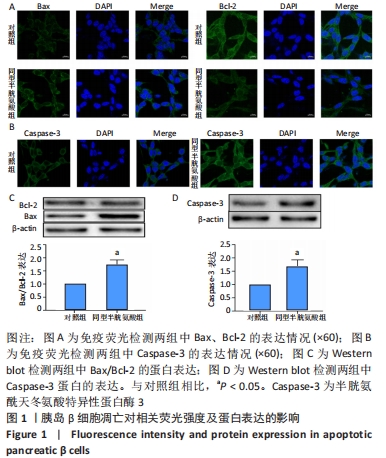
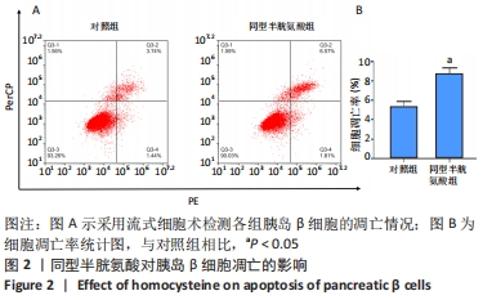
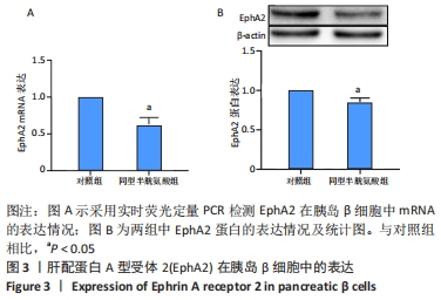
[1] LI Z, CHENG Y, WANG D, et al. Incidence Rate of Type 2 Diabetes Mellitus after Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis of 170,139 Women. J Diabetes Res. 2020;2020:3076463.
[2] ZHANG P, WANG AP, YANG HP, et al. Apelin-13 attenuates high glucose-induced calcification of MOVAS cells by regulating MAPKs and PI3K/AKT pathways and ROS-mediated signals. Biomed Pharmacother. 2020; 128:110271.
[3] COLE JB, FLOREZ JC. Genetics of diabetes mellitus and diabetes complications. Nat Rev Nephrol. 2020;16(7):377-390.
[4] FENG X, GAO X, JIA Y, et al. PPAR-α Agonist Fenofibrate Decreased RANTES Levels in Type 2 Diabetes Patients wit Hypertriglyceridemia. Med Sci Monit. 2016;22:743-51.
[5] ZARIC BL, OBRADOVIC M, BAJIC V, et al. Homocysteine and Hyperhomocysteinaemia. Curr Med Chem. 2019;26(16):2948-2961.
[6] XIE L, MA S, DING N, et al. Homocysteine induces podocyte apoptosis by regulating miR-1929-5p expression through c-Myc, DNMT1 and EZH2. Mol Oncol. 2021;15(11):3203-3221.
[7] 殷荷,王艳华,吴琪瑞,等.miR-5088-5p在子痫前期患者胎盘组织中的表达及其对滋养细胞自噬的影响[J].实用医学杂志,2020, 36(24):3312-3316+3322.
[8] 董小艳,刘达越,徐灵博,等. 脂肪酸结合蛋白4在同型半胱氨酸致大鼠心肌细胞焦亡中的作用[J].解放军医学杂志,2022,47(1):33-38.
[9] BAHARUDDIN WNA, YUSOFF AAM, ABDULLAH JM, et al. Roles of EphA2 Receptor in Angiogenesis Signaling Pathway of Glioblastoma Multiforme. Malays J Med Sci. 2018;25(6):22-27.
[10] ANDERTON M, VAN DER MEULEN E, BLUMENTHAL MJ, et al. The Role of the Eph Receptor Family in Tumorigenesis. Cancers (Basel). 2021; 13(2):206.
[11] TIAN D, QIN Q, LI M, et al. Homocysteine Impairs Endothelial Cell Barrier Function and Angiogenic Potential via the Progranulin/EphA2 Pathway. Front Pharmacol. 2021;11:614760.
[12] PI T, LIU B, SHI J. Abnormal Homocysteine Metabolism: An Insight of Alzheimer’s Disease from DNA Methylation. Behav Neurol. 2020; 2020:8438602.
[13] BANSAL A, PINNEY SE. DNA methylation and its role in the pathogenesis of diabetes. Pediatr Diabetes. 2017;18(3):167-177.
[14] AHMED SAH, ANSARI SA, MENSAH-BROWN EPK, et al. The role of DNA methylation in the pathogenesis of type 2 diabetes mellitus. Clin Epigenetics. 2020;12(1):104.
[15] LOVIC D, PIPERIDOU A, ZOGRAFOU I, et al. The Growing Epidemic of Diabetes Mellitus. Curr Vasc Pharmacol. 2020;18(2):104-109.
[16] LAAKSO M. Biomarkers for type 2 diabetes. Mol Metab. 2019;27S(Suppl): S139-S146.
[17] MOIN ASM, BUTLER AE. Alterations in Beta Cell Identity in Type 1 and Type 2 Diabetes. Curr Diab Rep. 2019;19(9):83.
[18] XU X, LAI Y, HUA ZC. Apoptosis and apoptotic body: disease message and therapeutic target potentials. Biosci Rep. 2019;39(1):BSR20180992.
[19] D’ORSI B, MATEYKA J, PREHN JHM. Control of mitochondrial physiology and cell death by the Bcl-2 family proteins Bax and Bok. Neurochem Int. 2017;109:162-170.
[20] VINCE JE, DE NARDO D, GAO W, et al. The Mitochondrial Apoptotic Effectors BAX/BAK Activate Caspase-3 and -7 to Trigger NLRP3 Inflammasome and Caspase-8 Driven IL-1β Activation. Cell Rep. 2018; 25(9):2339-2353.e4.
[21] NICHANI K, LI J, SUZUKI M, et al. Evaluation of Caspase-3 Activity During Apoptosis with Fluorescence Lifetime-Based Cytometry Measurements and Phasor Analyses. Cytometry A. 2020;97(12):1265-1275.
[22] KAPLAN P, TATARKOVA Z, SIVONOVA MK, et al. Homocysteine and Mitochondria in Cardiovascular and Cerebrovascular Systems. Int J Mol Sci. 2020;21(20):7698.
[23] NOOR A, RAHMAN MU, FARAZ N, et al. Relationship of Homocysteine With Gender, Blood Pressure, Body Mass Index, Hemoglobin A1c, and the Duration of Diabetes Mellitus Type 2. Cureus. 2021;13(11):e19211.
[24] LONDON M, GALLO E. The EphA2 and cancer connection: potential for immune based interventions. Mol Biol Rep. 2020;47(10):8037-8048.
[25] REZAIE E, AMANI J, BIDMESHKI POUR A, et al. A new scfv based recombinant immunotoxin against EPHA2-overexpressing breast cancer cells; High in vitro anti-cancer potency. Eur J Pharmacol. 2020; 870:172912.
[26] HUANG C, CHEN Z, HE Y, et al. EphA2 promotes tumorigenicity of cervical cancer by up-regulating CDK6. J Cell Mol Med. 2021;25(6): 2967-2975.
[27] WEI Q, ZHANG J, LI Z, et al. Serum Exo-EphA2 as a Potential Diagnostic Biomarker for Pancreatic Cancer. Pancreas. 2020;49(9):1213-1219.
[28] WANG M, NGO V, WANG W. Deciphering the genetic code of DNA methylation. Brief Bioinform. 2021;22(5):bbaa424.
[29] LAW PP, HOLLAND ML. DNA methylation at the crossroads of gene and environment interactions. Essays Biochem. 2019;63(6):717-726.
[30] ANGELONI A, BOGDANOVIC O. Enhancer DNA methylation: implications for gene regulation. Essays Biochem. 2019;63(6):707-715.
[31] EDWARDS JR, YARYCHKIVSKA O, BOULARD M, et al. DNA methylation and DNA methyltransferases. Epigenetics Chromatin. 2017;10:23.
[32] SINGH R, CHANDEL S, DEY D, et al. Epigenetic modification and therapeutic targets of diabetes mellitus. Biosci Rep. 2020;40(9): BSR20202160.
[33] INGROSSO D, PERNA AF. DNA Methylation Dysfunction in Chronic Kidney Disease. Genes (Basel). 2020;11(7):811.
[34] CHOI WY, HWANG JH, CHO AN, et al. DNA Methylation of Intragenic CpG Islands are Required for Differentiation from iPSC to NPC. Stem Cell Rev Rep. 2020;16(6):1316-1327.
[35] NASTESKA D, HODSON DJ. The role of beta cell heterogeneity in islet function and insulin release. J Mol Endocrinol. 2018;61(1):R43-R60.
[36] BRONNER C, ALHOSIN M, HAMICHE A, et al. Coordinated Dialogue between UHRF1 and DNMT1 to Ensure Faithful Inheritance of Methylated DNA Patterns. Genes (Basel). 2019;10(1):65.
[37] CHEN YT, LIN WD, LIAO WL, et al. NT5C2 methylation regulatory interplay between DNMT1 and insulin receptor in type 2 diabetes. Sci Rep. 2020;10(1):16087.
|