Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (2): 208-215.doi: 10.12307/2022.1015
Previous Articles Next Articles
Treadmill exercise improves cognitive dysfunction in diabetic rats
Wu Shuangshuang, Zhang Ying, Kou Xianjuan
- Hubei Provincial Key Laboratory of Sports Training and Monitoring, School of Health Sciences, Wuhan Sports University, Wuhan 430079, Hubei Province, China
-
Received:2021-12-29Accepted:2022-02-19Online:2023-01-18Published:2022-06-20 -
Contact:Kou Xianjuan, MD, Professor, Doctoral supervisor, Hubei Provincial Key Laboratory of Sports Training and Monitoring, School of Health Sciences, Wuhan Sports University, Wuhan 430079, Hubei Province, China -
About author:Wu Shuangshuang, Master candidate, Hubei Provincial Key Laboratory of Sports Training and Monitoring, School of Health Sciences, Wuhan Sports University, Wuhan 430079, Hubei Province, China -
Supported by:the National Natural Science Foundation of China, No. 81601228 (to KXJ); Humanities and Social Sciences Planning Fund of the Ministry of Education, No. 21YJA890014 (to KXJ)
CLC Number:
Cite this article
Wu Shuangshuang, Zhang Ying, Kou Xianjuan. Treadmill exercise improves cognitive dysfunction in diabetic rats[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 208-215.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
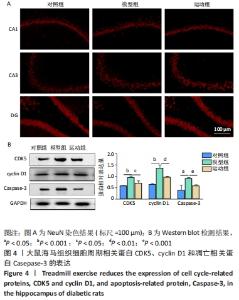
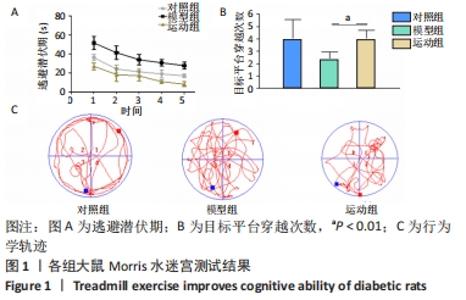
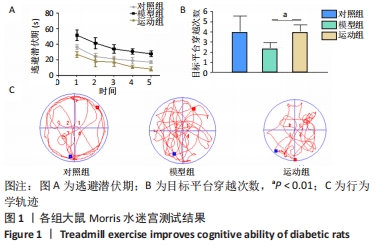
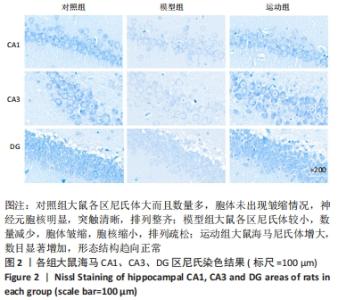
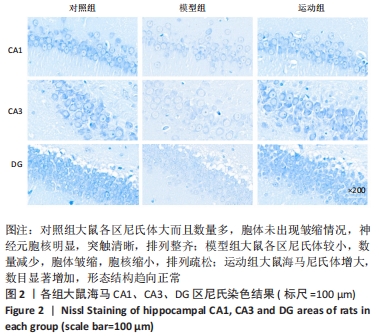
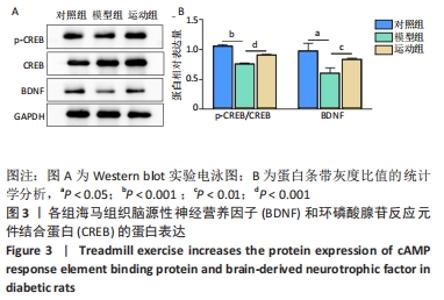
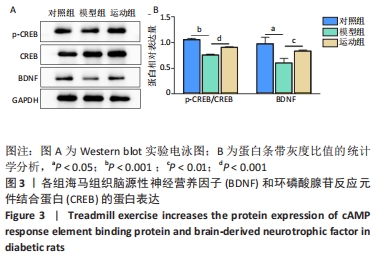
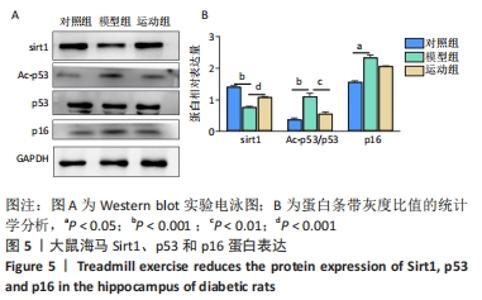
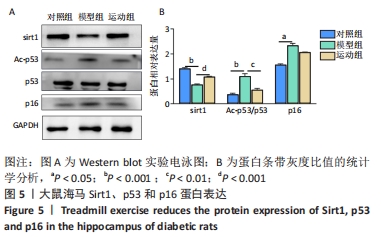
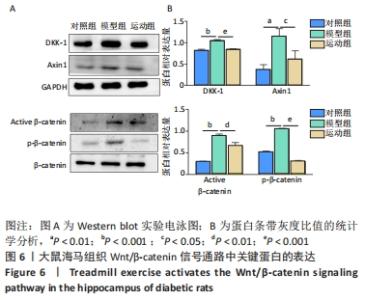
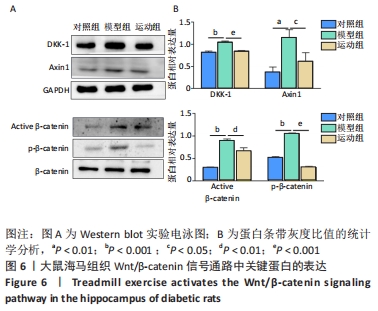
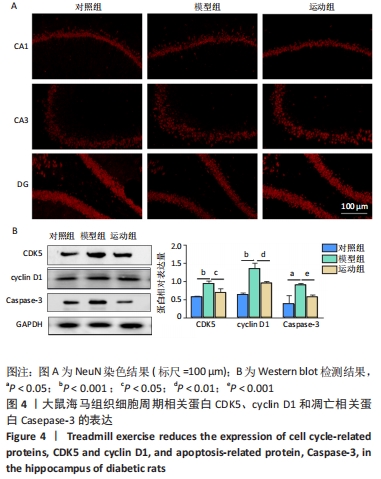
2.5 跑台运动降低糖尿病大鼠海马组织细胞周期和凋亡相关蛋白的表达 为了探讨跑台运动干预对SD大鼠海马组织细胞周期和神经细胞凋亡的保护作用,使用NeuN染色观察海马神经元形态结构改变和特异性数目变化,并通过Western blot实验进一步检测细胞周期相关蛋白CDK5、cyclin D1和凋亡相关蛋白Caspase-3的表达。 2.5.1 NeuN染色结果 对照组大鼠海马各区NeuN特异性标记的细胞数量多、胞核清晰、核仁明显、排列有序;相较于对照组,模型组糖尿病大鼠海马组织NeuN标记的细胞数量显著减少、核固缩、结构松散、排列紊乱。而相较于模型组,运动组大鼠海马组织NeuN标记细胞数量增多,细胞形态结构趋向正常,且排列更加紧密。说明跑台运动可有效减轻神经元损伤,见图4A。 2.5.2 Western blot结果 相较于对照组,模型组CDK5、cyclin D1和Caspase-3蛋白的表达明显上调(P < 0.05,P < 0.001);而8周跑台运动减少了细胞周期蛋白和促凋亡蛋白Caspase-3的表达(P < 0.05,P < 0.01,P < 0.001),结果说明8周跑台运动干预可以调节链脲佐菌素诱导的糖尿病大鼠细胞周期,并抑制海马神经元凋亡,见图4B。"
| [1] 王君佩, 杨吉春. 长非编码RNA与糖尿病及其并发症[J]. 生理科学进展, 2020,51(1): 1-7. [2] KAUTZKY-WILLER A, HARREITER J, PACINI G. Sex and Gender Differences in Risk, Pathophysiology and Complications of Type 2 Diabetes Mellitus. Endocr Rev. 2016;37(3):278-316. [3] RIEDERER P, KORCZYN AD, ALI SS, et al. The diabetic brain and cognition. J Neural Transm (Vienna). 2017;124(11):1431-1454. [4] TEO ZL, THAM YC, YU M, et al. Global Prevalence of Diabetic Retinopathy and Projection of Burden through 2045: Systematic Review and Meta-analysis. Ophthalmology. 2021;21(S0161-6420):32-13. [5] 牟严艳,叶中慧,林梅珍,等. 糖尿病流行病学研究进展[J]. 糖尿病新世界, 2019,22(4):196-198. [6] UMEGAKI H, HAYASHI T, NOMURA H, et al. Cognitive dysfunction: An emerging concept of a new diabetic complication in the elderly. Geriatr Gerontol Int. 2013;13(1):28-34. [7] NUSSE R, CLEVERS H. Wnt/β-Catenin Signaling, Disease, and Emerging Therapeutic Modalities. Cell. 2017;169(6):985-999. [8] NARVAES RF, FURINI CRG. Role of Wnt signaling in synaptic plasticity and memory. Neurobiol Learn Mem. 2021;187:107558. [9] LIBRO R, BRAMANTI P, MAZZON E. The role of the Wnt canonical signaling in neurodegenerative diseases. Life Sci. 2016;158:78-88. [10] HUANG P, YAN R, ZHANG X, et al. Activating Wnt/β-catenin signaling pathway for disease therapy: Challenges and opportunities. Pharmacol Ther. 2019;196(1):79-90. [11] INESTROSA NC, TAPIA-ROJAS C, CERPA W, et al. WNT Signaling Is a Key Player in Alzheimer’s Disease. Handb Exp Pharmacol. 2021;269(1):357-382. [12] GARCIA-VELAZQUEZ L, ARIAS C. The emerging role of Wnt signaling dysregulation in the understanding and modification of age-associated diseases. Ageing Res Rev. 2017;37(6):135-145. [13] AGHAIZU ND, JIN H, WHITING PJ. Dysregulated Wnt Signalling in the Alzheimer’s Brain. Brain Sci. 2020;10(12):902. [14] CHEN D, ZHANG Y, ZHANG M, et al. Exercise Attenuates Brain Aging by Rescuing Down-Regulated Wnt/β-Catenin Signaling in Aged Rats. Front Aging Neurosci. 2020;12(3):105-120. [15] MENET R, BOURASSA P, CALON F, et al. Dickkopf-related protein-1 inhibition attenuates amyloid-beta pathology associated to Alzheimer’s disease. Neurochem Int. 2020;141(4):104-114. [16] HERRANZ N, GIL J. Mechanisms and functions of cellular senescence. J Clin Invest. 2018;128(4):1238-1246. [17] QU J, XIONG X, HUJIE G, et al. MicroRNA-132-3p alleviates neuron apoptosis and impairments of learning and memory abilities in Alzheimer’s disease by downregulation of HNRNPU stabilized BACE1. Cell Cycle. 2021;20(21):2309-2320. [18] BARRETT T, STANGIS KA, SAITO T, et al. Neuronal Cell Cycle Re-Entry Enhances Neuropathological Features in AppNLF Knock-In Mice. J Alzheimers Dis. 2021; 82(4):1683-1702. [19] MALHOTRA N, GUPTA R, KUMAR P. Pharmacological relevance of CDK inhibitors in Alzheimer’s disease. Neurochem Int. 2021;148(1):105-115. [20] GUPTA R, JHA A, AMBASTA RK, et al. Regulatory mechanism of cyclins and cyclin-dependent kinases in post-mitotic neuronal cell division. Life Sci. 2021;285(2):1200-1216. [21] 刘申贝, 戴瑛, 赵军宁. 糖尿病脑病的研究进展[J].中国药理学与毒理学杂志, 2019,33(6): 444-450. [22] NAY K, SMILES WJ, KAISER J, et al. Molecular Mechanisms Underlying the Beneficial Effects of Exercise on Brain Function and Neurological Disorders. Int J Mol Sci. 2021;22(8):40-52. [23] CHOI DH, KWON KC, HWANG DJ, et al. Treadmill Exercise Alleviates Brain Iron Dyshomeostasis Accelerating Neuronal Amyloid-β Production, Neuronal Cell Death, and Cognitive Impairment in Transgenic Mice Model of Alzheimer’s Disease. Mol Neurobiol. 2021;58(7):208-3223. [24] BELAYA I, IVANOVA M, SORVARI A, et al. Astrocyte remodeling in the beneficial effects of long-term voluntary exercise in Alzheimer’s disease. J Neuroinflammation. 2020;17(1):271-290. [25] MATINFAR P, PEERI M, AZARBAYJANI MA. Swimming exercise attenuates anxiety-like behavior by reducing brain oxidative stress in type 2 diabetic mice. Physiol Behav. 2021;237:113449. [26] 税晓平, 李春莹, 曹艳霞, 等 . 有氧和抗阻运动干预2型糖尿病模型大鼠周围神经内质网应激相关蛋白的表达[J]. 中国组织工程研究,2021,25(11): 1693-1698. [27] PALOMER E, BUECHLER J, SALINAS PC. Wnt Signaling Deregulation in the Aging and Alzheimer’s Brain. Front Cell Neurosci. 2019;13(6):227-240. [28] ZHANG C, LI B. The correlation between LncRNA-17A expression in peripheral blood mononuclear cells and Wnt/β-catenin signaling pathway and cognitive function in patients with Alzheimer disease. Am J Transl Res. 2021;13(10):11981-11986. [29] TAPIA-ROJAS C, INESTROSA NC. Wnt signaling loss accelerates the appearance of neuropathological hallmarks of Alzheimer’s disease in J20-APP transgenic and wild-type mice. J Neurochem. 2018;144(4):443-465. [30] SALEM MA, BUDZYŃSKA B, KOWALCZYK J, et al. Tadalafil and bergapten mitigate streptozotocin-induced sporadic Alzheimer’s disease in mice via modulating neuroinflammation, PI3K/Akt, Wnt/β-catenin, AMPK/mTOR signaling pathway. Toxicol Appl Pharmacol. 2021;429:1156-1177. [31] TORICELLI M, PEREIRA A A R, SOUZA ABRAO G, et al. Mechanisms of neuroplasticity and brain degeneration: strategies for protection during the aging process. Neural Regen Res. 2021;16(1):58-67. [32] HERNANDEZ-ORTEGA K, QUIROZ-BAEZ R, ARIAS C. Cell cycle reactivation in mature neurons: a link with brain plasticity, neuronal injury and neurodegenerative diseases?. Neurosci Bull. 2011;27(3):185-196. [33] LUKASIK P, ZALUSKI M, GUTOWSKA I. Cyclin-Dependent Kinases (CDK) and Their Role in Diseases Development-Review. Int J Mol Sci. 2021;22(6):29-35. [34] ZHOU M, HUANG T, COLLINS N, et al. APOE4 Induces Site-Specific Tau Phosphorylation Through Calpain-CDK5 Signaling Pathway in EFAD-Tg Mice. Curr Alzheimer Res. 2016;13(9):1048-1055. [35] LIU S L, WANG C, JIANG T, et al. The Role of Cdk5 in Alzheimer’s Disease. Mol Neurobiol. 2016;53(7):4328-4342. [36] SHI J J, LIU H F, HU T, et al. Danggui-Shaoyao-San improves cognitive impairment through inhibiting O-GlcNAc-modification of estrogen α receptor in female db/db mice. J Ethnopharmacol. 2021;281:114562. [37] AIN Q, SCHMEER C, PENNDORF D, et al. Cell cycle-dependent and -independent telomere shortening accompanies murine brain aging. Aging (Albany NY). 2018; 10(11):3397-3420. [38] AL-KHALAF HH, NALLAR SC, KALVAKOLANU DV, et al. p16(INK4A) enhances the transcriptional and the apoptotic functions of p53 through DNA-dependent interaction. Mol Carcinog. 2017;56(7):1687-1702. [39] MODI PK, JAISWAL S, SHARMA P. Regulation of Neuronal Cell Cycle and Apoptosis by MicroRNA 34a. Mol Cell Biol. 2016;36(1):84-94. [40] CHAUHAN M, MODI PK, SHARMA P. Aberrant activation of neuronal cell cycle caused by dysregulation of ubiquitin ligase Itch results in neurodegeneration. Cell Death Dis. 2020;1(6):441-451. [41] SAH E, KRISHNAMURTHY S, AHMIDOUCH MY, et al. The Cellular Senescence Stress Response in Post-Mitotic Brain Cells: Cell Survival at the Expense of Tissue Degeneration. Life (Basel). 2021;11(3):229-240. [42] SOLTANIAN B, DEHGHAN SHASALTANEH M, RIAZI GH, et al. Alteration of gene expression in reactive astrocytes induced by Aβ1-42 using low dose of methamphetamine. Mol Biol Rep. 2021;48(8):6103-6112. [43] KIM SH, KO YJ, KIM JY, et al. Treadmill Running Improves Spatial Learning Memory Through Inactivation of Nuclear Factor Kappa B/Mitogen-Activated Protein Kinase Signaling Pathway in Amyloid-β-Induced Alzheimer Disease Rats. Int Neurourol J. 2021;25(Suppl 1):S35-43. [44] FARIOLI-VECCHIOLI S, TIRONE F. Control of the Cell Cycle in Adult Neurogenesis and its Relation with Physical Exercise. Brain Plast. 2015;1(1):41-54. [45] BIANCHI A, MARCHETTI L, HALL Z, et al. Moderate Exercise Inhibits Age-Related Inflammation, Liver Steatosis, Senescence, and Tumorigenesis. J Immunol. 2021; 206(4):904-916. [46] JIA L, PIÑA-CRESPO J, LI Y. Restoring Wnt/β-catenin signaling is a promising therapeutic strategy for Alzheimer’s disease. Mol Brain. 2019;12(1):104-120. [47] REN C, GU X, LI H, et al. The role of DKK1 in Alzheimer’s disease: A potential intervention point of brain damage prevention?. Pharmacol Res. 2019;144:331-335. [48] BAYOD S, FELICE P, ANDRÉS P, et al. Downregulation of canonical Wnt signaling in hippocampus of SAMP8 mice. Neurobiol Aging. 2015;36(2):720-729. [49] JIN N, ZHU H, LIANG X, et al. Sodium selenate activated Wnt/β-catenin signaling and repressed amyloid-β formation in a triple transgenic mouse model of Alzheimer’s disease. Exp Neurol. 2017;297:36-49. [50] WANG J, JING Y, SONG L, et al. Neuroprotective effects of Wnt/β-catenin signaling pathway against Aβ -induced tau protein over-phosphorylation in PC12 cells. Biochem Biophys Res Commun. 2016;471(4):628-632. [51] NING WJ, LV RJ, XU N, et al. Lycopene-Loaded Microemulsion Regulates Neurogenesis in Rats with Aβ-Induced Alzheimer’s Disease Rats Based on the Wnt/β-catenin Pathway. Neural Plast. 2021;2021:5519330. [52] 张楠, 王莉智, 杨桂姣, 等. 自主跑轮运动对阿尔兹海默病模型小鼠认知、情绪以及杏仁核炎性因子表达的影响[J]. 神经解剖学杂志,2017,33(6):741-747. |
| [1] | Shui Xiaoping, Li Chunying, Li Mingjuan, Li Shunchang, Sun Junzhi, Su Quansheng. Effects of aerobic and resistance exercise on antioxidant stress index and brain-derived neurotrophic factor expression in the hippocampus of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 264-269. |
| [2] | Zhu Miaomiao, Kong Fanming, Zhao Qian. Exercise regulates lactic acid metabolism [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 322-328. |
| [3] | Zhang Luyao, Yang Kang. Effects of different exercises on renal interstitial fibrosis in type 2 diabetic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 200-207. |
| [4] | Liu Siqi, Wu Mingrui, Qiao Lingran, Xie Liying, Chen Siyu, Han Zhibo, Zuo Lin. Effects of hydrogel loaded with human umbilical cord mesenchymal stem cells on diabetic wound repair in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 21-27. |
| [5] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [6] | Gao Lei, Qin Xinyuan, Nie Xin, Wang Lei, Wang Jiangning. Extracorporeal circulation compression perfusion in the reconstruction of limb microcirculation from the mechanism of mechanical and chemical signal transduction [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1334-1340. |
| [7] | Gu Zhengqiu, Xu Fei, Wei Jia, Zou Yongdi, Wang Xiaolu, Li Yongming. Exploratory study on talk test as a measure of intensity in blood flow restriction training [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1154-1159. |
| [8] | Lü Yiyan, Li Hanbing, Ma Xiaoqing, Zhang Han, Zhang Yuhang, Li Genlin. Establishment and characteristic analysis of interior heat and diabetes mouse model using compound factors [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1187-1193. |
| [9] | Chen Xianghe, Liu Bo, Yang Kang, Lu Pengcheng, Yu Huilin. Treadmill exercise improves the myocardial fibrosis of spontaneous type 2 diabetic mice: an exploration on the functional pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1210-1215. |
| [10] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 999-1004. |
| [11] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1027-1031. |
| [12] | Shui Xiaoping, Li Chunying, Li Shunchang, Sun Junzhi, Su Quansheng . Effects of aerobic and resistance exercises on brain-derived neurotrophic factor, nuclear factor-kappa B and inflammatory cytokines in skeletal muscle of type II diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 669-675. |
| [13] | Deng Shuang, Pu Rui, Chen Ziyang, Zhang Jianchao, Yuan Lingyan . Effects of exercise preconditioning on myocardial protection and apoptosis in a mouse model of myocardial remodeling due to early stress overload [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 717-723. |
| [14] | Wei Xing, Liu Shufang, Mao Ning. Roles and values of blood flow restriction training in the rehabilitation of knee joint diseases [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 774-779. |
| [15] | Tang Jiping, Zhang Yeting. Exercise regulates adult hippocampal neurogenesis in Alzheimer’s disease: mechanism and role [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 798-803. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||