Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (2): 200-207.doi: 10.12307/2022.933
Previous Articles Next Articles
Effects of different exercises on renal interstitial fibrosis in type 2 diabetic mice
Zhang Luyao1, Yang Kang2
- 1Department of Physical Education, Huaiyin Institute of Technology, Huaian 223023, Jiangsu Province, China; 2Department of Rehabilitation Medicine, Northern Jiangsu People's Hospital, Yangzhou 225001, Jiangsu Province, China
-
Received:2021-09-03Accepted:2021-10-14Online:2023-01-18Published:2022-06-20 -
Contact:Yang Kang, Master, Technician, Department of Rehabilitation Medicine, Northern Jiangsu People’s Hospital, Yangzhou 225001, Jiangsu Province, China -
About author:Zhang Luyao, Master, Associate professor, Department of Physical Education, Huaiyin Institute of Technology, Huaian 223023, Jiangsu Province, China -
Supported by:the Scientific Resarch Foundation of Northern Jiangsu Peoples Hospital, No. SBKY21010 (to YK)
CLC Number:
Cite this article
Zhang Luyao, Yang Kang. Effects of different exercises on renal interstitial fibrosis in type 2 diabetic mice[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 200-207.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
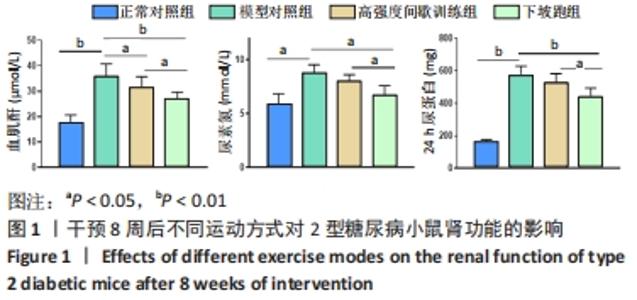
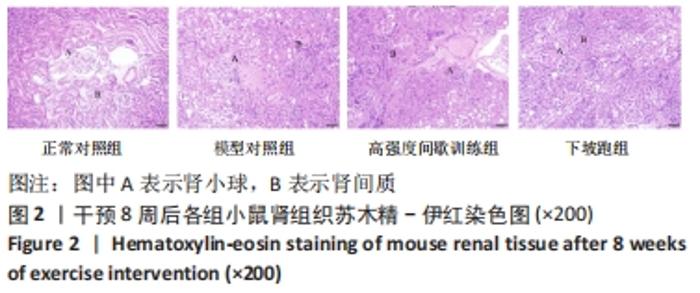
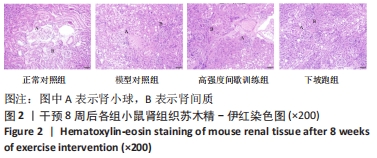
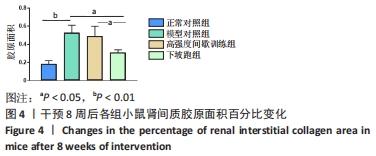
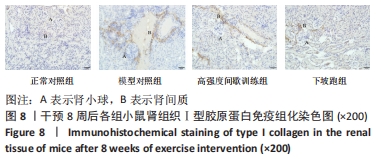
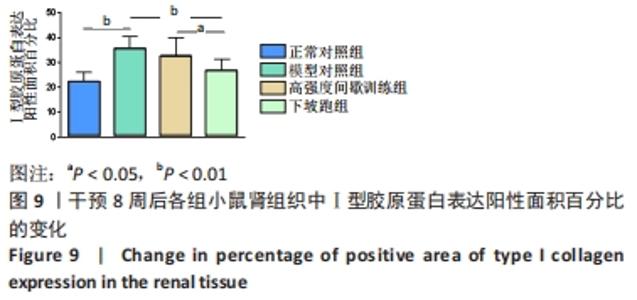
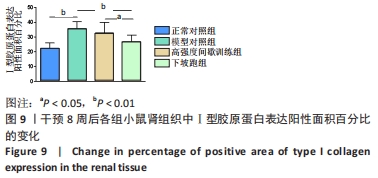
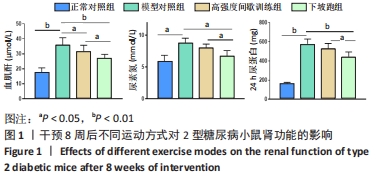
2.1 实验动物数量分析 实验期间,因糖尿病导致部分小鼠死亡及运动干预意外,最终各组纳入 9只样本统计。 2.2 不同方式运动对2型糖尿病小鼠肾功能的影响 干预8周后,与正常对照组相比,模型对照组血肌酐、尿素氮和24 h尿蛋白水平均显著升高(P < 0.05或P < 0.01)。与模型对照组相比,高强度间歇训练组血肌酐水平显著下降(P < 0.05),24 h尿蛋白、尿素氮水平下降但差异无显著性意义(P > 0.05);下坡跑组血肌酐、尿素氮和24 h尿蛋白水平均显著下降(P < 0.05或P < 0.01)。与高强度间歇训练组相比,下坡跑组血肌酐、尿素氮和24 h尿蛋白水平均显著下降(P < 0.05),见图1。"

2.5 不同方式运动对2型糖尿病小鼠肾组织中相关因子mRNA表达的影响 干预8周后,与正常对照组相比,模型对照组Klotho mRNA表达水平显著下调(P < 0.01),转化生长因子β1、Smad3、Ⅰ、Ⅲ型胶原蛋白 mRNA表达水平显著上调(P < 0.05或P < 0.01)。与模型对照组相比,高强度间歇训练组Klotho mRNA表达水平显著上调(P < 0.05),Ⅰ、Ⅲ型胶原蛋白 mRNA表达水平显著下调(P < 0.05),而转化生长因子β1和Smad3 mRNA表达水平下调但差异无显著性意义(P > 0.05);下坡跑组Klotho mRNA表达水平显著上调(P < 0.01),转化生长因子β1、Smad3、Ⅰ、Ⅲ型胶原蛋白 mRNA表达水平显著下调(P < 0.05或P < 0.01)。与高强度间歇训练组相比,下坡跑组Klotho mRNA表达水平显著上调(P < 0.05),转化生长因子β1、Ⅰ、Ⅲ型胶原蛋白 mRNA表达水平均显著下调(P < 0.05),Smad3 mRNA表达水平下调但差异无显著性意义(P > 0.05),见图5。"

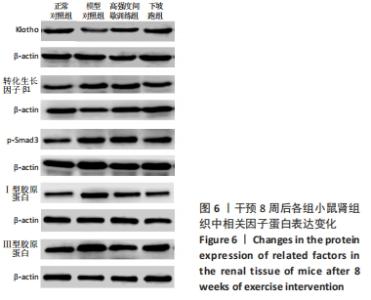
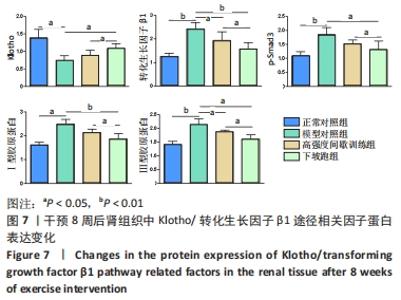
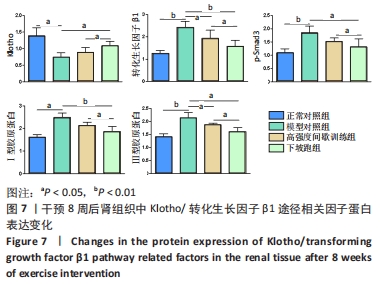
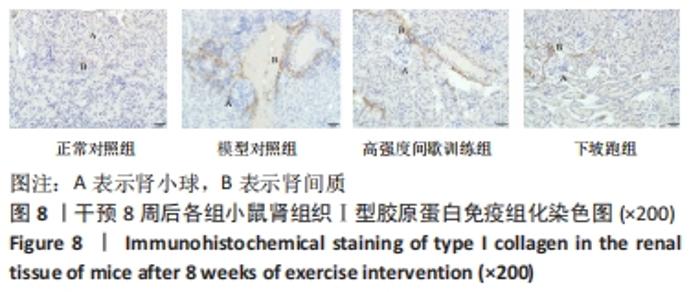
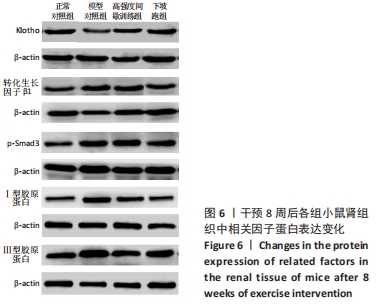
2.6 不同方式运动对2型糖尿病小鼠肾组织中相关因子蛋白表达的影响 干预8周后,与正常对照组相比,Klotho蛋白表达水平显著下调(P < 0.01),转化生长因子β1、p-Smad3、Ⅰ、Ⅲ型胶原蛋白的蛋白表达水平均显著上调(P < 0.05或P < 0.01)。与模型对照组相比, 高强度间歇训练组Klotho蛋白表达水平上调但差异无显著性意义(P > 0.05),转化生长因子β1、Ⅲ型胶原蛋白的蛋白表达水平均显著下调(P < 0.05),p-Smad3、Ⅰ型胶原蛋白的蛋白表达水平下调但差异无显著性意义(P > 0.05);下坡跑组Klotho蛋白的蛋白表达水平上调(P < 0.05),转化生长因子β1、p-Smad3、Ⅰ、Ⅲ型胶原蛋白的蛋白表达水平均显著下调(P < 0.05或P < 0.01)。与高强度间歇训练组相比,下坡跑组Klotho蛋白表达水平显著上调(P < 0.05),转化生长因子β1、p-Smad3、Ⅰ、Ⅲ型胶原蛋白的蛋白表达水平均显著下调(P < 0.05),见图6,7。"
| [1] HOU NN, KAN CX, HUANG N, et al. Relationship between serum Dickkopf-1 and albuminuria in patients with type 2 diabetes. World J Diabetes. 2021;12(1):47-55. [2] LYTVYN YL, BJORNSTAD P, RAALTE DH, et al. The new biology of diabetic kidney disease-mechanisms and therapeutic implications. Endocr Rev. 2020;41(2):202-231. [3] NI WJ, TANG LQ, ZHOU H, et al. Renoprotective effect of berberine via regulating the PGE 2-EP1-Gαq-Ca2+ signalling pathway in glomerular mesangial cells of diabetic rats. J Cell Mol Med. 2016; 20(8):1491-1502. [4] LIN Y, SUN ZJ. In Vivo Pancreatic β-Cell–specific expression of antiaging gene Klotho: A novel approach for preserving β-cells in type 2 diabetes. Diabetes. 2015;64(4):1444-1458. [5] HU Q, GAO LN, PENG B, et al. Baicalin and baicalein attenuate renal fibrosis in vitro via inhibition of the TGF-β1 signaling pathway. Exp Ther Med. 2017;14(4):3074-3080. [6] BEUSEKOM CD, ZIMMERING TM. Profibrotic effects of angiotensin II and transforming growth factor beta on feline kidney epithelial cells. J Feline Med Surg. 2019;21(8):780-787. [7] GUO J, ZHENG HJ, ZHANG WT, et al. Accelerated kidney aging in diabetes mellitus. Oxid Med Cell Longev. 2020;20(7):123-130. [8] HEYDARPOUR F, SAJADIMAJD S, MIRZARAZI E, et al. Involvement of TGF-β and autophagy pathways in pathogenesis of diabetes: A comprehensive review on biological and pharmacological insights. Front Pharmacol. 2020;30(11):498- 504. [9] 曹娇. 游泳运动通过TGF-β1/Smad3通路改善自发性高血压大鼠心肌间质纤维化研究[J]. 广州体育学院学报,2018,38(5):106-112. [10] TANG LX, WANG B, WU ZK. Aerobic exercise training alleviates renal injury by interfering with mitochondrial function in type 1 diabetic mice. Med Sci Monit. 2018;24(7):9081-9089. [11] 上官若男, 焦艺科, 尚画雨, 等. 8周游泳运动对2型糖尿病大鼠心肌间质纤维化的影响[J]. 中国康复医学杂志,2018,33(2):158-164. [12] YIN Q, LIU H. Connective tissue growth factor and renal fibrosis. Adv Exp Med Biol. 2019;35(12):365-380. [13] ITOH Y. Membrane-type matrix metalloproteinases: Their functions and regulations. Matrix Biol. 2015;44(5):207-223. [14] 姜辉. 耐力和抗阻运动对糖尿病大鼠肾脏间质纤维化的影响[D]. 成都:成都体育学院,2018. [15] LIU JX, ZHU L, LI PJ, et al. Effectiveness of high-intensity interval training on glycemic control and cardiorespiratory fitness in patients with type 2 diabetes: a systematic review and meta-analysis. Aging Clin Exp Res. 2019;31(5):575-593. [16] PHILIPPE M, GATTERER H, EDER EM, et al. The effects of 3 weeks of uphill and downhill walking on blood lipids and glucose metabolism in pre-diabetic men: a pilot study. J Sports Sci Med. 2017;16(1):35-43. [17] 陈祥和, 余慧琳, 徐帅, 等. AMPK介导运动改善2型糖尿病骨代谢紊乱的明星分子[J]. 中国骨质疏松杂志,2019,25(11):1650-1654. [18] LIAO P, HE QH, ZHOU X, et al. Repetitive bouts of exhaustive exercise induces a systemic inflammatory response and multi-organ damage in rats. Front Physiol. 2020; 31(11):685-694. [19] BARBALHO SM, FLATO UP, TOFANO RJ, et al. Physical exercise and myokines: relationships with sarcopenia and cardiovascular Complications. Int J Mol Sci. 2020;21(10):3607-3612. [20] PANIZO S, ARIAS LM, MONTES CA, et al. Fibrosis in chronic kidney disease: pathogenesis and consequences. Int J Mol Sci. 2021;22(1): 408-415. [21] AMARAL LS, SILVA FA, CORREIA VB, et al. Beneficial effects of previous exercise training on renal changes in streptozotocin-induced diabetic female rats. Exp Biol Med (Maywood). 2016;241(4):437-445. [22] 梁丽华, 唐海松, 秦明群, 等. 高脂高糖联合小剂量链尿佐菌素建立2型糖尿动物模型的研究[J]. 全科口腔医学电子杂志,2019, 20(6):163-165. [23] CHEN X, YANG K, JIN X, et al. Bone Autophagy: A Potential Way of Exercise-Mediated Meg3/P62/Runx2 Pathway to Regulate Bone Formation in T2DM Mice. Diabetes Metab Syndr Obes. 2021;4:2753-2764. [24] 陈祥和. 生长期下坡跑对去卵巢小鼠骨胶原蛋白、成骨细胞及相关因子的影响研究[D]. 上海:华东师范大学,2012. [25] SCHIMIZU H, MARUYAMA S, YUZAWA Y, et al. Anti-monocyte chemoattractant protein-1 gene therapy attenuated renal injury induced by protein-overload proteinuria. J Am Soc Nephrol. 2002; 14(6):1496-1505. [26] AMERICAN DIABETES ASSOCIATION. Microvascular complications and foot care: Standards of medical care in diabetes−2020. Diabetes Care. 2020;43(1):S135-S151. [27] KAN WC, HWANG JY, CHUANG LY, et al. Effect of osthole on advanced glycation end products-induced renal tubular hypertrophy and role of klotho in its mechanism of action. Phytomedicine. 2019;53:205-212. [28] HAYASHI D, UEDA S, YAMANOUE M, et al. Amelioration of diabetic nephropathy by oral administration of d-alpha-tocopherol and its mechanisms. Biosci Biotechnol Biochem. 2018;82(1):65-73. [29] RAPTIS AE, VIBERTI G. Pathogenesis of diabetic nephropathy. Ixp Clin Endocrinol Diabetes. 2001;109:S424-437. [30] 黎磊石,刘志红.中国肾脏病学[M].北京:人民军医出版社,2008: 706. [31] 张宝文, 雷香丽, 李瑾娜, 等. miR-21-5p靶向调控TIMP3抑制2型糖尿病肾病小鼠肾脏系膜细胞增殖及细胞外基质堆积[J].山东大学学报(医学版),2020,58(7):7-14. [32] XU SH, YI XP, LIU WY, et al. A review of nephrotoxicity of microcystins. Toxins (Basel). 2020;12(11):693-702. [33] YI XP, XU SH, HUANG FY, et al. Effects of chronic exposure to microcystin-LR on kidney in mice. Int J Environ Res Public Health. 2019; 16(24):5023-5030. [34] WANG J, ZHANG SW, LI XH, et al. LncRNA SNHG7 promotes cardiac remodeling by upregulating ROCK1 via sponging miR-34-5p. Aging (Albany NY). 2020;12(11):10441-10456. [35] LI HY, PENG X, WANG YT, et al. Atg5-mediated autophagy deficiency in proximal tubules promotes cell cycle G2/M arrest and renal fibrosis. Autophagy. 2016;12(9):1472-1486. [36] BAI XZ, LIU JQ, YANG LL, et al. Identification of sirtuin 1 as a promising therapeutic target for hypertrophic scars. Br J Pharmacol. 2016;173(10): 1589-1601. [37] MENCKE R, OLAUSON H, HILLEBRANDS J. Effects of Klotho on fibrosis and cancer: a renal focus on mechanisms and therapeutic strategies. Adv Drug Deliver Rev. 2017;12(1):85-100. [38] SUGIURA H, YOSHIDA T, SHIOHIRA S, et al. Reduced klotho expression level in kidney aggravates renal interstitial fibrosis. Am J Physiol Renal. 2012;30(2):1252-1264. [39] WU Y, XIE J, AN S, et al. Inhibition of trpc6 channels ameliorates renal fibrosis and contributes to renal protection by soluble klotho. Kidney Int. 2017;91(7):830-841. [40] DOI S, ZOU Y, TOGAO O, et al. Klotho inhibits transforming growth factor-β1 (TGF-β1) signaling and suppresses renal fibrosis and cancer metastasis in mice. J Biol Chem. 2011;286(10):8655-8665. [41] HUANG JS, CHUANG CT, LIU MH, et al. Klotho attenuates high glucose-induced fibronectin and cell hypertrophy via the ERK1/2-p38 kinase signaling pathway in renal interstitial fibroblasts. Mol Cell Endocrinol. 2014;39(10):45-53. [42] PAN Y, FENG TS, CHAN YC, et al. OR01-3 microRNA-34a-mediated FGF21 resistance in the adipose tissue contributes to insulin resistance and hypoadiponectinemia in diet-induced obesity. J Endocr Soc. 2019; 3(1):1-3. [43] YANG YL, XUE M, JIA YJ, et al. Long noncoding RNA NEAT1 is involved in the protective effect of Klotho on renal tubular epithelial cells in diabetic kidney disease through the ERK1/2 signaling pathway. Exp Mol Med. 2020;52(2):266-280. [44] RODRIGUES AM, SERRALHA RS, FARIAS C, et al. P2X7 receptor and klotho expressions in diabetic nephropathy progression. Purinergic Signal. 2018;14(2):167-176. [45] REFARDT J, WINZELER B, CRAIN MC. Copeptin and its role in the diagnosis of diabetes insipidus and the syndrome of inappropriate antidiuresis. Clin Endocrinol (Oxf). 2019;91(1):22-32. [46] STEIN SA, MALONEY KL, POLLIN TI. Genetic counseling for diabetes mellitus. Curr Genet Med Rep. 2014; 2(2):56-67. [47] KOU X, LI J, LIU X, et al. Swimming attenuates D-galactose- induced brain aging via suppressing miR-34a-mediated autophagy impairment and abnormal mitochondrial dynamics. J Appl Physiol. 2017;(122): 1462-1469. [48] SHEI RJ, MACKINTOSH KA, PEABODY JE, et al. Exercise physiology across the lifespan in cystic fibrosis. Front Physiol. 2019;23(10):1382-1391. [49] 张超, 吴广礼, 黄旭东, 等. PI3K/Akt信号通路在大鼠过度训练致急性肾损伤中的作用及山莨菪碱的干预效应[J]. 中国中西医结合肾病杂志,2018,19(1):3-6. [50] LIOU K, HO S, FILDES J, et al. High intensity interval versus moderate intensity continuous training in patients with coronary artery disease: a meta-analysis of physiological and clinical paramcters. Heart, lung circulation. 2016;25(2):166-174. [51] HUANG CC, LIN YY, YANG AL, et al. Anti-renal fibrotic effect of Exercise Training in Hypertension. Int J Mol Sci. 2018;19(2):613-622. [52] CHUNG KW, LEE EK, LEE MK, et al. Impairment of PPARα and the fatty acid oxidation pathway aggravates renal fibrosis during aging. J Am Soc Nephrol. 2018;29(4):1223-1237. [53] 丁华琳, 李扬扬, 于丰源, 等. 达格列净通过Klotho/TGF-β1通路抑制糖尿病肾病大鼠肾纤维化的作用[J]. 山东大学学报(医学版), 2020,58(3):75-80. [54] KIM DJ, KANG JM, PARK SH, et al. Diabetes aggravates post-ischaemic renal fibrosis through persistent activation of TGF-β1 and Shh signalling. Sci Rep. 2017;15(7):16782-16789. [55] YANG WQ, LIU L, WEI Y, et al. Exercise ameliorates the FGF21-adiponectin axis impairment in diet-induced obese mice. Endocr Connect. 2019;8(5):596-604. [56] SOUZA CS, OLIVEIRA BS, VIANA GN, et al. Preventive effect of exercise training on diabetic kidney disease in ovariectomized rats with type 1 diabetes. Exp Biol Med (Maywood). 2019;244(9):758-769. [57] LI J, QIU PC, HE QY, et al. Dencichine a meliorates kidney injury in induced type II diabeticnephr opathy via the TGF-B/Smad signalling pathway. Eur J Pharmacol. 2017;27(8):196-202. [58] JIA Q, YANG R, LIU XF, et al. Genistein attenuates renal fibrosis in streptozotocin-induced diabetic rats. Mol Med Rep. 2019;19(1):423-431. [59] 李燕. IRF-1通过抑制Klotho促进肾间质纤维化的机制研究[D]. 重庆:中国人民解放军陆军军医大学,2020. [60] 陈艳霞. LncRNA-SNHG29/miR-200b-3p调控Klotho/FGF23轴介导血管钙化的机制研究[D]. 南昌:南昌大学,2020. [61] KERKSICK CM, WILBORN CD, ROBERTS MD, et al. ISSN exercise & sports nutrition review update: research & recommendations. J Int Soc Sports Nutr. 2018;15(7):38-43. [62] SEDIGHI M, BALUCHNEJADMOJARAD T, FALLAH S, et al. The association between circulating Klotho and dipeptidyl peptidase-4 activity and inflammatory cytokines in elderly patients with alzheimer disease. Basic Clin Neurosci. 2020;11(3):349-357. [63] 李莎莎, 刘其锋, 何敖林. 表观遗传调控异常在慢性肾脏病Klotho减少中的作用[J]. 基础医学与临床,2019,39(9):1366-1370. |
| [1] | Wu Shuangshuang, Zhang Ying, Kou Xianjuan. Treadmill exercise improves cognitive dysfunction in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 208-215. |
| [2] | Shui Xiaoping, Li Chunying, Li Mingjuan, Li Shunchang, Sun Junzhi, Su Quansheng. Effects of aerobic and resistance exercise on antioxidant stress index and brain-derived neurotrophic factor expression in the hippocampus of type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 264-269. |
| [3] | Zhu Miaomiao, Kong Fanming, Zhao Qian. Exercise regulates lactic acid metabolism [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 322-328. |
| [4] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [5] | Gu Zhengqiu, Xu Fei, Wei Jia, Zou Yongdi, Wang Xiaolu, Li Yongming. Exploratory study on talk test as a measure of intensity in blood flow restriction training [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1154-1159. |
| [6] | Wu Cong, Jia Quanzhong, Liu Lun. Relationship between transforming growth factor beta1 expression and chondrocyte migration in adult articular cartilage after fragmentation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1167-1172. |
| [7] | Wang Qin, Shen Cheng, Liao Jing, Yang Ye. Dapagliflozin improves renal injury in diabetic nephropathy rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1216-1222. |
| [8] | Shui Xiaoping, Li Chunying, Li Shunchang, Sun Junzhi, Su Quansheng . Effects of aerobic and resistance exercises on brain-derived neurotrophic factor, nuclear factor-kappa B and inflammatory cytokines in skeletal muscle of type II diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 669-675. |
| [9] | Deng Shuang, Pu Rui, Chen Ziyang, Zhang Jianchao, Yuan Lingyan . Effects of exercise preconditioning on myocardial protection and apoptosis in a mouse model of myocardial remodeling due to early stress overload [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 717-723. |
| [10] | Wei Xing, Liu Shufang, Mao Ning. Roles and values of blood flow restriction training in the rehabilitation of knee joint diseases [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 774-779. |
| [11] | Tang Jiping, Zhang Yeting. Exercise regulates adult hippocampal neurogenesis in Alzheimer’s disease: mechanism and role [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 798-803. |
| [12] | Liu Yiyi, Qiu Junqiang, Yi Longyan, Zhou Cailiang. Effect of resistance training on interleukin-6 and C-reactive protein in middle-age and elderly people: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 804-812. |
| [13] | Mo Weibin, Huang Tianchang, Zeng Zhiwei, Yan Linbo. Effects of Pueraria lobata flavonoids on expressions of beta-catenin and glycogen synthase kinase 3beta in the brain of rats undergoing exhaustive exercise after long endurance exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 736-741. |
| [14] | Lin Jiayu, Huang Huibin, Liang Bo, Chen Lijun. Changes in irisin, leptin, adiponectin and visceral fat in obese rats undergoing high-intensity intermittent exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5583-5588. |
| [15] | Yang Ling, Huang Sen. Telomeres and telomerase: mechanism of exercise retarding aging telomeres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5733-5740. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||