Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (28): 4458-4465.doi: 10.12307/2022.298
Previous Articles Next Articles
Osteogenesis, angiogenesis and anti-aseptic loosening of europium-doped calcium polyphosphate bone tissue engineering scaffold
Wu Yuchong1, Peng Xu2, Yu Xixun1
- 1College of Polymer Science and Engineering, Sichuan University, Chengdu 610065, Sichuan Province, China; 2Laboratory Animal Center, Sichuan University, Chengdu 610065, Sichuan Province, China
-
Received:2021-02-27Accepted:2021-03-31Online:2022-10-08Published:2022-03-18 -
Contact:Yu Xixun, Professor, College of Polymer Science and Engineering, Sichuan University, Chengdu 610065, Sichuan Province, China -
About author:Wu Yuchong, Master candidate, College of Polymer Science and Engineering, Sichuan University, Chengdu 610065, Sichuan Province, China -
Supported by:the Key Research & Development Program of Sichuan Province, No. 2019YFS0121 (to YXX)
CLC Number:
Cite this article
Wu Yuchong, Peng Xu, Yu Xixun. Osteogenesis, angiogenesis and anti-aseptic loosening of europium-doped calcium polyphosphate bone tissue engineering scaffold[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4458-4465.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

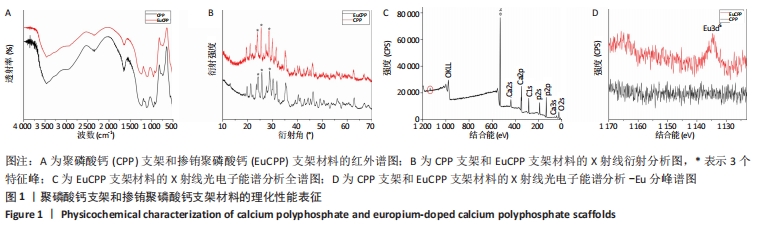
2.1 各组支架理化性质表征 从红外光谱图中可看到,EuCPP和聚磷酸钙的谱图没有明显差异,说明掺杂少量的铕离子并没有改变聚磷酸钙的原本主链结构,且EuCPP谱图中可见1 177 cm-1和1 235 cm-1处的O-P=O键的对称和非对称伸缩振动吸收峰,在732 cm-1处也有表示线性P-O-P键的峰出现,这说明EuCPP中具有直链[Ca(PO3)2]n结构,见图1A。X射线衍射分析图中EuCPP曲线中的特征峰与聚磷酸钙曲线完全相似,尤其是20°-30°之间的3个特征峰,通过与标准PDF卡(JCPD,77-1 953)进行比较表明EuCPP也为β晶型,见图1B。从X射线光电子能谱分析图中可见,5% EuCPP光谱内位于1 134.7 eV的结合能处有一个明显的Eu3d5峰,但在聚磷酸钙光谱中没有出现,这表明EuCPP材料中含有铕元素,即铕离子已经成功掺入,见图1C,D。"
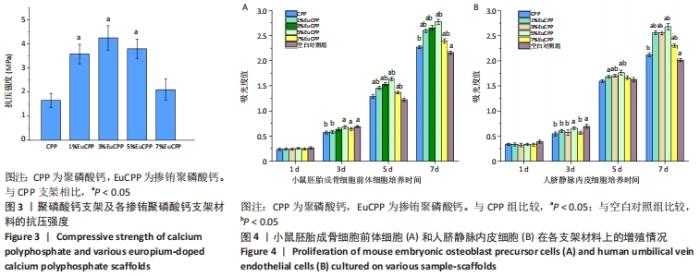
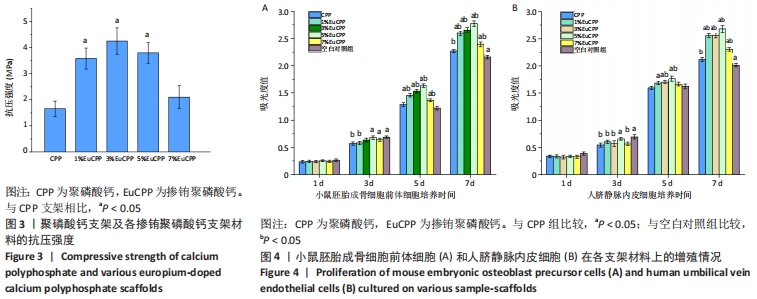
2.3 各组支架抗压强度测试结果 所有EuCPP支架的抗压强度均高于聚磷酸钙组,其中3% EuCPP的抗压强度最高(4.3 MPa),随着铕离子的掺杂含量进一步增加,EuCPP支架的强度略有下降;另外,5%EuCPP支架的抗压强度与3%EuCPP支架相当(P > 0.05),见图3。 2.4 各组支架上的细胞增殖情况 培养第1天,每组中的小鼠胚胎成骨细胞前体细胞数保持在相同水平,从第3天开始,所有EuCPP支架上培养的细胞数均高于聚磷酸钙支架,甚至在第5天和第7天显著高于空白对照组(P < 0.05);随着铕离子掺杂量的增加,每个时间点在相应支架上培养的小鼠胚胎成骨细胞前体细胞数也同步增加,在5%EuCPP组达到峰值,然后随着铕离子浓度的进一步升高,在EuCPP支架上培养的该细胞数量开始略有降低;在EuCPP支架上培养的人脐静脉内皮细胞也具有相似的增长趋势,见图4。"

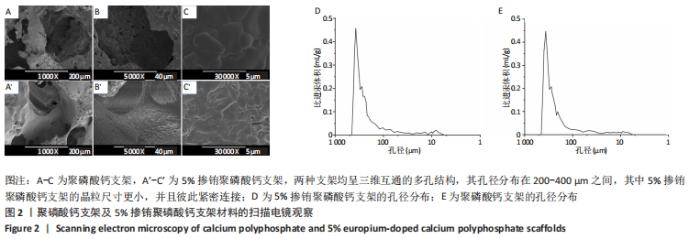
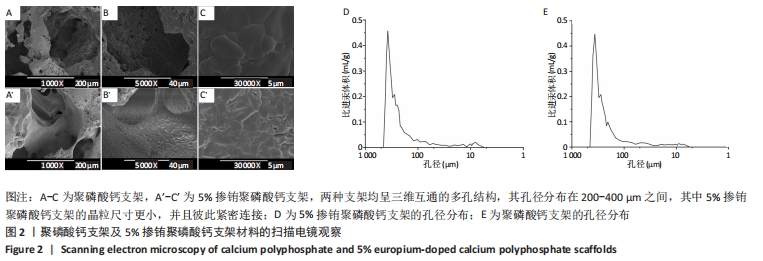
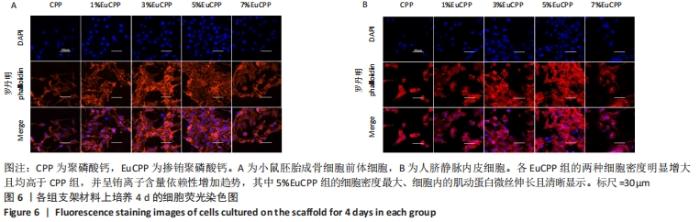
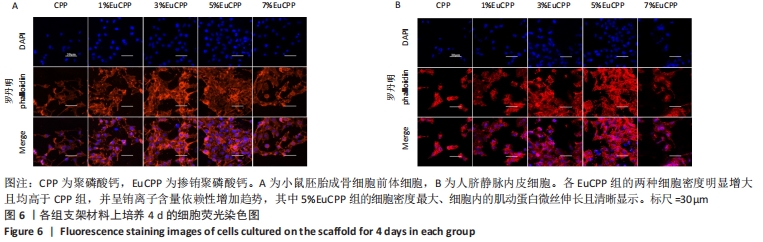
2.5 各组支架上的细胞形态和分布 因为扫描电镜和激光扫描共聚焦显微镜观察都可以直观反映细胞在支架上的增殖数量及附着状态,并且扫描电镜下观测的细胞增殖数量与CCK-8细胞增殖实验的结果是保持一致的,所以只选取了细胞增殖数量最多的5%EuCPP组与聚磷酸钙组进行比较。 从扫描电镜图片可见,5%EuCPP组的两种细胞数量均远高于聚磷酸钙组,聚磷酸钙支架上的小鼠胚胎成骨细胞前体细胞和人脐静脉内皮细胞彼此分离,细胞生存力较低,而在5%EuCPP支架上的小鼠胚胎成骨细胞前体细胞呈现出良好的成骨细胞形态,细胞伪足清晰可见且彼此紧密联系;5%EuCPP支架上的人脐静脉内皮细胞均为多角且生长良好,几乎形成连续的细胞层覆盖于材料表面,表现出良好的快速内皮化潜力,见图5。从激光扫描共聚焦显微镜图片也可见,各EuCPP组的两种细胞密度明显增大且均高于聚磷酸钙组,并呈铕离子含量依赖性增加趋势,其中5%EuCPP组的细胞密度最大,细胞内的肌动蛋白微丝伸长且清晰显示,见图6。"

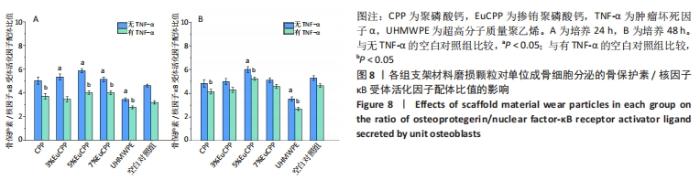
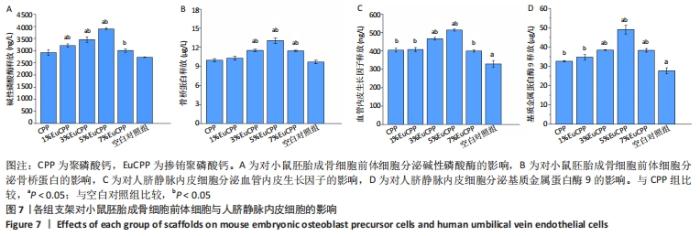
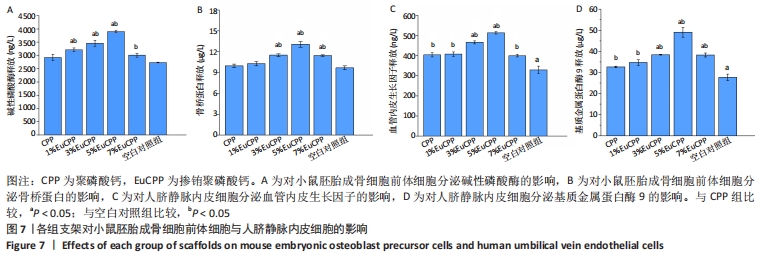
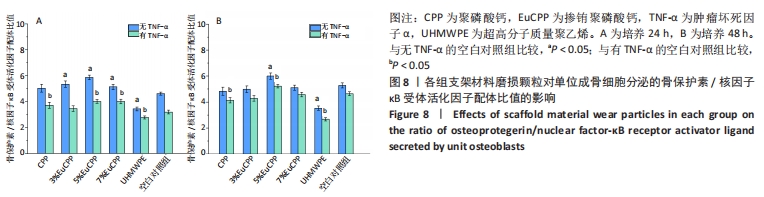
2.7 EuCPP磨损颗粒对小鼠胚胎成骨细胞前体细胞释放骨保护素和核因子κB受体活化因子配体的影响 因前文数据可得EuCPP的细胞相容性及对上述几种蛋白分泌的促进作用均呈铕剂量性依赖,在发现5% EuCPP表现出最佳性能后,想要探索它是否具有防治无菌性松动的潜能,故只选择了与其最接近的两个梯度分别为3%,7%EuCPP作为对照。 在24 h时,所有EuCPP组的骨保护素/核因子κB受体活化因子配体比值均高于空白对照组和超高分子质量聚乙烯组,而聚磷酸钙组和空白对照组之间无明显差异(P > 0.05),其中5% EuCPP组的比值最高,超高分子量聚乙烯组的比值最低;在各组加入肿瘤坏死因子α后,所有测试组中的骨保护素/核因子κB受体活化因子配体比值均降低,但是5% EuCPP组仍显示最高比值,超高分子质量聚乙烯组仍显示最低比例,见图8A。在48 h时,5%EuCPP组的骨保护素/核因子κB受体活化因子配体比值高于所有其他测试组,空白对照组的比值高于其他EuCPP组、聚磷酸钙组和超高分子量聚乙烯组,其中超高分子质量聚乙烯组仍显示最低比值;在加入肿瘤坏死因子α后,各组的骨保护素/核因子κB受体活化因子配体比值均略有下降,且趋势与没添加肿瘤坏死因子α时相似,见图8B。"
| [1] ANTALYA H, JOHANNA B, RUSTOM LE, et al. Bone regeneration strategies: engineered scaffolds, bioactive molecules and stem cells Current stage and future perspectives. Biomaterials. 2018;180:143-162. [2] HENCH LL, THOMPSON I. Twenty-first century challenges for biomaterials. J R Soc Interface. 2010;7(Suppl_4):379-391. [3] PILLIAR RM, FILIAGGI MJ, WELLS JD, et al. Porous calcium polyphosphate scaffolds for bone substitute applications — in vitro characterization. Biomaterials. 2001;22(9):963-972. [4] YOU C, LEE MH, LEE HJ, et al. The effect of macro/micro combination pore structure of biphasic calcium phosphate scaffold on bioactivity. Ceram Int. 2017;43(4):3540-3546. [5] KIM TG, SHIN H, DONG WL. Biomimetic Scaffolds for Tissue Engineering. Adv Funct Mater. 2012;22(12):2446-2468. [6] WU C, CHEN Z, YI D, et al. Multidirectional effects of Sr-, Mg-, and Si-containing bioceramic coatings with high bonding strength on inflammation, osteoclastogenesis, and osteogenesis. ACS Appl Mater Interfaces. 2014;6(6):4264-4276. [7] GUO B, LEI B, LI P, et al. Functionalized scaffolds to enhance tissue regeneration. Regen Biomater. 2015;2(1):47-57. [8] RATNAYAKE JTB, GOULD ML, SHAVANDI A, et al. Development and characterization of a xenograft material from New Zealand sourced bovine cancellous bone. J Biomed Mater Res B Appl Biomater. 2017; 105(5):1054-1062. [9] HETTICH G, SCHIERJOTT RA, EPPLE M, et al. Calcium Phosphate Bone Graft Substitutes with High Mechanical Load Capacity and High Degree of Interconnecting Porosity. Materials (Basel). 2019;12(21):3471. [10] BOSE S, FIELDING G, TARAFDER S, et al. Understanding of dopant-induced osteogenesis and angiogenesis in calcium phosphate ceramics. Trends Biotechnol. 2013;31(10):594-605. [11] WANG X, GU Z, JIANG B, et al. Surface modification of strontium-doped porous bioactive ceramic scaffolds via poly(DOPA) coating and immobilizing silk fibroin for excellent angiogenic and osteogenic properties. Biomater Sci. 2016;4(4):678-688. [12] VASCONCELOS DM, SANTOS SG, LAMGHARI M, et al. The two faces of metal ions: From implants rejection to tissue repair/regeneration. Biomaterials. 2016;84:262-275. [13] LI Q, CHEN H, HUANG X, et al. Effects of 12 metal ions on iron regulatory protein 1 (IRP-1) and hypoxia-inducible factor-1 alpha (HIF-1α) and HIF-regulated genes. Toxicol Appl Pharmacol. 2006;213(3): 245-255. [14] STEGEN S, DEPREZ S, EELEN G, et al. Adequate hypoxia inducible factor 1α signaling is indispensable for bone regeneration. Bone. 2016; 87:176-186. [15] QIU K, ZHAO XJ, WAN CX, et al. Effect of strontium ions on the growth of ROS17/2.8 cells on porous calcium polyphosphate scaffolds. Biomaterials. 2006;27(8):1277-1286. [16] WANG X, WANG Y, LI L, et al. Stimulations of strontium-doped calcium polyphosphate for bone tissue engineering to protein secretion and mRNA expression of the angiogenic growth factors from endothelial cells in vitro. Ceram Int. 2014;40(5):6999-7005. [17] PATRA CR, BHATTACHARYA R, PATRA S, et al. Pro-angiogenic Properties of Europium(III) Hydroxide Nanorods. Adv Mater. 2008;20(4):753-756. [18] PATRA CR, ABDEL MONEIM SS, WANG E, et al. In vivo toxicity studies of europium hydroxide nanorods in mice. Toxicol Appl Pharmacol. 2009; 240(1):88-98. [19] BARTA CA, SACHS-BARRABLE K, JIA J, et al. Lanthanide containing compounds for therapeutic care in bone resorption disorders. Dalton Trans. 2007;(43):5019-5030. [20] ZHONG M, ZHANG X, SHI X, et al. Halofuginone inhibits LPS-induced attachment of monocytes to HUVECs. Int Immunopharmacol. 2020; 87:106753. [21] 刘广建,岳文贞,陈荣春.UHMWPE用于生物工程的摩擦磨损研究[J].工程塑料应用,2002,30(1):36-38. [22] FANG HW, HO YC, YANG CB, et al. Preparation of UHMWPE particles and establishment of inverted macrophage cell model to investigate wear particles induced bioactivites. J Biochem Biophys Methods. 2006; 68(3):175-187. [23] BALOGH E, PARAGH G, JENEY V. Influence of Iron on Bone Homeostasis. Pharmaceuticals (Basel). 2018;11(4):107. [24] GARCÍA-GARETA E, COATHUP MJ, BLUNN GW. Osteoinduction of bone grafting materials for bone repair and regeneration. Bone. 2015;81: 112-121. [25] SOUS M, BAREILLE R, ROUAIS F, et al. Cellular biocompatibility and resistance to compression of macroporous beta-tricalcium phosphate ceramics. Biomaterials. 1998;19(23):2147-2153. [26] MEI L, HONG F, LI W, et al. Size-dependent microstructure and europium site preference influence fluorescent properties of Eu3+-doped Ca10(PO4)6(OH)2 nanocrystal. J Lumin. 2008;128(3):428-436. [27] MIAO G, CHEN X, MAO C, et al. Synthesis and characterization of europium-containing luminescent bioactive glasses and evaluation of in vitro bioactivity and cytotoxicity. J Solgel Sci Technol. 2014;69(2): 250-259. [28] SONG W, TIAN M, CHEN F, et al. The study on the degradation and mineralization mechanism of ion-doped calcium polyphosphate in vitro. J Biomed Mater Res B Appl Biomater. 2009;89(2):430-438. [29] ZHAO R, XIE P, ZHANG K, et al. Selective effect of hydroxyapatite nanoparticles on osteoporotic and healthy bone formation correlates with intracellular calcium homeostasis regulation. Acta Biomater. 2017; 59:338-350. [30] JENSEN T, BAAS J, DOLATHSHAHI-PIROUZ A, et al. Osteopontin functionalization of hydroxyapatite nanoparticles in a PDLLA matrix promotes bone formation. J Biomed Mater Res A. 2011;99(1):94-101. [31] LIU F, ZHANG X, YU X, et al. In vitro study in stimulating the secretion of angiogenic growth factors of strontium-doped calcium polyphosphate for bone tissue engineering. J Mater Sci Mater Med. 2011;22(3):683-692. [32] HEGDE PS, WALLIN JJ, MANCAO C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immunotherapeutics. Semin Cancer Biol. 2018;52(Pt 2):117-124. [33] ADYA R, TAN BK, PUNN A, et al. Visfatin induces human endothelial VEGF and MMP-2/9 production via MAPK and PI3K/Akt signalling pathways: novel insights into visfatin-induced angiogenesis. Cardiovasc Res. 2008;78(2):356-365. [34] WU C, XIA L, HAN P, et al. Europium-Containing Mesoporous Bioactive Glass Scaffolds for Stimulating in Vitro and in Vivo Osteogenesis. ACS Appl Mater Interfaces. 2016;8(18):11342-11354. [35] HOLLBORN M, STATHOPOULOS C, STEFFEN A, et al. Positive feedback regulation between MMP-9 and VEGF in human RPE cells. Invest Ophthalmol Vis Sci. 2007;48(9):4360-4367. [36] PARK JH, LEE NK, LEE SY. Current Understanding of RANK Signaling in Osteoclast Differentiation and Maturation. Mol Cells. 2017;40(10):706-713. [37] CHEN X, WANG Z, DUAN N, et al. Osteoblast-osteoclast interactions. Connect Tissue Res. 2017;59(2):99-107. [38] KENKRE JS, BASSETT J. The bone remodelling cycle. Ann Clin Biochem. 2018;55(3):308-327. [39] BOYCE BF, XING L. Functions of RANKL/RANK/OPG in bone modeling and remodeling. Arch Biochem Biophys. 2008;473(2):139-146. [40] 陈昱铭,曾晖,熊奡,等.TNF-α对人滑膜细胞RANKL、OPGmRNA表达的影响[J].中国现代医学杂志,2009,19(9):1301-1303,1307. [41] CHILDS LM, GOATER JJ, O’KEEFE RJ, et al. Efficacy of etanercept for wear debris-induced osteolysis. J Bone Miner Res. 2001;16(2):338-347. [42] BLADEN CL, TZU-YIN L, FISHER J, et al. In vitro analysis of the cytotoxic and anti-inflammatory effects of antioxidant compounds used as additives in ultra high-molecular weight polyethylene in total joint replacement components. J Biomed Mater Res B Appl Biomater. 2013; 101(3):407-413. [43] HACCHOU Y, UEMATSU T, UEDA O, et al. Inorganic polyphosphate: a possible stimulant of bone formation. J Dent Res. 2007;86(9):893-897. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [2] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1005-1011. |
| [3] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [4] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [5] | Lu Yunan, Zhang Xinzhao, Lin Binbin, Xu Gan, Chen Jingdi, Chen Shunyou. Naringin-chitosan/hydroxyapatite composite scaffold in repair of rat skull defect [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4441-4445. |
| [6] | He Jiachen, Liu Chang, Chen Chichi, Shi Qin. Preparation and in vitro evaluation of injectable microspheres loaded with cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4483-4488. |
| [7] | Lu Renpei, Zou Zhichen, Zhao Fengnian, Zheng Changming, Song Keguan. Role and application of polylactic acid-hydroxyacetic acid copolymer scaffolds in bone defect repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4525-4531. |
| [8] | Wang Jing, Yang Jiuju, Wang Ningning, Liu Chao. Effect of modified three-dimensional-printed titanium scaffold on osteogenic differentiation of adipose-derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4265-4271. |
| [9] | Sun Kai, Li Ruixin, Gao Lilan, Fan Meng, Zhang Xizheng, Li Hui. Biocompatibility of silk fibroin/type II collagen/hydroxyapatite three-phase composite scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4283-4287. |
| [10] | Chen Yue, Luo Feng, Su Dan, Yang Diyuan, Pu Qi, Zhang Min, Gui Wenjuan, Sun Xiaorong. Hemostatic efficacy, degradation in vivo and biological evaluation of fish scale collagen [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4353-4359. |
| [11] | Zhao Doudou, Lin Kaili. Application of multicellular construction of vascularized tissue engineered bone in bone repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(27): 4386-4392. |
| [12] | Zhang Zihan, Wang Wenli, Li Jinnuo, Li Yourui. Carvacrol: antibacterial activity, bone repair, and prevention and treatment in oral diseases [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4252-4257. |
| [13] | Chen Chichi, Zhang Yu, He Jiachen, Shi Qin. Osteogenic differentiation of bone marrow mesenchymal stem cells in obese mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3846-3851. |
| [14] | Chen Na, Wang Xiaohan, Zhang Yunke. Effect and mechanism of mesenchymal stem cells on aging-related ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3914-3920. |
| [15] | Han Zhi, Wang Zhimiao, Gaxi Sijia, Lu Qingling, Guo Tao. Tissue engineered cartilage constructed by polyurethane composite chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3455-3459. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||