Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (1): 106-111.doi: 10.12307/2022.018
Previous Articles Next Articles
Biological function of circular RNAs in osteogenic differentiation of mesenchymal stem cells
Kang Yue1, Liu Jie2
- 1Liaoning Cancer Hospital & Institute, Shenyang 110042, Liaoning Province, China; 2China Medical University, Shenyang 110001, Liaoning Province, China
-
Received:2020-02-27Revised:2020-03-05Accepted:2020-10-24Online:2022-01-08Published:2021-10-25 -
About author:Kang Yue, MD, Attending physician, Liaoning Cancer Hospital & Institute, Shenyang 110042, Liaoning Province, China -
Supported by:Science and Technology Project of Shenyang, No. F16-102-4-00 (to LJ)
CLC Number:
Cite this article
Kang Yue, Liu Jie. Biological function of circular RNAs in osteogenic differentiation of mesenchymal stem cells[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 106-111.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
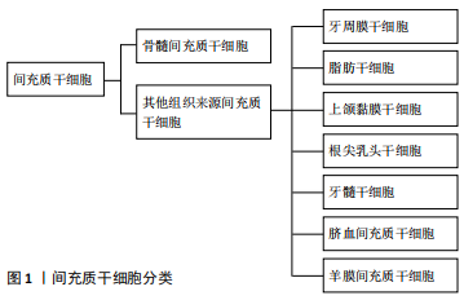
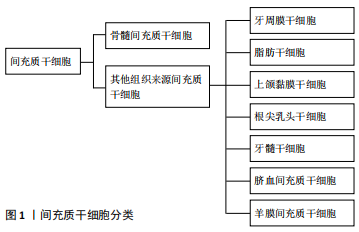
2.1 环状RNA简介 环状RNA是封闭的环状分子,不具有5'至3'极性的共价闭环结构或3′polyA末端[16]。SANGER等[2]在20世纪70年代发现某些高等植物中存在可致病的单链环状类病毒,这是人类首次发现环状RNA,继而陆续在丁型肝炎病毒、酵母线粒体中发现环状RNA。环状RNA是由一种非经典剪接方式-反向剪接产生;当反向剪接发生时,下游剪接位点与上游剪接位点共价连接闭合。随着生物技术的进步,特别是生物信息学和高通量测序技术的发展,人们已经鉴定了大量的环状RNA。事实上,环状RNA是丰富、多样和保守的分子,通常在组织表达和发育阶段具有特定的方式[17-19]。 多数环状RNA由蛋白编码基因产生,由单个外显子或者多个外显子构成,定位于细胞质中。也有部分环状RNA是由内含子环化而成的套索结构。内部的内含子保留可能导致产生含来自外显子和内含子序列的环状RNA,并且在经典的剪接过程中内含子套索的脱支失败导致环状内含子RNA产生;外显子-内含子环状RNA和环状内含子RNA位于细胞核中,这些环状RNA通过与U1小核核糖核蛋白的相互作用或通过正调节RNA聚合酶Ⅱ介导的转录促进其亲本基因的转录。 2.2 环状RNA的生成和种类 环状RNA区别于线性RNA的结构是具有闭合环状结构[20-21]。环状RNA主要分为3类,包括外显子-内含子环状RNA(EIciRNAs)、环状内含子RNA(ciRNA) 和外显子环状RNA(ecircRNAs)。环状RNA的产生机制和过程尚有待实验研究,其生成过程区别于一般线性RNA经典剪切方式,主要由反向剪接产生;当反向剪接发生时,下游剪接位点与上游剪接位点共价连接闭合[22]。环状RNA主要通过“内含子配对驱动环化”机制和“外显子跳读”机制合成[23-25]。另外,外显子跳跃期间形成套索(可替换的外显子从mRNA中剪接出来,并最终包含在切除的套索内),当套索被反向剪切时,可导致环状RNA的形成。最后,未被套索脱支酶水解的内含子套索会形成环状内含子RNA[26]。许多环状RNA中涉及反向剪接的外显子倾向于有很长的内含子侧翼,这些环状RNA一般来自具有高活性启动子的基因。另外,组蛋白和基因体区域上表观遗传的改变影响可变剪接,也可能会对环状RNA和生物发生有直接影响。总之,环状RNA的形成非常复杂,可能存在许多未知的形成机制。 2.3 环状RNA的生物学特征 2.3.1 环状RNA的分布及种类特点 环状RNA广泛存在于真核转录组中,且种类众多,是比mRNA更为主要的转录本,其丰度更高,在母基因转录本中有5%-10%为环状RNA[27-28]。 目前有研究表明环状RNA含量甚至可以达到其宿主基因表达量的几十倍[18]。近年来,高通量RNA测序(RNA-seq)和环状RNA特异性生物信息学算法已经在真核生物中鉴定了数千种环状RNA,包括真菌、原生生物、植物、蠕虫、鱼类、昆虫和哺乳动物,并且发现它们具有组织特定的表达模式。 2.3.2 环状RNA的生物特性 环状RNA不易被核酸外切酶 Rnase R降解,因此在人体内较线性RNA更具稳定性。外显子环状RNA在细胞中非常稳定,在大多数物种中的半衰期超过48 h,而mRNA的平均半衰期为10 h。但是,外显子环状RNA在血清中不稳定,半衰期<15 s,可能是由于循环RNA核酸内切酶引起的。由于这种稳定性,一些外显子环状RNA比线性RNA基因产物的含量更高[29]。 2.3.3 环状RNA的组织时序特异性 由于环状RNA在一些特定的疾病、组织或阶段有特异性的表达,可以充当临床疾病诊断评估的特异性指标。例如癌症的发生通常伴随着环状RNA表达谱的改变。CHEN等[30]通过测序和注释144个局部前列腺肿瘤,共鉴定出76 311种不同的环状RNA,这些环状RNA与癌细胞的侵袭性有关;此外,在76 311个环状RNA中,有171个环状RNA对前列腺癌细胞的增殖至关重要,这表明局限性前列腺癌受到环状RNA的广泛调控。另有报道,诸如动脉粥样硬化的心血管疾病受环状ANRIL(circANRIL)的调节。BURD等[31]发现含有邻近INK4/ARF外显子的ANRIL亚型的表达与动脉粥样硬化密切相关。环状RNA在哺乳动物神经元组织中高度富集,说明它们可能在中枢神经系统的调节中起重要作用。在阿尔茨海默病中,泛素结合酶E2A(UBE2A)是一种有自体吞噬作用的蛋白,其重要功能是清除阿尔茨海默病患者脑内的淀粉样蛋白。CDR1as作为miR-7的海绵,可提高miR-7靶基因表达水平。因此,CDR1as在大脑中表达减少时,会引起miR-7靶基因UBE2A表达水平下降,加重阿尔茨海默病的发展[32]。环状RNA在急性髓细胞性白血病中也起着关键作用。例如,WU等[33]研究提示在急性髓细胞性白血病组织和细胞中上调的circRNA-DLEU2通过抑制miR-496和促进PRKACB的表达,促进急性髓细胞性白血病细胞增殖并抑制细胞凋亡。 2.3.4 环状RNA的生物进化保守性 环状RNA数量庞大,研究显示大部分环状RNA在细胞进化过程中显示出很强的保守性。作为非编码RNA的另一种类型,内源性环状RNA保留了相似的基因组结构。外显子环状RNA、内含子环状RNA可以通过2'-5'接合点来进一步区分。外显子环状RNA不具有2'-5'连接而由3'-5'连接组成。这些特征表明特定的反向剪接使其具有高度保守性。 2.4 环状RNA的生物学功能 2.4.1 通过竞争性抑制参与miRNA功能的调节 miRNA是一类非编码RNA,约含20多个核苷酸,并与mRNA的3'-非翻译区结合以调控其翻译。有越来越多的证据表明,环状RNA作为miRNA的海绵来调节mRNA的表达[34-37]。首先被验证为miRNA海绵的环状RNA是CDR1as(ciRS-7)和circSry[38-39]。 HANSEN等[40]于2013年研究发现CDR1as在人和小鼠的大脑中高度表达,包含70多个保守的miR-7结合位点,它是miR-7的环状抑制剂,对miR-7起到负调控作用。CircSry是一种睾丸特异性环状RNA,包含16个特定的miR-138结合位点,可作为miR-138海绵[38]。近几年,环状 RNA 作为 miRNA 海绵在成骨分化中发挥作用的研究逐渐涌现。CircRNA436在牙周膜干细胞中显著下调,并且通过相互作用网络分析发现它与miR-107和miR-335均相关[41]。据报道,miR‐107可能通过靶向Wnt/β-catenin来下调Dkk-1以抑制骨肉瘤的发生和发展[27]。miR-335还通过Wnt/β-catenin途径调节间充质干细胞的成骨分化[42]。因此,circRNA436可能通过影响Wnt/β-catenin途径而成为牙周膜干细胞分化的关键调节因子之一。研究表明,hsa_circ_0127781可以与miR-210和miR-335相互作用[43]。miR-210是通过抑制1B型激活素A受体而成为成骨细胞分化的积极调节剂之一[44]。此外, miR-335通过下调Dkk-1来促进成骨分化,从而激活Wnt信号传导[45]。因此,hsa_circ_0127781可能在抑制成骨细胞分化中发挥作用。已验证CircIGSF11可以延缓骨髓间充质干细胞的成骨过程,并且与miR-199b-5p呈负相关。miR-199b-5p可以通过靶向GSK-3β刺激成骨细胞分化[46]。 2.4.2 环状RNA参与调控mRNA前体影响蛋白质合成 环状RNA通过与mRNA调节的结合蛋白结合,从而形成大的RNA-蛋白复合体(RPCs)。这些RNA-蛋白复合体可以与基因的线性转录物相互作用。例如,蛋白质复合体盲肌(muscleblind, MBL or MBNL1)通过结合其亲本基因第2外显子而环化形成circMbl,凭借其两侧翼上内含子包含的MBL结合位点再与MBL结合,参与调控环状RNA的合成。外显子环化与经典的pre-mRNA剪接之间是竞争关系,最终使环化与线性剪接处于一种平衡状态。这些证明环状RNA参与调解pre-mRNA的剪接,影响蛋白质的合成[47],其他环状RNA,例如circPABPN1和circANRIL也通过与靶蛋白结合起作用。circPABPN1通过螯合人宫颈癌HeLa细胞中的RBP Hu抗原R(HUR)来抑制核多聚(A)结合蛋白1(PABPN1)mRNA的翻译,而circANRIL通过与pescadillo同源1(PES1)结合而损伤rRNA前体加工和核糖体生物发生。PES1是人血管平滑肌细胞和巨噬细胞中一种重要的60S核糖前体组装因子。 circANRIL诱导核仁应激和p53的激活,可能是通过从动脉粥样硬化斑块中去除过度增殖细胞来防止动脉粥硬化形成 。 2.4.3 环状RNA参与调控基因的转录 内含子环状RNA的形成取决于其侧翼的内含子,内含子能形成套索结构而不同于其他分支结构,内含子环状RNA大量存在于细胞核内,主要以顺式反应参与调节宿主转录。外显子-内含子 RNA也主要存在于细胞核内,这种类型的环状 RNA可以通过与U1小细胞核RNA的RNA-RNA相互作用,同U1小细胞核核糖核蛋白相结合,促进了RNA聚合酶Ⅱ的聚集,由此提高亲本基因的转录水平。环状泛素蛋白连接酶E3是一种环状RNA,和miR-7、miR-17及miR-214的相互作用可提高泛素蛋白连接酶E3表达水平[48]。 2.4.4 环状RNA翻译成蛋白质 环状RNA缺乏帽依赖性翻译的必需元件,例如5'帽和poly(A)尾。然而,环状RNA的帽非依赖性翻译可以通过内部核糖体进入位点(IRES)或在5'非翻译区(UTR)中加入m6A RNA修饰后发生。尽管预计有数千种环状RNA含有假定的开放阅读框(ORF)和上游IRES,但迄今为止,只有少数内源性环状RNA已被证明可作为蛋白质模板,例如circ-ZNF609,circMb1,circFBXW7,circPINTexon2和circ-SHPRH。大多数环状RNA衍生肽的功能相关性仍然未知。由于环状RNA衍生的肽通常是缺乏必需功能结构域的标准蛋白的截短形式,它们可能作为替代蛋白复合物的显性-阴性蛋白变体、诱饵或调节剂。 2.5 人间充质干细胞的成骨分化研究进展 2.5.1 间充质干细胞的简介 间充质干细胞最早是被 FRIEDENSTEIN在骨髓中发现的,随后还发现存在于人体发生、发育过程的许多种组织中,并在机体损伤组织再生中发挥关键作用[49]。间充质干细胞在适宜的体内或体外环境下具有分化为肌细胞、肝细胞、成骨细胞、脂肪细胞、软骨细胞、基质细胞等多种细胞的能力[50-51]。在组织工程和再生医学中,间充质干细胞被认为是理想的细胞来源,可以在体外快速扩展为多个谱系,并保持其分化潜能[52]。此外,它们可以与不同的天然或合成生物材料联合应用于临床前和临床研 究[53-54]。研究表明,在生物包埋后,间充质干细胞可以保留其干性,这与其低免疫原性特征和异体抗原限制的表达有关[55]。 脂肪组织和骨髓是最容易获得的间充质干细胞来源,其他组织来源的间充质干细胞还包括牙周膜干细胞、上颌窦黏膜干细胞、根尖乳头干细胞、牙髓干细胞、脐血间充质干细胞、羊膜间充质干细胞等,见图1。"
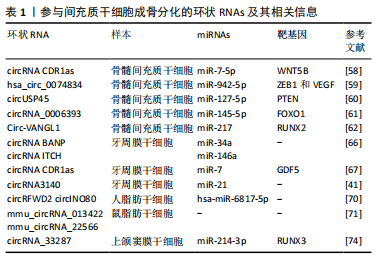
2.5.2 环状RNA参与骨髓间充质干细胞成骨分化的研究 骨髓间充质干细胞是一种骨髓来源的间充质干细胞,在一定诱导条件下可相应分化为成骨细胞、软骨细胞、脂肪细胞等,并且能维持和修复结缔组织[56]。大量研究表明骨髓间充质干细胞可以诱导分化为成骨细胞,修复受损的骨组织,实现骨再生。骨髓间充质干细胞在骨组织工程的应用广泛,与其他间充质干细胞相比,对骨髓间充质干细胞成骨机制的研究相对较多[57]。目前一些研究发现,环状RNA 参与并调控骨髓间充质干细胞的成骨分化。CHEN等[58]研究发现circRNA CDR1as可能通过CDR1as-miR-7-5p-WNT5B轴在激素性股骨头坏死来源骨髓间充质干细胞的成脂/成骨分化障碍中发挥关键作用。敲低CDR1as促进成骨分化而抑制成脂分化,过表达CDR1as抑制成骨分化而促进成脂分化。萤光素酶报告基因检测证实了CDR1as和WNT5B的miR-7-5p结合位点,为激素性股骨头坏死来源骨髓间充质干细胞成骨/成脂分化异常的分子机制提供新的见解,为激素性股骨头坏死的诊断和治疗提供新的生物标记。OUYANG等[59]分离骨质疏松患者骨髓间充质干细胞后,使用环状RNA微阵列检测环状RNA在骨髓间充质干细胞中的表达,其中hsa_circ_0074834的表达降低,其充当ceRNA,通过miRNA-942-5p调节ZEB1和VEGF的表达。Hsa_circ_0074834可促进骨髓间充质干细胞的成骨分化和骨缺损的修复,是治疗骨不连的关键靶标。KUANG等[60] 研究发现circUSP45的过表达降低了成骨基因的表达,抑制了骨髓间充质干细胞的增殖。此外,circUSP45主要位于细胞质中,并与miR-127-5p直接相互作用。MiR-127-5p 与其靶标PTEN共同作用来调节成骨作用。WANG等[61]研究证明了circRNA_0006393通过竞争性结合miR-145-5p并上调FOXO1来促进糖皮质激素诱发的骨质疏松症的成骨作用。YANG等[62]发现骨髓间充质干细胞中Circ-VANGL1通过竞争性结合miRNA-217调节RUNX2表达来促进骨质疏松症的发展。总之,骨髓间充质干细胞在特定条件下可分化为成骨细胞,可应用于骨组织工程,促进骨缺损的修复。 2.5.3 环状RNA参与牙周膜干细胞成骨分化的研究 牙髓干细胞是人类牙齿中最早分离出的间充质干细胞,具有在体外和体内分化为成牙本质细胞、成骨细胞、软骨细胞、肌细胞和脂肪细胞以及神经细胞的潜力。在特定的体外培养条件下,牙周膜干细胞能够分化为成脂细胞和成骨细胞,并且可以在体内介导牙周膜的再生,是牙周和牙槽骨组织再生的候选细胞[63-64]。ZHENG等[65]分离牙周膜干细胞并在成骨培养基中培养,然后进行RNA测序,以检测环状RNA、mRNA的表达谱,共检测到12 693个环状RNA转录本,并且环状RNA显示出阶段特异性表达,它们在调节细胞外囊泡的生物发生中发挥潜在作用。GU 等[66]对成骨分化前后的牙周膜干细胞进行高通量测序,通过生物信息分析构建环状RNA调控网络,并筛选出了编码为TCONS_00212979和TCONS_00212984的长链非编码RNA,以及circRNA BANP和circRNA ITCH,可能与miRNA-34a和miRNA-146a相互作用,通过MAPK途径调节牙周膜干细胞成骨分化。LI等[67]观察到在成骨分化过程中CDR1as显著上调,而miR-7显著下调。实验证明CDR1as 通过吸附 miR-7 增强了靶基因GDF5 表达,进而促进Smad1/5/8和p38 MAPK 磷酸化使牙周膜干细胞成骨向分化。WANG等[41] 研究发现circRNA3140与miRNA-21在机械力诱导牙周膜干细胞成骨分化中起关键作用,可以调节正畸牙齿移动过程和牙槽骨重塑。 2.5.4 环状RNA参与脂肪干细胞成骨分化的研究 脂肪干细胞是间充质干细胞中的一种,具有多向分化和自我更新能力。与骨髓间充质干细胞相比,脂肪干细胞来源广泛、含量较多、获取方式简单、患者承受的痛苦也较少。脂肪干细胞具有多向分化的能力和较明确的成骨潜能。在适当的环境下,脂肪干细胞可以分化为脂肪细胞、成骨细胞、成软骨细胞以及肌细胞等。脂肪干细胞是组织工程中理想的干细胞来源[68],并被认为是促进骨骼形成和再生的种子细胞[69]。HUANG等[70] 研究发现circRFWD2和circINO80在重组NELL-1诱导的脂肪干细胞成骨过程中被上调,而敲低它们会影响NELL-1对成骨的积极作用。CircRFWD2和circINO80可以与hsa-miR-6817-5p 相互作用,从而抑制成骨作用。沉默hsa-miR-6817-5p可以部分逆转si-circRFWD2和si-circINO80对成骨的负面影响。因此,circRFWD2和circINO80可以调控hsa-miR-6817-5p的表达,并影响重组NELL-1诱导的人脂肪干细胞的成骨分化。LONG等[71]对小鼠脂肪干细胞成骨分化过程的环状RNA进行芯片检测,发现mmu_circRNA_013422和mmu_circRNA_22566可能在成骨分化中发挥调控作用。目前对于环状RNA在脂肪干细胞成骨分化过程中的作用机制研究还相对较少。 2.5.5 环状RNA参与上颌窦黏膜干细胞成骨分化的研究 上颌窦黏膜来源的上颌窦黏膜干细胞具有成骨分化能力[72-73],在上颌窦底提升成骨过程中可能发挥重要作用。研究也证实了上颌窦黏膜干细胞具有成骨分化能力。PENG等[74]采用微阵列分析正常和骨形态发生蛋白2诱导的上颌窦黏膜干细胞成骨分化过程中circRNA的表达特征。circRNA_33287的过表达和沉默分别增加和降低成骨关键标志物的表达水平。此外,circRNA_33287充当miR-214-3p的分子海绵,它通过靶向Runx3的3’UTR来调节Runx3的表达。同时,circRNA_33287保护Runx3免受miR-214-3p介导的抑制。当与miR-214-3p抑制剂共转染时,circRNA_33287可增加体内异位骨的形成,并具有较强的刺激骨形成的能力。 2.5.6 环状RNA参与根尖乳头干细胞成骨分化的研究 自人类脱落的乳牙分离的间充质干细胞,具有与牙髓干细胞相似的成骨、成软骨、成脂和神经源性分化能力,且比骨髓间充质干细胞或牙髓干细胞的增殖活性更高。据报道,根尖牙乳头干细胞存在于发育中的根尖牙根乳头中,迁移、再生和增殖能力强并且能够在体内形成牙本质[75]。LI等[76]分离的根尖牙乳头干细胞分别在生长培养基或成骨培养基中培养并进行RNA测序,确定了环状RNA的表达谱和构建的circRNA-miRNA-mRNA网络,共检测到333个未调节的环状RNA和317个下调的环状RNA在成骨分化中的显著变化。生物信息学分析确定了几种生物途径可能参与根尖牙乳头干细胞的成骨分化。此外,选择了10个环状RNA,21个miRNAs和19个mRNAs来构建竞争性内源RNA网络。环状RNA在根尖牙乳头干细胞成骨分化的机制研究相对较少。参与间充质干细胞成骨分化的环状RNAs及其相关信息见表1。"
| [1] WILUSZ JE. A 360 view of circular RNAs: From biogenesis to functions. Wiley Interdiscip Rev RNA. 2018;9(4):e1478. [2] SANGER HL, KLOTZ G, RIESNER D, et al. Viroids are single-stranded covalently closed circular RNA molecules existing as highly base-paired rod-like structures. Proc Natl Acad Sci U S A. 1976;73(11):3852-3856. [3] HUANG S, YANG B, CHEN BJ, et al. The emerging role of circular RNAs in transcriptome regulation. Genomics. 2017;109(5-6):401-407. [4] CHEN H, MAO M, JIANG J, et al. Circular RNA CDR1as acts as a sponge of miR-135b-5p to suppress ovarian cancer progression. Onco Targets Ther. 2019;12:3869-3879. [5] LI X, YANG L, CHEN LL. The Biogenesis, Functions, and Challenges of Circular RNAs. Mol Cell. 2018;71(3):428-442. [6] DE FRAIPONT F, GAZZERI S, CHO WC, et al. Circular RNAs and RNA Splice Variants as Biomarkers for Prognosis and Therapeutic Response in the Liquid Biopsies of Lung Cancer Patients. Front Genet. 2019;10:390. [7] SUZUKI H, TSUKAHARA T. A view of pre-mRNA splicing from RNase R resistant RNAs. Int J Mol Sci. 2014;15(6):9331-9342. [8] CAI J, CHEN Z, ZUO X. circSMARCA5 Functions as a Diagnostic and Prognostic Biomarker for Gastric Cancer. Dis Markers. 2019;2019:2473652. [9] ZENG Y, DU WW, WU Y, et al. A Circular RNA Binds To and Activates AKT Phosphorylation and Nuclear Localization Reducing Apoptosis and Enhancing Cardiac Repair. Theranostics. 2017;7(16):3842-3855. [10] KUMAR L, SHAMSUZZAMA, HAQUE R, et al. Circular RNAs: the Emerging Class of Non-coding RNAs and Their Potential Role in Human Neurodegenerative Diseases. Mol Neurobiol. 2017;54(9):7224-7234. [11] CHEN BJ, HUANG S, JANITZ M. Changes in circular RNA expression patterns during human foetal brain development. Genomics. 2019;111(4):753-758. [12] ZHENG Q, BAO C, GUO W, et al. Circular RNA profiling reveals an abundant circHIPK3 that regulates cell growth by sponging multiple miRNAs. Nat Commun. 2016;7:11215. [13] 陈欣,张春林.组织工程骨修复骨缺损的研究进展及临床应用[J].中国组织工程研究,2010,14(24):4486-4490. [14] 王慎东.自体及异体骨软骨移植与骨缺损修复[J].中国组织工程研究, 2008,12(40):7905-7908. [15] HUANG X, CEN X, ZHANG B, et al. Prospect of circular RNA in osteogenesis: A novel orchestrator of signaling pathways. J Cell Physiol. 2019;234(12): 21450-21459. [16] CHEN LL, YANG L. Regulation of circRNA biogenesis. RNA Biol. 2015;12(4): 381-388. [17] JECK WR, SORRENTINO JA, WANG K, et al. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA. 2013;19(2):141-157. [18] RYBAK-WOLF A, STOTTMEISTER C, GLAŽAR P, et al. Circular RNAs in the Mammalian Brain Are Highly Abundant, Conserved, and Dynamically Expressed. Mol Cell. 2015;58(5):870-885. [19] SALZMAN J, CHEN RE, OLSEN MN, et al. Cell-type specific features of circular RNA expression. PLoS Genet. 2013;9(9):e1003777. [20] DANAN M, SCHWARTZ S, EDELHEIT S, et al. Transcriptome-wide discovery of circular RNAs in Archaea. Nucleic Acids Res. 2012;40(7):3131-3142. [21] SALZMAN J, GAWAD C, WANG PL, et al. Circular RNAs are the predominant transcript isoform from hundreds of human genes in diverse cell types. PLoS One. 2012;7(2):e30733. [22] LI Z, HUANG C, BAO C, et al. Exon-intron circular RNAs regulate transcription in the nucleus. Nat Struct Mol Biol. 2015;22(3):256-264. [23] CHEN I, CHEN CY, CHUANG TJ. Biogenesis, identification, and function of exonic circular RNAs. Wiley Interdiscip Rev RNA. 2015;6(5):563-579. [24] CHEN LL. The biogenesis and emerging roles of circular RNAs. Nat Rev Mol Cell Biol. 2016;17(4):205-211. [25] JECK WR, SHARPLESS NE. Detecting and characterizing circular RNAs. Nat Biotechnol. 2014;32(5):453-461. [26] ZHANG Y, ZHANG XO, CHEN T, et al. Circular intronic long noncoding RNAs. Mol Cell. 2013;51(6):792-806. [27] ZHANG XO, WANG HB, ZHANG Y, et al. Complementary sequence-mediated exon circularization. Cell. 2014;159(1):134-147. [28] WANG PL, BAO Y, YEE MC, et al. Circular RNA is expressed across the eukaryotic tree of life. PLoS One. 2014;9(6):e90859. [29] SUZUKI H, ZUO Y, WANG J, et al. Characterization of RNase R-digested cellular RNA source that consists of lariat and circular RNAs from pre-mRNA splicing. Nucleic Acids Res. 2006;34(8):e63. [30] CHEN S, HUANG V, XU X, et al. Widespread and Functional RNA Circularization in Localized Prostate Cancer. Cell. 2019;176(4):831-843.e22. [31] BURD CE, JECK WR, LIU Y, et al. Expression of linear and novel circular forms of an INK4/ARF-associated non-coding RNA correlates with atherosclerosis risk. PLoS Genet. 2010;6(12):e1001233. [32] ZHAO Y, ALEXANDROV PN, JABER V, et al. Deficiency in the Ubiquitin Conjugating Enzyme UBE2A in Alzheimer’s Disease (AD) is Linked to Deficits in a Natural Circular miRNA-7 Sponge (circRNA; ciRS-7). Genes (Basel). 2016; 7(12):116. [33] WU DM, WEN X, HAN XR, et al. Role of Circular RNA DLEU2 in Human Acute Myeloid Leukemia. Mol Cell Biol. 2018;38(20):e00259-018. [34] CHERUBINI A, BARILANI M, ROSSI RL, et al. FOXP1 circular RNA sustains mesenchymal stem cell identity via microRNA inhibition. Nucleic Acids Res. 2019;47(10):5325-5340. [35] WANG K, LONG B, LIU F, et al. A circular RNA protects the heart from pathological hypertrophy and heart failure by targeting miR-223. Eur Heart J. 2016;37(33):2602-2611. [36] ASHWAL-FLUSS R, MEYER M, PAMUDURTI NR, et al. circRNA biogenesis competes with pre-mRNA splicing. Mol Cell. 2014;56(1):55-66. [37] QIAN W, CAI X, QIAN Q, et al. Angelica Sinensis Polysaccharide Suppresses Epithelial-Mesenchymal Transition and Pulmonary Fibrosis via a DANCR/AUF-1/FOXO3 Regulatory Axis. Aging Dis. 2020;11(1):17-30. [38] HANSEN TB, JENSEN TI, CLAUSEN BH, et al. Natural RNA circles function as efficient microRNA sponges. Nature. 2013;495(7441):384-388. [39] MEMCZAK S, JENS M, ELEFSINIOTI A, et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature. 2013;495(7441):333-338. [40] HANSEN TB, KJEMS J, DAMGAARD CK. Circular RNA and miR-7 in cancer. Cancer Res. 2013;73(18):5609-5612. [41] WANG H, FENG C, JIN Y, et al. Identification and characterization of circular RNAs involved in mechanical force-induced periodontal ligament stem cells. J Cell Physiol. 2019;234(7):10166-10177. [42] LEGNINI I, DI TIMOTEO G, ROSSI F, et al. Circ-ZNF609 Is a Circular RNA that Can Be Translated and Functions in Myogenesis. Mol Cell. 2017;66(1):22-37.e9. [43] ZHANG M, JIA L, ZHENG Y. circRNA Expression Profiles in Human Bone Marrow Stem Cells Undergoing Osteoblast Differentiation. Stem Cell Rev Rep. 2019;15(1):126-138. [44] MIZUNO Y, TOKUZAWA Y, NINOMIYA Y, et al. miR-210 promotes osteoblastic differentiation through inhibition of AcvR1b. FEBS Lett. 2009;583(13):2263-2268. [45] ZHANG J, TU Q, BONEWALD LF, et al. Effects of miR-335-5p in modulating osteogenic differentiation by specifically downregulating Wnt antagonist DKK1. J Bone Miner Res. 2011;26(8):1953-1963. [46] ZHAO R, LI Y, LIN Z, et al. miR-199b-5p modulates BMSC osteogenesis via suppressing GSK-3β/β-catenin signaling pathway. Biochem Biophys Res Commun. 2016;477(4):749-754. [47] BAHN JH, ZHANG Q, LI F, et al. The landscape of microRNA, Piwi-interacting RNA, and circular RNA in human saliva. Clin Chem. 2015;61(1):221-230. [48] LI F, ZHANG L, LI W, et al. Circular RNA ITCH has inhibitory effect on ESCC by suppressing the Wnt/β-catenin pathway. Oncotarget. 2015;6(8):6001-6013. [49] FRIEDENSTEIN AJ, PETRAKOVA KV, KUROLESOVA AI, et al. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation. 1968;6(2):230-247. [50] PITTENGER MF, MACKAY AM, BECK SC, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143-147. [51] JIANG Y, JAHAGIRDAR BN, REINHARDT RL, et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature. 2002;418(6893):41-49. [52] GUILAK F, COHEN DM, ESTES BT, et al. Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell. 2009;5(1):17-26. [53] SI YL, ZHAO YL, HAO HJ, et al. MSCs: Biological characteristics, clinical applications and their outstanding concerns. Ageing Res Rev. 2011;10(1):93-103. [54] REN G, CHEN X, DONG F, et al. Concise review: mesenchymal stem cells and translational medicine: emerging issues. Stem Cells Transl Med. 2012; 1(1):51-58. [55] BARRY FP, MURPHY JM, ENGLISH K, et al. Immunogenicity of adult mesenchymal stem cells: lessons from the fetal allograft. Stem Cells Dev. 2005;14(3):252-265. [56] XIE XJ, WANG JA, CAO J, et al. Differentiation of bone marrow mesenchymal stem cells induced by myocardial medium under hypoxic conditions. Acta Pharmacol Sin. 2006;27(9):1153-1158. [57] NOËL D, DJOUAD F, JORGENSE C. Regenerative medicine through mesenchymal stem cells for bone and cartilage repair. Curr Opin Investig Drugs. 2002;3(7):1000-1004. [58] CHEN G, WANG Q, LI Z, et al. Circular RNA CDR1as promotes adipogenic and suppresses osteogenic differentiation of BMSCs in steroid-induced osteonecrosis of the femoral head. Bone. 2020;133:115258. [59] OUYANG Z, TAN T, ZHANG X, et al. CircRNA hsa_circ_0074834 promotes the osteogenesis-angiogenesis coupling process in bone mesenchymal stem cells (BMSCs) by acting as a ceRNA for miR-942-5p. Cell Death Dis. 2019;10(12):932. [60] KUANG MJ, XING F, WANG D, et al. CircUSP45 inhibited osteogenesis in glucocorticoid-induced osteonecrosis of femoral head by sponging miR-127-5p through PTEN/AKT signal pathway: Experimental studies. Biochem Biophys Res Commun. 2019;509(1):255-261. [61] WANG XB, LI PB, GUO SF, et al. circRNA_0006393 promotes osteogenesis in glucocorticoid‑induced osteoporosis by sponging miR‑145‑5p and upregulating FOXO1. Mol Med Rep. 2019;20(3):2851-2858. [62] YANG L, ZENG Z, KANG N, et al. Circ-VANGL1 promotes the progression of osteoporosis by absorbing miRNA-217 to regulate RUNX2 expression. Eur Rev Med Pharmacol Sci. 2019;23(3):949-957. [63] SEO BM, MIURA M, GRONTHOS S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004;364 (9429):149-155. [64] OHTA S, YAMADA S, MATUZAKA K, et al. The behavior of stem cells and progenitor cells in the periodontal ligament during wound healing as observed using immunohistochemical methods. J Periodontal Res. 2008; 43(6):595-603. [65] ZHENG Y, LI X, HUANG Y, et al. The Circular RNA Landscape of Periodontal Ligament Stem Cells During Osteogenesis. J Periodontol. 2017;88(9):906-914. [66] GU X, LI M, JIN Y, et al. Identification and integrated analysis of differentially expressed lncRNAs and circRNAs reveal the potential ceRNA networks during PDLSC osteogenic differentiation. BMC Genet. 2017;18(1):100. [67] LI X, ZHENG Y, ZHENG Y, et al. Circular RNA CDR1as regulates osteoblastic differentiation of periodontal ligament stem cells via the miR-7/GDF5/SMAD and p38 MAPK signaling pathway. Stem Cell Res Ther. 2018;9(1):232. [68] ZUK PA, ZHU M, ASHJIAN P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12):4279-4295. [69] ZUK PA, ZHU M, MIZUNO H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211-228. [70] HUANG X, CEN X, ZHANG B, et al. The roles of circRFWD2 and circINO80 during NELL-1-induced osteogenesis. J Cell Mol Med. 2019;23(12):8432-8441. [71] LONG T, GUO Z, HAN L, et al. Differential Expression Profiles of Circular RNAs During Osteogenic Differentiation of Mouse Adipose-Derived Stromal Cells. Calcif Tissue Int. 2018;103(3):338-352. [72] LI X, CHEN SL, ZHU SX, et al. Guided bone regeneration using collagen membranes for sinus augmentation. Br J Oral Maxillofac Surg. 2012;50(1): 69-73. [73] MAHLER D, LEVIN L, ZIGDON H, et al. The “dome phenomenon” associated with maxillary sinus augmentation. Clin Implant Dent Relat Res. 2009;11 Suppl 1:e46-51. [74] PENG W, ZHU S, CHEN J, et al. Hsa_circRNA_33287 promotes the osteogenic differentiation of maxillary sinus membrane stem cells via miR-214-3p/Runx3. Biomed Pharmacother. 2019;109:1709-1717. [75] 马学娟,陈旭.牙源性干细胞应用于牙髓牙本质再生研究进展[J].中国实用口腔科杂志,2017,10(10):635-638. [76] LI Z, LI N, GE X, et al. Differential circular RNA expression profiling during osteogenic differentiation of stem cells from apical papilla. Epigenomics. 2019;11(9):1057-1073. |
| [1] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1316-1322. |
| [2] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1323-1329. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1183-1190. |
| [4] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1191-1197. |
| [5] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1033-1039. |
| [6] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1040-1046. |
| [7] | Gao Yujin, Peng Shuanglin, Ma Zhichao, Lu Shi, Cao Huayue, Wang Lang, Xiao Jingang. Osteogenic ability of adipose stem cells in diabetic osteoporosis mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1047-1052. |
| [8] | Hou Jingying, Guo Tianzhu, Yu Menglei, Long Huibao, Wu Hao. Hypoxia preconditioning targets and downregulates miR-195 and promotes bone marrow mesenchymal stem cell survival and pro-angiogenic potential by activating MALAT1 [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1053-1059. |
| [9] | Zhou Ying, Zhang Huan, Liao Song, Hu Fanqi, Yi Jing, Liu Yubin, Jin Jide. Immunomodulatory effects of deferoxamine and interferon gamma on human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1060-1067. |
| [10] | Liang Xuezhen, Yang Xi, Li Jiacheng, Luo Di, Xu Bo, Li Gang. Bushen Huoxue capsule regulates osteogenic and adipogenic differentiation of rat bone marrow mesenchymal stem cells via Hedgehog signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1074. |
| [11] | Wang Jifang, Bao Zhen, Qiao Yahong. miR-206 regulates EVI1 gene expression and cell biological behavior in stem cells of small cell lung cancer [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1075-1079. |
| [12] | Liu Feng, Peng Yuhuan, Luo Liangping, Wu Benqing. Plant-derived basic fibroblast growth factor maintains the growth and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1080-1085. |
| [13] | Wen Dandan, Li Qiang, Shen Caiqi, Ji Zhe, Jin Peisheng. Nocardia rubra cell wall skeleton for extemal use improves the viability of adipogenic mesenchymal stem cells and promotes diabetes wound repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1086-1092. |
| [14] | Zhu Bingbing, Deng Jianghua, Chen Jingjing, Mu Xiaoling. Interleukin-8 receptor enhances the migration and adhesion of umbilical cord mesenchymal stem cells to injured endothelium [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1098. |
| [15] | Luo Xiaoling, Zhang Li, Yang Maohua, Xu Jie, Xu Xiaomei. Effect of naringenin on osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1099-1104. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||