Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (25): 3981-3987.doi: 10.12307/2021.008
Previous Articles Next Articles
Comparison on the curative effect of human placenta-derived mesenchymal stem cells and induced islet-like cells in gestational diabetes mellitus rats
Gao Shan1, Huang Dongjing1, Hong Haiman1, Jia Jingqiao2, Meng Fei1
- 1Department of Obstetrics, Affiliated Central Hospital of Shenyang Medical College, Shenyang 110024, Liaoning Province, China; 2WellCare Stem Cell Biotech (Liaoning) Co., Ltd., Benxi 110024, Liaoning Province, China
-
Received:2020-06-17Revised:2020-06-24Accepted:2020-08-04Online:2021-09-08Published:2021-03-24 -
Contact:Meng Fei, Master’s supervisor, Professor, Department of Obstetrics, Affiliated Central Hospital of Shenyang Medical College, Shenyang 110024, Liaoning Province, China -
About author:Gao Shan, Master, Associate chief physician, Department of Obstetrics, Affiliated Central Hospital of Shenyang Medical College, Shenyang 110024, Liaoning Province, China -
Supported by:the Science and Technology Fund of Shenyang Medical College, No. 20182040 (to GS); the Scientific Research Project of Shenyang Health and Family Planning Commission (to GS)
CLC Number:
Cite this article
Gao Shan, Huang Dongjing, Hong Haiman, Jia Jingqiao, Meng Fei. Comparison on the curative effect of human placenta-derived mesenchymal stem cells and induced islet-like cells in gestational diabetes mellitus rats[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3981-3987.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
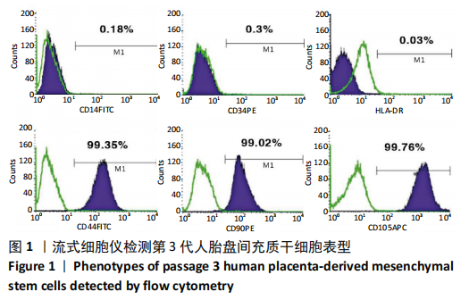
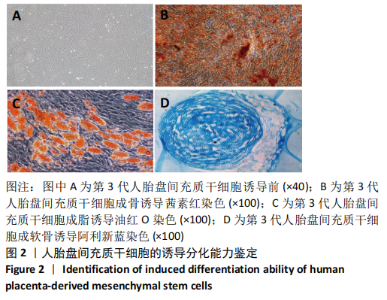
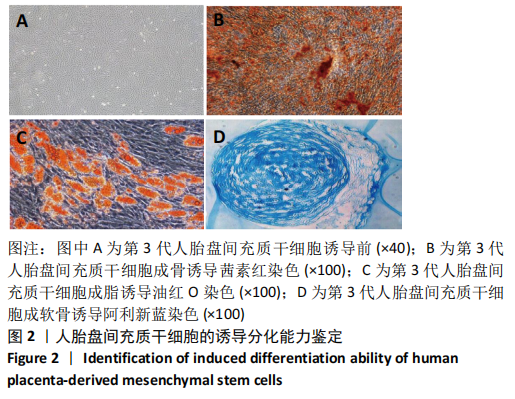
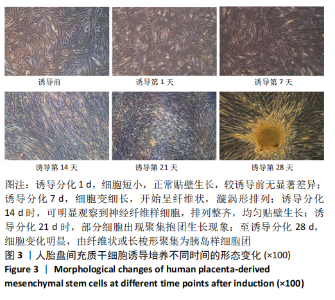
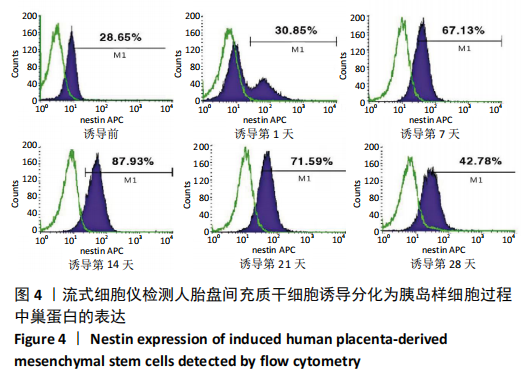
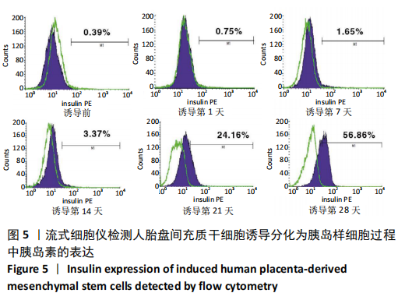
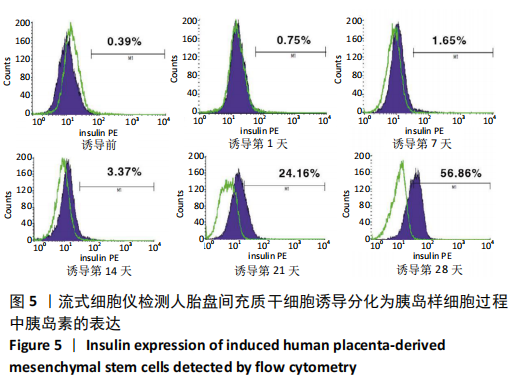
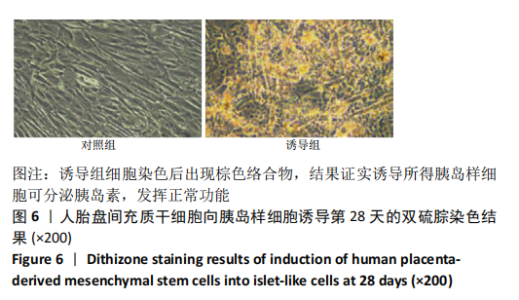
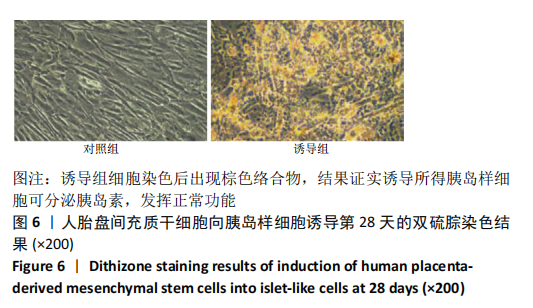
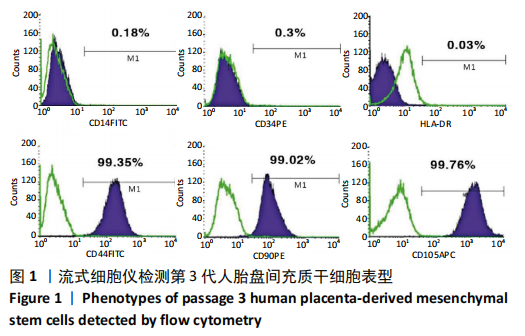
2.1 诱导前人胎盘间充质干细胞形态学特征 胎盘组织在培养瓶中培养至14 d时,可见细长短小梭形细胞,待80%细胞融合后进行传代培养至第3代,大多数细胞表现为长梭形,排列整齐,贴壁均匀,细胞增殖活力良好,可进行下一步鉴定。 2.2 人胎盘间充质干细胞鉴定结果 流式细胞仪检测第3代人胎盘间充质干细胞阴性表达CD14-FITC(0.18%)、CD34-PE (0.30%)、HLA-DR-APC (0.03%),阳性表达CD44-FITC (99.35%)、CD90-PE(99.02%)、CD105-APC(99.76%),见图1,上述结果与胎盘干细胞表面标志相符[23]。第3代人胎盘间充质干细胞成骨诱导21 d经茜素红染色呈阳性,有大小不等橘红色钙结节;成脂肪诱导21 d经油红O染色呈阳性,细胞为红色,核为蓝色,脂肪颗粒呈红色;成软骨诱导21 d经甲苯胺蓝染色呈阳性,细胞呈蓝色,局部呈球状,见图2。"
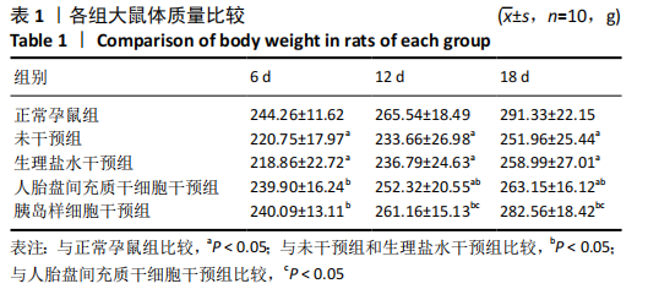
2.7 各组大鼠体质量变化 各组大鼠妊娠后体质量均随时间增加,但不同处理组增加程度不同,见表1。 妊娠第6天:各妊娠期糖尿病组大鼠体质量均低于正常孕鼠组,组间总体差异有显著性意义(F=41.19,P=0.000 1)。LSD法两两比较显示,未干预组和生理盐水干预组大鼠体质量显著低于正常孕鼠组(P < 0.05),人胎盘间充质干细胞干预组和胰岛样细胞干预组体质量显著高于未干预组和生理盐水干预组(P < 0.05),人胎盘间充质干细胞干预组、胰岛样细胞干预组和正常孕鼠组间差异无显著性意义,未干预组、生理盐水干预组间差异无显著性意义。 妊娠第12天:各妊娠期糖尿病组大鼠体质量均低于正常孕鼠组,组间总体差异有显著性意义(F=41.19,P=0.001)。LSD法两两比较显示,未干预组和生理盐水干预组大鼠体质量显著低于正常孕鼠组(P < 0.05),人胎盘间充质干细胞干预组和胰岛样细胞干预组体质量显著高于未干预组和生理盐水干预组(P < 0.05),人胎盘间充质干细胞干预组体质量显著低于正常孕鼠组(P < 0.05),胰岛样细胞干预组体质量显著高于人胎盘间充质干细胞干预组(P < 0.05),但正常孕鼠组、胰岛样细胞干预组间差异无显著性意义,未干预组、生理盐水干预组间差异无显著性意义。 妊娠第18天:各组间总体差异有显著性意义(F=125.67,P=0.000 1)。LSD法两两比较显示,未干预组和生理盐水干预组大鼠体质量显著低于正常孕鼠组(P < 0.05),人胎盘间充质干细胞干预组和胰岛样细胞干预组体质量显著高于未干预组和生理盐水干预组(P < 0.05),人胎盘间充质干细胞干预组体质量显著低于正常孕鼠组(P < 0.05),胰岛样细胞干预组体质量显著高于人胎盘间充质干细胞干预组(P < 0.05),但正常孕鼠组、胰岛样细胞干预组间差异无显著性意义,未干预组、生理盐水干预组间差异无显著性意义。"
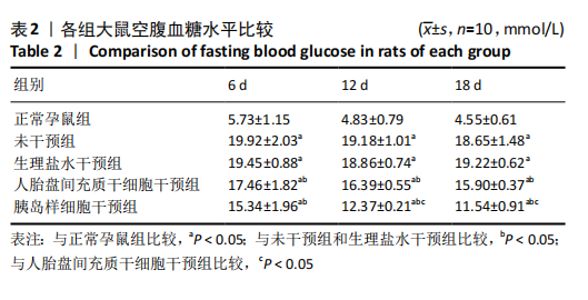
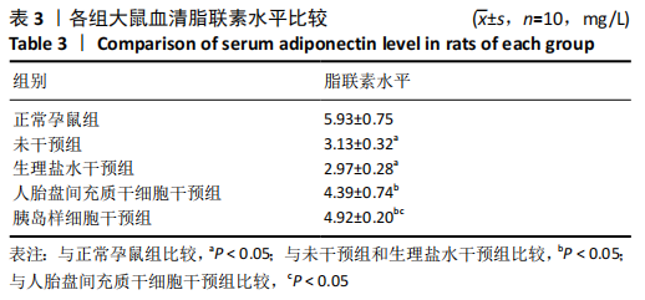
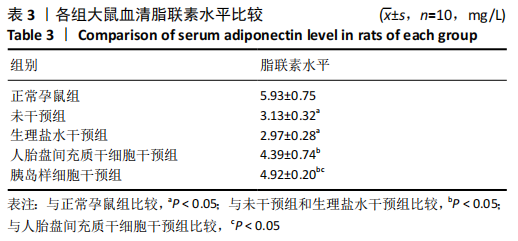
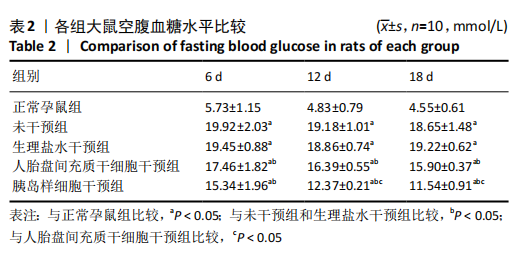
2.8 各组大鼠空腹血糖水平 注射人胎盘间充质干细胞和胰岛样细胞后,妊娠期糖尿病大鼠空腹血糖均随时间延长而逐渐降低,见表2。 妊娠第6天:各妊娠期糖尿病组大鼠空腹血糖均高于正常孕鼠组,总体差异有显著性意义(F=29.47,P=0.000 1),组间两两比较结果表明,人胎盘间充质干细胞干预组和胰岛样细胞干预组大鼠空腹血糖分别显著低于未干预组和生理盐水干预组(P < 0.05),但人胎盘间充质干细胞干预组、胰岛样细胞干预组间差异无显著性意义,未干预组、生理盐水干预组间差异也无显著性意义。妊娠第12天:各妊娠期糖尿病组大鼠空腹血糖显著高于正常孕鼠组,总体差异有显著性意义(F=47.13,P=0.000 1),组间两两比较结果表明,人胎盘间充质干细胞干预组和胰岛样细胞干预组血糖分别显著低于未干预组和生理盐水干预组(P < 0.05),人胎盘间充质干细胞干预组、胰岛样细胞干预组血糖均较妊娠第6天有所降低,且胰岛样细胞干预组血糖显著低于人胎盘间充质干细胞干预组(P < 0.05)。妊娠第18天:各妊娠期糖尿病组大鼠空腹血糖仍显著高于正常孕鼠组,总体差异有显著性意义(F=66.36,P=0.000 1),组间两两比较结果表明,人胎盘间充质干细胞干预组和胰岛样细胞干预组血糖显著低于未干预组和生理盐水干预组(P < 0.05),人胎盘间充质干细胞干预组、胰岛样细胞干预组血糖均较妊娠第12天有所降低,且胰岛样细胞干预组血糖依旧显著低于人胎盘间充质干细胞干预组 (P < 0.05)。"
| [1] DENNEY JM, QUINN KH. Gestational Diabetes: Underpinning Principles, Surveillance, and Management. Obstet Gynecol Clin North Am. 2018; 45(2):299-314. [2] 李艺,段丽娜,陈静,等.孕前体重指数与妊娠期糖尿病发生风险及妊娠结局的相关性研究[J].现代预防医学,2019,46(2):262-264, 273. [3] WANG Y, LUO BR. The association of body composition with the risk of gestational diabetes mellitus in Chinese pregnant women: A case-control study. Medicine (Baltimore). 2019;98(42):e17576. [4] LANE W, BAILEY TS, GERETY G. Insulin Analogues and Hypoglycemia in Patients With Type 1 Diabetes-Reply. JAMA. 2017;318(18):1828-1829. [5] 吴珊珊,顾俊菲,张永明.干细胞治疗1型糖尿病的研究进展[J].医学综述,2020,26(13):2647-2653. [6] DANNE T, HEINEMANN L, BOLINDER J. New Insulins, Biosimilars, and Insulin Therapy. Diabetes Technol Ther. 2018;20(S1):S55-S70. [7] QI Y, MA J, LI S, et al. Applicability of adipose-derived mesenchymal stem cells in treatment of patients with type 2 diabetes. Stem Cell Res Ther. 2019;10(1):274. [8] SORIA-JUAN B, ESCACENA N, CAPILLA-GONZÁLEZ V, et al. Cost-Effective, Safe, and Personalized Cell Therapy for Critical Limb Ischemia in Type 2 Diabetes Mellitus. Front Immunol. 2019;10:1151. [9] CAPLAN AI. Mesenchymal stem cells. J Orthop Res. 1991;9(5):641-650. [10] NITKIN CR, BONFIELD TL. Concise Review: Mesenchymal Stem Cell Therapy for Pediatric Disease: Perspectives on Success and Potential Improvements. Stem Cells Transl Med. 2017;6(2):539-565. [11] PELLEGRINI S, PIEMONTI L, SORDI V. Pluripotent stem cell replacement approaches to treat type 1 diabetes. Curr Opin Pharmacol. 2018;43:20-26. [12] 牛婷,李爱斌,曹景云,等.胎盘间充质干细胞的应用研究[J]. 中国组织工程研究,2015,19(32):5236-5242. [13] 项杰,张劼.间充质干细胞治疗糖尿病足:问题与更好的应用[J].中国组织工程研究,2018,22(1):146-151. [14] LENG ZK, GAO ZC, HE XJ, et al. Cultivation, screening, identification and transplantation of Muse cell from human umbilical cord-derived for spinal cord injury in rats. Zhongguo Gu Shang. 2019;32(4):327-334. [15] XU M, LIU G, JIA Y, et al. Transplantation of human placenta mesenchymal stem cells reduces the level of inflammatory factors in lung tissues of mice with acute lung injury. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi. 2018;34(2):105-109. [16] 黄启林,杨屹,罗晨,等.胎盘间充质干细胞移植对大鼠重症急性胰腺炎的保护作用[J].第三军医大学学报,2020,42(7):646-655. [17] 马晨,李晓国,徐明均,等.人胎盘间充质干细胞的扩增与鉴定[J].中国组织工程研究,2019,23(33):5293-5299. [18] LI XY, WU SY, LEUNG PS. Human Fetal Bone Marrow-Derived Mesenchymal Stem Cells Promote the Proliferation and Differentiation of Pancreatic Progenitor Cells and the Engraftment Function of Islet-Like Cell Clusters. Int J Mol Sci. 2019;20(17):4083. [19] 于健,周燕,黎萍,等.妊娠糖尿病大鼠视黄醇结合蛋白4水平的变化及与胰岛素抵抗的关系[J].山西医药杂志,2013,42(6):606-608. [20] 林峰,吴洁,王晓,等.黄连素改善妊娠期糖尿病大鼠胰岛素抵抗及其机制研究[J].中华全科医学,2019,17(10):1647-1651. [21] RETNAKARAN R, YE C, CONNELLY PW, et al. Serum apoA1 (Apolipoprotein A-1), Insulin Resistance, and the Risk of Gestational Diabetes Mellitus in Human Pregnancy-Brief Report. Arterioscler Thromb Vasc Biol. 2019;39(10):2192-2197. [22] BOHÁČOVÁ P, HOLÁŇ V. Mesenchymal stem cells and type 1 diabetes treatment. Vnitr Lek. 2018;64(7-8):725-728. [23] LIAU LL, MAKPOL S, AZURAH AGN, et al. Human adipose-derived mesenchymal stem cells promote recovery of injured HepG2 cell line and show sign of early hepatogenic differentiation. Cytotechnology. 2018;70(4):1221-1233. [24] MITANCHEZ D, YZYDORCZYK C, SIDDEEK B, et al. The offspring of the diabetic mother--short- and long-term implications. Best Pract Res Clin Obstet Gynaecol. 2015;29(2):256-269. [25] SNEDDON JB, TANG Q, STOCK P, et al. Stem Cell Therapies for Treating Diabetes: Progress and Remaining Challenges. Cell Stem Cell. 2018; 22(6):810-823. [26] BERNAL A, ARRANZ L. Nestin-expressing progenitor cells: function, identity and therapeutic implications. Cell Mol Life Sci. 2018;75(12): 2177-2195. [27] NAKAHARA F, BORGER DK, WEI Q, et al. Engineering a haematopoietic stem cell niche by revitalizing mesenchymal stromal cells. Nat Cell Biol. 2019;21(5):560-567. [28] 谢幸,孔北华,段涛.妇产科学[M].9版.北京:人民卫生出版社, 2018:105-109. [29] SHEN Y, JIA Y, LI Y, et al. Genetic determinants of gestational diabetes mellitus: a case-control study in two independent populations. Acta Diabetol. 2020;57(7):843-852. [30] NGUYEN-NGO C, SALOMON C, QUAK S, et al. Nobiletin exerts anti-diabetic and anti-inflammatory effects in an in vitro human model and in vivo murine model of gestational diabetes. Clin Sci (Lond). 2020; 134(6):571-592. [31] ZHANG H, WANG Q, HE S, et al. Ambient air pollution and gestational diabetes mellitus: A review of evidence from biological mechanisms to population epidemiology. Sci Total Environ. 2020;719:137349. [32] DE GENNARO G, PALLA G, BATTINI L, et al. The role of adipokines in the pathogenesis of gestational diabetes mellitus. Gynecol Endocrinol. 2019;35(9):737-751. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [5] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [6] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [7] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [8] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [9] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [10] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [11] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [12] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [13] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [14] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [15] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||